Abstract
Chronic lung diseases, such as chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF), involve progressive and irreversible destruction and pathogenic remodeling of airways and have become the leading health care burden worldwide. Pulmonary tissue has extensive capacities to launch injury-responsive repairing programs (IRRPs) to replace the damaged or dead cells upon acute lung injuries. However, the IRRPs are frequently compromised in chronic lung diseases. In this review, we aim to provide an overview of somatic stem cell subpopulations within distal airway epithelium and the underlying mechanisms mediating their self-renewal and trans-differentiation under both physiological and pathological circumstances. We also compared the differences between humans and mice on distal airway structure and stem cell composition. At last, we reviewed the current status and future directions for the development of targeted therapeutics on defective distal airway regeneration and repairment in chronic lung diseases.
Introduction
The lung tissue had been considered highly quiescent with limited regenerative capacities; however, large body of evidences has revealed that the lung tissue possesses remarkable reparative capacities responsive to varieties of distal airway injuries 1 . The distal airway comprises terminal bronchioles (TB) and alveoli that perform essential respiratory functions. The bronchioles branch off from a hierarchy of bronchi that are stemmed from the trachea, and the alveoli are the basic units for gas exchange. The distal airway epithelium is composed of a wide variety of epithelial cells2,3, several of which have been reported as somatic stem cells (SSCs) (Fig. 1).
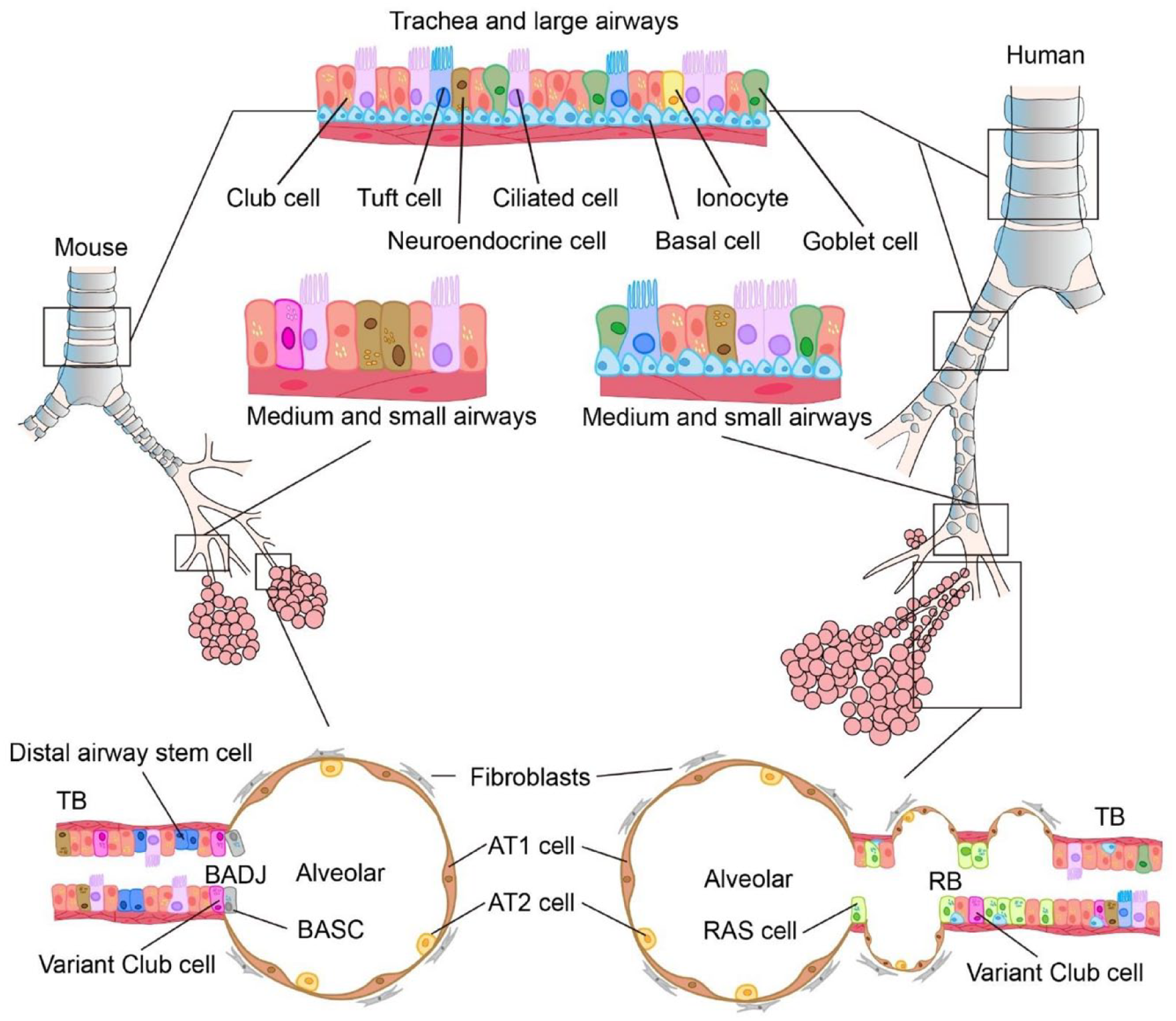
The anatomical structures of human and mouse airways and their epithelial components. TB: terminal bronchioles; BADJ: bronchioalveolar duct junctions; BASC: bronchioalveolar stem cells; AT1: alveolar type I; AT2: alveolar type II; RAS: respiratory airway secretory; RB: respiratory bronchioles.
Different SSCs have their own unique niches, including SSCs and their progenies, but also a variety of heterogeneous cell types and extracellular matrix (ECM). Under physiological conditions, SSCs are largely in dormant state, maintaining low level of self-renewal and differentiation. Once the tissue is injured, the SSCs receive signals from surrounding microenvironment that subsequently drive the regeneration or reparative process. However, the reparative capacities of SSCs are frequently compromised if the niche environment is not supportive 4 .
Human and mouse lungs have similar histological structure. The lung is composed of trachea, bronchi, bronchioles, and alveoli from the proximal to the distal airways. Trachea and bronchi are mainly composed of club cells, goblet cells, multiciliated cells, basal cells, and a small number of neuroendocrine cells, brush cells, and ionocytes. Unlike human, mouse small airways do not contain basal cells or goblet cells. The human distal airway is connected to the alveolar through a transitional airway region (also known as the respiratory bronchiole [RB]). In this region, there are rare newly discovered stem cells called respiratory airway secretory (RAS) cells 5 . There are no RB in mice, which directly transit to the alveolar area through the middle airway. The distal bronchioalveolar duct junction (BADJ) region in the mouse lung contains bronchioalveolar stem cell (BASC) population, whose counterparts have not been found in human lung. Both mouse and human alveoli contain two cell populations, alveolar type II (AT2) and alveolar type I (AT1) cells (Table 1).
Identification of Pulmonary Epithelial Stem Cells.

Distal Airway SSCs
SSCs in the Bronchi
Basal cells
Human lung basal cells are distributed throughout the main airways to the terminal and respiratory bronchioles (TRB), and owing to the lack of counterpart RB, the mouse basal cells are mainly distributed in the main bronchi only 6 . Basal cells occupy ~6% of the epithelium in the smallest conducting airways and are commonly identified as KRT5+TP63+ at steady state 7 . Lineage-tracing experiments validated that Krt14+ basal cells are multipotent airway progenitors that could give rise to ciliated cells and secretory cell types following naphthalene-induced lung injury (Box 1)8,9. In vitro sphere-forming assays (Box 2) demonstrated that both human and mouse airway basal cells can self-renew and generate luminal cells 7 . Gene expression analysis and histomorphometric studies with both steady-state and naphthalene-injured mouse trachea have demonstrated that there are two basal cell pools (Krt14+ and Krt14−) that restore the tracheobronchial epithelium coordinately10,11. More recently, a study pointed out that NOTCH signaling is required for the differentiation, but not self-renewal of basal cells12,13. Another study showed that immediately following injury, one p63+ basal cell population activates intracellular NOTCH2 signaling and generates secretory cells directly, while the other basal cell population expresses c-Myb and directly yields ciliated cells 14 . In addition, a rare population of SOX9+/P63+/KRT5+ basal cells has been shown to be able to regenerate human alveoli 15 .
Models of lung injuries.

Lung Organoids Culture Models.

AT2: alveolar type II; AT1: alveolar type I; iPSCs: induced pluripotent stem cells.
Club cells
With specific expression of the secretoglobins Scgb1a1 and Scgb3a2, club cells are known as predominant progenitors for the maintaining and repair of bronchial epithelium. Genetic lineage tracing study demonstrated that Scgb1a1+club cells can self-renew and differentiate into ciliated cells 16 . Of note, studies of dioxide-induced injury and influenza viral infection in mice demonstrated that club cells can also dedifferentiate into basal cells when basal cells are ablated 17 . However, it remains unclear whether this process also occurs in physiological condition.
Variant club cells
Naphthalene is an aromatic hydrocarbon that is used to induce the ablation of club cells 18 . A pollutant-resistant subpopulation of club cells called variant club cells expressing Clara cell secretory protein (CCSP/SCGB1A1) and Upk3a were identified in a study of naphthalene-induced club cell ablation19,20. This variant club cell subpopulation lacks the expression of cytochrome P450-2F2 (CYP-2F2), which generates highly cytotoxic 1R, 2S-naphthalene epoxide19,20. Variant club cells normally locate around neuroendocrine bodies (NEBs) and within the BADJs, and these cells can replenish secretory and ciliated cells under both physiological and pathological (club cell ablation) conditions. In addition, variant club cells can also give rise to AT1 and AT2 cells after bleomycin-induced alveolar injury21,22.
Distal airway stem cells (DASCs)
Emerging number of reports suggest that alternative subsets of distal airway epithelial cells may serve as facultative progenitors and promote regeneration of injured alveolar epithelium. Lineage tracing study revealed that Trp63+stem cells originated from the bronchiolar epithelium can be detected in alveolar regions and assemble into Krt5+ pods following pulmonary infection of H1N1 influenza virus 23 . Kumar et al. 23 defined this facultative progenitor cells as DASCs. Controversially, earlier studies have shown that DASCs can differentiate into AT1, AT2 cells as well as bronchiolar secretory cells following H1N1 infection 24 . However, subsequent articles showed that DASCs are unable to repair alveolar epithelium after influenza virus infection in mice25,26.
Bronchioalveolar stem cells
At the BADJs in the mouse, a rare population of stem cells expressing markers of both club cells (Scgb1a1) and AT2 cells (Sftpc) was called BASCs. BASCs are resistant to bronchiolar and alveolar damage and proliferate during epithelium restoration27–30. However the role of BASCs during lung repair and regeneration in vivo remains controversial: Rawlins et al. 16 concluded that BASCs do not contribute into postnatal maintaining of bronchioles and alveoli; in contrast, recent two studies demonstrated that BASCs could generate the majority of distal lung airway cells including club cells, ciliated cells, AT1, and AT2 cells after bronchiolar damages and rarely contribute to cellular turnover under homeostatic conditions by using lineage-tracing system31,32. However, the structure of BADJ has not been found in the human lung. Therefore, it is important to confirm whether BASCs or similar cell subsets exist in human lung.
RB cells
Human distal airways contain a unique structure of RB that are absent in mice 6 . RB are frequently injured in several chronic human respiratory diseases, such as chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF) 33 . Since the mouse lung lacks the structure of RB, it remains one of the least known regions of the human lung. Surprisingly, two back-to-back papers recently revealed that a new lineage of stem cells is present in this mysterious region of humans distal airways5,34. Morrisey et al. identified a distinct population of RAS cells expressing high SCGB3A2 level. Results of organoid model (Box 2) showed that these RAS cells are unidirectional progenitor cells that can rapidly differentiate into AT2 cells, a process controlled by coordinated NOTCH and Wnt signaling 5 . Using spatial transcriptomics and single-cell profiling of distal airways, Tata et al. identified several novel TRB cell types, including airway-associated LGR5+ fibroblasts, TRB-specific alveolar type-0 (AT0) cells, and TRB secretory cells (TRB-SCs). They showed a new differentiation program that AT2 cells transiently acquire an AT0 state from which they can differentiate into either AT1 cells or TRB-SCs. Although more research is needed to unveil how the RB structures and related stem cells impact human lung function and regeneration, these two articles have milestone significance in the field of human alveolar research.
Alveolar SSCs
Pulmonary gas exchange occurs in alveolar sacs lined by two epithelial cell types: AT2 and AT1. AT1 cells are long flat squamous cells covering majority of the gas-exchange alveolar surface, while AT2 cells are small, cuboidal cells secreting surfactants that prevent alveolar collapse18,19. AT2 cells have been reported as SSCs35,36. Recently, it has been reported that AT1 cells also have stem cell plasticity during alveolar development and regeneration37,38 (Fig. 2).

Trans-differentiation network among somatic stem cells in distal airways. BASC: bronchioalveolar stem cells; AT2: alveolar type II; RAS: respiratory airway secretory; AT1: alveolar type I.
AT2
AT2 cells are the most thoroughly studied alveolar regenerative progenitors to date. After acute lung injury induced by NO2 exposure in rats, AT2 cells can self-renew and differentiate into AT1 cells35,36. These data support that AT2 cells are the progenitor cells for AT1 cells in rodents 39 . Moreover, a medical study first provided evidence that new lung growth occurred in an adult human after a right-sided pneumonectomy (PNX) (Box 1) 40 . Genetic lineage-tracing experiments showed that AT2 cells self-renew and differentiate at a low rate over 1 year. Moreover, when the lung epithelial cells are ablated after injury, the remaining AT2 cells can proliferate rapidly 41 . More details on AT2 functioning at the alveolar stem cells will be reviewed in the following section.
AT1
AT1 cells have long been considered as terminally differentiated cells. However, recent studies have shown that AT1 cells also possess stem cell plasticity. In 2015, Jain et al. 42 discovered that Hopx+ AT1 cells proliferated and dedifferentiated into AT2 cells during adult alveolar regeneration after PNX (Box 1), and this process was regulated by tumor growth factor-beta (TGF-β) signaling. Moreover, a subsequent study further explored the changes of AT1 cells at the transcriptome level and found that there are two types of postnatal AT1 cells: Igfbp2+ Hopx+ and Igfbp2− Hopx+ AT1 cells. The findings of this study supported that there is an “immature” period of Igfbp2− Hopx+ AT1 cells during the process of AT2-to-AT1 differentiation in alveolar regeneration. The stem cell like “immature” Igfbp2− Hopx+ AT1 cells are bidirectional and capable to trans-differentiate into AT2 cells under pathological conditions, while Igfbp2+ Hopx+ AT1 cells represent the terminally differentiated population of AT1 cells 38 . In the latest study, AT1 cells can be reprogrammed into AT2 cells to promote alveolar regeneration after neonatal injury, and this plasticity is retained during alveolar maturation, while the ability of AT2 cells to differentiate into AT1 cells is restricted in the adult mouse lung 37 . Up to now, understanding of AT1 cell development and heterogeneity is very limited in the field of lung regeneration. Future investigations are needed to validate whether the reported AT1 plasticity can be extrapolated to other species or other lung injury models.
Basal cells, club cells, and AT2s are the main SSCs in distal airways. SSCs transdifferentiate into each other to maintain lung homeostasis under physiological and pathological states.
Repairing Mechanisms for Distal Airways
Acute pulmonary injuries rapidly induce the intrinsic repairing programs of the SSCs to restore the structural and functional homeostasis. In general, majority of our knowledge for repairing mechanisms of distal airways were obtained from studies of acute pulmonary injuries, during which SSC-intrinsic and -extrinsic factors work orchestrally to facilitate balanced self-renewal and differentiation of SSCs.
Alveolar Repairing and Regeneration
Transitional AT2 states and related regulatory signaling pathways during alveolar regeneration
In the alveolar epithelium, only small proportions (~20%) of AT2 are alveolar epithelial progenitor (AEP) cells that are quiet stable during physiological homeostasis 43 . When acute lung injuries occur, the AEP pool expands rapidly to regenerate the damaged alveolar epithelium by replenishing large number of AT2 and AT1; however, the AEP pool shrinks back to physiological state once the repairing process is accomplished35,43. The single-cell omics technique has characterized three transitional states of AT2 during the alveolar repairing process: self-renew (proliferating), self-renew cessation (cell cycle arrest), and transdifferentiating (AT2-to-1) 44 . Several groups have coincidentally defined a AT2 state as pre-AT1 transitional state (PATS) or damage-associated transient progenitor (DATP) cells, which shares the similar transcriptomic features as AT1, but its expressions of canonical AT1 markers including Pdpn, Hopx, and Cav-1 are much lower45,46–48.
Numerous molecular and genetic studies have identified multiple pathways involved in the regulation of alveolar regeneration, including Wnt, FGF, BMP, NOTCH ligands, TGF-β and retinoid acid (RA). Synergistic activation of these signaling pathways occur dynamically and orderly at different stages of the alveolar repair process. The Wnt/β-catenin, FGF/FGFR2 and ETS variant transcription factor 5 (Etv5) signaling facilitate AT2 self-renew but inhibit AT2-to-1 trans-differentiation35,43,49,50, while BMP signaling promotes AT2-to-1 transdifferentiation 51 . For TGF-β signaling, it was initially considered that the autocrine TGF-β1/Smad pathway is important for AT2-to-1 transdifferentiation 52 , further study precisely revealed that TGF-β signaling needs to be temporally upregulated for cell cycle arrest in AT2, but downregulated afterwards for AT2-to-1 trans-differentiation 44 . The NOTCH pathway is activated in AT2 for AT2 proliferation in response to acute injuries, while subsequent expression of Dlk1 is required to temporally modulate NOTCH signaling for AT2-to-1 transition 52 . RA indirectly promote alveolar repair by rejuvenating regenerative fibroblasts 53 .
Alveolar microenvironment fuels alveolar repair
Wide variety of components in the alveolar niches, such as fibroblasts, endothelial and immune cells, produce and secrete supporting factors such as Wnt ligands, FGF, BMP, NOTCH ligands, TGF-β, RAs, and so on, to cultivate the AEPs to launch the repairing programs in response to injuries (Fig. 3).

Reparative niche for alveolar regeneration: (A) Pdgfrα+ fibroblasts maintain the stemness of AT2 cells by secreting short-range Wnt ligands. PCECs regulate epithelial homeostasis by modulating the expression of HGF and EGF. Macrophages promote alveolar repair by producing inflammatory cytokines (IL-1β and TNF-α) and HGF. Damaged AT2s secrete the “alarmin” molecules IL-33 that in turn activates ILC2s to release IL-13 and IL-5, which indirectly facilitate AT2 proliferation by promoting differentiation of alternately activated macrophages. (B) A transitional AT2-to-1 state exists during alveolar regeneration. Persistence of aberrant AT2-to-1 transient cell state leads to impaired alveolar regeneration. AT2: alveolar type II; PCECs: pulmonary capillary endothelial cells; HGF: hepatocyte growth factor; EGF: epidermal growth factor; IL: Interleukin; TNF-α: tumor necrosis factor alpha; AT1: alveolar type I.
Fibroblasts serve as the main source of the Wnt ligands
Three fibroblast subpopulations are present in the alveoli: retinol-storing Pdgfrα+ lipofibroblasts, cytokine-producing stromal fibroblasts, and mechanically contractile myofibroblasts54–57. The AEPs are located near the single Pdgfrα+ lipofibroblast niches, which are essential for alveolar development by secreting short-range Wnt ligands to facilitate AT2 stemness maintaining and also producing RAs and lipids to promote surfactant synthesis in AT235,54–57. Persistent or repeated fibroblast activation leads to pathogenic airway remodeling and impaired capacity to produce supporting factors for SSCs regeneration under chronic pathogenic conditions54,58.
Pulmonary capillary endothelial cells promote lung alveologenesis
Growing evidence has proved that epithelial-vascular interactions are essential for tissue patterning59,60. Alveolar epithelial cells are surrounded by pulmonary capillary endothelial cells (PCECs) 61 . PCECs are not just passive conduits for the delivery of oxygen, but also promote alveologenesis by providing angiocrine growth signals59,60. Unilateral PNX induce VEGFR2 and FGFR1 activation within PCECs of the remaining lung lobes, leading to increased production of matrix metalloprotease MMP14, which subsequently promotes regenerative alveolarization by releasing epidermal growth factor (EGF)-like ligands 62 . Pulmonary capillaries regulate epithelial morphogenesis by modulating the expression of hepatocyte growth factor (HGF), which is necessary for distal airway development 60 . Besides, endothelial cell derived HGF also indirectly facilitating lung regeneration through suppressing tissue fibrosis by preventing aberrant activation of the NADPH Oxidase 4 (NOX4) in perivascular fibroblasts63–65. Recent articles have shown that the Car4+PCECs receive reparative signals from adjacent AT1 cells during influenza infection induced alveolar revascularization66,67.
Immune niches for alveolar epithelium cells
Macrophages/monocytes eliminate foreign substances by engulfing pathogens and triggering immune response68,69. Growing evidence has shown that macrophages and bone marrow-derived monocytes contribute into acute injury-induced airway epithelium regeneration70–72. Mechanistically, macrophages promote alveolar repair by producing inflammatory cytokines (IL-1β and TNF-α) and growth factors (HGF)70,73–75. Damaged AT2s secrete the “alarmin” IL-33, which in turn activates group 2 innate lymphoid cells (ILC2s) that release IL-13 and IL-5. The aforementioned type II cytokines indirectly facilitate AT2 expansion by promoting differentiation of alternatively activated macrophages71,76. Controversially, another study reported that IL-13 disrupts AT2 stem cell activity 72 . Future studies are important for dissecting other immune cell subpopulations that mediate alveolar regeneration.
Regeneration of Bronchi Epithelium
The distal airways mainly contain two types of SSCs, basal cells and club cells, which function cooperatively to maintain distal airway homeostasis and regeneration8,9,16 (Fig. 4).

Reparative niche for bronchi epithelium regeneration. MCs: mesenchymal cells; ASMCs: airway smooth muscle cells; IL: interleukin.
NOTCH and TGF-β/BMP/SMAD signals promote basal cell differentiation and pseudostratified epithelium regeneration
Adult KRT5+ basal stem cells turnover slowly to maintain airway epithelial integrity during homeostasis 77 . When the airway epithelium is injured, basal cells proliferate rapidly and differentiate into all cell types of pseudostratified epithelium to repair the damaged airways14,78. Fate determination of basal cell differentiation is controlled by NOTCH signaling pathway. Basal cells tend to differentiate into club cells when NOTCH2 signal is active; otherwise, they differentiate into ciliated cells 14 . In addition, activation of the TGF-β/BMP/SMAD signaling pathway promotes basal cell differentiation and luminal regeneration after injury, while inhibition of this signal leads to basal cell hyperplasia 79 .
BMP and NOTCH signals are essential for club stem cell self-renewal
Club cells mainly exist in the noncartilaginous region of the distal airway, especially in the bronchioles. Club cells can self-renew and differentiate into ciliated cells, making club cells the main progenitors for the maintenance and repair of bronchial epithelium 16 . BMP and NOTCH are involved in the self-renewal and differentiation of club stem cells. Bmpr1a-mediated nonclassical BMP signaling pathway is essential for the expansion of regenerating club cells 80 . Loss of Bmpr1a, Tak1, or Mapk14 in adult mouse club cells leads to defective bronchial regeneration and repair. Activation of NOTCH signaling can maintain club cell fate and prevent differentiation of club cells into ciliated cells81,82. p53 deficiency promoted club cell self-renewal 83 .
Mesenchymal stromal cells regulate bronchi epithelium regeneration
Mesenchymal cells (MCs), in proximity to the SSCs in the distal airways, have been shown to regulate the stemness and differentiation of SSCs through FGF, Hippo, Wnt, NOTCH signaling pathways. Under steady state, MCs maintain basal stem cell status in cartilaginous airways by providing FGF10 to basal cells to inhibit Hippo signal (increase nuclear yap proteins)84,85, while upon airway injuries, increased expression of FGF10 in airway smooth muscle cells (ASMCs) and Wnt7b in airway epithelium allow basal cells to move into noncartilage-conducted airways for epithelial regeneration 84 . The Wnt ligands produced by MCs do not contribute much to basal cell proliferation under physiological condition but are essential for epithelium repair post injuries 54 . Following pulmonary injuries, MCs produce Wnt ligands to promote proliferation of basal cells, which further secrete Wnt ligands and subsequently regulate ciliated cells generation from basal cells 54 . Similarly, upon airway injury, IL-6 produced by MCs activates STAT3 in basal stem cells, which in turn inhibits NOTCH signaling and promotes basal stem cell differentiation into ciliated cells 86 . LGR6+ ASMCs in bronchiolar epithelium can also help club stem cells repair damaged airway epithelium through Wnt-FGF10 signaling 87 . Hedgehog signaling in Scgb1a1+ club cells of the distal airways control postnatal lung mesenchyme quiescence and regulates distal airway epithelium homeostasis and regeneration 88 .
MCs maintain basal stem cell status in cartilaginous airways by providing FGF10 to basal cells to inhibit Hippo signal, while upon airway injuries, increased expression of FGF10 in ASMCs and Wnt7b in airway epithelium allow basal cells to move into noncartilage-conducted airways for epithelial regeneration. IL-6 produced by MCs activates STAT3 in basal stem cells, which in turn inhibits NOTCH signaling and promotes basal stem cell differentiation into ciliated cells.
Impaired Injury-Induced Reparative Programs of SSCs in Chronic Lung Diseases
Chronic lung diseases, such as COPD and IPF, involve long-term and irreversible airway destructions and pathogenic airway remodeling, leading to dampened capacities of lung tissue to launch the “injury-induced reparative programs” 89 ; however, the underlying mechanisms remain largely unknown. In most cases, the only treatment for chronic lung disease is tissue transplantation, and the 5-year survival rate post transplantation is only 59% 90 . Understanding of causal mechanisms responsible for the impaired reparative programs is of great significance for the development of targeted therapeutics for chronic lung diseases. Currently speaking, it is suggested that the airway SSCs under repeated injury-repair cycles during the development of chronic lung diseases possess exhausted SSCs stemness caused by SSC-intrinsic and extrinsic mechanisms91,92.
SSC-Intrinsic
Accumulating evidence suggests AT2 cell injury is an early event in the development of chronic lung diseases93,94. The common features of COPD and IPF are great loss of functional AT2s accompanied with increased AT2s aging and senescence89,95–98. Genetic studies demonstrate that senescence of AT2s is sufficient to initiate progressive IPF-like pathologies 99 . Abnormal telomerase expression and misfolded surfactant proteins within AT2s are also closely associated with AT2s senescence and apoptosis100,101. Therefore, elucidating the underlying mechanisms of AT2s senescence is essential for the development of effective therapies for alveolar repair.
Recent studies have shown that persistence of aberrant AT2-to-1 transient cell state controlled by TP53 signaling fails to repair damaged alveoli and plays important roles in the pathologies of IPF45,46. The AT2-to-1 transitional cells are trapped in the intermediate stage of abnormal differentiation and are incapable to terminally differentiate into AT1 cells in IPF, leading to destroyed alveolar repairment45,46. The aberrant PATs are characterized by enriched p53 and TGF-β signaling, cell senescence and DNA damage 45 .
SSC-Extrinsic
Functional reparative capacities of SSCs need permissive niche environment, which is frequently compromised in chronic lung diseases. Progress of lung tissue fibrosis and airway remodeling are accompanied with activation of mesenchymal stromal cells and dysregulated lung endothelial cells, which are incapable of producing enough growth factors and cytokines (HGF, FGF7. . .) to facilitate distal airway regeneration and repairment102–104. Zepp et al. 54 identified two MC subpopulations in mouse lung tissue: one is mesenchymal alveolar niche cells (MANCs), which secrete FGF7 to promote AT2s regeneration and differentiation upon lung injury; the other is Axin2+ myofibrogenic progenitors (AMPs), which tend to proliferate uncontrollably to form fibrotic scars. Lee et al. 102 reported that decreased expression of the thrombospondin 1 (TSP1) by lung endothelial cells suppresses distal airway regeneration.
Therapeutic Strategies for Distal Airway Repair in Chronic Lung Diseases
Development of targeted therapeutics for distal airway regeneration and repairment in chronic lung diseases has been very challenging, and most of the attempts are preliminary and pre-clinical. The general rationale is to rejuvenate SSCs stemness or/and restore the reparative niche environments (Tables 2 and 3, Fig. 5).
Stem Cell Therapy in Preclinical Trials.

AT2s: alveolar type II cells; BLM: bleomycin; iPSCs: induced pluripotent stem cells; MSCs: mesenchymal stem cells; ESCs: embryonic stem cells.
Stem Cell Therapy in Clinical Trials.

COPD: chronic obstructive pulmonary disease; MSCs: mesenchymal stem cells; IPF: idiopathic pulmonary fibrosis; LSCs: lung stem cells.

Overview of current therapeutic strategies for distal airway repair in chronic lung diseases. SSCs: somatic stem cells; DQ: dasatinib and quercetin; AAV: adeno-associated virus; iPSCs: induced pluripotent stem cells; hESCs: human embryonic stem cells; MSCs: mesenchymal stem cells; LSCs: lung stem cells.
Therapeutic Interventions on SSCs
Antisenescence
SSCs in the distal airways of chronic lung diseases often express aging-related indicators89,95,96,105, making anti-senescence strategy an attractive option to rescue SSCs stemness. Delivery of telomerase by adeno-associated virus (AAVs) vectors in AT2s increased telomere length and restored regenerative capacity of AT2s in mice with pulmonary fibrosis 106 . Senolytic drugs such as dasatinib and quercetin have been shown to delay the progression of pulmonary fibrosis in aged mice and reduce the aging of alveolar epithelial cells and fibroblasts105,107. The first published clinical trial of Senolytic therapy (dasatinib plus quercetin, DQ) included 14 patients with IPF to evaluate feasibility of implementing a senolytic intervention (ClinicalTrials.gov ID: NCT02874989). DQ therapy had significantly improved the physical functions of the patients, (including 6-min walk distance, 4-m gait speed and chair-stands time). Based on these preliminary data, researchers are planning to carry out larger clinical trials of DQ therapy.
Growth factor applications
We could also induce the distal airway SSCs stemness by supplementing the essential stem cell growth factors and cytokines, whose availabilities are greatly reduced in chronic lung diseases. Exogeneous FGF7 (also known as keratinocyte growth factor) application could promote compensatory lung growth by alveolar proliferation post PNX in mice 108 . A randomized controlled phase-I clinic trial showed that intravenous application of FGF7 attenuated alveolar injury in patients with acute respiratory disease syndrome (ARDS) 109 . However, FGF7 application did not efficiently ameliorate any of the disease manifestations of ARDS in a phase-II clinical trial 110 .
Human populations with haplo-insufficiencies for FGF10 have higher incidence to develop COPD, 111 and FGF10 expression is suppressed in mesenchymal stromal cells from patients with IPF 103 . Rodent studies have proven the therapeutic values of FGF10 in pulmonary epithelial regeneration in various lung disease models, such as bleomycin-induced alveolar injuries 112 , naphthalene injury, 113 and high-altitude pulmonary edema 114 . Well-designed clinical trials are needed to demonstrate the curative effects of FGF10 on distal airway regeneration in patients with chronic lung diseases.
Therapeutics Interventions on SSCs Microenvironment
As progress of tissue fibrosis limits the supportive functions of mesenchymal stromal cells to SSCs in distal airways, antifibrotic agents could be instrumental to distal airway epithelium repair. Quercetin induces apoptosis of senescent fibroblasts in aged mice with pulmonary fibrosis 107 . Another study used a combination of quercetin and dasatinib (a tyrosine kinase inhibitor, widely known for its role in the treatment of myeloproliferative syndrome) to treat age-related morbidity in mice. The quercetin-dasatinib therapy significantly suppressed lung tissue fibrosis and aging of alveolar epithelial cells in aged mice 105 . Navitoclax, a specific inhibitor of anti-apoptotic BCL-2 and BCL-XL, can selectively kill senescent cells in vitro. Navitoclax treatment effectively eliminated radiation-induced senescent fibroblasts and inhibited senescence-associated secretory phenotype (SASP) in mice 115 .
Growth factors, such as HGF and FGF1/2/7/10, can inhibit lung tissue fibrosis by reducing collagen accumulation in mice with pulmonary fibrosis112,116–119.
All-trans retinoid acid (ATRA) is known as a key regulator of both embryonic lung development and postnatal alveolarization, promoting alveolar septum and lung growth 120 . In vitro, ATRA stimulates human pulmonary angiogenesis and induces lung fibroblast elastin synthesis 121 . Preclinical studies using in vivo animal models of emphysema have demonstrated the incredible ability of ATRA to improve the state of emphysema and repair the alveolar structure of emphysema mice 122 . There was another trial showing that ATRA treatment of elastase-induced emphysema (Box 1) mice increased the number of reparative bone marrow-derived cells in the alveoli 123 . Moreover, a recent study also supports some novel retinoid X receptor (RXRs) agonists as potential therapeutic strategies for the treatment of porcine pancreatic elastase and cigarette smoke extract (CSE)-induced emphysema 124 .
Stem Cell Therapies
Stem cell transplantation has been proposed as an alternative cell therapy for IPF and COPD125–127. Preclinical studies have shown that induced pluripotent stem cells (iPSCs), human embryonic stem cells (hESCs), mesenchymal stem cells (MSCs), or lung stem cells (LSCs) could be used to treat COPD and IPF127–129. Transplantation of AT2s into bleomycin-induced IPF mice can reduce collagen deposition, restore pulmonary surfactant protein production, and alleviate the severity of pulmonary fibrosis 125 . Similarly, lung injury was abrogated in bleomycin-induced IPF mice after transplantation of hESCs-derived AT2 cells 128 . IPSCs transplantation into IPF mice could significantly improve the pulmonary functions 130 . Furthermore, IPSCs transplantation bypasses possible ethical concerns and has a broader application prospect in comparison with ESCs. In a cigarette-smoke-stimulated COPD model, intrapulmonary injection of MSCs can alleviate airway inflammation and emphysema by down-regulating COX-2/PGE2 in alveolar macrophages 131 .
Challenges and Perspectives
The human respiratory system is anatomically different in details from that of mouse. The human lung has two left lobes and three right lobes, while the mouse lung has one left lobe and four right lobes. In mice, the conducting airway directly transits to the alveoli at the junction of the bronchial alveolar duct, while in humans and other mammals such as ferrets, the proximal airway gradually transitions to the farthest alveolar space through RB and alveolar tubules 5 . The pseudostratified columnar airway epithelium is present in the trachea and mainstem bronchus in mice, while it even extends to the distal bronchioles in human 132 .
The composition and proportion of SSCs within the distal airway epithelium varies between humans and mice. Most of the epithelial in the trachea are ciliated cells in human, but are nonciliated cells in mice 133 . There are more goblet cells and fewer club cells in human airways than in mice. The newly discovered RAS cells within human-specific RB are absent in mice 5 . The distal BADJ region in the mouse lung contains BASCs population, whose presence has not been determined in human lung 32 . Since most of our studies and knowledge on lung homeostasis and regeneration are derived from mouse lungs, the differences between human and mouse lung tissues have limited our understanding of molecular and cellular heterogeneity in human lungs and hindered the investigations of pathogenesis of human chronic lung diseases and the development of related targeted therapeutics. We need to find and use other animal models, such as ferrets, whose lung structures and SSCs are closely related to human lungs 5 .
We still know little about the RB, which are particularly vulnerable to damages that are associated with chronic lung diseases. Distal airways are the target region of early injuries during COPD pathogenesis, and RB are the main obstruction sites of COPD91,134, reminding us of the importance to unveil the pathogenesis of distal airways during early disease stages.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Professor Pixin Ran from Guangzhou National Laboratory and Professor Jiekai Chen from Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences for the constructive comments to the manuscript.
Author Contributions
JW and HX conceived and wrote the manuscript. GP drew the graphics.
Ethical Approval
This study was approved by our Institutional Review Board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (grant no. 32070918), Distinguished Young Scholars in Guangdong Province (grant no. 2022B1515020109), Start-up funding for the Pediatric Research Institute of Guangzhou Women and Children’s Medical Center (grant no. 3001082).
