Abstract
Periventricular-intraventricular hemorrhage (PIVH) is common in extremely low gestational age neonates (ELGAN) and leads to motor and behavioral impairments. Currently there is no effective treatment for PIVH. Whether human nonhematopoietic umbilical cord blood-derived stem cell (nh-UCBSC) administration reduces the severity of brain injury and improves long-term motor and behavioral function was tested in an ELGAN-equivalent neonatal rat model of PIVH. In a collagenase-induced unilateral PIVH on postnatal day (P) 2 model, rat pups received a single dose of nh-UCBSCs at a dose of 1 × 106 cells i.p. on P6 (PIVH + UCBSC group) or were left untreated (Untreated PIVH group). Motor deficit was determined using forelimb placement, edge-push, and elevated body swing tests at 2 months (N = 5–8). Behavior was evaluated using open field exploration and rearing tests at 4 months (N =10–12). Cavity volume and hemispheric volume loss on the PIVH side were determined at 7 months (N = 6–7). Outcomes were compared between the Untreated PIVH and PIVH + UCBSC groups and a Control group. Unilateral motor deficits were present in 60%–100% of rats in the Untreated PIVH group and 12.5% rats in the PIVH + UCBSC group (P = 0.02). Untreated PIVH group exhibited a higher number of quadrant crossings in open field exploration, indicating low emotionality and poor habituation, and had a cavitary lesion and hemispheric volume loss on the PIVH side. Performance in open field exploration correlated with cavity volume (r2 = 0.25; P < 0.05). Compared with the Untreated PIVH group, performance in open field exploration was better (P = 0.0025) and hemispheric volume loss was lower (19.9 ± 4.4% vs 6.1 ± 2.6%, P = 0.018) in the PIVH + UCBSC group. These results suggest that a single dose of nh-UCBSCs administered in the subacute period after PIVH reduces the severity of injury and improves neurodevelopment in neonatal rats.
Keywords
Introduction
Preterm infants are at risk for periventricular-intraventricular hemorrhage (PIVH) with an incidence of 25%–30%1,2. Extremely low gestational age neonates (ELGAN; <28 weeks of gestation at birth) are especially vulnerable2,3. PIVH begins as hemorrhage in the germinal matrix, a fragile vascular region between the striatum and thalamus, with extension of hemorrhage into the adjacent ventricle and brain parenchyma1,4. Brain parenchymal hemorrhage occurs in 10%–15% of PIVH cases and is typically unilateral5,6. PIVH is a major cause of death and disability in ELGAN. Cerebral palsy, blindness, deafness, and cognitive deficits are seen in 50%–75% of the survivors3,7. Approximately 25% of nondisabled survivors develop psychiatric disorders and executive function deficits8,9. Gray and white matter injury, inflammation, posthemorrhagic ventriculomegaly, and arrest of brain development are responsible for these adverse outcomes 6 . Currently, PIVH cannot be prevented, and treatment is limited to supportive care.
Umbilical cord blood (UCB)-derived mononuclear cell (MNC) administration as treatment for PIVH has a strong appeal. UCB is a rich source of stem cells, immunoregulatory cells, and neurotrophic factors10,11. A recent meta-analysis of preclinical studies evaluating the effects of human UCB-derived stem cells (UCBSCs) on perinatal brain injury demonstrated that UCBSC administration decreases the severity of brain injury, astrocytosis, microglial activation, and neuroinflammation; increases the number of neuron and oligodendroglia; and improves motor function 12 . Subgroup analysis showed that neuroprotection with UCBSCs was greater for PIVH, compared with neonatal hypoxic-ischemic (HI) injury 13 . Previous studies of PIVH in neonatal animal models have demonstrated that administration of human UCBSCs reduces neuronal apoptosis, microgliosis, and astrocytosis; attenuates ventriculomegaly; stabilizes the blood brain barrier; restores thalamocortical plasticity; and improves myelination and short-term motor and behavioral deficits14–22. The long-term effects on brain injury and functional outcomes were not evaluated in these studies.
The objective of this study was to determine the effects of human nonhematopoietic UCBSC (nh-UCBSC) administration in the subacute period (4 days) after PIVH on long-term survival, motor function, behavior, and brain structure at adulthood in an ELGAN-equivalent neonatal rat model of PIVH. These unique CD34-negative cells derived from the MNC fraction of human UCB express stem cell transcription factors, Oct-4, Rex-1, and Sox-2 23 . Immunophenotype using flow cytometry demonstrates that these cells are positive for CD10, CD13, CD44, CD49, CD90, and major histocompatibility complex (MHC) class I and negative for CD34, CD45, and MHC class II 23 . The cells retain neuroprotective efficacy even after several population duplications 24 . Our previous studies in adult rats have demonstrated that administration of nh-UCBSCs decreases the severity of brain injury and improves functional outcome in hemorrhagic and ischemic stroke23–26.
Materials and Methods
Animals
Experiments were performed using male and female Sprague Dawley rat pups. Pregnant dams were purchased (Charles River Laboratory, Wilmington, MA, USA) and allowed to deliver spontaneously. Pups were weaned on postnatal day (P) 21. Dams and pups were maintained under standard laboratory conditions under a 12h:12h light:dark cycle (lights out at 1800) and allowed to feed and drink ad lib. All experimental procedures were approved by the Institutional Animal Care and Use Committee. Timeline of the experiments is given in Fig. 1.

Experimental timeline. Collagenase-induced PIVH was induced on P2. Rats with PIVH received either nh-UCBSCs at a dose of 1 × 106 cells in PBS or PBS intraperitoneally on P6. Motor function was tested at 2 months and behavioral function was tested at 4 months. The brain was harvested at 7 months for histochemical assessment of brain injury. P: postnatal day; PIVH: periventricular-intraventricular hemorrhage; UCBSCs: umbilical cord blood-derived stem cells; PBS: phosphate-buffered saline.
Induction of PIVH
Unilateral PIVH was induced on P2 using the neonatal collagenase-induced PIVH model with minor modifications27–29. Rat brain development on P2 is comparable to that of a human ELGAN 30 . Collagenase injection into the subventricular ganglionic eminence causes vascular leak and local hemorrhage that extends into the adjacent ventricle and periventricular brain parenchyma, thus modeling severe PIVH in human ELGAN27–29. Using hypothermia for sedation and the eye-to-ear line as landmark 27 , type VII-S collagenase was injected at a dose of 0.75 U in 2-µl normal saline into the right ganglionic eminence (approximately 2 mm rostral to the right ear and 2 mm deep from the surface) using a micro-syringe with 31-gauge needle. The average surgical time was 20 min. Pups were returned to the dam after recovery. A group of littermates without PIVH served as the Control group. To avoid a litter of origin effect, pups were randomized to all the three (Control, Untreated PIVH, and PIVH + UCBSC) groups in each litter.
Preparation and Administration of nh-UCBSCs
Cell preparation
CD34-negative human nh-UCBSCs were isolated and grown as in our previous publications23–26. Briefly, UCB was collected after delivery, and within 4 h, MNCs were separated by centrifugation at 500 × g for 30 min in a Ficoll-hypaque density gradient (1.077 g/cm3) (Sigma, St. Louis, MO, USA). The cells were then grown in Dulbecco’s modified Eagle medium DMEM (Invitrogen, Waltham, MA, USA)/MCDB-201(Sigma) mix with 10% fetal bovine serum (Invitrogen), 10−4 M of
nh-UCBSC administration
Pups subjected to PIVH were randomized to the PIVH + UCBSC group or Untreated PIVH group. Pups randomized to the PIVH + UCBSC group received a single dose of nh-UCBSCs at a dose of 1 × 106 cells in 0.2-ml PBS intraperitoneally (i.p.) 4 days after the induction of PIVH (ie, on P6). Pups randomized to the Untreated PIVH group received an equivalent volume of PBS i.p. Pups were returned to their dams after the injection.
Outcome Assessment
Motor and behavioral assessments were tested at adulthood in a blinded manner. Motor and behavioral assessments were performed in separate cohorts of animals to avoid the confounding effects of multiple tests on the outcomes.
Motor Assessment
Unilateral motor deficit was assessed at 2 months of age by testing for forelimb placement, edge-push, and elevated body swing tests as in our previous study 25 . The tests are briefly described below:
Forelimb placement test
Forelimb placement test was performed by holding the rat in midair by its tail and slowly lowering it toward the benchtop. Normal rats extend both forelimbs as their body approaches the surface. Rats with unilateral brain injury are unable to extend the forelimb on the side contralateral to the injured side. The test was repeated three times, and the average performance of each rat was determined.
Edge-push test
In this test, the rat was placed horizontally on the benchtop and pushed nose first toward the edge. Normal rats resist by placing both forelimbs under their body to prevent falling over the edge. Rats with unilateral brain injury are unable to use the forelimb contralateral to the injured side, which drops over the edge. The test was repeated three times, and the average performance of each rat was determined.
Elevated body swing test
This test determines bias in side-to-side lateral flexion of the trunk. The rat was suspended vertically by its tail above the benchtop with its nose pointing downward. Normal rats reach upward by truncal flexion to the left and right sides in equal frequencies without bias toward one side. Rats with unilateral brain injury demonstrate bias to the side ipsilateral to the injured side. The test was repeated three times, and the average of each rat was determined.
Behavioral Assessment
Behavior was assessed using the open field exploration and rearing tests at 4 months of age. These tests measure exploratory locomotor behavior and general activity in rodents 31 . Both tests were performed using a cylindrical chamber measuring 39.5 cm in diameter and 34 cm in height and consisting of four quadrants drawn on the floor. The number of quadrants crossed by a rat during a 15-min open field exploration (quadrant crossings) was recorded. The number of times a rat stood on its hindlimbs against the wall of the chambers (rearing) during this period was also recorded. Both tests were performed on three consecutive days. Higher quadrant crossing and rearing scores indicate low emotionality and poor habituation due to impaired information processing32–34, and correlate with the size of the cavitary lesion at the site of parenchymal brain injury 35 .
Histochemical Assessment
Severity of brain injury was determined using histochemistry at 7 months of age. Rats were deeply anesthetized using isoflurane. The brain was removed and processed for histochemistry. A cavitary lesion due to cystic changes at the site of parenchymal hemorrhage (porencephaly) and enlargement of the adjacent ventricle is a hallmark of severe PIVH in human ELGAN and neonatal animal models, and is a risk factor for poor neurodevelopment1,6,14,15,19,36. Therefore, cavity volume at the site of PIVH and hemispheric volume loss on the ipsilateral side were determined. Consecutive coronal brain sections (50 µm) were collected using a cryostat. Brain sections were stained with cresyl violet acetate solution to identify Nissl substance in the cytoplasm of neurons 37 . Every sixth brain section was visualized under a light microscope (Leica DM6 B; Leica Microsystems Inc, Deerfield, IL, USA) and images were captured at 1.25× magnification. The boundaries of the cavitary lesion and both cerebral hemispheres were manually outlined, and the surface area was determined using Leica Application Suite X software (LAS X; Leica Microsystems Inc). The volume was calculated using the following formula: sum of all surface areas measured × 0.05 mm (section thickness) × 6 (number of sections between each stained section). As the cavitary lesion extended to the non-PIVH hemisphere (ie, left side) in some rats in the Untreated PIVH group, the ratio of total cavity volume (ie, sum of the cavity volumes on the right and left sides) to the total brain volume (sum of the right and left hemisphere volumes) was also determined. Hemispheric volume loss on the PIVH side was determined using the following formula: (volume of the contralateral hemisphere − volume of the ipsilateral hemisphere)/volume of the contralateral hemisphere × 100.
Statistical Analysis
Data were analyzed using GraphPad Prism 9 software (graphpad.com). Continuous variables were compared using analysis of variance (ANOVA). Intergroup differences were determined using Tukey’s multiple comparisons tests. Categorical variables were compared using chi-square or Fisher’s exact test as applicable. The relationship between performance in a behavioral test and the cavity volume was determined using Pearson’s R. Data are presented as mean ± SD. Statistical significance was set at P < 0.05.
Results
Survival
Survival to adulthood was 100% (10/10) in the Control group, 70.6% (12/17) in the Untreated PIVH group, and 86.4% (19/22) in the PIVH + UCBSC group. The intergroup differences were not statistically significant [χ2(2, N = 49) = 4.33, P = 0.11]. Body weight at adulthood did not differ among the three groups in either females or males (F = 1.85, P = 0.21 for females and F = 0.85, P = 0.45 for males; Supplemental Table 1).
Motor Outcome
There was an overall effect of group on forelimb placement [χ2(2, N = 21) = 7.54, P = 0.02], edge-push [χ2(2, N = 21) = 16.71, P = 0.0002], and elevated body swing [χ2(2, N = 21) = 16.71, P = 0.0002] tests (Table 1). Unilateral deficits in forelimb placement (60%), edge-push (100%), and elevated body swing (100%) tests on the side contralateral to the PIVH side were present in the Untreated PIVH group (N = 5) (all Ps < 0.005 vs Control group; Table 1). In contrast, only one out of eight (12.5%) rats in the PIVH + UCBSC group (N = 8) demonstrated deficits in the three motor tests (Table 1). The difference between Untreated PIVH and the PIVH + UCBSC groups was statistically significant for edge-push and elevated body swing tests (both Ps < 0.02). None of the rats in the Control group (N = 8) exhibited motor deficits (Table 1).
Motor Deficits in the Control, Untreated PIVH, and PIVH + UCBSC Groups.

Values are number and percent of animals in a group with motor deficits. There is an effect of group for forelimb placement [χ2(2, N = 21) = 7.54, P = 0.02], edge-push [χ2(2, N = 21) = 16.71, P = 0.0002], and elevated body swing [χ2(2, N = 21) = 16.71, P = 0.0002] tests. *P < 0.005 versus Control group; **P < 0.02 versus Untreated PIVH group (Fisher’s exact test). There are no differences between Control and PIVH + UCBSC groups for any of the three tests. PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell.
Behavioral Outcome
Untreated PIVH group (N = 9) exhibited higher number of quadrant crossings in the open field exploration test, compared with the Control group (N = 11) (P = 0.006; Fig. 2A). Trend for a similar effect was present for the rearing test (P = 0.07; Fig. 2B). Compared with the Untreated PIVH group, the PIVH + UCBSC group (N = 10) exhibited fewer quadrant crossings in the open field exploration test (P = 0.0025; Fig. 2A). Performance of the PIVH + UCBSC group in both behavioral tests was comparable to the Control group.

Performance of rats in the open field exploration (A) and rearing (B) tests. Values are mean ± SD number of open field quadrant crossings (A) or rearing events (B) during a 15 min of open field exploration on three consecutive days. N = 11, Control group; N = 9, Untreated PIVH group; and N = 10, PIVH + UCBSC group. PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell. *P < 0.05 versus Control and PIVH + UCBSC groups.
Histochemical Outcome
Hemispheric atrophy on the PIVH side was evident on gross examination in 75% of rats in the Untreated PIVH group and 21% of rats in the PIVH + UCBSC group [χ2(2, N = 28) = 8.02, P = 0.018]. Histochemical examination demonstrated a cavitary lesion due to brain tissue loss (porencephaly) at the site of PIVH and enlargement of the adjacent ventricle (Fig. 3).

Cavitary lesion and hemispheric volume loss at adulthood. Coronal brain sections (50 µm) from representative rats from the Control group (A), Untreated PIVH group (B), and PIVH + UCBSC group (C) stained with cresyl violet for Nissl substance are shown. Arrows in B and C point to the cavitary lesion at the site of PIVH; bar = 1 mm. PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell.
There was an effect of group on cavity volume (ANOVA, P = 0.017; Table 2). Compared with the Control group (N = 7), cavity volume was larger in the Untreated PIVH group (N = 6) (Table 2). Cavity volume was smaller in the PIVH + UCBSC group (N = 7), relative to the Untreated PIVH group (67.17 ± 74.13 mm3 vs 18.22 ± 42.85 mm3). However, the difference was not statistically significant. There was a correlation between the cavitary volume and performance in the open field exploration test (r2 = 0.25; P < 0.05; Fig. 4A) but not in the rearing test.
Cavity Volume in the Control, Untreated PIVH, and PIVH + UCBSC Groups.
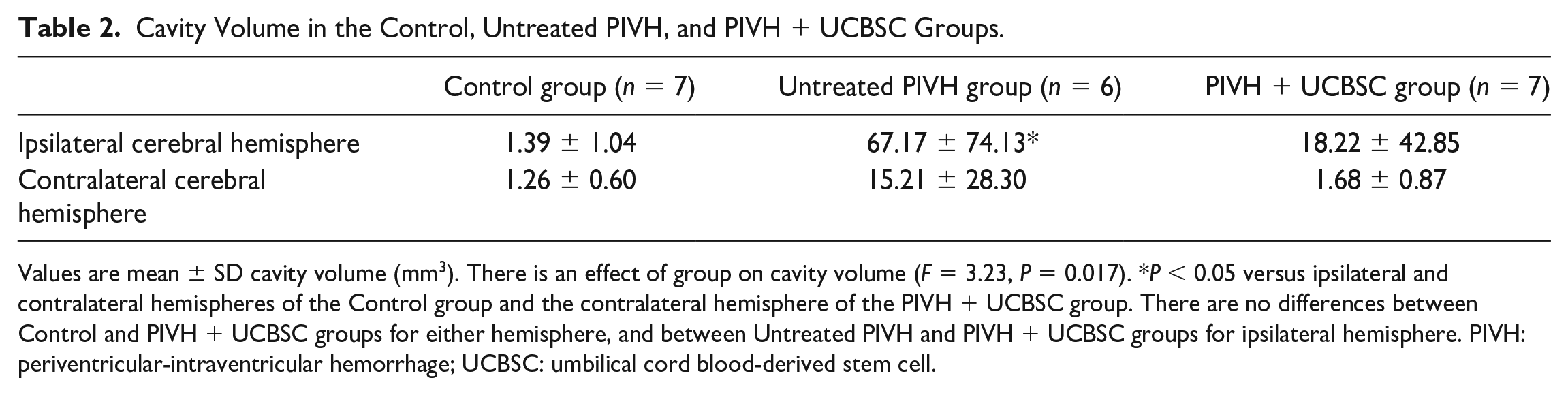
Values are mean ± SD cavity volume (mm3). There is an effect of group on cavity volume (F = 3.23, P = 0.017). *P < 0.05 versus ipsilateral and contralateral hemispheres of the Control group and the contralateral hemisphere of the PIVH + UCBSC group. There are no differences between Control and PIVH + UCBSC groups for either hemisphere, and between Untreated PIVH and PIVH + UCBSC groups for ipsilateral hemisphere. PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell.

Relationship between cavity volume (A) and cavity to hemispheric ratio (B) and performance in the open field exploration test. Values are number of quadrant crossings during a 15 min of open field exploration; N = 6, Control group and N = 5, Untreated PIVH and PIVH + UCBSC groups. r2 = 0.25; P < 0.05 in A and r2 = 0.26; P < 0.05 in B (Pearson’s R). PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell.
As cavitary lesion extended to the contralateral side (ie, non-PIVH side) in some rats in the Untreated PIVH group, we also determined the ratio between total cavity volume and total brain volume. There was an effect of group on total cavity volume/total brain volume ratio (ANOVA, P = 0.03; Fig. 5A). The ratio was higher in the Untreated PIVH group, relative to the Control group (10.5 ± 10.0 vs 0.30 ± 0.14, P = 0.03; Fig. 5A). The ratio was smaller (2.7 ± 6.0) in the PIVH + UCBSC group (Fig. 5A). However, the difference between Untreated PIVH and PIVH + UCBSC groups was not statistically different. As with cavity volume, the total cavity volume/total brain volume ratio correlated with performance in the open field exploration test (r2 = 0.26; P < 0.05; Fig. 4B), but not in the rearing test.

Total cavity volume to total brain volume ratio (A) and hemispheric volume loss on the PIVH side relative to the non-PIVH hemisphere (B) at adulthood. Values are mean ± SD; N = 7, Control and PIVH + UCBSC groups and N = 6, Untreated PIVH group. There is an overall effect of group on both (P < 0.05). *P < 0.05 versus Control group and **P < 0.05 versus Untreated PIVH group. PIVH: periventricular-intraventricular hemorrhage; UCBSC: umbilical cord blood-derived stem cell.
There was an effect of group on hemispheric volume loss (ANOVA, P = 0.01; Fig. 5B). Hemispheric volume loss on the PIVH side was present in both Untreated PIVH and PIVH + UCBSC groups (both P < 0.02; Fig. 5B). However, compared with the Untreated PIVH group, hemispheric volume loss was lower in the PIVH + UCBSC group (19.9 ± 10.7% vs 6.1 ± 6.9%, P = 0.018; Fig. 5B).
Discussion
Advances in neonatal care over the past two decades have led to a steady improvement in the survival of ELGAN without a parallel decrease in the incidence of severe PIVH4,38. There has also been no significant progress in treatment for the condition, which is limited to supportive care and surgical drainage of posthemorrhagic hydrocephalus. This study demonstrates that a single dose of nh-UCBSCs administered 4 days after PIVH reduces the severity of brain injury and improves motor and behavioral outcomes at adulthood in an ELGAN-equivalent neonatal rat model of PIVH.
A recent meta-analysis of 55 eligible studies evaluating the effects of human UCBSCs in preclinical models of perinatal brain injury reported that UCBSC administration decreases white matter apoptosis, astrocytosis, microglial activation, and neuroinflammation; increases neuronal and oligodendroglia numbers; and improves motor function 12 . Subgroup analysis showed that the beneficial effects of UCBSCs on white matter apoptosis and neuroinflammation were greater for PIVH than for HI injury 13 . Overall, neuroprotection with UCBSCs was greater with administration 24 to 72 h postinjury, compared with administration within 24 h of injury 13 .
Short-term beneficial effects with human UCBSCs in neonatal animal models of PIVH have been reported in previous studies4,14–17,19–22. Majority of these studies used UCB-derived mesenchymal stem cells (MSCs). In a neonatal rat model of intraventricular hemorrhage induced by injecting maternal blood into the ventricle on P4, intracerebroventricular (ICV) administration of UCB-derived MSCs at a dose of 1 × 105 or intravenous (IV) administration at a dose of 5 × 105 two days after injury reduced neuronal apoptosis, microgliosis, and astrocytosis; attenuated ventriculomegaly; stabilized blood brain barrier; restored thalamocortical plasticity; and improved myelination and motor and behavioral deficits at 32 days14–18. Outcomes were better with early UCBSC administration (2 vs 7 days) 19 . In a preterm rabbit model of intraventricular hemorrhage induced by i.p. administration of glycerol 3–4 h after birth, either ICV or IV administration of UCB-derived nonhematopoietic multipotent unrestricted somatic stem cells at a dose of 1 × 106 cells decreased cell death and microglial infiltration, restored aquaporin expression toward baseline, reduced ventriculomegaly, and improved locomotion and myelination at 14 days21,22. Suppression of proinflammatory cytokines, immunomodulation, and provision of neurotrophic factors, and not engraftment of the administered UCBSCs, were considered responsible for the beneficial effects in these studies14–17,19–22.
This study differs from the previous studies in several aspects. We induced PIVH on P2, which is neurodevelopmentally comparable to human ELGAN 39 , the population at highest risk for PIVH 3 . The collagenase-induced PIVH model in this study closely models the pathogenesis and neuropathology of PIVH in human ELGAN 5 . Unlike UCBSC administration in the acute phase of injury employed in the previous studies14–17,19–22, we administrated nh-UCBSCs in the subacute period (4 days post-PIVH). Demonstrating the effectiveness of UCBSC administration in the subacute period has translational relevance as it suggests a wider therapeutic window. Many human ELGAN with PIVH are born at facilities that lack resources for cell therapies and have to be transferred to a higher level neonatal intensive care unit for treatment. Unlike the invasive ICV administration employed in some of the previous studies15–17,19, we administered nh-UCBSCs systemically (i.p.), further supporting translational relevance. Finally, we evaluated the long-term benefits at adulthood. The duration of assessment was limited to 14–32 days in previous studies14,15,17.
The beneficial effect of nh-UCBSCs on motor outcome in this study is similar to the effect we have previously reported in adult rat models of hemorrhagic and ischemic stroke23–26. Compared with the previous study in adult rats 25 , improvement in motor function was better in this study. This could be due to the greater neuroplasticity of the developing brain 40 or a reflection of progressive motor improvement following nh-UCBSC administration. In our previous study in adult rats, performance in forelimb placement and elevated body swing tests were better at 13 days after nh-UCBSC administration, compared with the performance at 6 days 25 . Progressive improvement over time could explain the better motor outcome at 2 months in this study. However, this possibility remains conjectural as we did not test motor function at earlier time points.
PIVH is associated with disorganized and poorly modulated behavioral regulation and habituation in human ELGAN and animal models41,42. Consistent with this, rats in the Untreated PIVH group demonstrated increased number of quadrant crossings and trend for a similar effect in rearing, indicating low emotionality and dishabituation due to impaired information processing32,33. Similar neurobehavioral deficits have been demonstrated in other neonatal brain injury models (eg, unilateral HI injury on P7)34,35,43. Compared with the Untreated PIVH group, rats in the PIVH + UCBSC group demonstrated fewer quadrant crossings, suggesting beneficial effects of nh-UCBSCs on this behavior. Other studies have also demonstrated improvement in hyperactivity with UCBSC administration in adult rats with stroke44,45.
Cavity size at the site of PIVH correlates with worse habituation in human ELGAN 42 . Consistent with this observation, cavity volume correlated with exploratory activity (quadrant crossings) in this study. A similar relationship between cavity size and increased exploratory activity in open field exploration at adulthood is reported in neonatal rats with unilateral HI injury 35 . Although the cavity volume was smaller in the PIVH + UCBSC group, relative to the Untreated PIVH group, the difference was not statistically significant, likely due to the wide range of values. However, compared with the Untreated PIVH group, hemispheric volume loss on the PIVH side was lower in the PIVH + UCBSC group, suggesting better brain tissue preservation, which might have led to improved behavioral performance.
We administered nh-UCBSCs intraperitoneally in this study. In a recent meta-analysis of 55 preclinical studies assessing the effects of UCBSCs in perinatal brain injury, 27% of the studies used the intraperitoneal route 13 . A beneficial effect with intraperitoneal administration of human UCBSCs has been reported by others46,47. While some studies have demonstrated the presence of transplanted cells in the brain after intraperitoneal administration48,49, other studies using molecular methods were unable to find evidence of transplanted cells in the brain or any other organ 50 . As with other routes of administration, an immunomodulatory or anti-inflammatory effect is likely responsible for neuroprotection with intraperitoneal UCBSC administration 46 . All UCB cell types administered intraperitoneally produce an immunomodulatory effect 47 . Intraperitoneal UCBSC administration is associated with increased number of mature oligodendrocytes, and decreased immune cell infiltration, reactive astrocytosis, and microglial activation at the site of injury47–49,51,52. Neuroprotection with intraperitoneal UCBSC administration was comparable to that with IV administration in a neonatal rat model of HI injury 49 . Subgroup analysis of the above-mentioned meta-analysis found that intraperitoneal UCBSC administration was inferior to intraventricular/intrathecal (local) administration for decreasing astrogliosis in the white matter, while an opposite effect favoring intraperitoneal administration over local administration was present for motor improvement 13 . There are some concerns that intraperitoneal administration of high doses of human UCBSCs could be associated with systemic inflammation and worsen excitotoxic white matter injury 50 . Therefore, the intraperitoneal route may not be suitable for high-dose UCBSC administration.
This study has limitations that must be acknowledged. We did not investigate the mechanism of neuroprotection with nh-UCBSCs. Our previous studies in adult rats demonstrated immune modulation as the mechanism of neuroprotection with nh-UCBSCs24–26. In those studies, nh-UCBSC administration reduced macrophage infiltration and the number of activated microglia and T cells at the site of injury, and normalized the gene transcripts of cell death; blood brain barrier and extracellular matrix function; cytokine and chemokine production; and M2 macrophages, CD4+ T cells, and regulatory T cells in the brain24,26. Similar mechanisms may be involved in this study but require further exploration. We did not test for the presence of nh-UCBSCs in the brain. In our previous study in adult rats, relatively few of nh-UCBSCs were present in the brain 14 days after administration 25 . Prior studies in neonatal animal models of IVH also have demonstrated that UCBSCs are rarely seen after 14–32 days even when administered via the ICV route15,21. Additional limitations are not testing the effects of sex, dose escalation, other routes of administration, and administration at other time points after PIVH, all of which are known to influence efficacy 13 .
In summary, administration of a single dose of nh-UCBSCs in the subacute period after PIVH improved motor and behavioral function and decreased the severity of brain injury at adulthood in an ELGAN-equivalent neonatal rat model. A recent Cochrane review found no randomized controlled trials on benefits or harms of stem cell–based therapies for prevention or treatment of PIVH in preterm infants 53 . A phase I dose escalation trial of UCB-derived MSCs administered via the ICV route in ELGAN (N = 9) with severe PIVH was not associated with increased mortality or serious adverse effects 54 . Neurodevelopmental assessment of these infants at 2 years of age is ongoing (NCT02673788). A phase IIa trial of these cells versus normal saline in 23–34 week gestation preterm infants (N = 22) with severe PIHV is currently active (NCT02890953). There are other ongoing clinical trials evaluating the efficacy and safety of UCBSCs for preterm brain injury, but not specifically for PIVH 53 . The results of our study provide the proof-of-concept for considering nh-UCBSCs in human infants with PIVH. However, several additional studies are necessary prior to clinical application. A thorough understanding of the mechanism of neuroprotection and additional data on safety, optimal dose, timing, and route of administration from age-appropriate large animal models and phase I trials in ELGAN are needed before nh-UCBSCs can be considered for treatment of PIVH in human preterm infants.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231189301 – Supplemental material for Nonhematopoietic Umbilical Cord Blood Stem Cell Administration Improves Long-term Neurodevelopment After Periventricular-Intraventricular Hemorrhage in Neonatal Rats
Supplemental material, sj-docx-1-cll-10.1177_09636897231189301 for Nonhematopoietic Umbilical Cord Blood Stem Cell Administration Improves Long-term Neurodevelopment After Periventricular-Intraventricular Hemorrhage in Neonatal Rats by Raghavendra B. Rao, Maple L. Shiao, Kathleen M. Ennis-Czerniak and Walter C. Low in Cell Transplantation
Footnotes
Acknowledgements
The authors acknowledge the assistance of Derek Chen and Alexandra Burt with the experiments.
Author Contributions
RBR and WCL developed the concept and designed the study; MLS and KME-C performed the experiments; KME-C and RBR analyzed the data; RBR wrote the manuscript; MLS, KME-C, and WCL edited the manuscript; and RBR takes complete responsibility for data integrity and the accuracy of data analysis.
Availability of Data and Materials
All data required to evaluate the conclusions in the study are present in the article and/or the Supplemental material. Additional data related to this article may be available to researchers upon request.
Ethical Approval
The study was approved by the Institutional Animal Care and Use Committee (Protocol number: 2112-39658A).
Statement of Human and Animal Rights
All the experimental procedures involving animals were conformed to the guidelines of the National Institutes of Health.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from Regenerative Medicine Minnesota (Grant# RMM 101617 DS 005); a Minnesota State Legislature allocated funding for research, commercialization, and clinical translation initiatives that improve or increase access to scientifically proven regenerative medicine throughout the state. The funding agency had no influence on study design, data interpretation, and manuscript preparation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
