Abstract
Human cord blood–endothelial progenitor cells (hCB-EPCs) isolated from the human umbilical cord can be used to repair damaged arteries. In this study, we used an animal model with pathological changes that mimics artery wall damage caused by stent retrievers in humans. We injected hCB-EPCs to investigate their effect on endothelial hyperplasia and dysfunction during intimal repair. Four groups were established based on the length of reperfusion (3 and 28 days), as well as the presence or absence of hCB-EPC therapy. Damage to the internal carotid artery was evaluated by hematoxylin-eosin and immunohistochemical staining. Stroke volume was not significantly different between non-EPC and EPC groups although EPC treatment alleviated intimal hyperplasia 28 days after intimal damage. Vascular endothelial growth factor (VEGF) and eNOS expression were significantly higher in the EPC-treated group than in the non-EPC group 3 days after intimal damage. In addition, MMP9 and 4HNE expression in the EPC-treated group was significantly lower than in the non-EPC group. Ultimately, this study found that venous transplantation of hCB-EPCs could inhibit neointimal hyperplasia, alleviate endothelial dysfunction, suppress intimal inflammation, and reduce oxidative stress during healing of intimal damage.
Keywords
Introduction
Ischemic stroke (IS) is a leading global cause of disability and death 1 . In recent years, mechanical thrombectomy (MT) has become established as the therapy of choice for large-vessel occlusion (LVO). However, it can also cause serious arterial damage, vascular inflammation, and lipid deposition, contributing to neointimal hyperplasia and vessel wall remodeling 2 .
The vascular intima consists primarily of a single layer of endothelial cells (ECs). EC regrowth occurs after intima damage, but it is insufficient to prevent platelet adhesion and smooth muscle cell proliferation, causing intimal thickening and decreased blood flow 3 . Endothelial progenitor cells (EPCs) are circulating cells that express many cell surface markers such as CD31, CD34, and CD105 4 and can reduce intimal hyperplasia via a paracrine mechanism. The concentration of EPCs circulating in peripheral blood is quite low. Human cord blood–endothelial progenitor cells (hCB-EPCs) isolated from the human umbilical cord can also be used to repair damaged arteries. They are more common, accessible, and have a higher proliferative capacity than EPCs from other sources 5 , and are more easily transplanted intravenously 6 . Human umbilical vein endothelial cells (HUVECs) are isolated from the vein of the umbilical cord and have similar traits as EPCs, such as vascular healing, but their capacity to proliferate in vitro is limited7,8. Therefore, hCB-EPCs may be an alternative therapy option for treating vascular endothelial damage.
To test this possibility, in this study, we used an animal model with pathologic changes that mimics artery wall damage caused by stent retrievers in humans 9 . hCB-EPCs were injected into the tail vein to investigate their effect on endothelial hyperplasia and dysfunction during intimal repair.
Materials and Methods
Cell Culture
hCB-EPCs for cell culture were purchased from StemBioSys® Inc. (EPC-500-T75, San Antonio, CA, USA). The cells were grown in Iscove’s Modified Dulbecco’s Medium (IMDM) supplemented with 20% fetal bovine serum (FBS), 1% antibiotic/antimycotic, 1%
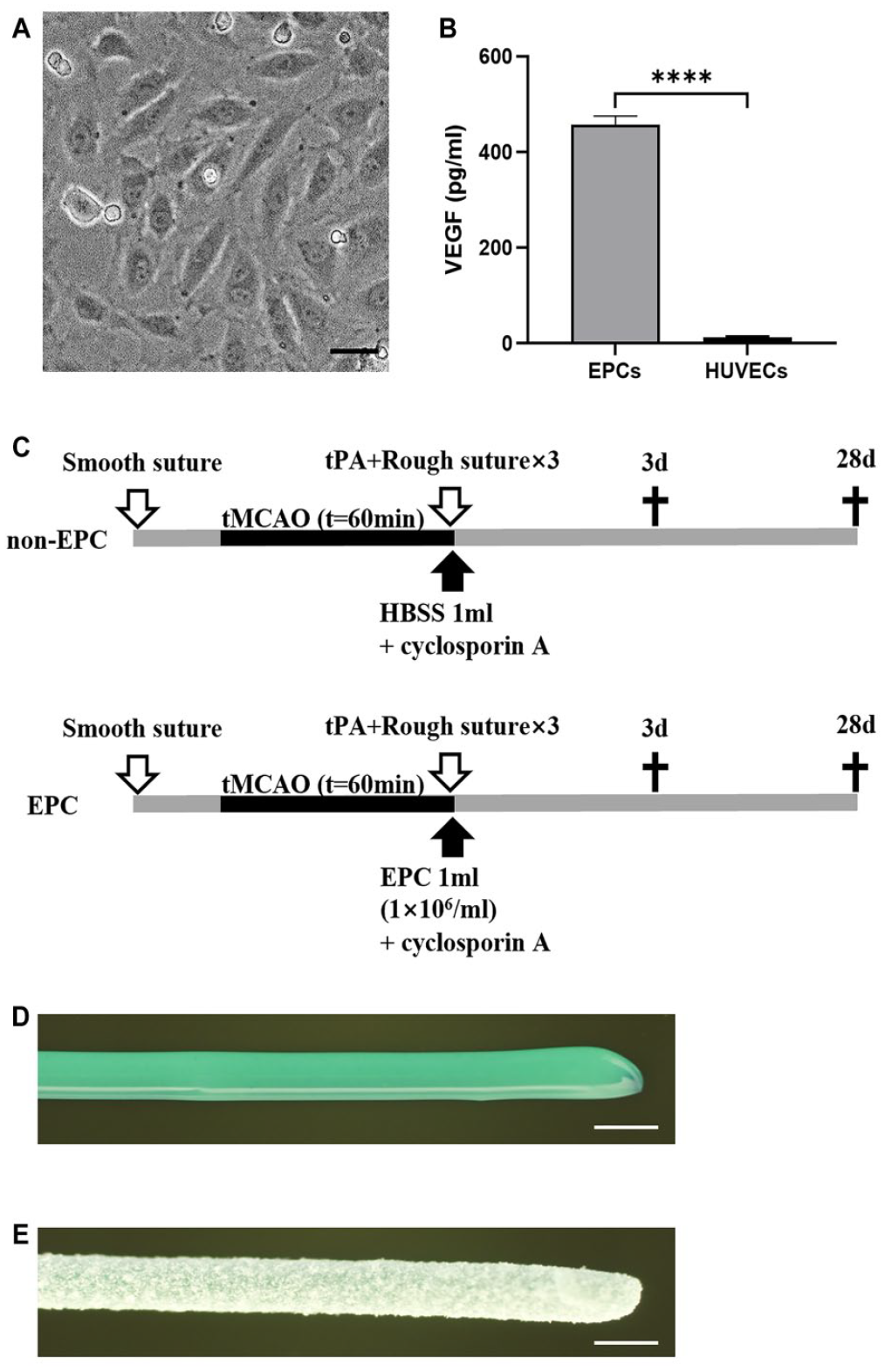
(A) The majority of hCB-EPCs became spindle-shaped on the third day of culture. Scale bar = 400 μm. (B) HUVECs and EPCs were incubated with equal quantities of fresh medium for 72 h before the medium was centrifuged. The release of VEGF was measured in the medium of hCB-EPCs and HUVECs by ELISA. n = 3, P < 0.0001. (C) Experimental groups included the non-EPC group (1 ml HBSS) and the EPC groups (1.0 × 106 EPC). White arrows indicate the insertion of smooth and rough sutures. Black arrows indicate tail vein injection of HBSS or EPCs. (D) A smooth suture was utilized to occlude the middle cerebral artery. (E) A rough suture with a fused alumina layer was used to moderately damage the artery intima. Scale bar = 500 μm. ELISA: enzyme-linked immunosorbent assay; HBSS: Hank’s balanced salt solution; hCB-EPC: human cord blood–endothelial progenitor cell; HUVECs: human umbilical vein endothelial cells; VEGF: vascular endothelial growth factor.
HUVECs were purchased from LONZA (C2517A, Kanagawa, Japan) and cultured in endothelial cell growth medium 2 (EGM-2; CC-3162, LONZA) according to the manufacturer's instructions. 2,500 cells/cm2 were implanted for passage. HUVECs were also cultured to the third generation and the cell supernatant was used for ELISA.
Analysis of hCB-EPC Paracrine Function
The paracrine function of hCB-EPCs was evaluated by assaying vascular endothelial growth factor (VEGF) concentration in the supernatant of cultured cells. On day 4 of the culture of hCB-EPCs and HUVECs, medium was changed to Dulbecco’s Modified Eagle’s Medium (DMEM) without growth factors but containing 10% FBS, and cultured continuously for 72 h. After centrifuging collected medium at 1000 × g for 10 min, the supernatant was used to detect VEGF by ELISA (DVE00, R&D Systems) according to the manufacturer’s instructions.
Animals and Groups
All animal experiments were run following protocols authorized by the Animal Committee of the Graduate School of Medicine and Dentistry, Okayama University (OKU#2021407) and the Okayama University Guidelines for the Care and Use of Laboratory Animals. Male Wistar rats (11 weeks old, body weight 250–330 g; SLC, Shizuoka, Japan) were acclimatized for 2 weeks in standard rat cages under conventional laboratory conditions with a 12/12 h light–dark cycle, constant humidity, and room temperature. The animals were fed rat pellets (MF, Oriental Yeast, Tokyo, Japan), and water was provided ad libitum. Each week, cages were cleaned and lined with paper pulp bedding (Oriental Yeast). Male rats were used because estrogen has been shown to improve post-ischemic stroke neurogenesis and behavioral recovery in female animals10,11. Four groups were established based on the reperfusion duration of 3 and 28 days, as well as the presence or absence of hCB-EPC therapy: non-EPC 3 group (n = 8), EPC 3 group (n = 6), non-EPC 28 group (n = 9), and EPC 28 group (n = 6) (Fig. 1C).
Focal Cerebral Ischemia, Carotid Artery Balloon Injury Model
A mixture of nitrous oxide, oxygen, and isoflurane (69%:30%:1%) was delivered to all rats via an inhalation mask to anesthetize them. The right carotid bifurcation was revealed, and the external carotid artery was ligated proximal to the bifurcation. Next, a smooth suture (silicone-coated 4-0 nylon thread with a diameter of 380 µm and a smooth surface; Fig. 1D) was introduced through the stump of the external cerebral artery and gently advanced for middle cerebral artery (MCA) occlusion. We delivered tPA (intravenously, 3 mg/kg, Grtpa, Mitsubishi Tanabe Pharma Corporation, Osaka, Japan) after 1 h of occlusion. To restore blood flow within the MCA region, the nylon embolus was removed. A specific rough suture, which was similarly inserted at the same position as the smooth suture, was promptly withdrawn, and the same procedure was repeated three times. This rough suture was a silicone-coated 4-0 nylon thread (Cat: C-23s-N3-5040s, Natume) with a 380 µm diameter, 2 cm length, and a surface fully coated with rough white fused alumina (20 µm in diameter; Naniwa Abrasive Manufacturing, Osaka, Japan) 9 . The suture was checked under a light microscope (Fig. 1E). The incision was then closed. As indicated previously 12 , hCB-EPCs (1.0 × 106) in 1 ml of Hank’s balanced salt solution (HBSS, pH = 7.4) or 1 ml of HBSS was injected intravenously into the tail vein. Intraperitoneal injections of the immunosuppressive agent cyclosporin A (10 mg/kg/day, Novartis International, Basel, Switzerland) were administered every other day. At 3 and 28 days after receiving an injection of hCB-EPCs, all animals were sacrificed. To determine group sizes necessary for adequate statistical power, power analysis was performed by G*Power 3.1 using a provisional dataset of rat carotid intima-media ratios. The data for the EPC group (0.49 ± 0.19) and the non-EPC group (1.19 ± 0.5) were calculated using a two-sided two-sample t test with a significance level (alpha) of 0.05. The analysis revealed that a sample size of six in each group was necessary to achieve 80% power.
Neurobehavioral Analysis
In each group, the body weight of rats was assessed daily after tMCAO. The neurological tests that were administered utilized Bederson's score. Rats were examined on days 0, 1, 2, and 3 for non-EPC 3 and EPC 3 groups, and on days 0, 1, 2, 3, 4, 5, 6, 7, 14, and 28 for non-EPC 28 and EPC 28 groups following tMCAO. The Bederson score with modest revisions13,14 was evaluated as follows: (1) unable to extend the left forepaw, (2) rotating to the contralateral side, (3) falling to the left, and (4) inability to move spontaneously.
Tissue Preparation
After 3 and 28 days after reperfusion, deep anesthesia was administered by allowing rats to inhale isoflurane through a mask and perfusing cold phosphate-buffered saline (PBS) and paraformaldehyde in 0.1 mol/l phosphate buffer (pH = 7.4) through the heart. The whole brain and a portion of the internal carotid artery were removed and submerged for 12 h at 4°C in the same fixative solution. After being washed in PBS, tissues were placed into 10%, 20%, and 30% (w/v) sucrose solutions, embedded in dry ice, and frozen at −80°C. Tissues were sliced (15 µm thick) with a cryostat at −24°C, plated on silicon-coated glass slides, and kept at −80°C.
Histology and Immunohistochemistry
Brain sections were cut at 2, 0, −2, −4, and −6 mm from the bregma, and infarct volume was calculated by multiplying the infarct area by a thickness of 2 mm 15 . Brain sections were immunostained with mouse monoclonal antibody against mouse microtubule-associated protein 2 (MAP-2, 1:500, Sigma, St. Louis, MO, USA) and examined under a light microscope (SZX-12; Olympus Optical, Tokyo, Japan). The loss of staining infarct areas was measured in sections using Photoshop (version 2021). Each brain section’s infarct volume was calculated by integrating the coronally oriented infarct area. When calculating cerebral infarct volume, the influence of cerebral edema was excluded using correction formulas. First, percentage cerebral edema was calculated as: ([volume of the infarcted hemisphere / volume of the normal hemisphere] / [volume of the normal hemisphere]) × 100. Corrected infarct volume was calculated as: (actual measurement infarct volume) × (100 / [100 + % cerebral edema]). Relative infarct volume (% I) was calculated as 100 × (corrected infarct volume / volume of normal hemisphere × 2) 16 .
Cross sections of the internal carotid artery (ICA) were cut at 2 to 3 mm distal to the bifurcation, and hematoxylin-eosin (H&E) staining was performed to evaluate ICA vessel wall structural damage and examined with a light microscope (Olympus BX-51; Olympus Optical). Photoshop was used to measure the thickness of intima and media at six randomly selected locations in the cross-section of arterial vessels. The intima/media (I/M) ratio was calculated, and the average was used for data analysis and comparisons among groups.
For immunohistochemistry, the following primary antibodies were utilized: rabbit anti-VEGF antibody (1:200; bs-0279R, Bioss Antibodies, Woburn, MA, USA), rabbit anti-endothelial nitric-oxide synthase (eNOS) antibody (1:200; bs-0136R, Bioss Antibodies), rabbit anti-matrix metalloproteinase-9 (MMP9) antibody (1:200; ab19016, Millipore, Billerica, MA, USA), and mouse anti-4-HNE antibody (1:200, MHN-020P, JaICA, Shizuoka, Japan) The sections were then treated with a biotin-labeled secondary antibody (1:500, Vector Laboratories Inc., CA, USA), followed by an ABC kit (PK-6101, Vector Laboratories Inc.) and 3,3-diaminobenzidine (DAB). Negative control sections were also stained using the same procedure but without the primary antibody.
Statistical Analyses
All statistical analyses were conducted with GraphPad Prism (version 9.2). All data are expressed as the mean ± standard deviation (SD). The analysis of the result of VEGF level in hCB-EPCs and HUVECs groups was performed using a t test (Fig. 1B). The Log-rank (Mantel-Cox) test was used to analyze the probability of survival (Fig. 2A, D). Two-way repeated-measures analysis of variance (ANOVA) followed by Tukey’s post hoc test was used to analyze weight (Fig. 2B, E) and Bederson scores (Fig. 2C, F). Data of the nonnormal distribution and unequal variance parameters of the infarct volume (Fig. 3B), H&E staining (Fig. 4C), VEGF (Fig. 5B), eNOS (Fig. 5D), MMP9 (Fig. 6B), and 4HNE (Fig. 6D) were compared using the Kruskal–Wallis test and Bonferroni test for post hoc comparisons. Results were considered statistically significant at P < 0.05.
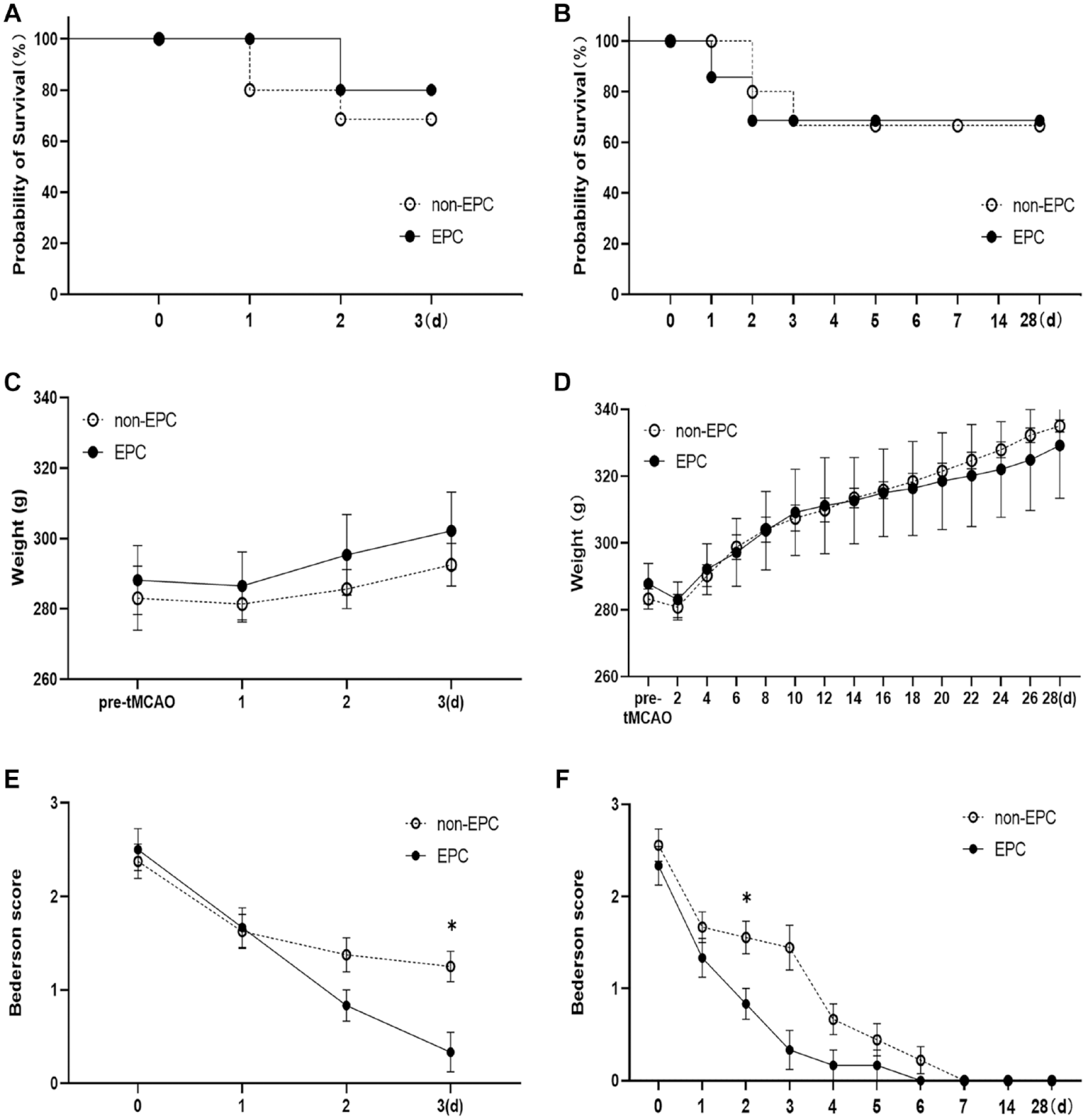
Neurobehavioral analysis of rats after transient middle cerebral artery occlusion (tMCAO). (A) After 3 days, the survival rates of the sacrificed group after tMCAO in the non-EPC 3 and EPC 3 groups were 72.7% and 85.7%, respectively. There was no significant differences between both groups. (B) There were no significant differences in body weight between the EPC 3 and non-EPC 3 groups. (C) The Bederson score of the EPC 3 group was significantly lower than that of the non-EPC 3 group on the third day (*P = 0.0307, vs non-EPC 3 group). (D) After 28 days, the survival rates of the sacrificed group after tMCAO in the non-EPC 28 and EPC 28 groups were 75 and 75%, respectively. (E) There was no variation in body weight between the two groups throughout the experimental period. (F) EPC groups showed significantly milder neurological deficits in Bederson scores on the third day (*P = 0.0460, vs non-EPC 28 group). EPC: endothelial progenitor cell.

Infarct volume of rats after transient middle cerebral artery occlusion (tMCAO). (A) MAP-2 staining of rat brain sections of 3 and 28 days groups after tMCAO. Scale bar = 2 mm. (B) Quantitative analysis of MAP-2 staining. There were substantial variations in cerebral infarction volume between the EPC 3 and EPC 28 groups (**P = 0.006, vs EPC 3 ), as well as between the non-EPC 3 and non-EPC 28 groups (# P = 0.0495, vs non-EPC 3 ), but there was no significant difference between the EPC and non-EPC groups at 3 days or 28 days. Values are means ± SD. EPC: endothelial progenitor cell; MAP-2: microtubule-associated protein 2.
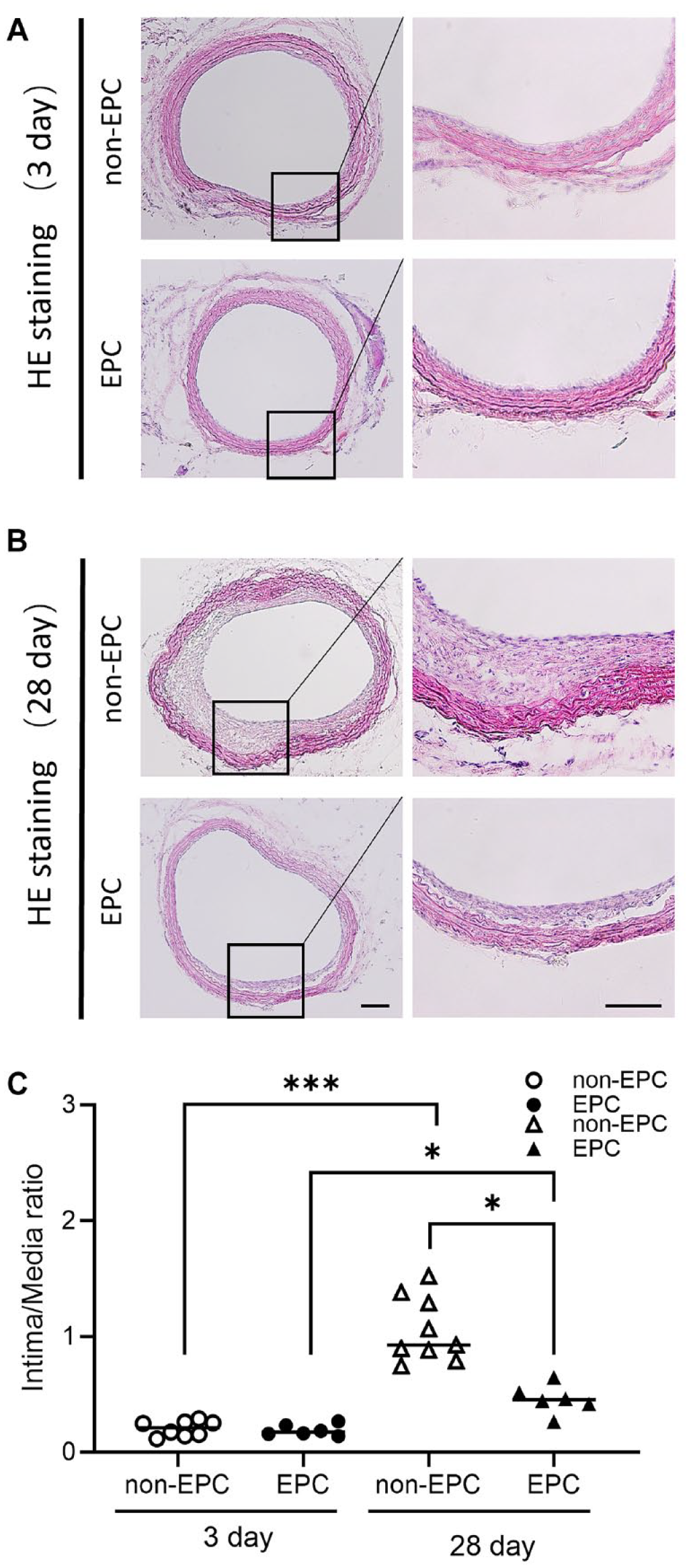
H&E staining of common carotid artery (CCA) cross sections to evaluate neointimal hyperplasia. (A) There was no significant difference in neointimal thickness between the EPC 3 and non-EPC 3 groups at 3 days. (b) Neointimal thickness of the EPC 28 group was significantly less than that of the non-EPC 28 group at 28 days. Scale bar = 10 μm. (c) The non-EPC 28 group displayed a marked increase in intimal thickness to media thickness ratio (I/M) than the EPC 28 group (*P = 0.032, vs EPC 28 ). A comparison of the I/M ratio between the EPC 3 and EPC 28 groups ($ P = 0.0272, vs EPC 3 ), as well as between the non-EPC 3 and non-EPC 28 groups, showed significant differences (### P = 0.0005, vs non-EPC 3 ). Values are means ± SD. EPC: endothelial progenitor cell.

Representative immunohistochemical photomicrographs of carotid artery sections with (A) VEGF and (C) eNOS staining of each group. Scale bar = 10 µm. (B) The VEGF signal was significantly stronger in the EPC 3 group than in the non-EPC 3 group (ϕP = 0.0326, vs non-EPC 3 ). VEGF expression differed significantly between the EPC 3 and EPC 28 groups (**P = 0.0059, vs EPC 3 ) and between the non-EPC 3 and non-EPC 28 groups (# P = 0.0320, vs non-EPC 3 ). (D) eNOS signal intensity was significantly higher in the EPC 3 group than in the non-EPC 3 group (ϕP = 0.0260, vs non-EPC 3 ). eNOS expression did differ significantly between the non-EPC 28 and EPC 28 groups ($$P = 0.003, vs non-EPC 28 ). eNOS expression was also significantly different between the EPC 3 and EPC 28 groups (*P = 0.0488, vs EPC 3 ). Values are means ± SD. eNOS: endothelial nitric-oxide synthase; EPC: endothelial progenitor cell; VEGF: vascular endothelial growth factor.
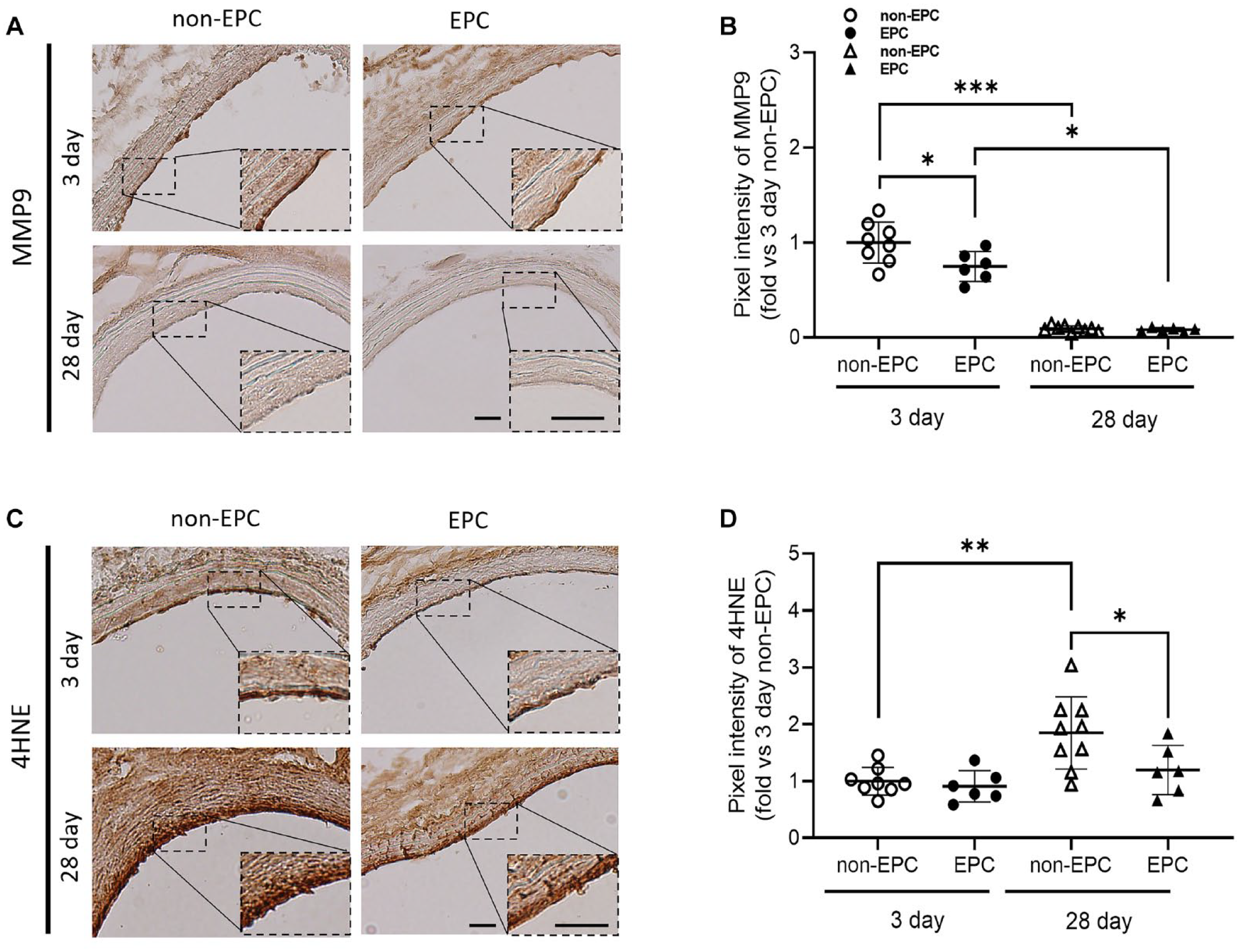
Representative immunohistochemical photomicrographs of carotid artery sections with (A) MMP9 and (C) 4HNE staining of each group. Scale bar = 10 µm. (B) The signal intensity of MMP9 in the EPC 3 group was significantly lower than that in the non-EPC 3 group (ϕ P = 0.0403, vs non-EPC 3 ). Comparisons of MMP9 signal intensity between the EPC 3 and EPC 28 groups (*P = 0.0398, vs EPC 3 ) and the non-EPC 3 and non-EPC 28 groups (### P = 0.0007, vs non-EPC 3 ) were statistically significant. (D) 4HNE expression level of the non-EPC 28 group was considerably higher than that of the EPC 28 group ($ P = 0.0473, vs non-EPC 28 ). There was a significant difference in 4HNE signal intensity between the non-EPC 3 and non-EPC 28 groups (## P = 0.0030, vs non-EPC 3 ). Values are means ± SD. EPC: endothelial progenitor cell.
Results
HCB-EPC-Mediated Release of VEGF In Vitro
We measured the concentration of VEGF in the supernatants of hCB-EPCs and HUVECs by ELISA. As show in Fig. 1B, the release of VEGF increased significantly in the supernatant of the hCB-EPC cell culture (457.56 ± 17.57 pg/ml, n = 3) relative to that of HUVECs (13.34 ± 1.28 pg/ml, n = 3, P < 0.0001).
Motor Function and Infarct Volume
The survival rate of rats sacrificed at 3 days after reperfusion in the non-EPC 3 and EPC 3 groups was 72.7% (n = 8) and 85.7% (n = 6), respectively (Fig. 2A). Over a 3-day period, there was no significant difference in body weight between the two groups (Fig. 2B) although the Bederson score between them was significantly different on the third day (P = 0.0307, two-way repeated-measures ANOVA; Fig. 2C).
The survival rates of rats sacrificed 28 days after reperfusion were 75% (n = 9) in the non-EPC 28 group and 75% (n = 6) in the EPC 28 group (Fig. 2D). There was no difference in body weight between the two groups throughout the 28-day period (Fig. 2E). On the third day, the two groups had significantly different Bederson scores (P = 0.0460, two-way repeated-measures ANOVA; Fig. 2F).
Infarct volume was evaluated by anti-MAP2 staining (Fig. 3A). There were significant differences in the amount of cerebral infarction between the EPC 3 (25.35 ± 4.78, n = 6) and EPC 28 groups (8.17 ± 1.43, n = 6, P = 0.006, vs EPC 28 , Kruskal–Wallis test), as well as between the non-EPC 3 (27.43 ± 5.26, n = 8) and non-EPC 28 groups (11.68 ± 3.22, n = 9, P = 0.0495, vs non-EPC 28 , Fig. 3B). Differences between EPC and non-EPC groups at 3 or 28 days were not significant.
Effect of hCB-EPC Transplantation on Neointimal Hyperplasia of Carotid Artery
A portion of common carotid artery (CCA) cross sections 2- to 3-mm distal to the bifurcation was used. After washing in PBS, tissues were placed into 10%, 20%, and 30% (wt/vol) sucrose solutions, allowing the tissue to be sufficiently dehydrated, embedded in dry ice, and frozen at −80°C. Slices were made with a cryostat at −24°C, and these were placed on silicon-coated glass slides that were kept at −80°C. CCA cross sections stained with H&E were then analyzed. After 3 days of ICA tunica intima damage, the tunica medium structure seemed to be intact (Fig. 4A), and the I/M ratio in the non-EPC 3 group (0.20 ± 0.07, n = 8) suggested a thicker tunica (nonsignificant finding) than that in the EPC 3 group (0.19 ± 0.05, n = 6, P = 0.8991). After 28 days of ICA intima damage, the arterial intima of the non-EPC 28 group was thickened with stenotic lumen (Fig. 4B). The non-EPC 28 group (1.05 ± 0.28, n = 9) had a significantly higher I/M ratio than the EPC 28 group (0.46 ± 0.13, n = 6, P = 0.032, vs EPC 28 ). The difference in I/M ratio between EPC 3 and EPC 28 groups (P = 0.0272, vs EPC 28 ), as well as between non-EPC 3 and non-EPC 28 groups (P = 0.0005, vs non-EPC 28 ), was insignificant (Fig. 4C).
Effect of hCB-EPCs on the Repair of the Intima
Since VEGF is the predominant factor released by EPCs, we decided to assess the paracrine action of hCB-EPCs by examining the expression of VEGF in the damaged intima (Fig. 5A). The VEGF signal was significantly stronger in the EPC 3 group 3 days after ICA intimal damage compared with the non-EPC 3 group (P = 0.0326, vs EPC 3 ). However, there was no significant change in VEGF expression between the EPC 28 and non-EPC 28 groups 28 days after ICA intimal damage (P = 0.9134). Expression of VEGF differed significantly between the EPC 3 and EPC 28 groups (P = 0.0059, vs EPC 28 ) and between the non-EPC 3 and non-EPC 28 groups (P = 0.0320, vs non-EPC 28 , Fig. 5B).
eNOS is a crucial regulator of endothelial cell proliferation, migration, and survival17. Thus, after ICA intimal damage, we evaluated the effect of EPC on endothelial cells through the expression of eNOS during intimal repair (Fig. 5C). On the third day, the signal intensity of eNOS was significantly higher in the hCB-EPCs (1 × 106)-treated group than in the untreated group (P = 0.0488, vs EPC 3 ). After 28 days, the expression of eNOS was not significantly different between the EPC 28 and non-EPC 28 groups (P = 0.4166, vs EPC 28 ). eNOS expression was significantly different between EPC 3 and EPC 28 groups (P = 0.0260, vs EPC 28 ) but not between non-EPC 3 and non-EPC 28 groups (P = 0.0723, Fig. 5D).
A substantial number of neutrophils are attracted to the damaged intimal surface following ICA intimal injury 17 . To demonstrate the anti-inflammatory action of EPCs during the repair of arterial intima, we evaluated the expression of MMP9 on the arterial surface in a set of experiments (Fig. 6A). The findings of MMP9 staining revealed that MMP9 signal intensity in the EPC 3 group was significantly lower than that in the non-EPC 3 group (P = 0.0403, vs EPC 3 ), but there was no significant difference between the EPC 28 and non-EPC 28 groups (P = 0.9881). MMP9 signal intensity was significantly different between the EPC 3 and EPC 28 groups (P = 0.0398, vs EPC 28 ) and between the non-EPC 3 and non-EPC 28 groups (P = 0.0007, vs non-EPC 28 ) (Fig. 6B).
The production of lipid peroxidation products at a site of injury is a common phenomenon after mechanical damage to the internal carotid artery intima 18 . Using 4HNE, variations in the expression of peroxidation stress products in each group following artery intima damage were observed (Fig. 6C). Three days after ICA intimal damage, there was no significant difference in the expression of 4HNE between the EPC 3 and non-EPC 3 groups (P = 0.9126). At 28 days after intimal damage, the level of 4HNE expression of the non-EPC 28 group was significantly higher than that of the EPC 28 group (P = 0.0473, vs EPC 28 ). The 4HNE signal intensity between the non-EPC 3 and non-EPC 28 groups was significantly different (P = 0.0030, vs non-EPC 28 ), but not between the EPC 3 and EPC 28 groups (P = 0.6826, Fig. 6D).
Discussion
Currently, human cord blood is the primary source of active EPCs 19 . Previous research demonstrated that hCB-EPCs had a beneficial effect on stroke, allowing final neurological function to recover 20 as a result of a decrease of neointimal hyperplasia 21 . Separately, an in vitro study indicated that hCB-EPCs could secrete VEGF (Fig. 1B). In clinical settings, it has been shown that stent retrievers cause certain structural changes in the artery wall, including endothelial denudation and internal elastic lamina fracture 22 . To detect the effect of hCB-EPCs on intimal damage, we used an animal model 9 that can mimic intimal damage after using stent retrievers (Fig. 1E), and found that venous transplantation of hCB-EPCs could inhibit neointimal hyperplasia (Fig. 3) and alleviate endothelial dysfunction (Fig. 5C, D) by secreting VEGF (Fig. 5A, B), suppressing intimal inflammation (Fig. 6A, B), and reducing oxidative stress (Fig. 6C, D) during the process of intimal damage healing.
In this study, we demonstrated that hCB-EPCs stimulated VEGF production (Fig. 5A, B) and inhibited intimal hyperplasia (Fig. 4). Various kinds of EPCs, including hCB-EPCs, repair arterial intima by activating resident endothelial cells to release paracrine factors such as VEGF or hepatocyte growth factor (HGF) 23 rather than by differentiation into new endothelial cells 24 . In the damaged regions, VEGF stimulates endothelial cell proliferation and promotes intimal healing 25 while the proliferation of exposed vascular smooth muscle cells decreased 26 .
A previous study reported that VEGF could stimulate eNOS activity of ECs in a PL3K-dependent manner via Akt phosphorylation 27 . Therefore, the enhanced expression of eNOS in ECs in the EPC 3 group after intimal injury (Fig. 5C, D) may be related to an increased in the expression of VEGF. eNOS stimulates an increase in nitric oxide (NO) activity 28 , and NO inhibits the generation of reactive oxygen species (ROS) that accelerates neutrophil migration 29 . MMP9 is mainly secreted by neutrophils and is involved in the regulation of inflammation 30 . In this study, we found that the expression of MMP9 at sites of intimal injury was reduced in the EPC3 group compared with the non-EPC 3 group (Fig. 6A, B). This finding suggests that hCB-EPCs restricted the migration of inflammatory cells to the injured arterial wall. This study demonstrates that hCB-EPCs reduced 4HNE expression in ECs of the carotid artery (Fig. 6C, D), indicating that lipid peroxidation was reduced. Amplified oxidative stress induces an increase in lipid peroxidation 31 , which contributes to arterial restenosis 32 .
This study has several limitations. According to previous research, transplanted fluorescently-labeled EPCs were observed to adhere to damaged arteries 12 . However, hCB-EPCs were not detected on ICA sections in this investigation, thus the participation of hCB-EPCs in intima repair was only shown indirectly by four markers, namely, VEGF, eNOS, MMP9, and 4HNE. There are several possible reasons why the hCB-EPCs were not detected on damaged arteries. First, there may be some challenges in recognizing hCB-EPCs in sections of the rat’s internal carotid artery by staining with an anti-human mitochondrial antibody. Second, human-derived hCB-EPCs might be rejected by the rat’s immune system because cyclosporine was injected every other day, and not every day. Third, hCB-EPCs may have moved to ischemic areas or to other organs. In the future, we will employ fluorescein labeling or molecular probes into cells to trace the migration of implanted hCB-EPCs more effectively. In addition, according to the behavioral data, the model's behavioral abnormalities diminished chronically, as the rats returned to near-normal activities at 28 days after tMCAO, which is another limitation of this study.
In conclusion, transplantation of hCB-EPCs can suppress neointimal hyperplasia and prevent restenosis after intimal injury, so it may be a potential option for the treatment of cerebrovascular diseases after endovascular thrombectomy.
Footnotes
Ethical Approval
This study was approved by the Animal Committee of the Graduate School of Medicine and Dentistry, Okayama University (OKU#2021407).
Statement of Human and Animal Rights
All animal experiments were run following protocols authorized by the Okayama University Guidelines for the Care and Use of Laboratory Animals (OKU#2021407).
Statement of Informed Consent
There were no human subjects in this study. Hence, informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by a Grant-in-Aid for Scientific Research (B) 17H0419611, (C) 20K09370, 17H0975609, and 17K1082709 and by Grants-in-Aid from the Research Committees (Kaji R, Toba K, and Tsuji S) from the Japan Agency for Medical Research and Development 7211700121, 7211800049, and 7211800130.
