Abstract
Bone marrow stromal cells (BMSCs) have emerged as a potential therapy for sepsis, yet the underlying mechanisms remain unclear. In this study, we investigated the effects of BMSCs on serum inflammatory cytokines in a rat model of lipopolysaccharide (LPS)-induced sepsis. Sepsis was induced by intravenous injection of LPS, followed by transplantation of BMSCs. We monitored survival rates for 72 h and evaluated organ functions, histopathological changes, and cytokines expression. Sepsis rats showed decreased levels of white blood cells, platelets, lymphocyte ratio, and oxygen partial pressure, along with increased levels of neutrophil ratio, carbon dioxide partial pressure, lactic acid, alanine aminotransferase, and aspartate aminotransferase. Histologically, lung, intestine, and liver tissues exhibited congestion, edema, and infiltration of inflammatory cells. However, after BMSCs treatment, there was improvement in organ functions, histopathological injuries, and survival rates. Protein microarray analysis revealed significant changes in the expression of 12 out of 34 inflammatory cytokines. These findings were confirmed by enzyme-linked immunosorbent assay. Pro-inflammatory factors, such as interleukin-1β (IL-1β), IL-1α, tumor necrosis factor-α (TNF-α), tissue inhibitor of metal protease 1 (TIMP-1), matrix metalloproteinase 8 (MMP-8), Leptin, and L-selectin were upregulated in sepsis, whereas anti-inflammatory and growth factors, including IL-4, β-nerve growth factor (β-NGF), ciliary neurotrophic factor (CNTF), interferon γ (IFN-γ), and Activin A were downregulated. BMSCs transplantation led to a decrease in pro-inflammatory cytokines and an increase in anti-inflammatory and growth factors. We summarized relevant molecular signaling pathways that resulted from cytokines in BMSCs for treating sepsis. Our results illustrated that BMSCs could promote tissue repair and improve organ functions and survival rates in sepsis through modulating cytokine networks.
Introduction
Sepsis is a life-threatening response to infection, affecting millions worldwide with a mortality rate of approximately 26%1,2. Excessive inflammatory responses, involving various cytokines and mediators, play a key role in sepsis development and pathogenesis. In severe infections, local inflammatory cells, including mononuclear macrophages, neutrophils, and vascular endothelial cells are activated and release a multitude of factors, such as tumor necrosis factor-α (TNF-α), interleukins-6 (IL-6), interleukins-8 (IL-8), high mobility group protein B1, and interleukin-1β (IL-1β) 3 . This activation of the inflammatory cells and mediators further responds to produce the inflammatory amplification reaction to exacerbate organ damage. Elevated levels of serum inflammatory cytokines have gained attention as important indicators of sepsis.
Bone marrow stromal cells (BMSCs) are promising stem cell candidates for transplantation therapy 4 . They possess self-renewal and proliferative abilities, as well as the potential to differentiate into various cell types. BMSCs exhibit unique features, including low ethical concerns and minimal tissue rejection. Their therapeutic potential has been explored in immune-associated diseases and tissue restoration, with positive outcomes observed 5 . BMSCs transplantation promotes neovascularization and potentially reduces inflammation in sepsis through modulation of the host’s immune responses6,7. However, the underlying molecular mechanisms of their actions after transplantation remain unclear. Further research is needed to fully understand BMSCs’ therapeutic effects and signaling pathways involved in sepsis treatment8,9.
In this study, we investigated whether BMSCs could promote the improvement in histopathological injuries, organ functions and survival rates, and modulating serum inflammatory cytokines in lipopolysaccharide (LPS)-induced sepsis in rats.
Methods
Animals and Groupings
Female Sprague Dawley (SD) rats (4–5 weeks old, weighing 180–220 g) were obtained from Animal Experimental Center of Kunming Medical University. The rats were kept in controlled conditions with a 12-h light/12-h dark cycle, with temperature of 20°C to 25°C, and humidity between 50% and 70%. They had free access to food and water. The experiments followed the guidelines for the Care and Use of Laboratory Animals by the National Institute of Health, China, and were approved by the Animal Ethical Committee of Kunming Medical University (Approval No. kmmu2020503).
Rats were divided randomly into three groups, each consisting of 40 rats: Sepsis group received intravenous injection of LPS (Sigma-L2880, O55: B5) at a dose of 3 mg/kg, Sham group received intravenous administration of sterile phosphate buffer saline, and BMSCs group received LPS injection followed by infusion of BMSCs through the caudal vein at a dose of 1 × 107 cells/kg, 2 h after sepsis induction.
Establishment of Rat Sepsis Model
In this study, the rat sepsis model was induced by injecting 3 mg/kg LPS through the femoral vein. To confirm the success of the sepsis model, a combination of diagnostic criteria for systemic inflammatory reaction syndrome in animals and sepsis model diagnostic standards were used10–12. The diagnostic criteria included (1) a total white blood cell (WBC) count twice as high or reduced by 50% compared with reference values, (2) hepatic insufficiency indicated by alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels double the reference values, and (3) renal insufficiency defined by serum creatinine levels ranging from 133 to 442 µmol/L. The sepsis rat model was confirmed as successful, based on the above indicators.
Cultivation and Transplantation of BMSCs
Bone marrow cells were extracted from the femur metaphyseal ends, using serum-free Dulbecco’s modified Eagle’s medium (DMEM)/nutrient mixture F12 (DMEM/F12, 1:1, Gibco-C11330500BT), and then centrifuged at 1,000 rpm for 5 min. The cells were resuspended and cultured in DMEM/F12 medium supplemented with 10% fetal bovine serum (BioInd, 04-001-1ACS) and 1% penicillin/streptomycin (Hyclone-SV30010) at 37°C in a 5% CO2 incubator with saturated humidity. Non-adherent cells were removed after 2 days and fresh culture medium was added. The medium was changed every 3 days. When the cells reached 70% to 80% confluence, adherent cells were harvested, expanded in T-75 Cell Culture Flasks (Sigma, SIAL0641-100EA, Austin, TX, USA), and used for subsequent experiments after undergoing 3 to 4 generations of subculturing. The morphological changes of the cultured bone marrow cells were observed and the cells exhibited typical mesenchymal stem cell morphology and differentiation potential. These cells were identified by immunohistochemistry, using CD44 antibody. The fourth-passage BMSCs, which were confirmed by flow cytometry (BD FACSCalibur, San Jose, CA, USA) analysis, were then transplanted into the sepsis rat model, 2 h after LPS induction at a dose of 1 × 107 cells/kg through intravenous injection.
Biochemical Assays
Twenty-four hours after the establishment of the sepsis model and transplantation of BMSCs, various assessments were performed, including blood routine analysis, blood gas analysis, serum biochemistry tests, and calculation of mortality rate. Serum levels of WBCs, platelets (PLTs), lymphocytes percentage (L%), neutrophils percentage (N%), AST, ALT, partial pressure of oxygen (PO2), partial pressure of carbon dioxide (PCO2), and lactate were measured, using a standard clinical automated analyzer. These measurements provided insights into hepatic dysfunction, lung dysfunction, and tissue hypoperfusion.
Histological Evaluation
Samples of lung, intestine, and liver were harvested and fixed in 10% formalin solution to assess morphological changes after 24 h following injection of LPS and/or BMSCs. Then, all the samples were embedded in paraffin and sliced into a serial of 5 mm thicknesses stained with hematoxylin and eosin to assess the morphological changes and histopathological injury scores.
Cytokines Antibody Arrays
The SD rats were intraperitoneally anesthetized with phenobarbital sodium at a dose of 40 mg/kg. The blood samples were then collected through the abdominal aorta. To assess the expression of 34 cytokines at different time points (6 h, 1 day, 3 days, and 7 days) after BMSCs transplantation, Rat Cytokine Antibody Arrays (RayBio Rat Cytokine Antibody Array G-Series 2) were employed. The signals on the BMSCs membrane were detected, using a chemiluminescence imaging system. Densitometry was used to quantify signal intensity, and RayBio AAR-CYT-G2 Software was utilized for data analysis. Differential expression factors were determined based on a fold change ratio exceeding 1.5 or below 0.66. To normalize signal values across different cell membranes, a positive control was established.
Enzyme-Linked Immunosorbent Assay (ELISA) for Serum Cytokines
ELISA was employed to validate the significant changes observed in cytokines expression during the study. Following the manufacturer’s instructions of the ELISA kit (RapidBio, Plymouth, Michigan), a standard curve was generated, using different dilutions of the four cytokines as the standard samples. The levels of these cytokines were determined by calculating their concentrations based on the standard curves. All samples and standards were measured in duplicate to ensure accuracy and reproducibility of the results.
Statistical Analysis
All data were presented as the mean ± SD and analyzed by the Log-Rank test, one-way analysis of variance, and Student–Newman–Keuls test to determine the differences between the Sham, Sepsis, and BMSCs groups. SPSS 22.0 software program was used to analyze the experimental data. A level of P < 0.05 was considered significant difference.
Results
BMSCs Transplantation Enhanced Survival Rates and Organs Functional Recovery
The survival rates in the 72 h following rat operation were 100% in the Sham group, 58.3% in the Sepsis group, and 75.0% in the BMSCs group (Fig. 1A), indicating a significant difference between each two groups (P = 0.0441, P < 0.05). Analysis of blood routine, blood gas, and biochemical parameters 24 h after operation revealed reduced WBC, PLT count, lymphocyte percentage(L%), and elevated neutrophil percentage (N%) in the Sepsis group compared with the Sham group. ALT and AST levels were also significantly higher in the Sepsis group. Moreover, the Sepsis group exhibited decreased arterial partial PO2 and PCO2 levels with increased lactate levels. Following BMSCs transplantation, there was distinct improvement in organ functions, including the liver, lung, blood, and circulation system (Fig. 1B–D).

The survival rates and organs function in the Sham, Sepsis, and BMSCs groups. (A) The survival rates. (B) Blood routine test, the levels of WBC, PLT, L%, and N%. (C) AST and ALT concentrations. (D) Blood gas analysis: PO2, PCO2, and Lac. BMSCs: bone marrow stromal cells; WBC: white blood cell; PLT: platelets; AST: aspartate aminotransferase; ALT: alanine aminotransferase; PO2: pressure of oxygen; PCO2: pressure of carbon dioxide.
BMSCs Transplantation Promoted Injured Organs Morphological Repair
There was noticeable relief from pathological changes, such as hyperemia, edema, and inflammatory cell infiltration in organs such as the lung, small intestine, and liver of sepsis rats after BMSCs administration, compared with the Sepsis group at 24 h after operation (Fig. 2A–I). Histopathological injury scores of the lung, intestine, and liver were substantially higher in the Sepsis group compared with the Sham group. However, following BMSCs transplantation, these organ injury scores were significantly reduced (Fig. 3A–C).
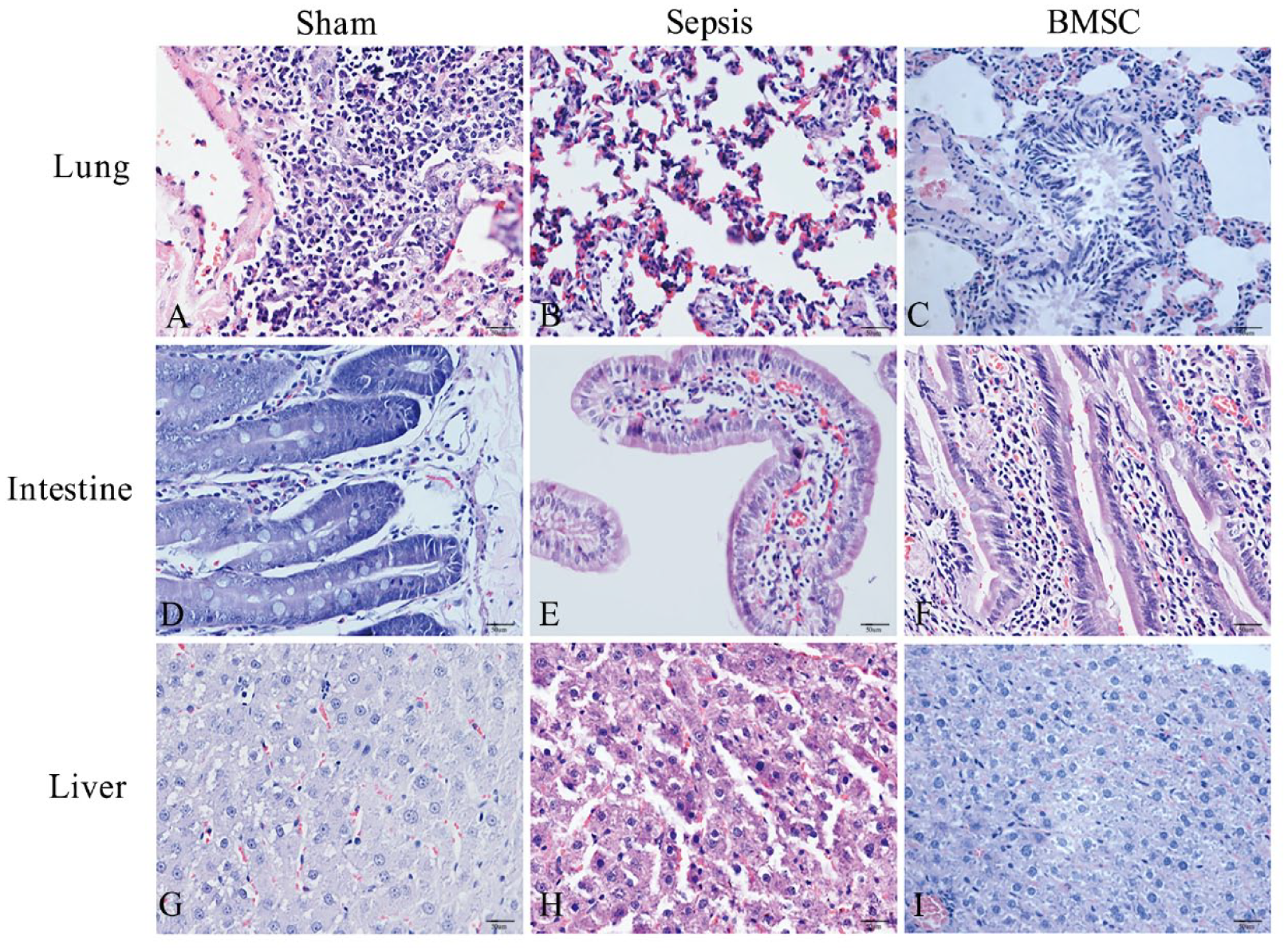
Histopathological evaluation of the lung, intestine, and liver (original magnification ×400). (A to C) the lung, (D to F) the intestine, (G to I) the liver. BMSC: bone marrow stromal cell.
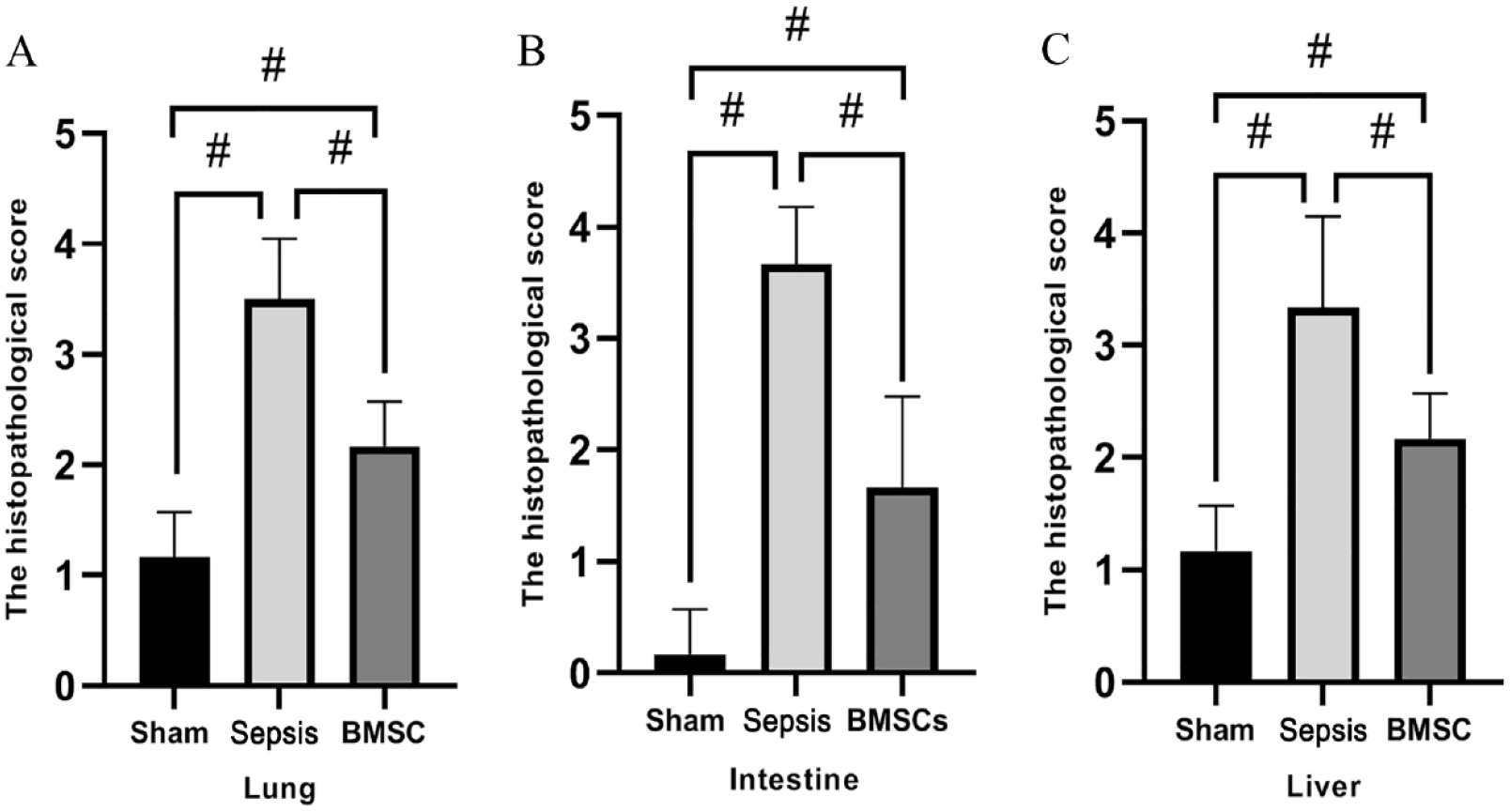
Histopathological injury scores of the lung, intestine, and livers in sham, sepsis, and BMSCs groups. (A) Lung injury scores, (B) intestine injury scores, and (C) liver injury scores. BMSCs: bone marrow stromal cells.
BMSCs Modulated the Expressions of Systemic Inflammatory Cytokines by Protein Microarray Analysis
Out of the 34 cytokines assessed on the protein chip, 12 showed significant changes based on the criteria of Fold Change >1.5 or <0.66 (Fig. 4). In the Sepsis group, pro-inflammatory factors, such as IL-1β, interleukin-1α (IL-1α), and TNF-α, displayed an upward trend, whereas they were all substantially downregulated in the BMSCs group at the same time point. Both the Sepsis and BMSCs groups exhibited similar patterns of change for interleukin −4 (IL-4), β-nerve growth factor (β-NGF), ciliary neurotrophic factor (CNTF), interferon γ (IFN-γ), and Activin A, but with more pronounced elevation seen in the Sepsis group. Other factors, including tissue inhibitor of metal protease 1 (TIMP-1), matrix metalloproteinase 8 (MMP-8), Leptin, and L-selectin were significantly increased in the Sepsis group but showed a decreasing trend in the BMSCs group.

The expressions of inflammatory cytokines by protein microarray analysis in the Sham, Sepsis, and BMSCs groups at the same time points. β-NGF: β-nerve growth factor; IL: interleukin; BMSC: bone marrow stromal cell; IFN-γ: interferon γ; TNF-α: tumor necrosis factor-α; TIMP-1: tissue inhibitor of metal protease 1; MMP-8: matrix metalloproteinase 8; CNTF: ciliary neurotrophic factor.
Notably, in the Sepsis group, there was a significant peak in Activin A and IFN-γ expression at 6 h post-operation (hpo) compared with the Sham group. This was followed by a gradual decline, with levels returning to baseline by 3 days post-operation (dpo). The expression of IL-1β and TNF-α displayed double peaks on 1 dpo and 7 dpo under sepsis conditions.
BMSCs Regulated Inflammatory Response by ELISA Validation for Serum Cytokines
During the changes in protein chip analysis, four inflammatory factors with significant changes, including IL-1, TNF-α, β-NGF, and Activin A were measured using ELISA for validation. As illustrated in Fig. 5A–D, the concentrations of IL-1 and TNF-α were significantly elevated in the Sepsis group, but were reduced in the BMSCs group. Conversely, the levels of β-NGF and Activin A were markedly upregulated in the rats receiving BMSCs transplantation when compared with the Sepsis group.

ELISA validation for serum cytokines in the Sham, Sepsis, and BMSCs groups. (A-D) Activin A, IL-1, TNF-α, and β-NGF. ELISA: enzyme-linked immunosorbent assay; BMSC: bone marrow stromal cell; IL: interleukin; TNF-α: tumor necrosis factor-α; β-NGF: β-nerve growth factor.
Molecular Signaling Pathways Involved in Sepsis Following BMSCs Transplantation
We highlight a few examples of specific signaling pathways that are induced by cytokines, including the nuclear factor kappa B (NF-κB), c-Jun N-terminal kinase/p38 mitogen-activated protein kinase (JNK/p38MAPK), janus kinase/signal transducer and activator of transcription (JAK/STAT), and phosphatidylinositol 3-kinase/Protein Kinase B (PI3K/Akt) pathways (Fig. 6). These pathways are implicated in a wide range of biological effects, encompassing inflammation and immune responses, cell proliferation and differentiation, cellular apoptosis, tissue remodeling and regenerative repair, as well as energy metabolism. They hold promising potential as targets for BMSCs transplantation treatment in sepsis.

Cytokine-mediated signaling pathways involved in sepsis following BMSCs transplantation. Cytokines bind to their respective receptors and activate associated signaling pathways, including the NF-κB, JNK/p38 MAPK, JAK/STAT, and PI3K/Akt pathways, which are involved in various biological effects, including inflammation and immune responses, cell proliferation and differentiation, cellular apoptosis, tissue remodeling and regenerative repair, as well as energy metabolism. BMSCs: bone marrow stromal cells; NF-κB: nuclear factor kappa B; CNTFR: ciliary neurotrophic factor receptor; INFGR: interferon-gamma receptor; JNK/p38MAPK: c-Jun N-terminal kinase/p38 mitogen-activated protein kinase; JAK/STAT: janus kinase/signal transducer and activator of transcription; LEPR: leptin receptor; PI3K/Akt: phosphatidylinositol 3-kinase/Protein Kinase B; IL: interleukin; TNF-α: tumor necrosis factor-α; TIMP-1: tissue inhibitor of metal protease 1; TNFR: tumor necrosis factor receptor; MMP-8: matrix metalloproteinase 8; β-NGF: β-nerve growth factor; CNTF: ciliary neurotrophic factor; IFN-γ: interferon γ.
Discussion
Accumulating evidences reveals that the severity of sepsis may depend more on the specificity and intensity of the host response rather than on the offending pathogen itself. Uncontrolled inflammatory reactions can cause detrimental effects on endothelial and epithelial cells, leading to apoptosis and cellular activation. These responses are accompanied by an upregulation of anti-inflammatory mediators, leukocyte adhesion, transmigration, and activation of the coagulation and complement systems13,14. Typically, sepsis is characterized by a pronounced pro-inflammatory reaction in the early stage. However, in cases of severe sepsis, septic shock, and multiple organ dysfunction syndrome (MODS), an anti-inflammatory response progressively dominates. This immune paralysis leaves the body prone to adverse outcomes and even higher mortality rates 15 . Extensive evidence supports the notion that the lung is the primary organ impacted by sepsis, followed by the liver, intestine, kidney, hematological system, and cardiovascular system 16 . So we selected to investigate the structural and functional alterations of the lung, liver, and intestine associated with sepsis-induced MODS. The results illustrate that BMSCs promote tissue repair and improve organ function in sepsis by modulating cytokines expression.
Our study revealed an imbalance between pro-inflammatory and anti-inflammatory cytokines in a rat model of LPS-induced sepsis. Early pro-inflammatory cytokines IL-1 and TNF-α significantly were increased within 24 h of sepsis induction. In response, the anti-inflammatory mediator IL-4, β-NGF, showed compensatory increases, playing an important role in mitigating inflammation. BMSCs transplantation has been demonstrated to effectively suppress systemic inflammation in sepsis animal models 17 . Consistent with these findings, our results showed that BMSCs transplantation significantly elevated the expression levels of anti-inflammatory cytokines (IL-4, β-NGF, CNTF, IFN-γ, and Activin A), while reducing the levels of pro-inflammatory factors (TNF-α, IL-1β, and IL-1α) and other molecules (TIMP-1, MMP-8, Leptin, and L-selectin). These outcomes collectively indicate that the anti-inflammatory factors and products induced by BMSCs transplantation, along with their immune response-modulating effects, contribute to the creation of an immune-regulation microenvironment for inflammation modulation.
BMSCs transplantation is known for its beneficial effects, primarily attributed to the release of various immuno-regulatory cytokines that aid in tissue repair. Our research found that, compared with the Sham group, the Sepsis group exhibited a significant peak in Activin A and IFN-γ expression at 6 h post-operation (hpo) during a 7-day period. These levels gradually declined and returned to baseline levels by 3 days post-operation (dpo). These findings shed light on the temporal changes in Activin A and IFN-γ expression within 7 days of sepsis and the bidirectional modulation achieved through BMSCs transplantation. Activin A, a member of the TGF-β superfamily, was elevated in sepsis patients and associated with disease severity and prognosis. It plays a crucial role in regulating immune system response, impacting immune cell function and proliferation, and contributing to organ damage and dysfunction18–20. In our study, we initially expounded the bidirectional regulatory effect of BMSCs transplantation on Activin A expression, promoting inflammation balance and restoring organ functions. Activin A shows promising potential as a novel therapeutic target for controlling inflammatory diseases. IFN-γ, a critical mediator of cellular immunity, orchestrates multiple protective functions to enhance immune responses during infections. It not only initiates immune response activation and pathogen elimination but also safeguards against excessive immune system activation and tissue damage21–23. Our study illustrated that BMSCs could suppress excessive IFN-γ expression, alleviating inflammation and tissue damage, while also promoting moderate IFN-γ expression to aid in pathogen clearance, immune regulation, and tissue homeostasis.
Importantly, IL-1β exhibited double peaks in expression on 1 dpo and 7 dpo under sepsis conditions. This novel finding suggests that the inflammatory response is a dynamic process that evolves over time, indicating a continuous struggle for the body to maintain inflammatory balance. The initial increase (on 1 dpo) in IL-1β during early sepsis is associated with immune cell activation and initiation of the inflammatory response. Studies have shown that, upon infection or injury, IL-1β is rapidly released to initiate the inflammatory response and recruit other immune cells 24 . The second peak of IL-1β in the later stages (on 7 dpo) of sepsis may be related to immune modulation and cellular damage. As sepsis progress, the negative feedback regulation of the inflammatory response gradually fails, leading to excessive release of cytokines and tissue damage 25 . Our research demonstrated that BMSCs effectively suppress the dual peak expression of IL-1β. First, during the early stages, BMSCs inhibit the first peak of IL-1β by suppressing immune cells activation and inflammatory mediators release. Second, in the later stages, BMSCs promote immune regulation and reduce the second peak of IL-1β to limit cellular damage. These findings highlight the ability of BMSCs to regulate the immune system, prevent sustained amplification of inflammatory response, and protect tissues from further damage.
Inflammatory cytokines, including IL-1, TNF-α, and MMP-8, activate the NF-κB signaling pathway to exert their effects on inflammation and immune modulation26–31. The MAPK signaling pathway, consisting of JNK, p38 MAPK, and ERK branches, can be activated by various factors such as IL-1, TNF-α, TIMP-1, MMP-8, L-selectin, IL-4, β-NGF, and Activin A19,32–39. Activation of these pathways contributes to cellular stress responses, proliferation, cell migration, and tissue repair processes. IL-1, TNF-α, Leptin, IL-4, CNTF, and IFN-γ induce cellular apoptosis through the JAK/STAT signaling pathway22,23,40–44. The PI3K/Akt signaling pathway, implicated in the effects of TIMP-1, Leptin, β-NGF and IFN-γ, regulates cell survival and proliferation36,41,45–48. These signaling pathways hold promise as potential targets for BMSCs therapy in sepsis and provide valuable insights for further mechanistic research.
However, several limitations exist in this study. First, the timing of BMSCs transplantation at 2 h after LPS administration may not accurately represent the clinical practice, where infections may not have reached the peak of inflammation during that period. Second, the potential dose-dependent and long-term effects of BMSCs in treating sepsis were not investigated in this study. In addition, it is important to note that rodent animals differ significantly from clinical patients with comorbidities, necessitating cautious interpretation of these results. Further research using relevant clinical models is necessary to validate our findings.
In summary, sepsis is a life-threatening organ dysfunction caused by a dysregulated host response to infection. It is a complex pathological process, involving various cytokines that form a network structure. BMSCs transplantation could regulate the expressions of cytokines involved in sepsis, thereby modulating the cascade of uncontrolled inflammation and promoting tissue repair and improvement of organ functions.
Footnotes
Ethical Approval
All procedures involving animals in this study were approved by the Ethics Committee of Experimental Animals, Kunming Medical University (Approval No. kmmu2020503), Kunming, China.
Statement of Human and Animal Rights
All the experimental procedures were carried out in accordance with the Ethics Committee of Experimental Animals, Kunming Medical University, Kunming, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Association Foundation Program of Yunnan Provincial science and Technology Department and Kunming Medical University (202001AY070001-166,202001AY070001-123), National Natural Science Foundation of China (81901950, 82260380), Yunnan Applied Basic Research Project Foundation (2019FB099), Young and Middle-aged Academic and Technical Talents of Yunnan Province, Yunnan Health Training Project of High-Level Talents (H-2017060), Yunnan University Donglu Talent Young Scholar, Yunnan Universities Scientific and Technical Innovation Team, and Yunnan Province Clinical Research Center for Gynecological and Obstetric Disease (202002AA100199-2022YJZX-FC19).
