Abstract
The immense neuroinflammation induced by multiple sclerosis (MS) promotes a favorable environment for ischemic stroke (IS) development, making IS a deadly complication of MS. The overlapping inflammation in MS and IS is a prelude to the vascular pathology, and an inherent cell death mechanism that exacerbates neurovascular unit (NVU) impairment in the disease progression. Despite this consequence, no therapies focus on reducing IS incidence in patients with MS. To this end, the preclinical and clinical evidence we review here argues for cell-based regenerative medicine that will augment the NVU dysfunction and inflammation to ameliorate IS risk.
Introduction
Ischemic stroke (IS), the most common cause of death and long-term disability worldwide 1 , and multiple sclerosis (MS), the number one neurodegenerative disease in young adults 2 , are two debilitating disorders with an unexpected relationship. MS is commonly described as a chronic autoimmune disorder targeting the myelin axon sheaths in the central nervous system (CNS). In contrast, IS involves acute cell death following nutrient and oxygen deprivation 3 . Despite the variety in onset, the common symptoms of neurological dysfunction seen in IS and MS can be explained by comparable demyelination, axonal injury, vascular impairment, neurodegeneration, glial cell activation, and immune cell proliferation. Furthermore, both MS and IS induce neurovascular unit (NVU) permeability due to inflammatory cytokines inducing changes within the NVU components 4 . NVU permeability permits further immune cell infiltration and even greater subsequent neuroinflammation. Considering a similar inflammatory pathophysiology, it is understandable that MS has been correlated to increased risk of IS2,5. In fact, MS has been shown to increase the risk of IS by 6.09× 2 .
Another devastating link between these pathologies is the lack of a cure. While many pharmaceuticals have been developed for MS, they only stabilize or delay symptom onset 6 . Considering the significant neuroinflammation of MS contributing to IS, a prominent goal for future treatment development should target and rapidly reverse this inflammation. The established anti-inflammatory effects 7 of stem cells (SCs) and recent SC advancements for treatment of IS and MS prompt discussion on the use of early SCs to lessen the risk of stroke in patients with MS. Furthermore, SCs are shown to restore NVU permeability, and thus may offer even greater therapeutic benefit in this respect. In this review, we will explore the underlying neuroinflammation in IS and MS, the SC advancements for both pathologies, and consider the early administration of anti-inflammatory cell-based therapy as a prophylactic measure against MS-induced stroke (Figure 1).

This figure exemplifies the role of MS-induced neuroinflammation in prompting atherosclerosis and IS via pro-inflammatory immune cell and cytokine infiltration. On the left, MS induces inflammatory cytokines, Th17, Th1, and inflammatory microglia proliferation in the circulation. These mediators induce atherosclerosis and eventual ischemic stroke as shown on the right. The middle panel demonstrates how stem cells can reduce this inflammation by increasing Treg and anti-inflammatory microglial proliferation, ultimately reducing stroke and MS damage. Once MS neuroinflammation is downregulated, the relationship between atherosclerosis, stroke, and MS would be theoretically negligible, as described in the middle panel. MS: multiple sclerosis; IS: ischemic stroke.
Neuroinflammation in Multiple Sclerosis
The defining pathological characteristic associated with MS presents as demyelinating lesions formed by the breakdown of the blood–brain barrier (BBB), an aspect of the NVU, allowing for trans-endothelial migration of activated leukocytes to the CNS that trigger demyelination and neuroinflammation 8 . Perhaps most significant to such early pathogenesis of MS is the leakage of fibrin into the CNS, which precedes leukocyte diapedesis but coincides with microglial activation. Other hemostatic elements enhance neuroinflammation by promoting leukocyte extravasation or play a potential neuroprotective role (reviewed in Abbadessa et al. 9 ). Ultimately, later neuroinflammation in MS is characterized by the migration of activated lymphocytes, reactive gliosis, neuronal death, and axonal destruction.
Lymphocytic infiltration is a significant driver of neuroinflammation in MS, and among the T-cell lineage, self-reactive T helper (Th) cells are the primary modulators of inflammation. Th1 cells, the most frequently identified in experimental autoimmune encephalomyelitis (EAE) animal models of MS, are hypothesized to target microglial activation through the pro-inflammatory interleukin (IL)-12/ interferon (IFN-γ) axis 10 . Meanwhile, Th17 cells are involved in MS pathogenesis through promotion of BBB disruption and activation of astrocytes and microglia to amplify neuroinflammation 10 . Another subpopulation of T lymphocytes, T regulatory cells (Tregs) (an anti-inflammatory derivative of T cells) also contribute to MS through certain genetic abnormalities, such as intrinsic Treg defects or the presence of suppressive markers preventing Tregs from downregulating the pro-inflammatory response 10 . Regarding B-cell involvement, intrathecal immunoglobulin is a critical determinant of MS diagnosis and management. Animal models have demonstrated that B cells aid in Th17 (a pro-inflammatory derivative of T cells) differentiation, inhibit development of Tregs, and produce pro-inflammatory markers 11 . While bidirectional B-cell trafficking between the CNS and periphery is poorly understood, B-cell infiltrates are abundant in patients with active MS 12 .
Disruption of the BBB and lymphocytic infiltrates in the CNS activate microglia to induce synaptic pruning and secrete inflammatory mediators that result in an overall neurotoxic effect. Focal MS lesions are driven by microgliosis triggering inflammation, demyelination, neurodegeneration, while microglial nodules are observed in white matter proximal to the plaques promoting axonal degeneration, oligodendrocyte stress, and complement product deposition 13 . Reactive astrogliosis is an additional contributor to persistent neuroinflammation, characterized by the release of pro-inflammatory cytokines and neurotoxic reactive oxygen/nitrogen species to increase BBB permeability, as well as glial scar formation, prevention of axonal regeneration, and inhibition of oligodendrocyte proliferation 14 . Furthermore, remyelination failure is enhanced by extracellular matrix molecule deposition of proteins which impair oligodendrocyte recruitment and enhance inflammation 15 .
Understanding the precise mechanism behind neuroinflammation in MS is of great therapeutic interest. Inflammasomes of innate immunity have also risen to prominence as mediators of such neuroinflammation through EAE models and patients with genetic deficiencies in inflammasome regulatory genes 16 . Mitochondrial function and accumulation of mitochondrial DNA damage are also hypothesized to contribute to neuroinflammation associated with MS (reviewed in Bargiela and Chinnery 17 ). An emerging target underlying neuroinflammation in MS manifests as altered synapses, occurring independent of demyelination and axonal destruction, that contribute to significant excitotoxic damage; stabilization, repair, and regeneration of excitotoxic synaptic abnormalities offers potential to reduce neuroinflammation throughout the stages of disease 18 . Overall, neuroinflammation in MS represents a complex interplay of mediators at both a cellular and molecular level that define and influence the course and morbidity of disease.
Risk of Ischemic Stroke Due to Neuroinflammation in the Setting of Multiple Sclerosis
Many theories explaining the relationship between IS and MS have been hypothesized (reviewed in Hong et al. 2 ). For instance, early childhood obesity increases the risk of not only MS but also atherosclerosis and stroke. The continuous alteration in myelination characteristic of MS is also related to atherosclerosis. Furthermore, lifestyle risks for stroke, such as smoking and sedentary lifestyles, are more common among patients with MS. Glucocorticoids, a standard treatment for MS, are also related to cardiovascular and cerebrovascular diseases. Surprisingly, even a genetic overlap of 24 genes is reported between individuals with MS and individuals with IS 1 .
One of the most pertinent contributors to stroke risk in patients with MS, however, is the chronic inflammatory state 19 . In MS, the classic demyelinated lesions are filled with CD4+ T cells, CD8+ T cells, and plasma cells, demonstrating an active and continuous pro-inflammatory environment (reviewed in Lopes Pinheiero et al. 3 ). In addition, the lesions show macrophages full of myelin, neurodegeneration, oligodendrocyte death and malfunction, and activated microglia and astrocytes. These immune cells not only destroy myelin sheaths with phagocytosis but also release IFN-γ and IL-17, propagating a toxic inflammatory environment. Inflammation and immune cell migration to ischemic regions is similarly observed in stroke, with neutrophils inducing an inflammatory cascade worsened by the later arrival of macrophages and lymphocytes. This invasion into the CNS leads to immense levels of pro-inflammatory cytokines and similar microglial and astrocytic activations 20 . In fact, anti-inflammatory medications used to lessen MS symptoms are also effective to mitigate stroke damage, such as Fingolimod and Natalizumab21–23.
Cytokines such as IL-1β, IL-6, IL-17, and tumor necrosis factor (TNF)-α are common to lesions secondary to both MS and IS 24 . Besides the injury to nervous tissue, chronic inflammation (like in MS) also induces detrimental vessel changes and instigates atherogenesis 25 . Circulating monocytes home to atherosclerotic plaques and differentiate into lipid-laden macrophages 26 . These macrophages then overproduce matrix metalloproteinases to degrade the plaque’s collagen architecture, increasing the risk of plaque rupture and thrombosis. In addition, CD40 ligand stimulates macrophages to express procoagulant tissue factor 26 . Beyond macrophages, platelet aggregation has also been associated with MS neuroinflammation 27 . Altogether, pathological manifestations of MS-induced chronic inflammation may lead to a hypercoagulative environment, possibly resulting in subsequent cerebral ischemia. Interestingly, similar to how Fingolimod and Natalizumab21–23 reduce stroke damage, thrombolytic therapies, such as tissue plasminogen activator, has shown benefit in patients with MS 3 .
A systematic review and meta-analysis incorporating nine studies with 380,000 patients revealed that patients with MS had a significant risk in overall stroke at 1 year and at 10 to 13 years. Of particular interest, there was a 6.09× increased risk of IS in patients with MS compared with those without 2 . Risk factors for IS are more common in patients with MS compared with matched controls; notably hypertension and hyperlipidemia are most commonly reported with prevalence >10% 5 . In all, 27.1% of patients with MS are comorbid with diabetes mellitus, which itself increases the relative risk of stroke 3–5× in the general population 5 . Understanding the possible neurological sequelae in the setting of MS, amelioration of neuroinflammation is critical and may be accomplished with SCs.
In an acute ischemic cerebrovascular insult, cytokines released by the innate immune system result in increased permeability of the BBB and subsequent extravasation of neutrophils; lymphocytes are dominant in the later phases of stroke 28 . There is an ongoing debate on intrathecal B-cell recruitment and subsequent oligoclonal band synthesis in the setting of an acute IS. In MS, however, this relationship is clearly established. B cells are also important mediators of MS; interestingly, lymphoid organs similar to lymph nodes are formed within the meninges in the setting of secondary progressive MS, and it is suggested that B cells are responsible for the initiation 29 . In fact, B cells are utilized in both the treatment and diagnosis of MS. Anti-CD20 agents such as rituximab are utilized in clinical practice to prevent exacerbations, and intrathecal IgG synthesis is screened for in cerebrospinal fluid (CSF) as a diagnostic tool. In 2012, Prüss et al. 30 reported from their patients who had an IS that 25% had intrathecal IgG and 18% had oligoclonal bands, with no significant difference whether the patient’s stroke was first-time versus recurrent. These findings prompted Laichinger et al. to investigate further; on the contrary, they reported that, of their 212 patients, only 6% had intrathecal IgG, 5% had isolated oligoclonal bands, and only 3% had both intrathecal IgG and oligoclonal bands. Intrathecal IgG was more common in patients who had previously suffered a stroke. Importantly, a third of the patients had a concomitant inflammatory nervous system disease such as MS or neurosarcoid. They concluded that these CSF findings are likely not related to acute IS, and indeed secondary to patients with neuroinflammation such as in MS or previous strokes 31 . This proves to be an important distinction between ISs and MS.
Stem Cell Therapies for Multiple Sclerosis
SCs are frequently appreciated for their significant anti-inflammatory and regenerative properties 7 , making them an ideal therapeutic for a diagnosis defined by cell destruction and death and neuroinflammation (Table 1).
Anti-inflammatory Stem Cell Studies for Multiple Sclerosis.

This table outlines cell-based preclinical trials aimed at reducing neuroinflammation seen in MS. EAE: experimental autoimmune encephalomyelitis; CCR5: chemokine receptor-5; NSC: neural stem cell; BMSC: bone marrow-derived mesenchymal stem cell; NT: neurotrophin; NPC: neural progenitor cell; CSF: cerebrospinal fluid; NMSC: neuralized mesenchymal stem cell; TMEV: Theiler’s murine encephalomyelitis virus; CNS: central nervous system; TGF: transforming growth factor; IL: interleukin; IFN: interferon; EV: extracellular vesicle; MSC: mesenchymal stem cell; TMP: tetramethylpyrazinze; ROS: reactive oxygen species; BBB: blood–brain barrier.
Mesenchymal SCs
Among the multiple mechanisms mediating the therapeutic effects of SCs, the cells’ ability to reduce neuroinflammation closely accompanies functional recovery in MS models. Mesenchymal stem cells’ (MSC) anti-inflammatory properties have been demonstrated in preclinical studies of MS (reviewed by Gugliandolo et al. 6 ). In animal models of MS, MSCs affect adaptive immune cells by balancing Th17, Th1, and Tregs; inhibit CD4+ and CD8+ T-cell inflammatory actions; and encourage innate microglial and astroglial differentiation away from the pro-inflammatory M1 and A1 phenotypes and toward the anti-inflammatory M2 and A2 phenotypes6,50. Moreover, MSCs reduce pro-inflammatory mediators while increasing protective signals. MSCs derived from the placenta offer even greater neuroprotection and neurogenesis by the production of human brain–derived neurotrophic factor (BDNF), nerve growth factor and neurotrophin 3 (NT-3) in MS rat models 35 . Astrogliosis and microgliosis, both detrimental reactions to MS pathophysiology, are also reduced by MSCs 42 . In EAE mice, MSCs become neural cells and reduce inflammation, demyelination, and pro-inflammatory mediators 48 . Neuralized MSCs given to EAE mice are better at reducing T-cell infiltration, increasing Treg differentiation, and promoting oligodendrocyte formation than MSCs. These neuralized MSCs effectively reduced MS symptomology as well, possibly via the release of neurotrophic factors 40 .
Oligodendrocytes, enhanced with ephrin, a protein which aids in cell migration, have also been shown to have therapeutic benefits in animal MS models 51 . Moreover, in mice models, the conditioned medium containing MSC-derived oligodendrocytes offers immense immunoregulation 52 . By implementing a neuroprotective environment against MS inflammation and oxidation, BBB integrity is restored and even protects against inflammatory and immune cell infiltration. MSCs may even further strengthen the BBB by downregulating aquaporin channels 44 .
Strategies designed to further optimize MSCs’ anti-inflammation machinery may enhance the cells’ therapeutic outcomes. MSCs have been paired with anti-inflammatory agents for MS, such as Fingolimod, Minocycline, and Rapamycin33,36,47, showing synergistic effects. Priming MSCs with tetramethylpyrazinze (TMP) also shows amplified benefit for MS, likely by activating the anti-oxidant and anti-inflammatory nuclear factor-erythroid 2-related factor-2 (Nrf2)/heme oxygenase 1(HO-1) signaling pathway 45 . The notion of priming MSCs has been demonstrated to enhance their therapeutic potential against stroke damage 53 . There is also a plethora of evidence supporting amplification of the Nrf2/HO-1 pathway to ameliorate stroke damage 54 .
Neural SCs
While MSCs have clear therapeutic benefits, neural stem cells (NSCs) in EAE mice are also shown to reduce MS symptoms, promote neurogenesis, and enhance myelination. In fact, they may be even more effective than MSCs. For instance, NSCs may play a critical role in restoring balance between the adaptive lymphocytes Th17 and Tregs; Th17 restoration is accomplished through the inhibition of IL-6 expression (a pro-inflammatory cytokine inducing pathogenic Th17 cells for demyelination) and Th17/Treg balance is achieved through inhibition of TNF-α and IFN-γ 46 . NSCs’ therapeutic potential can be further enhanced by modulating factors like NT-3 (promoting remyelination through enhanced differentiation into oligodendrocytes and neurons while inhibiting differentiation into astrocytes), chemokine receptor-5 (promoting early-stage migration of NSCs to inflammatory foci) TGFβ-1 (upregulating Tregs and the microglial M2 phenotype while downregulating Th17 activity) and TGF-β2 (reprogramming infiltrative monocytes to anti-inflammatory myeloid cells)32,34,37,38. NSCs and MSCs show impressive potential for MS treatments and may also be beneficial in preventing later IS incidence by reducing neuroinflammation early.
Extracellular Vesicles
A major benefit of using extracellular vesicles (EVs) as opposed to cells is their cell-free configuration, circumventing the logistical problem of graft rejection inherent in transplanting “cells.” MSC EVs are shown to improve motor symptoms, reduce neuronal death, increase neurogenesis, shift T-cell and microglia 41 populations toward an anti-inflammatory phenotype, and decrease inflammation in the spinal cord of MS models 39 . When obtained from MSCs cultured with anti-inflammatory microglia, EVs promote oligodendrocyte differentiation, migration, and anti-inflammation 43 . Excitingly, these EVs also show significant treatment potential when administered intranasally via immunomodulation 49 . Thus, avoiding the graft rejection risks associated with SC transplantation, EVs offer an exciting research direction for MS treatment and immunomodulation.
Clinical Applications
With this wide array of studies supporting SC in MS, it is logical to apply these concepts in clinical trials 55 . MSCs derived from adipose tissue and umbilical cord tissue and NSCs are safe and demonstrate neuroprotection, anti-inflammation, and improved clinical presentation in patients with MS. In patients with MS, hematopoietic stem cells treatment (HSCT) is more likely to show no evidence of disease after 3 years compared with patients treated with alemtuzumab, an anti-inflammatory MS therapy. This SC treatment had a higher risk of complications for the first 100 days after transplantation, but after 100 days, elevated risk was then surpassed by alemtuzumab 56 . These studies support further investigation of SCs in clinical settings, specifically in efforts to reduce neuroinflammation induced complications, such as IS.
Stem Cell Therapies for Stroke
Utility of SC Therapy for Stroke
The inflammation-mediated secondary cell death that accompanies stroke is very much akin to MS, a chronic neurodegenerative disorder. SC therapy represents a novel treatment approach for stroke damage mediated by such neuroinflammation due to its potential for an extended therapeutic window in the post-stroke inflammatory state compared with current standards of care (Table 2).
Anti-Inflammatory Stem Cell Studies for Stroke.
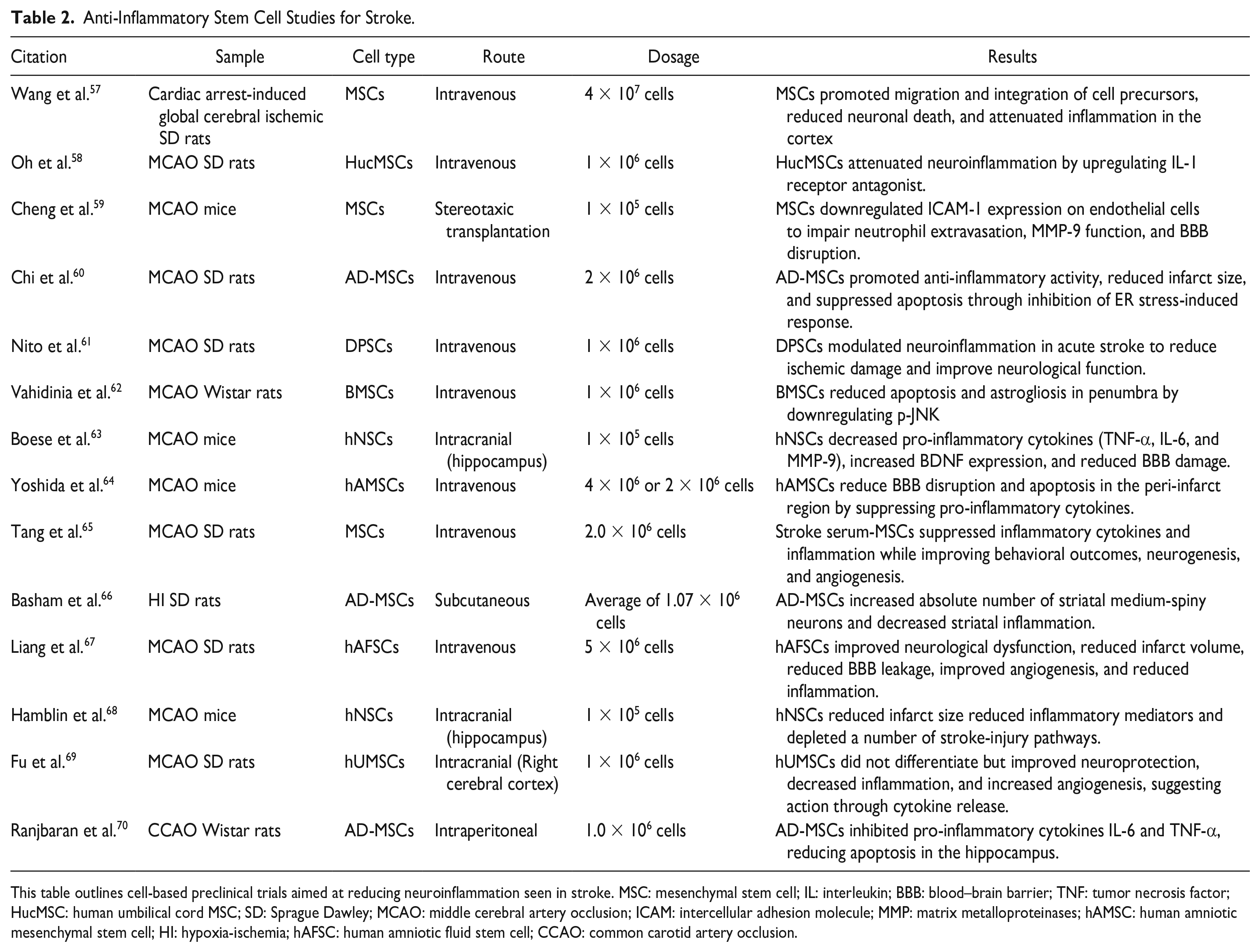
This table outlines cell-based preclinical trials aimed at reducing neuroinflammation seen in stroke. MSC: mesenchymal stem cell; IL: interleukin; BBB: blood–brain barrier; TNF: tumor necrosis factor; HucMSC: human umbilical cord MSC; SD: Sprague Dawley; MCAO: middle cerebral artery occlusion; ICAM: intercellular adhesion molecule; MMP: matrix metalloproteinases; hAMSC: human amniotic mesenchymal stem cell; HI: hypoxia-ischemia; hAFSC: human amniotic fluid stem cell; CCAO: common carotid artery occlusion.
Effector Mechanisms and SC Targets
NSCs have been found to exercise a broad range of effects in mediating tissue recovery and improving neurological function. The timing of SC delivery in IS appears to be critical to the mechanism of therapy, with administration in the early stroke phase upregulating neuroprotective factors, including BDNF, vascular endothelial growth factor (VEGF), ciliary neurotrophic factor, neural growth factor, and glial cell line–derived neurotrophic factor, while NSCs delivered in the chronic stroke phase amplify neurorestoration and cellular proliferation 71 .
SC therapy has been shown to decrease immune cell activation through human-induced pluripotent stem cell (iPSC)-derived NSC transplantation, in addition to the downregulation of pro-inflammatory cytokines 72 . In addition, MSCs increase secretion of anti-inflammatory cytokines which serve to inhibit the proliferation and differentiation of immune cell lines 73 .
Downregulation of oxidative stress is another approach for SC treatment. MSCs are documented to reduce intracellular calcium and reactive oxygen species (ROS) in ischemic neurons 73 . The anti-oxidant response promotes SC survival and reduces neuronal ischemic injury. Nrf2, a transcription factor involved in the anti-oxidant response, is strongly associated with endogenous NSC survival, and thus drug-based therapies focused on the Nrf2 pathway may enhance the effects of exogenous SC transplantation 54 .
Stimulation of endogenous NSCs has also been investigated in promoting stroke recovery and combating neuroinflammation. Adjunct therapies designed to enhance endogenous NSCs focus on delivery of neurogenic factors, including BDNF, VEGF, glial-derived neurotrophic factor, granulocyte colony-stimulating factor, basic fibroblast growth factor, bone morphogenetic protein, and TGF-α to modulate survival pathways 74 . Transplanted NSCs augment neuroplasticity through action on endogenous NSCs, promoting neurogenesis, reorganization, and angiogenesis 72 .
Extracellular Vesicles
As discussed previously, EVs of SCs offer an ethical alternative to the induction of anti-inflammatory effects and BBB repair. EVs are attractive in treating IS as they possess a reduced risk of blood vessel blockage and thrombosis, and furthermore are rather effective at crossing the BBB 75 . Administration of MSC-derived EVs promotes neuroprotection, neurogenesis, and angiogenesis in IS models, with preconditioning of MSCs with either normal or ischemic brain extract further ameliorating recovery 76 . In ischemic rats, miRNA-17-92 overexpressing EVs show improved outcomes in oligodendrogenesis and dendritic plasticity compared with standard MSC-derived EVs, while mi-R-133b overexpression results in neurorestoration and enhanced neuroplasticity77,78. EVs are also strongly characterized by interaction with innate microglia, with EVs derived from a number of CNS lineages, including NSCs, promoting the transition to the microglial anti-inflammatory M2 phenotype (reviewed in Spellicy and Stice 79 ). The overall therapeutic potential of EVs was confirmed in a recent systematic review, supporting SC-derived EVs as being associated with reduced infarct volume and improved recovery from stroke, thus clarifying the future of such therapy in human trials 80 .
Clinical Applications
The ever-growing body of evidence supporting the beneficial outcomes of SC treatment as a stroke therapeutic ultimately supports a transition to clinical medicine. To date, clinical trials for cell-based stroke therapies have employed MSCs, bone marrow–derived mononuclear cells, peripheral blood SCs, and adipose-derived stromal vascular fractions (MSCs among other lineages)81–91. A recent meta-analysis of 11 clinical trials including 368 patients demonstrated the safety and viability of SC therapy for stroke treatment 92 . In the context of MS-induced stroke, SCs can ameliorate the chronic inflammatory phase that has significant pathological overlap in both IS and MS.
Peripheral Contributions
While we have mainly focused on the role of SCs centrally, a multitude of studies have shown the vital role of peripheral organs in neuroinflammation 93 . As discussed, the BBB is impaired in stroke and MS, facilitating the entry of peripheral inflammatory cells into the typically immunoprivileged CNS. After stroke, notable spleen and thymus shrinkage has been observed alongside amplified lymphocytes release 94 . Substantial bone marrow changes are seen after stroke as well, suggesting B lymphopoiesis amplifies peripheral inflammatory responses to stroke as well95,96. In addition, bone marrow–derived MSCs have made incredible impacts on stroke-induced neuroinflammation, further supporting this primary lymphoid organ’s role in stroke pathogenesis97–99. In MS, the T cells perpetuating autoimmunity similarly amplify the bone marrow’s contribution to neuroinflammation, and autologous hematopoietic SC transplantation demonstrates successes in trials100–102. Important immune cell contributions are notable from the gut as well, a concept known as the gut–brain axis. After stroke and in MS, the gut becomes more permeable, and dysbiosis is seen103,104. Furthermore, cervical lymph nodes are activated due to cerebral antigens escaping from the permeable BBB after stroke105,106. Discrepancies between lymph node sizes in treated versus non-treated patients with MS suggests cervical lymph nodes may play a role in MS pathophysiology as well 107 . Not only are these organs notable for their peripheral inflammatory contributions following stroke, but MSC treatments also show SC migration to these organs and subsequent reduction in systemic inflammation93,98,99,108. The immense influence of peripheral organs on neuroinflammation suggests these organs may be notable targets for reducing MS inflammation and reducing stroke risks as well.
Conclusion
There is vast evidence prompting clinical use of SCs to reduce neuroinflammation in MS and IS independently; however, no studies have employed SCs and measured their abilities to reduce MS-induced IS. Given the role of the MS neuroinflammatory environment in the incidence of IS, alongside the promising studies in SCs, the use of SCs to reduce IS risk is a logical and relevant future research direction. Furthermore, another role for SCs in ameliorating IS occurrence secondary to MS may involve restoring the NVU integrity, as the inflammation seen in both pathologies induces detrimental BBB permeability and SCs can regulate BBB stability 109 .
Footnotes
Acknowledgements
Author Contributions
C.V.B., M.M., J.G., and G.L. contributed to the idea generation, writing, and editing of this manuscript. M.M. designed the figure. J.G. and G.L. designed the tables.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.V.B. declared leadership position with University of South Florida, patents holder and patent applications on stem cell biology and its therapeutic applications, consultant to a number of stem cell–based companies, and research funding from the NIH. All the other authors declared no potential conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
