Abstract
Cell expansion of human pluripotent stem cells (hPSCs) commonly depends on Matrigel as a coating matrix on two-dimensional (2D) culture plates and 3D microcarriers. However, the xenogenic Matrigel requires sophisticated quality-assurance processes to meet clinical requirements. In this study, we develop an innovative coating-free medium for expanding hPSCs. The xenofree medium supports the weekend-free culture and competitive growth of hPSCs on several cell culture plastics without an additional pre-coating process. The pluripotent stemness of the expanded cells is stably sustained for more than 10 passages, featured with high pluripotent marker expressions, normal karyotyping, and differentiating capacity for three germ layers. The expression levels of some integrins are reduced, compared with those of the hPSCs on Matrigel. This medium also successfully supports the clonal expansion and induced pluripotent stem cell establishment from mitochondrial-defective MELAS (mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes) patient’s peripheral blood mononuclear cells. This innovative hPSC medium provides a straightforward scale-up process for producing clinical-orientated hPSCs by excluding the conventional coating procedure.
Introduction
Human pluripotent stem cells (hPSCs), such as embryonic stem cells (ESCs), germline stem cells, and induced pluripotent stem cells (iPSCs), have the potential to produce unlimited cell sources for cell therapy and regenerative medicine1–4. Traditional amplification of the hPSCs requires mouse feeder cells or pre-coating the culture devices with extracellular matrix (ECM), such as Matrigel and Geltrex, for promoting cell adhesion and cell proliferation5,6. Nevertheless, mouse feeder cells and Matrigel are xenogenic and potentially risky for clinical use. The current protocols for producing clinical-grade hPSCs apply defined ECM molecules, such as vitronectin, laminin (laminin 511), recombinant laminin, or laminin fragments (laminin-511 E8) to facilitate cell attachment on cell culture-grade plastics7,8. Adding laminin fragments, vitronectin, or inter-alpha inhibitor (IαI) in the culture medium can promote the hPSC adhesion onto cell culture vessels9,10. However, the production of these recombinant proteins requires sophisticated mammalian cell–based manufacturing. The high cost limits their applications for mass PSC production.
In addition to the ECM coating, surface-modified properties of culture plates were also intensively investigated11,12. Several synthetic polymers have been developed to coat culture substrates for enhancing hPSC attachment13–18. Researchers also demonstrated that providing artificial nanostructured materials, such as nanopillar-fabricated or plasma-irradiated substrates, facilitates the hPSC adhesion19,20. Although these modifications may be practical on two-dimensional (2D) cell culture for research use, these approaches nevertheless face several obstacles for large-scale hPSC cultures in clinical studies, such as long-term safety validation, good manufacturing practice (GMP) production, costs of synthetic polymers and instruments, and compatibility with existing cell manufacturing procedures.
To simplify the establishment of GMP-compatible hPSCs, we develop a coating-free medium for the competitive growth of hPSCs. The maintenance of the pluripotency is validated by the cell and colony morphology, the expressed pluripotent markers, and the competency for three germ layers differentiation. Whether this medium supports the primary iPSC establishment from peripheral blood mononuclear cells (PBMCs) of disease-bearing patients is also investigated. We mainly address whether the newly developed DuoESy medium stably supports both human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs) expansion and stemness without pre-coating treatment on general cell culturewares in the absence of vitronectin, synthetic laminin fragments, and IαI protein.
Materials and Methods
hPSC Culture with ECM Coating
The hESC and iPSC lines, including the H9 hESCs (XX; WiCell, Madison, WI, USA) and 253G1 hiPSCs (XX; HPS0002; RIKEN BRC, Tsukuba, Ibaraki, Japan), were cultured in Essential 8 medium (E8 medium, A1517001, Gibco, Thermo-Fisher, Waltham, MA, USA), mTeSR1 medium (85850, Stemcell Technology, Vancouver, Canada), on 1.0% hESC-qualified Matrigel (CLS354277, Corning, Glendale, AZ, USA)-coated cell culture dishes (Falcon/Corning, USA), or six-well cell plates (Z707759, TPP, Trasadingen, Swissland; 353046, Falcon; CLS3516, Corning, USA). After 5 days of culture, we dissociated the cells with Accutase (AT104, Innovative Cell Technologies, San Diago, CA, USA) and passaged the cells at a 1:5 or 1:10 ratio. The E8 and mTeSR1 media were refreshed every day.
hPSC Culture Without a Pre-coating Step
We cultured hPSCs in the xenofree DuoESy medium without a pre-coating step (ERL001, DuoGenic StemCells Corporation, Taichung, Taiwan). DuoESy medium contains both thermal-stable basic human fibroblast growth factor (bFGF, or named FGF-2)21,22 and human insulin-like growth factor-1 (IGF-1) to promote cell proliferation. DuoESy medium does not contain the recombinant vitronectin, laminin fragments, or purified IαI proteins.
When the confluence of the cells reached up to 80% in a six-well cell culture plate, we dissociated the hPSCs (5-10 × 105 cells) with 1 ml Accutase or Versene solution (15040066, Gibco, USA) per well for 3 min at room temperature. The cells were gently pipetted and mixed with 1 ml DuoESy α medium before the transfer into a 15 ml centrifuge tube. The suspended cells were harvested and concentrated after the centrifugation at 1,000 rpm (800 × g, Kubota 2420, Kubota, Osaka, Japan) for 2 min. The concentrated cells were resuspended with 1 ml DuoESy α medium by gently pipetting. We adjusted the seeding cell density to be 0.5-1.0 × 105 cells/ml in a new well of the culture plate. After 1 day of culture, the medium was replaced with 2 ml DuoESy mixture (α/β = 1/5, V/V) to support the exponential cell growth without medium refreshment during the next 2 to 3 days incubation period. Evaluation of the cell growth rate and adjusting the optimal seeding density are prerequisites for weekend-free culture.
Alkaline Phosphatase Activity
The alkaline phosphatase (AP) activities of the cultured cells were determined by an AP staining kit at 24 h culture (8288; Sciencell Research Laboratories; Carlsbad, CA, USA). The AP enzyme activities were examined with p-nitrophenyl phosphate substrate reaction, following the manufacturer’s protocol (37621, Thermo-Fisher, USA). We determined the optical density (OD) values of the hydrolyzed p-nitrophenol at 405 nm by a full-spectrum enzyme-linked immunosorbent assay (ELISA) reader (Multiskan GO, N10588, Thermo Scientific, USA).
Plasmid-Based Generation of hiPSCs
The establishment of mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) patient-derived iPSCs in this study followed the policy instructions on the ethics of human embryos and ESC research in Taiwan. The blood sample collection was approved by the ethics institutional review board (IRB) in Tungs’ Taichung Metroharbor Hospital (Taichung, Taiwan; IRB number: 109038). We also obtained the patient’s informed consent for blood collection and iPSC production. The patient’s chief complaints were muscle weakness and pain, recurrent headaches, and anorexia. The PBMCs of the donor were separated from 10 ml peripheral blood by Ficoll-Paque premium (17544203, Cytiva) and then cryopreserved in frozen vials with Stem-Cellbanker (11897, Zenoaq, Fukushima, Japan). Four plasmids with pCXLE vector backbone carry six modified reprogramming factors (Oct4/shp53, Sox-2/hKlf-4, and L-Myc/Lin28) and an episomal replicating regulator, Epstein-Barr virus nuclear antigen 1 (EBNA1; Addgene, Watertown, MA, USA). The thawed PBMCs were recovered with the maintenance medium (5% human platelet lysate in RPMI-1640) by 1-day culture. The 106 PBMNCs were electroporated with the four plasmids (total 3 µg) by Amaxa Nucleofector II (program V-024, Lonza; Cologne, Germany) in a 100 µl homemade buffer23,24. After the electroporation, 800 µl DuoESy α medium with 50 U/ml interleukin-2 (IL-2, 200-02, PeproTech/Thermo-Fisher, USA) and 5 µg/ml phytohaemagglutinin P (PHA-P, L1668, Sigma-Aldrich/Merck MilliporeSigma, Burlington, MA, USA) were added to the cuvette and the cells were transferred to seed on non-coated culture vessel for 1 day. We added 1.5 ml DuoESy β medium and 100 ng/ml bFGF (100-18b, PeproTech; DuoESy/bFGF) to a six-well plate on day 2. Half media in the well were refreshed with the DuoESy/bFGF on day 4 and day 6 culture. From culture day 8, the culture media were replaced with 2 ml DuoESy/bFGF every 2 days. We observed iPSC-like colonies on day 12. The individual iPSC colonies were isolated and subcultured on day 20.
Three Germ Layers Differentiation from hPSCs
Neural differentiation
We steered the dissociated hPSCs differentiation to neural lineages by embryoid body (EB) formation25–27. In brief, the 1-2 × 105/ml cells were cultured in Essential 6 medium (E6 medium, A1516401, Gibco) to form EB within the first 2 days. On days 3 and 4, we initiated the neural differentiation by culturing the cells with refreshed E6 medium (A1516401, Gibco) combined with 10 ng/ml bFGF (PeproTech), 10 µM SB431542 (S1067, Selleckchem, Houston, TX, USA), and 0.5 µM CHIR99021 (4423, Tocris Bioscience/Biotechne, Minneapolis, MN, USA). From day 5, the cells were cultured with neural basal medium (21103049, Gibco) and B-27 supplement (17504044, Gibco) to support the growth of neuroepithelial cells. The EBs were attached on a Matrigel-coated cover slide on day 7 and cultured for additional 9 days for neural maturation. The cell characterizations of the differentiating cells were validated by immunocytochemical (ICC) staining.
Cardiac differentiation
The hESCs with knock-in cardiac reporter MYH6:mCherry were dissociated with 0.5 mM ethylenediaminetetraacetic acid (15575038, Gibco) at 37°C for 4 min. The cells were differentiating into cardiac cell lineage by a method using Wnt-modulating molecules28,29. In brief, hESCs were cultured in hESC medium until they attained 80% to 90% confluency. The hESCs were transiently treated with 12 µM CHIR99021 (a Wnt agonist molecule, Tocris Bioscience) in RPMI-1640 (11875085, Gibco), supplemented with B27 minus insulin (A1895601, Gibco), 2 mM GlutaMAX (35050061, Gibco) and 100 U/ml Penicillin/Streptomycin (30002CL, Corning) for 1-day differentiation. The culture medium was refreshed on day 2 by the same medium on day 1 without CHIR99021. Consequently, differentiating cells were treated with 5 µM IWR-1 (a WNT antagonist molecule, Tocris Bioscience) on days 3 and 4 but the IWR-1 was removed on day 5. From day 7, insulin was provided in the differentiation medium using complete B27 supplements (17504044, Gibco) to replace the B27 supplement minus insulin. The hESC-derived cardiac cells were replated on day 15 on cover slides and analyzed on day 20 by ICC staining.
Endoderm differentiation
The hPSCs were first seeded on a Matrigel-coated cover slide and maintained with DuoESy β medium for 2 days. Endoderm differentiation was steered by treating the cells with 3 µM CHIR9902130–32 and 2 ng/ml transforming growth factor-β (TGF-β, 100-21, Peprotech) in E6 medium. Differentiating cells were fixed on day 4 and validated by ICC staining.
Flow Cytometry Analysis
The expression of pluripotent markers on the hPSC surface was analyzed by flow cytometry. On day 4, the hPSCs were fully dissociated with Accutase and adjusted to 5 × 106 cells/ml in 0.1 ml phosphate-buffered saline (PBS). The cells were double-stained with the antibodies against stage-specific embryonic antigen 4 (SSEA4; 1:100, fluorescein isothiocyanate [FITC]-conjugated, 330410, Biolegend) and Tra-1-60 (1:50, Alexa Fluor 647-conjugated, 560850, Beckton-Dickinson, BD, Franklin Lake, NJ, BD; 1:100, PE-conjugated, 60064PE.1, Stemcell Technologies) for 30 min at room temperature. Nonspecific bound antibodies were removed by twice PBS washes. The cells were kept on ice and filtered with 0.45 µm mesh before the flow cytometry analyses (Accuri C6 plus, BD).
Immunocytochemical Staining
Cells on cover slides of microscope glass were fixed with 4% paraformaldehyde (PFA, Sigma-Aldrich) and washed twice with PBS. The cells were permeabilized with 0.3% Triton-100 (93443-500 ml, Sigma-Aldrich) for 10 min on ice and then treated with BlockPro blocking buffer (BP01-1l, Visual Protein, Taipei, Taiwan). Primary antibodies were diluted and applied as per the suggestion of the manufacturing companies. The primary antibodies were against Oct-3/4 (SC-8628, Santa Cruz Biotechnology, Dallas, TX, USA), Sox2 (AB5603, Merck Millipore), Nanog (sc-374103, Santa Cruz Biotechnology), c-Myc (GTX103436, GeneTex, Irvine, CA, USA), nestin (841901, BioLegend, San Diago, CA, USA), β-III tubulin (TuJ1, 801201, BioLegend), Actinin (A7811, Sigma-Aldrich), MYH6 (MAB8979, R&D Systems/Biotechne), Troponin T type 2 (TNNT2; ab45932, Abcam, Cambridge, UK), TTN-C (TTN-9, Myomedix, Neckargenmuend, Germany), and Sox17 (ab84990, Abcam). The primary antibodies were incubated at 4°C overnight. The secondary antibodies against mouse or rabbit immunoglobins were detected by their conjugated Alexa-488 or Alexa-647 fluorophores. The secondary antibodies (Thermo-Fisher, USA) were diluted 1,000-fold and reacted with hPSC-derived cells for 30 min at room temperature. Fluorescent images were captured using an upright microscope with a charge-coupled device camera (80i, Nikon, Tokyo, Japan) and processed with the Nikon Elements software (Nikon).
Integrin Gene Expression
We collected 5 × 106 hPSCs (H9 and 253G1 cells) cells after 3 days of cultivation with DuoESy medium alone or E8 medium and Matrigel combination (E8/M). The total ribonucleic acid (RNA) of the cells was harvested by REzol C&T (KP200CT, Protech Systems, Taipei, Taiwan). We detected the gene expression of the integrin subunits in 1 µg RNA with real-time reverse transcription polymerase chain reaction (real-time RT-PCR). The cDNA was synthesized by using Moloney murine leukemia virus reverse transcription kit (PT-RT-KIT, Protech Biosystems) and 20 ng of the cDNA was mixed with the specific primers (0.4 µM for each primer, listed in Table 1). We determined the integrin expressions using qPCRBIO SyGreen Mix (PB20.15-01, PCR Biosystems, London, UK) and amplified them with a QPCR machine (QuantStudio 6 Pro, A43180, Applied Biosystems/Thermo-Fisher, USA).
The Primers of the Subunits of Human Integrins.

Cell Proliferation Assay With Integrin-Neutralizing Antibodies
The hPSCs (H9 and 253G1 cells) were plated 2 × 104 cells/well in 24-well plates (353047, Falcon) with DuoESy medium. We inhibited the integrin binding activity by treating 1 μg/ml integrin-specific blocking antibodies. The neutralizing antibodies were all obtained from Chemicon company (Merck MilliporeSigma, USA)33,34. The clones of the monoclonal antibodies were P1D1 for α5 (MAB1956Z), 6S6 for β1 (MAB2253Z), P1F6 for αvβ5 (MAB1961Z), and NKI-GoH3 for α6 (MAB1378). We determined the cell proliferation by colorimetric MTS assay (G2581, Promega, USA) at 24, 48, 72, and 96 h post-plating with an ELISA reader (Multiskan Go, Thermo Scientific). DuoESy medium and the blocking antibodies were refreshed on day 2 culture.
Statistical Analysis
Data were collected from at least three independent experimental results and shown as the mean value ± SD. Statistical analyses were conducted using the Student t test between two groups or one-way/two-way analysis of variance (ANOVA) with Tukey’s post hoc test. A P value less than 0.05 was considered to be statistically significant.
Results
hPSCs Growth in DuoESy Medium
Two hPSCs, H9 hESCs and 253G1 hiPSCs, were stably maintained on Matrigel-coating or non-coating dishes (Falcon) in Essential 8 (E8) medium and DuoESy medium, respectively (Fig. 1A). Without Matrigel coating, both cells in E8 medium failed to adhere to the culture dishes instead and formed cell spheres on day 1 culture. Notably, using DuoESy medium effectively steered the cell adhesion on non-coating dishes, and the cultured cells showed hPSC-like morphology (Fig. 1A). The cells were amplified for 40 days after 10 passages and still kept in undifferentiated morphology. The population doubling times (PDT) of the hESCs and the hiPSCs in the DuoESy medium were 25.88 ± 1.06 and 27.93 ± 1.32 h, respectively, similar to the results in E8 (25.4 ± 0.9, 26.2 ± 0.7 h, respectively) and mTeSR1 medium (26.6 ± 1.2, 27.0 ± 1.1 h, respectively), with Matrigel-coating conditions (E8/M vs DuoESy; mTeSR/M vs DuoESy; ns, P > 0.05, one-way ANOVA; Fig. 1B).

The DuoESy medium supports cell adhesion and cell proliferation. (A) H9 ESCs and 253G1 hiPSCs were dissociated and cultured with Essential 8 (E8) medium on non-coating (E8/non-coating) or Matrigel (E8/M) coating six-well plates. Both cells were also cultured with DuoESy medium without additional extracellular matrix coating. The remaining cells on the culture wells were illustrated. (B) The cell proliferation of both cells was calculated, and the population doubling time was estimated in E8/M and DuoESy culture conditions. Another culture medium for hPSCs, the mTeSR medium, was also examined. (C) The hPSCs were analyzed for the AP activities, and the cellular AP staining results were shown. (D) The comparative ratios of the AP staining’s OD revealed that DuoESy-cultured hPSCs exhibited significantly higher OD than that of E8/non-coating group (*P < 0.05; **P < 0.01; ***P < 0.001, One-way ANOVA). However, no significance (ns) exists between the E8/M and DuoESy groups. ANOVA: analysis of variance; AP: alkaline phosphatase; hESC: human embryonic stem cell; hiPSC human induced pluripotent stem cells; OD: optical density.
The adhered cells were further stained with an AP staining kit at 24 h culture (Fig. 1C) and the total cells were harvested with alkaline lysis buffer to determine the enzyme activities (Fig. 1D). We illustrated that few E8 medium-cultured cells have remained on the non-coating dishes, but DuoESy culture medium robustly supported the cell attachment, cell growth, and stemness maintenance (Fig. 1C). Comparable activities of AP, one of the indicators of the pluripotency, were detected in both E8/M and DuoESy culture conditions for both hESCs and hiPSCs (E8/M vs DuoESy; ns, P > 0.05, one-way ANOVA; Fig. 1D). These results demonstrated that the DuoESy medium is a novel product for enhancing the hPSC binding activity and supporting cell growth without requiring a pre-coating matrix.
Weekend-Free Culture for hPSCs
Due to the rapid degradation of the bFGF at 37°C, a fresh culture medium is generally supplied daily for stable hPSC proliferation. The thermal-stable bFGF mutant is a solution for extending the medium expiration from 1 day to 3 days at 37°C 35 . The DuoESy medium also contains a synthetic thermal-stable bFGF. We examine whether the medium supports the weekend-free culture for hPSCs. The confluent hiPSCs on culture plates were routinely dissociated with accutase and seeded at 0.5-1.0 × 105 cells/cm2 of six-well plates in 2 ml DuoESy α medium (day 1, D1). After 1 day of the hiPSCs culture (day 2, D2, Fig. 2A), the medium was replaced with 2 ml DuoESy mixture (α/β = 1/5, V/V) to support the exponential cell growth without medium refreshment during the next 3 days of incubation (D3 to D5, Fig. 2A). We illustrated that the PDTs were 22.5 ± 3.8 h for hiPSCs, respectively, and the cells exhibited classical hPSC morphology (Fig. 2A). These results demonstrate that the DuoESy medium can support stable and efficient cell expansion without fresh medium feeding for 3 days.

The DuoESy medium supports weekend-free culture. (A) The hiPSCs were cultured on non-coating six-well plates (Falcon) with DuoESy-α medium on day 1, and the medium was replaced with DuoESy αβ mixture (α/β = 1/5, V/V) on day 2 (D2). The cell proliferation was maintained without medium refreshment during the next 3 days of culture (D3–D5). (B) The DuoESy fitness to commercial culturewares was also evaluated. The hiPSCs were cultured on six-well plates from TPP, Falcon, and Corning companies for 4 days. The size of the colonies and the cell morphology of the cultured cells showed high similarity. hiPSC: human induced pluripotent stem cells.
The Fitness to Cell Culturewares
We next examined whether the DuoESy medium generally supports the hPSC growth on commercial cell culture dishes. The hiPSCs were dissociated with accutase and seeded on Matrigel-coating or non-coating culture plates at 104 cells/cm2 density. The hiPSCs were cultured on six-well plates of TPP, Falcon, and Corning companies for 4 days. The cell morphology was stably maintained, and the PDT showed no significant difference among the tested groups (TPP, Falcon, and Corning; 24.4 ± 4.9, 23.8 ± 2.7, 25.8 ± 2.63, respectively; P > 0.05, one-way ANOVA; Fig. 2B, C). This evidence demonstrates that the source of the culturewares might not be a limiting factor for applying the DuoESy medium.
The Maintenance of Pluripotency With DuoESy Medium
To explore the pluripotency maintenance using the DuoESy medium, we cultured the hESCs and hiPSCs for 40 days after 10 passages and examined for the expressions of pluripotent markers without the pre-coating process. ICC staining revealed that Sox2 and Oct4 transcription factors were strongly detected in most cells (Fig. 3A). FACS analyses revealed that more than 90% of cells expressed SSEA4 and TRA-1-60 on the cultured hPSC cell surfaces (Fig. 3B).

The DuoESy medium maintains the pluripotency of hPSCs. (A) The pluripotency of the cultured H9 hESCs and 253G1 hiPSCs were examined by immunocytochemical (ICC) staining of Oct4 and Sox2 expression. DuoESy-cultured cells show the intense expression of both genes in the nuclei, as E8/M and TeSR/M culture conditions. (B) DuoESy medium, as E8/M and TeSR/M, supported the high expression of SSEA4 and Tra-1-60 surface markers in both hESCs and hiPSCs. (C) The DuoESy-cultured hESCs and hiPSCs showed classical compact cell colonies with strong ZO-1 tight junction protein expression. The F-actin intracellular architecture was similar to the ZO-1 expression pattern. The nuclei of the cells were counterstained with DAPI. (D) The hESCs were maintained with the DuoESy medium for more than 10 passages. The normal karyotyping result of the amplified cells was illustrated. DAPI: 4’,6-diamidino-2-phenylindole; hESC: human embryonic stem cell; hiPSC: human induced pluripotent stem cells; hPSCs: human pluripotent stem cells.
The DuoESy-cultured hESCs and hiPSCs showed classical compact colonies (Fig. 3C) and the intense expression of a tight junction protein, ZO-1 (Fig. 3C). The intracellular F-actin architecture, revealed by phalloidin-iFluor staining, is similar to the ZO-1 expression pattern (Fig. 3C) 36 . In addition to the rapid proliferation and pluripotent gene expression, we examined the chromosome integrity and differentiating pluripotency in DuoESy-cultured hPSCs. We illustrated that the DuoESy-cultured hESCs at 10 passages showed normal karyotype configuration (Fig. 3D). This result indicated that the DuoESy medium could maintain chromosome integrity and might not have genotoxicity for triggering the genetic aberration.
Three Germ Layers Differentiation of hPSCs
We further cultured the hiPSCs under neural, cardiac, and endoderm differentiation medium, and the derived cells’ fates were identified by ICC staining. The derived neuroectodermal, endodermal, and cardiac mesodermal cells were robustly induced and featured with nestin+/β-III tubulin+ (Fig. 4A), Sox17+ (Fig. 4B), and TNNT2+/ actinin alpha 2 (ACTN2)+ proteins (Fig. 4C), respectively. We demonstrated that the DuoESy-cultured hiPSCs could commit into three germ layer cell lineages effectively.

DuoESy-cultured cells differentiate into three germ layer cells. The DuoESy-cultured hiPSCs were induced to differentiate toward neural, cardiac, and endoderm cells. (A) The differentiating neural precursors and mature neural cells were characterized by nestin and βIII-tubulin (TuJ1) expressions, respectively. (B) The differentiating endodermal cells expressed the Sox17 transcription factor. Only a few Sox2-positive cells, representing the undifferentiated cells, were detected. (C) The hiPSC-derived cardiac cells were featured with the expression of TNNT2 and ACTN2 proteins and muscular fibers. hiPSC: human induced pluripotent stem cells; TNNT2: Troponin T type 2; ACTN2: actinin alpha 2.
The Expressions of Integrin Messenger RNAs in the DuoESy-Cultured hPSCs
The integrin-associated intracellular signal is essential for hPSC attachment, proliferation, and pluripotency14,34,37. Previous studies have shown that integrin subunits α5, α6, αv, β1, and β6 were expressed on hPSC surface when the cells adhered onto Matrigel or vitronectin-coating culturewares33,34. The active downstream signals of both αvβ5 and β1 integrins of hPSCs are essential for cell adhesion and proliferation33,34. Here, in DuoESy-cultured hESCs cells, we found that the ITGA2, ITGA3, ITGA5, ITGA6, and ITGAB5 messenger RNA (mRNA) expressions were reduced, but ITGA7, ITGAV, ITGB1, and ITGB3 were not affected, compared with those of the E8/M cultured cells (Fig. 5A). In DuoESy-cultured hiPSCs, all the examined integrin subunits’ expressions were decreased by about half (Fig. 5B). Blocking the binding of α5, α6, β1, and αvβ5 attenuated the cell growth of both hESCs and hiPSCs (Fig. 5C, D). Notably, blocking the binding of αvβ5 rapidly and consistently decreased both hiPSC growths at 48 h posttreatment. These results indicate that the integrin-associated signals are conserved for the attachment and proliferation of DuoESy-cultured cells, especially for the α5, α6, β1, and αvβ5 molecules.
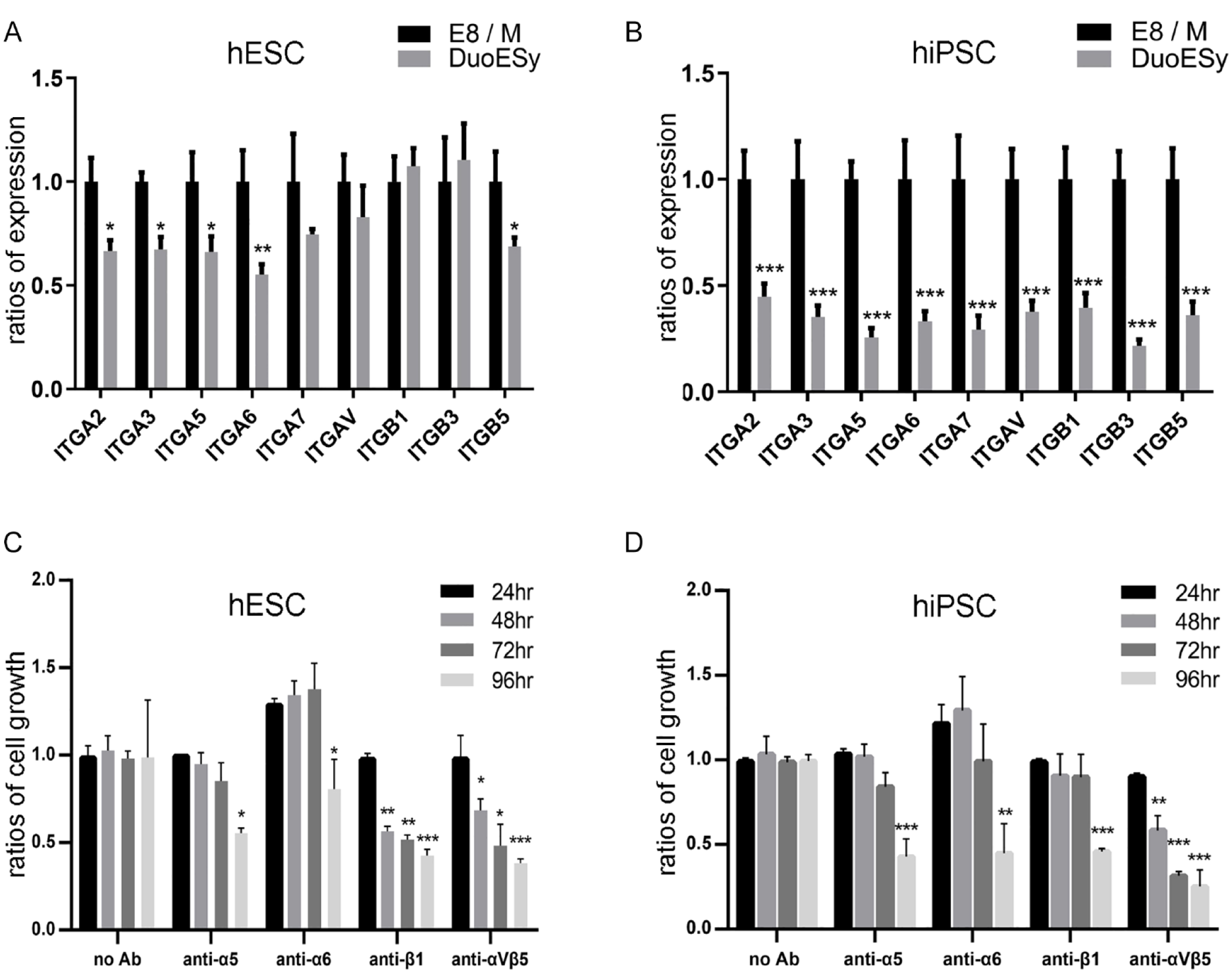
The integrin expression profiles of the DuoESy-cultured cells. The mRNA of integrin subunits in H9 hESCs (A) and 253G1 hiPSCs (B) were analyzed by quantitative RT-PCR under E8/M and DuoESy culture conditions. The significant reduction of the integrin expression was validated from triplicate experiments by one-way ANOVA. The hESCs (C) and hiPSCs (D) were cultured with DuoESy medium in the presence of integrin-blocking antibodies. The cell numbers were estimated by colorimetric MTS assay. The hPSC cell numbers with E8/M at 24 h were set as 1.0, and the growth inhibitory effects of the blocking antibodies on the hPSCs were expressed as comparative ratios with that of the E8/M condition. We conducted triplicate independent tests of the integrin-blocking antibodies, and the significance was validated by two-way ANOVA and Tukey’s post hoc test. ANOVA: analysis of variance; hESC: human embryonic stem cell; hiPSC: human induced pluripotent stem cells; mRNA: messenger ribonucleic acid; RT-PCR: real-time polymerase chain reaction.
DuoESy Medium Supporting the Clonal Expansion and iPSC Establishment
We next examined whether the DuoESy medium provides sufficient growth stimuli for the single-cell clonal expansion. We seeded 10,000 hESCs cells in 10-cm culture dishes at a low cell density and applied the AP staining to count clonal numbers. We discovered that in the E8/Matrigel condition, 183.3 ± 25.1 AP positive colonies were identified and, in the DuoESy condition, 198.7 ± 46.5 AP positive colonies were detected on day 14 culture (P = 0.32; Student t test). These results indicate that using the DuoESy medium alone exhibits a competitive performance of clonal expansion as using E8/Matrigel combination.
The validated clonal formation ability of the DuoESy medium promoted us to investigate the potential for patient iPSC establishment from PBMCs. MELAS disease, a hereditary mitochondrial genetic disorder, mainly affects the nervous system and muscles in adolescence or early adulthood 38 . MT-TL1 mitochondrial gene is the most commonly affected target, and the m3243A>G mutant is found in 80% of patients 38 . We recruited a mutant MT-TL1-carrying MELAS patient, and her PBMNCs were purified after Ficoll-mediated density-gradient separation. We transfected the 1.0 × 106 PBMNCs with Yamanaka factors and cultured them with the DuoESy medium without ECM coating. Thirteen iPSC-like clones (Fig. 6A) were isolated, and three clones were successfully amplified after more than 15 passages with DuoESy medium. The iPSCs clones were SSEA4 positive and expressed the pluripotent Oct4 and Nanog proteins (Fig. 6B–D). Interestingly, the established iPSC clones converted to the iPSCs carrying normal MT-TL1 genotypes (m3243A). This genetic correction in MELAS iPSCs has been reported and may be caused by the heterogeneity of mitochondria in donor PBMNCs and the metabolic selection pressure during the cell reprogramming period39–41. Together, these results illustrate that the DuoESy medium is suitable for clonal cell expansion and for the iPSC establishment without the requirement of ECM pre-coating.

DuoESy medium supports iPSC clonal expansion and establishment. We collected the peripheral blood mononuclear cells from a symptomatic MELAS patient with a point mutation at the MT-TL1 mitochondrial gene (m3243G). (A) The MELAS iPSCs were established by the DuoESy culture with non-coating Falcon 6-cm dishes, and the iPSC-like clone at 19 passages (MELAS iPSC P19) was illustrated. The iPSCs clones were SSEA4 positive (B), and more than 90% of cells expressed the pluripotent Oct4 (C) and Nanog proteins (D). DNA sequencing data showed that the expanded MELAS iPSCs at P19 carried a normal MT-TL1 genotype (m3243A) but not the mutant one. DAPI: 4’,6-diamidino-2-phenylindole; FITC: fluorescein isothiocyanate; iPSC: induced pluripotent stem cells; MELAS: mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes; SSEA4: stage-specific embryonic antigen 4.
Discussions
This study introduces a coating-free medium for hiPSC expansion on conventional cell culturewares, compared with the popular TeSR or E8 medium. This GMP-compatible medium supports the fast adhesion of hiPSCs on polystyrene culture plates and retains a normal karyotype and pluripotency for at least 10 passages. In addition, the working hours, labor, and consumable costs will be substantively reduced for exponential cell scale-up by excluding the coating processing. Furthermore, this medium could integrate well with existing CMC (chemistry, manufacturing, and controls) standards for hiPSC culture without requiring additional procedures, manufacturing, or specialized instruments.
The extraordinary regenerative potential of hPSC inspires academic and industrial researchers to find suitable solutions for efficient scale-up hPSCs. Laminin fragments and vitronectin have been shown as culture medium ingredients to facilitate the hPSC adhesion in non-coating cell culture plates8,42. Alternatively, surface modifications with ECM or polymers were discovered for the replacement of Matrigel, such as vitronectin, laminin, and their derivatives, glycosaminoglycan-binding peptides, fibrin-based hydrogels, and synthetic polymers12,43,44. Nevertheless, these approaches have not been extensively implemented and most researchers still use Matrigel or vitronectin as the supporting matrix for hPSC growth. The restriction of these alternatives may be high cost, lacking reproducibility, complicated ingredients, or noncompatible with existing CMC protocols. This study demonstrates that using the DuoESy medium is a simple and time-efficient method for hPSC culture. These characteristics may enable this product to support a straightforward scale-up approach for producing clinical-orientated hPSCs on commercial culturewares or microcarriers without additional coating substrates.
For the clinical-grade hPSC culture, recombinant vitronectin is commonly used for coating tissue culture plastics. Vitronectin is chemically defined and xenofree, contributing to the consistency of CMC and product safety, respectively. However, vitronectin is thermal-unstable and must be aliquoted and stored at −20°C or −80°C45,46. The coating procedure must be pre-validated for successful iPSC establishment and expansion. In addition to the coating matrix, vitronectin could be supplied as a medium ingredient for promoting cell adhesion. Miyazaki et al. added the recombinant vitronectin, iMatrix-511, and laminin-521 in the E8 or StemFit medium 9 . They found that both iMatrix-511 and vitronectin, but not laminin 521, supported the cell adhesion and exponential hPSC cell growth on non-precoating plates. Especially, at a lower concentration, the iMatrix-511 exhibited superior performance than vitronectin for cell adhesion. Adding either iMatrix or vitronectin in the culture medium provides a coating-free hPSC medium that excludes these quality-assurance works for the protein shelf life and the coating efficacy examinations. Nevertheless, combining iMatrix/vitronectin with E8/StemFit media costs too high to culture hPSCs for current academic and clinical applications.
A human serum–derived protein, IαI, activates pluripotent signals in mouse PSCs and steers hPSC attachment in E8 medium in a non-coating tissue culture device10,47. The IαI supplement also supports clonal expansion and long-term culture of hPSCs without rho-activated kinase (ROCK) inhibitor 10 . The size of IαI is 225 kDa and can be isolated from human plasma with high purity. These promising results make E8/IαI an excellent solution for scale-up clinical-orientated hPSCs. Nevertheless, the E8/IαI product has not been publicly commercialized since the investigators published it in 2016.
Cells from patients with MELAS disease, featured with mitochondrial DNA mutation and high heterogeneity, have shown significantly lower efficiency of iPSC establishment when the patient’s cells harbor a high ratio of mitochondrial DNA mutants 48 . In this study, we demonstrated that using nonviral episomal vectors and simple electroporation can successfully produce several MELAS iPSC lines from the PBMCs of a patient. This result shows that the DuoESy medium might offer a reliable solution to support single iPSC cloning and exponential cell growth of iPSC lines. The reprogramming-mediated genetic correction with DuoESy culture might help the production of normal tissue or organs for cell therapy in MELAS patients.
Here we introduce an innovative xenofree medium to support the coating-free culture of hPSC growth on tissue culture devices. The genomic stability and the pluripotent stemness of the expanded cells are stably sustained. This medium also supports the iPSC establishment from patients’ primary PBMCs. Although the complete ingredients of the DuoESy medium are not revealed due to the trade secret, we claim that the formula is innovative and does not violate the published patents to our best knowledge. The key ingredients of the medium, including the factors for promoting cell growth and adhesion, are synthesized in the E. coli system under ISO13485 regulation. The DuoESy medium also does not contain the aforementioned vitronectin, synthetic laminin, and IαI proteins.
This study demonstrated that the DuoESy-cultured hPSC could differentiate into neuronal cells, cardiomyocytes, and primitive endodermal cells. Mature endodermal lineages, organoid formation, and teratoma induction in vivo are still required to strengthen the claim of pluripotency of DuoESy-cultured hPSCs. Accumulative evidence for exploring the pluripotency with the DuoESy medium will benefit the reliability of this product. We hope this discovery will help basic stem cell research, drug screening, organoid research, and even clinical therapies by simplifying the hPSC culture procedure.
Footnotes
Acknowledgements
The authors are grateful to Professor Hong-Lin Su’s lab members for their technical support in this study. The authors thank the staff of the DuoGenic StemCells Corporation, Taichung, Taiwan, for assisting with the experiments.
Author Contributions
C.Y.L. and H.L.S. contributed to conceptualization. C.Y.L., Y.Y.C., S.F.W., Y.K.L., L.Y.S., S.Y.T., C.I.S., and H.L.S. contributed to in vitro experiments and analyses. H.C.F., S.Y.T., Y.C.C., and H.L.S. contributed to resources. C.Y.L. and H.L.S. contributed to writing—original draft. C.Y.L., H.C.F., S.Y.T., and H.L.S. contributed to writing, review, and editing of the draft.
Ethical Approval
Ethical issues of the study were approved by the Research Ethics Committee of the ethics institutional review board in Tungs’ Taichung Metroharbor Hospital (Taichung, Taiwan) (IRB number: 109038).
Statement of Human and Animal Rights
All procedures involving human subjects were conducted in accordance with the Clinical Trials Act and the tenets set down in the Declaration of Helsinki and with the ethical guidelines for medical and health research involving human subjects of Taiwan policies, adopted by the Research Ethics Committee of Tungs’ Taichung Metroharbor Hospital (Taichung, Taiwan; institutional review board [IRB] number: 109038).
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information in accordance with the Declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors have the following competing interests. The technology transfer office of National Chung Hsing University has received consultancy, speaker fees, and research grants on behalf of H.L.S. from DuoGenic StemCells Corporation and Hualien Tzu Chi Medical Center. C.Y.L., Y.Y.C., W.S.F., Y.K.L., and C.I.S. are employees of Duogenic StemCells Corporation. C.Y.L., Y.Y.C., W.S.F., Y.K.L., C.I.S., and H.L.S. are shareholders of DuoGenic StemCells Corporation. H.C.F., L.Y.S., S.Y.T., and Y.C.C. have no conflict of interest with respect to the research, authorship, and publication of this article. There are patents, products in development, and marketed products associated with this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by DuoGenic StemCells Corporation, Taiwan. This work was also supported by the Industry–Academic cooperation project (108-D-595, 109-D-525) between the DuoGenic StemCells Corporation and National Chung Hsing University.
