Abstract
Intrauterine adhesions (IUAs) often occurred after common obstetrical and gynecological procedures or infections in women of reproductive age. It was characterized by the formation of endometrial fibrosis and prevention of endometrial regeneration, usually with devastating fertility consequences and poor treatment outcomes so far. Telocytes (TCs), as a novel interstitial cell type, present in female uterus with
Keywords
Introduction
Intrauterine adhesions (IUAs) are partial or total occlusions of uterine cavity caused by trauma or infection in basal layer of endometrium and irreversible damaged self-repair capacity. IUAs are frequently accompanied by various complications, such as hypomenorrhea, amenorrhea, recurrent miscarriage, or even devastating infertility, either of these will severely impact female reproductive health.1–3 Endometrial stromal cells (ESCs) are mainly located in basal layer of endometrium and are of great significance for self-renew or regeneration repair of endometrium after cyclic physiological menstruation, trauma, or infection-induced pathological endometrial damage.4,5 Wnt/β-catenin is a key signaling pathway involved in ESCs decidualization and mesenchymal–epithelial transition (MET), both were essential for ESCs maturation and endometrium reparation.6–9 Damage in number and function of ESCs, including abnormal Wnt/β-catenin signal pathway, impaired decidualization, and imbalanced epithelial–mesenchymal transition (EMT)/MET process, are all risk factors in the onset or recurrence of IUAs.10–12
Telocytes (TCs) are a newly discovered type of interstitial cell with multiple proposed functions, including paracrine effects, maintaining proliferation and homeostasis, anti-fibrosis in multiple tissues and organs, and so on.13–16 Some of these functions were based on experimental evidence, such as exosomes (Exo) derived from cardiac TCs containing multiple abundant microRNAs (miRNAs), which can decrease cardiac fibrosis, increase angiogenesis, and improve cardiac function in rat model of myocardial infarction.
17
Whereas, other proposed functions were highly speculative, merely based on characteristic ultrastructure, and intercellular junctions, thus need extensive investigations. More recently, TCs was found to provide essential non-epithelial Wnt ligand signals for self-renewal and differentiation of local stem cell (SC) niches, maintaining SC-mediated intestinal mucosal regeneration and repair.18,19 Our team previously found that Wnt ligands secreted by isolated uterine TCs have the capacity to activate Wnt/β-catenin pathway, subsequently enhance decidualization and MET process in ESCs
Based on known involvement of TCs in intestinal mucosa self-renewal18,19 and in ESCs enhancement
Materials and methods
Maintenance of animals
Animal experiments were reviewed and approved by the Ethics Committee of Soochow University (ECSU2019000163). Eight-week-old BALB/c-specific pathogen-free female virgin mice, weighing 20–25 g, were purchased from the Laboratory Animal Center of Soochow University. The animals were raised with chow and fresh water in an animal facility with the same controlled conditions (22°C, 14 h:10 h light/dark cycle).
Isolation and primary culture of uterine ESCs
ESCs were isolated, and cultured, as described previously.7,20 The uterine tissues of female BALB/c mice were collected under aseptic conditions and washed three times with phosphate-buffered saline (PBS) containing 100 U/ml penicillin and 0.1 mg/ml streptomycin (Beyotime, Shanghai, China), then minced into 0.5–1 mm3 pieces. Prepared uterine tissue was digested for 90 min at 4°C in 0.5% trypsin (Beyotime, Shanghai, China). The remaining tissue was incubated in Dulbecco’s modified eagle medium (DMEM)/F12 (Hyclone, Logan, UT, USA) supplemented with 0.1% type-II collagenase (Sigma-Aldrich China, Inc., Shanghai, China) at 37°C, and dissociated mechanically using a pipette every 15 min, followed by termination of the digestion reaction after 90 min. Subsequently, the resulting suspension was filtered through 100 µm and 40 sterile nylon-mesh filters (BD Falcon, Heidelberg, Germany). Following centrifugation at 302 ×
Isolation and primary culture of uterine TCs
The isolation and primary culture of uterine TCs were performed according to our previous procedures.7,20–22 The uterine was cut longitudinally, and incubated in 0.1% type-II collagenase in DMEM for 60 min at 37°C. Then, the tissue was placed with the luminal side facing up, and the endometrial layer of the uterus was scraped off using a razor, and then minced into 0.5–1 mm3 pieces. The tissue was incubated in 0.1% type-II collagenase for an additional 60 min. Subsequently, the resulting suspension was filtered through 40 µm sterile filters. The cells were washed with 10% FBS to stop the enzymatic reaction and then washed in PBS. Cells were cultured using DMEM/F12 medium supplemented with 10% FBS in a 10 cm dish for 2 h. The media containing slowly adhering cells were re-plated into a new dish and maintained in culture. After 48 h of incubation, the medium was replaced with serum-free DMEM/F12 medium and then cultured for an additional 24 h. Supernatants were collected and filtered through a 0.45-μm filter, referred to as TC-conditioned medium (TCM), stored at −80°C for subsequent experiments.
Development of IUA cell model (transforming growth factor-beta 1-treated ESCs)
Mouse transforming growth factor-beta 1 (TGF-β1; 0, 5, 10, 15, 20, and 25 ng/ml) (BioLegend, San Diego, USA) was administrated to ESCs for 72 h. The protein expression of fibrosis markers collagen type 1 alpha 1 (COL1A1), fibronectin (FN), and α-smooth muscle actin (α-SMA) was detected by Western blotting (WB). Preliminary results showed that the protein expression level was most prominent within ESCs after 15 ng/ml TGF-β1 exposure.
Then, 15 ng/ml TGF-β1 was chosen to treat ESCs for 0, 12, 24, 48, and 72 h respectively, with the same WB determination of COL1A1, FN, and α-SMA. The results showed that the combination of 15 ng/ml TGF-β1 and 48 h can reach the highest expression level of three fibrosis markers within ESCs, and therefore, develop the optimal IUA cell model, rendered for experiments that followed.
Treatment with TCM
To observe the reversal potential of TCM on fibrotic ESCs, the established TGF-β1-treated ESCs IUA model was seeded in six-well plates at a density of 3 × 105 cells/well and cultured in DMEM/F12 (served as control group: TGF-β1-ESCs + DMEM/F12) or in TCM (TCM group: TGF-β1-ESCs + TCM) supplemented with 10% FBS
Cell immunofluorescence
To observe the morphology of primary TCs and ESCs, double immunofluorescence staining was performed as described previously7,21 Briefly, primary TCs and ESCs were individually seeded at a suitable density on slides. After being fixed with 4% paraformaldehyde and blocked with 3% bovine serum albumin (BSA). TCs and ESCs were incubated with the first primary antibodies overnight at 4°C. Rabbit anti-vimentin and mouse anti-CD34 antibodies for TCs, rabbit anti-vimentin antibody, and mouse anti-pan cytokeratin for ESCs. After washing three times with PBS, both cells were incubated with fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit immunoglobulin G (IgG) (H + L) or Cy3-conjugated goat anti-mouse IgG (H + L) as secondary antibodies. Nuclei were stained with 4’6-diamidino-2-phenylindole (DAPI; Servicebio, Wuhan, China). Then, observed and imaged under an inverted fluorescent microscope (Nikon, Tokyo, Japan).
Meanwhile, similar immunofluorescence was also performed to observe fibrosis markers among three groups (blank, control, and TCM groups) as described above. The secondary antibody was Cy3-conjugated goat anti-mouse IgG (H + L) or Cy3-conjugated goat anti-rabbit IgG (H + L). The antibodies and labels were listed in Table 1.
List of Antibodies and Labels for WB, Cell IF, and Tissue IHC.

WB: Western blotting; Cell IF: Cell immunofluorescence; IHC: immunohistochemistry; CST: cell signaling technology; COL1A1: collagen type 1 alpha 1; FN: fibronectin; α-SMA: α-smooth muscle actin; VEGF: vascular endothelial growth factor; HRP: horse radish peroxidase.
Extraction, determination, and identification of TC-derived Exo
After the effectiveness of reversal on fibrosis was confirmed in TCM, Exo from TCM was extracted for further investigation using exoEasy Maxi Kit (Qiagen, Hilden, Germany) according to manufacturer’s instruction. Briefly, TCM was mixed with a binding buffer called XBP buffer in a 1:1 ratio. The buffer media were then loaded into exoEasy spin columns and centrifuged at 500 ×
Protein contents in Exo were determined for the subsequent quantification of WB experiments and administration dosage using PierceTM BCA Protein Assay Kit (Thermo Scientific, USA) according to the instruction manual. Briefly, Exo sample mixed with BCA working solution was incubated at 37°C for 30 min, then optical density (OD) value was tested at the wavelength of 562 nm. Size and concentration of Exo particles were determined using nanoparticle tracking analysis (NTA) with ZetaView PMX 110 (ZetaView software edition 8.04.02, Particle Metrix, Meerbusch, Germany). Briefly, as described previously,23,24 isolated Exo samples were appropriately diluted using PBS buffer (Biological Industries, Israel), and NTA measurement was analyzed and recorded at 11 positions. The ZetaView system was calibrated using 110 nm polystyrene particles. The temperature was maintained at around 28°C.
The morphology of Exo was observed under transmission electron microscope (TEM). A 20 µl suspension with Exo was negatively stained by phosphotungstic acid and fixed on copper mesh for 10 min at room temperature.25,26 The mesh was thoroughly dried before viewing and then observed under a TEM (JEOL, JEM-1200EX, Japan).
Meanwhile, WB was used to examine expression of CD63 and TSG101 in three independent samples, both markers are specific surface proteins pertaining to Exo.27,28
Vascular endothelial growth factor production in ESCs
Vascular endothelial growth factor (VEGF) is a specific substance for angiogenesis within endometrium which is produced by ESCs and is relevant to cyclic menstruation and endometrial regeneration. 29 TC-derived Exo was separately added into culture medium of TGF-β1-treated ESCs (3 × 105 cells/well) with a final concentration of 5 µg/ml, served as TC-Exo group (TGF-β1-ESCs + TC-Exo). Meanwhile, TGF-β1-treated and non-TGF-β1-treated regular ESCs served as control (TGF-β1-ESCs) and blank group (regular ESCs). After 48-h incubation, VEGF in three groups (blank, control, and TC-Exo group) was determined by quantitative real-time polymerase chain reaction (qRT-PCR) for mRNA and WB for protein in ESCs. And total VEGF in the culture medium released from ESCs was measured by a mouse VEGF Quantikine ELISA kit (R&D Systems, MN, USA) according to manufacturer’s instructions. The absorbance of samples was read at 450 nm using a microplate reader (BioTek Instruments Inc., USA).
Role of Wnt ligands and Wnt/β-catenin pathway
To investigate whether the observed
Meanwhile, to observe involvement of Wnt pathway in reversal of fibrotic ESCs, XAV939 (MCE, Shanghai, China), a small molecule inhibitor of Wnt/β-catenin pathway, which is capable of blocking Wnt signaling through upregulating degradation of β-catenin by stabilizing the axin protein, was used to inhibit Wnt/β-catenin signaling pathway within ESCs.33,34 A final concentration of 1 μM XAV939 was applied to reaction system containing TGF-β1-treated ESCs and TC-derived Exo with a final concentration of 5 µg/ml, served as TC-Exo + XAV939 group (TGF-β1-ESCs + TC-Exo + XAV939). Group settings (blank, control, and TC-Exo group) and procedures were the same as above-mentioned. Four groups of ESCs were incubated in the same conditions for 48 h, and fibrosis markers (COL1A1, FN, and α-SMA) and pathway protein (β-catenin) were determined with WB.
Quantitative real-time polymerase chain reaction
To measure mRNA expression of fibrosis marker COL1A1, FN, and α-SMA (Acta2), total RNA was lysed and extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to manufacturer’s instructions. The concentration and purity of the RNA were determined using a UV spectrophotometer (260 nm, Thermo Scientific, MA, USA). Briefly, 1 μg of cellular RNA was reverse transcribed into cDNA using a PrimeScriptTM RT Master Mix kit (Takara, Kyoto, Japan) to a final volume of 10 μl. Then, 1 μl of cDNA was added into PowerUpTM SYBRTM Green Master Mix (Thermo Scientific, Waltham, MA, USA) to a final volume of 20 μl. Amplification was performed on an ABI QuantStudio3 Detection System (Applied Biosystems, Carlsbad, CA, USA). The 2–ΔΔCt method was used to determine the relative quantification in each sample. All reactions were triple duplicated under the same conditions. The house-keeping gene GAPDH (glyceraldehyde-3-phosphate dehydrogenase) was used as an internal reference gene to normalize individual samples. The primer sequences are provided in Table 2.
List of qRT-PCR Primers.

qRT-PCR: quantitative real-time polymerase chain reaction; COL1A1: collagen type 1 alpha 1; FN: fibronectin; α-SMA: α-smooth muscle actin.
Western blotting
WB was performed as previously described7,21 to measure the protein expression. Briefly, total proteins were extracted using RIPA (radioimmunoprecipitation assay) lysis buffer (Beyotime, Shanghai, China) containing a protease inhibitor cocktail (BBI, Shanghai, China). After determining the concentration using a PierceTM BCA Protein Assay Kit (Thermo Scientific, USA), the proteins were resolved by 5%–12% sodium dodecyl-sulfate (SDS)-polyacrylamide gel, and then electrotransferred to 0.45 μm polyvinylidene fluoride (PVDF) membrane (Millipore, Billerica, MA, USA), followed by immunoblotting with the corresponding primary antibodies. Labeled with secondary antibodies, then, detected with an enhanced chemiluminescence kit (Absin Bioscience Inc., Shanghai, China). Results were photographed using a gel imaging system (Tanon, Shanghai, China) and analyzed by ImageJ software (ImageJ 1.53k, NIH). The expression levels of individual proteins were expressed as a ratio relative to the internal reference protein expression of β-tubulin. The antibodies and labels were listed in Table 1.
IUA mouse model
The mouse model of endometrial fibrosis or IUAs was established by the combination of mechanical uterine curettage and inflammatory lipopolysaccharides (LPSs) injection into uterine cavity as described in the literature.35,36 Briefly, daily vaginal smears proved estrum mice were anesthetized with an intraperitoneal injection of 1.25% avertin. Through vertical dorsal incision, the uterus was exposed, and uterine horn was mechanically injured with a rough surface needle inserted through the lumen and scratched up and down 50 times until the uterine inner wall became rough. A single dose of 20 μl LPS (10 mg/ml; Beyotime, Shanghai, China) was then injected into the uterine cavity to induce inflammatory damage, with both ends of the punctuation site sealed by tweezers for 5 min to avoid any liquid leakage. A pseudo-operation that received only vertical dorsal incision without any mechanical and inflammation intervention on uterine tissues served as sham control. After surgery, the mice received a prophylactic intramuscular injection of a single dose of 20,000 U penicillin (Solarbio, Beijing, China). Then, 5 days later, six mice were randomly assigned at 1:1 ratio into two groups, with three in each group: (A) IUA-TC-Exo group, (B) IUA group. Together with three in pseudo-operation served as the sham group (C).
Briefly, groups A and B received three doses of intrauterine injections (5 days apart) through the original incision with TC-derived Exo (200 μg/ml) for group A and 20 μl PBS for group B, with both ends of the punctuation site sealed by tweezers for 5 min to prevent any liquid leakage. The mice were maintained with the same procedure after each injection. Then, after the third injection, mice in all groups were sacrificed and uterine tissues were collected at the estrum.
Hematoxylin–eosin, Masson staining, and immunohistochemistry staining
Mouse uterine samples were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned at 5 mm. Hematoxylin–eosin (HE) and Masson trichrome staining were performed to observe endometrial histopathology and collagen content to evaluate IUA, respectively. Masson trichrome staining was performed using a Masson staining kit (Servicebio, Wuhan, China). For results evaluation, endometrial thickness measurements were obtained from the lateral sides of each cross-section under high-magnification fields. Briefly, a total of eight high-power fields (HPFs) (×400) with 45° intervals in each cross-section were chosen, measured, and averaged for each HE-stained section. Similarly, the glands numbers in three HPF (×400) were randomly selected, counted, and averaged to determine endometrial gland abundance. On the other hand, the fibrosis area ratio was calculated as follows: total area of endometrial fibrosis per field/the sum area of endometrial stroma and gland in Masson stained slice. 37 Images were analyzed using the ImageJ software (ImageJ 1.53k, NIH, USA).
For immunohistochemistry (IHC) staining, the slides were dewaxed and rehydrated, endogenous peroxidase activity was blocked with 3% hydrogen peroxide. Then, after blocking with 3% BSA for 1 h at room temperature, the slides were incubated at 4°C overnight with primary antibodies, including FN, E-cadherin, N-cadherin, VEGF, CD-31 (Table 1). FN is a fibrosis maker, E-cadherin and N-cadherin are closely related to EMT/MET phenotype. VEGF and CD31 were performed to assess angiogenesis and microvessel density (MVD). After being washed with PBS (PH = 7.4), slices were incubated with horse radish peroxidase (HRP)-conjugated goat anti-mouse/rabbit lgG (Table 1) as the secondary antibody for 50 min at room temperature. Then, DAB Substrate System (Servicebio, Wuhan, China) was used to reveal the IHC staining. Images were captured by an inverted microscope (CIC XSP-C204, China).
Results of immunostaining (FN, E-cadherin, N-cadherin, VEGF) were evaluated using IHC score, 38 which was calculated by multiplying the staining intensity score and percentage score. The staining intensity was divided into four scores: 0 (negative, no staining), 1 (weak, light brown), 2 (medium, brown), and 3 (strong, dark brown). Scores were combined to generate each IHC score (min, 0; max, 4). The analysis was performed by ImageJ software (ImageJ 1.53k, NIH, USA).
MVD was recorded as the number of CD31-positive endothelial cells or endothelial cell clusters per HPF (×400) from at least four areas with the highest vascularization. 39
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8 (GraphPad Software, La Jolla, CA, USA). Data are expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used to evaluate differences between three or multiple groups, followed with Tukey’s test for post hoc pairwise comparisons. The Shapiro–Wilk test was implemented to validate normal distribution. Statistical significance was set at
Results
Morphological and immunophenotype of ESCs and TCs
Primary ESCs show typical fusiform morphology with clear outline, sparse intercellular links, and vimentin (+)/cytokeratin (–) immunofluorescence staining (Fig. 1A), consistent with our previous results. 7

Morphology and immunophenotype of mouse uterine ESCs and TCs observed by immunofluorescence staining and inverted fluorescent microscope. Nuclei were counterstained with DAPI (blue). (A) Immunofluorescence staining of ESCs was positive for Vimentin (green) and negative for cytokeratin. Scale bar = 100 μm. (B) TCs were typical mesenchymal cells with a characteristic oval cellular body and long extensions named telopodes (Tps), composed of alternating thin (podomer) and thick (podom) segments under high-magnification fields. Double positive for Vimentin (green) and CD34 (red). In the merged image, both immunofluorescence signals overlapped each other along entire cellular body and Tps, with the podomer, and podom indicated. Scale bar = 20 μm. (C) Immunofluorescence staining of TCs was positive for Vimentin (green) and CD34 (red) under low-magnification fields. Scale bar = 100 μm. ESC: endometrial stromal cell; TC: telocyte; DAPI: 4’6-diamidino-2-phenylindole.
Primary uterine TCs show irregular or spindle cellular body with long and characteristic telopodes (Tps), which displayed alternating thin podomers and thick podoms segments. TCs show a typical immunophenotype of double-positive vimentin/CD34, with green and red immunofluorescence staining overlapping each other along the whole length of the cellular body and Tps (Fig. 1B, C).
Successful development of IUA cell model (TGF-β1-induced ESCs)
To induce cell fibrosis, primary ESCs were exposed to TGF-β1 with a combination of different concentrations and treatment times. WB results showed fibrosis markers (COL1A1, FN, and α-SMA) were in a concentration-dependent and time-dependent manner. For different concentrations, 15 ng/ml TGF-β1 can yield peak values of three markers (ANOVA, all

Time and dose-dependent effects of TGF-β1 on fibrosis markers expression (COL1A1, FN, and α-SMA) in regular ESCs analyzed by WB. β-Tubulin served as loading control for WB. Error bars indicated standard deviation (SD) from three independent experiments (biological replicates). #
TCM reversed fibrosis in IUA cell model
The reversal effect of TCM on TGF-β1-induced ESCs fibrosis was evidenced by qRT-PCR and WB determination of fibrosis markers (COLIA1, FN, and α-SMA). Results showed that when exposed to TCM, mRNA and protein expression decreased significantly in TGF-β1-induced ESCs than in control group (ANOVA, post hoc paired test, all

TCM exposure reversed and downregulated fibrosis markers (COL1A1, FN, and α-SMA) in TGF-β1-treated ESCs. Error bars indicated SD from three independent experiments (biological replicates). *
Identification of TC-derived Exo
The morphology of Exo was observed under TEM. Exo was elliptical or round vesicles with intact capsules and uneven sizes (Fig. 4A). The mean diameter of particle measured by NTA was 159.4 ± 54.9 nm, which was confirmed by TEM as well. The original concentration of Exo was 3.5 × 109 particles/ ml, with peak distribution when particle size was 138.6 nm (Fig. 4B). This was within the size of extracellular vesicles ranging from 45 nm to 1 μm reported before. 40 Meanwhile, WB analysis confirmed expression of CD63 and TSG101 (Fig. 4C), which were specific surface proteins and expected to be presented in Exo.27,28

Identification and characterization of TC-derived Exo. (A) Transmission electron micrographs of exosomes. Scale bar (from left to right): 500, 200, 100 nm, respectively. (B) NTA of TC-derived exosomes. Relationship between mean diameter (159.4 ± 54.9 nm) and original concentration of Exo (3.5 × 109 particles/ ml), with peak distribution when particle size being 138.6 nm. (C) WB was used to identify the expression of CD63 and TSG101, both were specific surface proteins pertaining to Exo. Lanes 1, 2, and 3 represent three repeats. TC: telocyte; NTA: nanoparticle tracking analysis; WB: Western blotting.
TC-derived Exo reversed fibrosis and enhanced VEGF secretion in IUA cell model
Reversal of fibrosis by TC-derived Exo in IUA cell model was evidenced by WB determination of fibrosis markers (COLIA1, FN, and α-SMA). Results showed that protein expression increased in the control group (TGF-β1-ESCs) than in blank (regular ESCs), but decreased significantly in the TC-Exo group (TGF-β1-ESCs + TC-Exo) (ANOVA, post hoc paired test, all
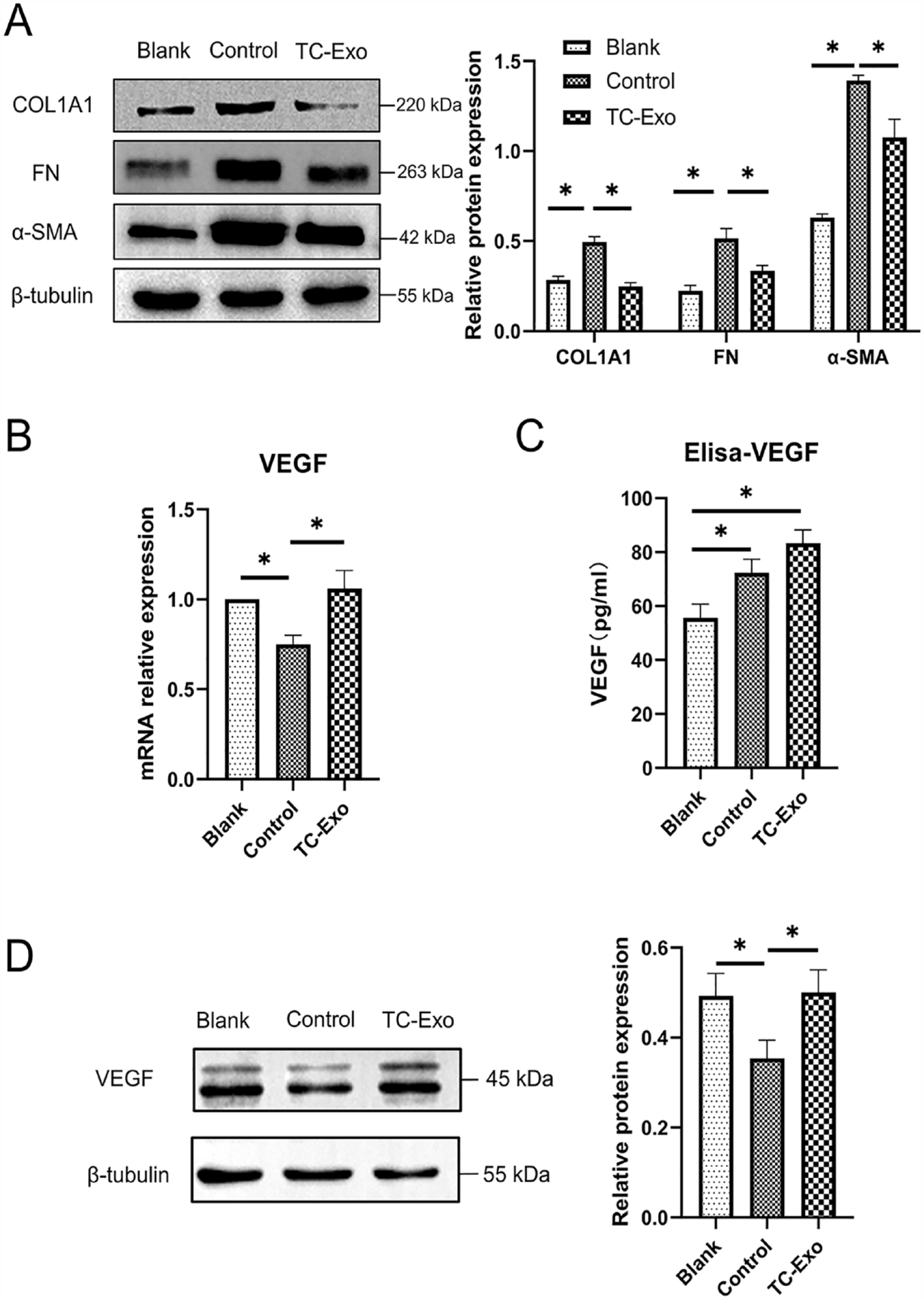
Reversal of fibrosis and enhanced secretion of VEGF in TGF-β1-treated ESCs when exposed to TC-derived Exo. Error bars indicate SD from three independent experiments (biological replicates). *
VEGF in groups of ESCs was determined by qRT-PCR for mRNA, WB for protein, and ELISA for culture media, respectively. Results showed VEGF mRNA and protein expression in the control group (TGF-β1-ESCs) decreased significantly than in blank (regular ESCs) but elevated significantly after TC-derived Exo salvage in TC-Exo group (TGF-β1-ESCs + TC-Exo) (ANOVA, post hoc paired test, all
Role of Wnt ligands and Wnt/β-catenin pathway
Involvement of Wnt/β-catenin in reversal of fibrotic ESCs was proved either in TCs blocked by ETC-159 or in ESCs blocked by XAV939, with the previous one aimed to block production of Wnt ligands, and the later one function to block Wnt/β-catenin pathway.
For ETC-159 experiment, TCs were pre-treated with ETC-159 and followed with Exo collection from cultural medium, named as ETC-159 treated TC-derived Exo (ETC-TC-Exo), together with the collection of regular TC-derived Exo. Four groups were designed: blank group (regular ESCs), control group (TGF-β1-ESCs), TC-Exo group (TGF-β1-ESCs + TC-Exo), and ETC-TC-Exo group (TGF-β1-ESCs + ETC-TC-Exo). Results showed that ETC-TC-Exo experienced a significant loss of fibrosis reversal when compared with regular TC-derived Exo (ANOVA, post hoc paired test, all

Reversal of fibrosis in TGF-β1-treated ESCs was antagonized by ETC-159 or XAV939 through blocking of Wnt ligands in TCs or Wnt/β-catenin pathway in ESCs. β-tubulin served as loading control. Error bars indicated SD from three independent experiments (biological replicates). *
For XAV939 experiment, four groups were designed: blank group (regular ESCs), control group (TGF-β1-ESCs), TC-Exo group (TGF-β1-ESCs + TC-Exo), and TC-Exo + XAV939 group (TGF-β1-ESCs + TC-Exo + XAV939). ESCs samples from all groups were collected for WB determination of fibrosis markers and β-catenin. Results showed that β-catenin increased in TC-Exo group (ANOVA, post hoc paired test, all
Morphometric analyses of endometrial thickness, glands, and fibrosis
Morphometric analyses indicated a successfully established IUA mouse model and the exact reversal effect on fibrosis by TC-Exo. First, histopathology observation (Fig. 7A) showed markedly decreased endometrium thickness and density of glands in IUA model group. These were totally different from the normal and intact endometrial structure in the Sham group, which have glandular epithelial cells, glands duct and capillaries fully developed. As shown in Fig. 7A, endometrial thicknesses were 519.80 ± 15.20, 384.70 ± 21.58, and 440.70 ± 18.84 µm, with endometrial glands numbers being 14.330 ± 3.283, 6.667 ± 0.333, and 8.889 ± 1.711 per HPF in the sham, IUA, and IUA-Exo groups, respectively. When compared with the sham group, a statistically thinner endometrium and a lesser number of glands were observed in IUA group (ANOVA, post hoc paired test, all

Morphometric analyses of endometrial thicknesses, number of endometrial glands, and fibrotic areas in IUA mouse model after TC-derived Exo treatment. Scale bar (from upper to lower enlarged images): 200, 20 μm, respectively. (A) HE staining to observe endometrial thicknesses and glands (arrow). (B) Masson trichrome staining of endometrium, with blue-stained collagen fibers and red-stained mucosa, submucosa, muscles, and blood vessels. (C) Statistical analysis of endometrial thicknesses, number of endometrial glands, and fibrotic areas in each group. *
Then, quantitative analysis showed TC-derived Exo treatment obviously decrease fibrosis area of the endometrium (ANOVA, all
Finally, as we know, FN constitutes a key component in extracellular matrix (ECM) and plays important role in the process of endometrium fibrosis.
41
IHC results demonstrated a significant difference of FN in endometrium among three groups (ANOVA, all

IHC of FN, CD31, VEGF, E-cadherin, and N-cadherin in uterine tissues from IUA mouse model. (A) IHC of FN, CD31, VEGF, E-cadherin, and N-cadherin in endometrial tissues. Arrow: CD31-positive endothelial cells. Scale bar = 20 μm. (B) Semi-quantitative analysis of FN, MVD, VEGF, E-cadherin, and N-cadherin in each group. *
TC-derived Exo enhanced angiogenesis in IUA model
MVD, an indicator of angiogenesis, was evaluated by immunostaining quantification of endothelial marker CD31. Results showed that neovascularization in IUA group (31.50 ± 2.136) significantly decreased compared with the sham group (54.33 ± 8.032), whereas TC-Exo treatment significantly increased CD31 immunostaining (47.33 ± 3.538) (ANOVA, all
Meanwhile, VEGF, a pro-angiogenic marker, was analyzed by IHC. The IHC score was 1.304 ± 0.1672, 1.183 ± 0.09087, and 1.304 ± 0.1833 in the sham, IUA, and IUA-Exo groups, respectively. Semi-quantitative analysis showed a similar trend as MVD, but lack of statistical difference among three groups (ANOVA, all
TC-derived Exo enhanced E-cadherin
E-cadherin, an epithelial adhesion molecule, is functionally linked to generation and maintaining of polarized epithelial phenotype and regulating tissue homeostasis.42–45 As shown in Fig. 8, IHC analysis showed E-cadherin expression in the IUA group (1.188 ± 0.0473) was significantly decreased compared with that in the sham group (1.665 ± 0.1966). Whereas TC-Exo treatment significantly enhanced E-cadherin expression (1.507 ± 0.0427) (ANOVA, all
Interestingly, no significant differences were observed for N-cadherin expression among three groups (sham: 1.466 ± 0.1918; IUA: 1.406 ± 0.1045; IUA-Exo: 1.408 ± 0.0985). This can be attributed to a prevalent expression of N-cadherin in non-epithelial tissues, including different types of cells, such as neural cells, endothelial cells, stromal cells, osteoblasts, and so on.46,47 Affections from TC-derived Exo might be overlapped and therefore cannot be significant reflected in non-epithelial tissues.
Discussion
The occurrence of IUAs has increased dramatically due to more frequent intrauterine interventions nowadays. The fundamental pathology of IUAs is endometrial fibrosis/scarring in response to various traumatic and inflammatory intrauterine alterations 1 . Basically, re-epithelialization of ESCs by means of MET will periodically regenerate the endometrium after regular menstruation. 12 However, failure of such MET process will potentially cause endometrial defect, followed with initiation of ESCs being transformed into fibroblasts and myofibroblasts, excessive deposition and reorganization of ECM, finally leading to endometrial fibrosis or IUAs.10,11 A recent systematic review reported that among women with various types of miscarriage, the incidence of IUAs ranges from 6% to 30%. 48 Currently, fertility-sparing hysteroscopic adhesiolysis is the mainstream recommendation for treatment of IUAs, with ancillary various physical barriers and hormone therapy.49,50 Emerging new options include hyaluronic acid gel, 51 improved hormone therapy, 52 and platelet-rich plasma perfusion. 53 However, faced with adhesion recurrence rate being as high as 62.5%, 54 and pregnancy and live birth rates being 66.1% and 64.0%, 55 these unsatisfied outcomes were main obstacles ahead of current treatment opinions. More recently, cell therapy demonstrates tremendous prospects for clinical application in treatment of IUAs. SC or SC-derived Exo therapy, received extensive investigations both in lab and pre-clinic stage.56,57 Such as umbilical cord-derived mesenchymal stromal cells, which have been proved to be a safe and effective therapeutic approach for recurrent IUAs after adhesiolysis surgery. 58 In this study, for the first time, we reported that TC-derived Exo demonstrated exciting results to reverse fibrosis and enhance regeneration and repair both in cellular and IUA models.
TCs are newly found interstitial cells that have extremely long cellular prolongations known as Tps with alternating thin (podomers) and thick segments (podoms). Tps was the key feature that distinguished TCs from any other cell and considered to be the ultrastructural hallmark. 59 Although TCs have no definite immunophenotypic characteristics, several reports claim that they have the ability to bind different antibodies. However, according to the literature, it was considered that the double-positive immunostaining with CD34/PDGFRα and CD34/vimentin may be appropriate when referring to these cells.13,59–61 And in this study, the particular morphological characteristics and the double-positive immunostaining with CD34/vimentin were used to refer to TCs as earlier.7,20,21 TCs form a complex 3D network within interstitial tissues, with intercellular junctions between its long Tps and various adjacent cells, thus influencing their activities under different pathophysiological conditions. 13 Apart from its 3D structural junctions in interstitial apartment, TCs also release rich amount of paracrine signaling substances, such as extracellular vesicles to regulate nearby cells.15,62 TCs play key roles in maintaining structural integrity of SC niches within intestinal mucosa crypts, providing different concentrations of Wnt ligands and Wnt inhibitors along the length of intestinal crypts. These signals were essential for nursing, self-renewal, differentiation of resident SC niche, and maintaining SC-mediated intestinal mucosal regeneration and repair.18,19 TC-driven Exo reduced cardiac fibrosis, improved heart function, increased angiogenesis in rats with myocardial infarction, 17 and had been shown to benefit acute lung injury. 63 TCs have been found in female reproductive system as well, including vagina, uterus, uterine tube, ovary, and placenta.40,64 After TC loss and ensuing microenvironmental deregulation, premature ovarian failure may develop. 65 The lack of TC is related to the occurrence of leiomyomas, and Aleksandrovych et al. 66 provided immunohistochemical evidence of a diminished count of uterine TCs in leiomyomas. Placental TCs were located in the chorionic villi next to fetal blood vessels and myofibroblasts, and if TCs lost, the affected patients might be at a higher risk of developing preeclampsia. 67 TCs may be considered as the possible pathogenesis of endometriosis. 68 Loss or damage of TCs is closely related to fibrotic diseases in many other systems or organs, such as systemic sclerosis, ulcerative colitis, Crohn’s disease, and liver fibrosis.16,69–73 Meanwhile, transplantation of TCs contributed to alleviation of fibrosis through reduction of ECM in disease-affected animal models.74,75
In this study, TGF-β1, a classic pro-fibrogenesis factor, at a concentration of 15 ng/ml was administrated to ESCs for 48 h to successfully develop fibrotic or IUA cell model, as identified by elevation of fibrotic markers (COL1A1, FN, and α-SMA). All of which were involved in cellular or tissue fibrogenesis.76,77
Then, the fibrotic ESCs received TC-derived Exo treatment to observe reversal of fibrosis after such effectiveness was confirmed in TCM. The results showed that VEGF was significantly elevated with inhibited fibrotic markers, indicating salvage or reversal of fibrosis phenotype by TCs (either TCM or TC-derived Exo). Interestingly, the total secretion of VEGF in control ESCs was higher than the blank (culture medium for 48 h), but in the cells, the VEGF in control ESCs was lower than the blank (at 48 h). The reason for this difference may be that VEGF production was enhanced when initially exposed to TGF-β1. Similar results were reported before.78–80 But with the progression of fibrosis, VEGF secretion decreased in TGF-β1 treated ESCs. Many factors can affect the expression of VEGF. In contrast to the cell model, there was no statistical difference in the level of VEGF expression in the animal model. This may be due to the more connections and interactions between different cell types in the uterus. Previously, our group reported TCM can induce elevation of E-cadherin and decreased N-cadherin in ESCs via Wnt/β-catenin signaling pathway, subsequently enhancing decidualization and MET process in cultured ESCs. 7 As we know, MET serves as one of the mechanisms of endometrial re-epithelialization after natural or artificial decidualization, which was the opposite direction against fibrosis process. 12 Currently, E-cadherin was elevated in IUA tissues after TC-derived Exo treatment. Therefore, combined with previous results, 7 we speculate that the ability of TCs to reverse fibrosis may be achieved through the MET process. Similar result was observed in rat model of renal fibrosis, in which TC transplantation inhibited EMT (the opposite process of MET) to attenuate renal fibrosis. 75
Further pathway study showed that, after being administrated with ETC-159 and XAV939, which function to conditionally deplete Wnt ligands product within TCs and block Wnt/β-catenin pathway within ESCs, TC-derived Exo lost its salvage effect on fibrosis. Interestingly, by means of bioinformatics analysis in a more recent study, Wnt signaling pathway was suggested to be involved in TCs mediated treatment of IUAs. 81 Therefore, combined with previous pathway mechanisms,7,18,19,81 this study further confirms the participation of Wnt ligands and Wnt/β-catenin pathway in the observed reversal effect on fibrotic ESCs. As we know, Wnt signaling is crucial for maintenance of stemness properties of SC, tissue homeostasis, and tissue development, especially for self-renewal of colon SC and endometrial mesenchymal stem-like cells.82–84 Besides, Wnt signaling pathway regulates tissue fibrosis, renewal, and regeneration in various tissues, including endometrium, lungs, kidney, and heart.85–87 Our work is in agreement with previous studies noting intestinal subepithelial Foxl1 and GLI1 positive TCs being an important non-epithelial source of Wnt ligands that supports stemness within intestinal crypts,18,19 uterine TCs were also proved to be an important source of Wnts that supports endometrium regeneration and fibrosis-resistant.
Conclusion
In conclusion, our results confirmed that TC-derived Exo provided an important source of Wnts that inhibit fibrosis in fibrotic ESCs model and promote regeneration repair by relieving fibrosis and enhancing angiogenesis in the IUA mouse model. This study provided new evidence for therapeutic potential of TCs in fibrosis of endometrium, trauma, tissue engineering, or congenital-related uterine diseases.
Footnotes
Acknowledgements
The authors thank the experimental platform provided by the Institute of Blood and Marrow Transplantation of Soochow University.
Author Contributions
Conceptualization, T-QC and X-JY; methodology, T-QC, X-JW, and S-HZ; software, T-QC and X-JW; validation, T-QC and X-JY; formal analysis, T-QC, X-JY, and S-HZ; investigation, T-QC, H-YL, and X-JY; resources, X-JY; data curation, T-QC; writing—original draft preparation, T-QC and X-JY; writing—review and editing, H-YL, S-HZ, and X-JY; visualization, T-QC; supervision, H-YL, S-HZ, and X-JY; project administration, X-JY; funding acquisition, X-JY. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
The research procedures involving animal were approved by the Ethics Committee of Soochow University (ECSU2019000163).
Statement of Human and Animal Rights
All of the experimental procedures used in this study were conducted in accordance with the Institutional Animal Care Guidelines of Soochow University. The animal welfare system was strictly followed during the research, and all efforts were made to reduce the suffering of the animals.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number: 81971335).
