Abstract
Previous studies transplanted human-induced pluripotent stem cells (hiPSCs)-derived mesenchymal stem cells (iMSCs) into thyroid cartilage defect of X-liked severe combined immunodeficiency (X-SCID) rats and confirmed transplanted cell survival and cartilage regeneration. Thus, this study aimed to investigate the contribution of iMSC transplantation to thyroid cartilage regeneration of nude rats. iMSCs were induced from hiPSCs via a neural crest cell lineage. Then, clumps formed from an iMSC/extracellular matrix complex were transplanted into thyroid cartilage defects in nude rats. The larynx was removed and histological and immunohistochemical analyses were performed 4 or 8 weeks after the transplantation. Human nuclear antigen (HNA)-positive cells were observed in 11 of 12 (91.7%) rats, which indicated that transplanted iMSCs survived in thyroid cartilage defects in nude rats. HNA-positive cells co-expressed SOX9, and type II collagen was identified around HNA-positive cells in 8 of 12 rats (66.7%), which indicated cartilage-like regeneration. Cartilage-like regeneration in nude rats in this study was comparable to the previous report on X-SCID rats (HNA-positive cells were observed in all 14 rats and cartilage-like regeneration was observed in 10 of 14 rats). This result suggests that nude rats could be an alternative to X-SCID rats in thyroid cartilage regeneration experiments using iMSCs, and this nude rat cartilage transplantation model may develop cartilage regeneration research concerning fewer problems such as infection due to immunosuppression.
Keywords
Introduction
Laryngeal and tracheal cartilages consist of an airway structural framework for breathing but are sometimes impaired due to malignant tumor surgical resection and inflammatory lesions. Large defects of cartilage in clinical practice cause problems in airway patency; thus, cartilaginous framework reconstruction is often necessary to maintain airway strength. Over the years, autologous cartilage from other organs has been used to repair cartilage defects 1 , but autografts are limited in the amount of recovered cartilage. Cartilage regeneration by autologous 2 or allogeneic 3 chondrocytes with scaffold materials has been studied in animals but without clinical application.
Mesenchymal stem cells (MSCs) are broadly accepted cell sources for cartilaginous regeneration 4 because they can differentiate into chondrocytes and have paracrine effects to promote tissue regeneration 5 . MSCs are found at a low ratio compared with other bone marrow mononuclear cells and their limitation in proliferation capacity; thus, the problem of the limited cell supply cannot be overcome. However, the human-induced pluripotent stem cell (hiPSC) establishment provided a new approach to solve this problem. hiPSCs may be obtained from the patient or a human leukocyte antigen–matched donor and are capable of unlimited proliferation capacity and pluripotency; thus, hiPSCs are an ideal cell source in regenerative medicine.
Our group focused on the unlimited proliferation capacity of hiPSCs and differentiation potency of MSC into cartilage, thereby inducing hiPSCs-derived MSCs (iMSCs) via neural crest cells (NCCs), transplanting clumps of iMSC/extracellular matrix complex (C-iMSCs) into defective thyroid cartilage of immunodeficient rats, and confirming the survival of transplanted cells and cartilage regeneration 6 . This study is a major step in cartilage regeneration research in the field of laryngotracheal reconstruction, but immunodeficient rats used in this study were X-liked severe combined immunodeficiency (X-SCID) rats established by gene knockout of interleukin (IL)-2 receptor gamma (Il2rg) and its immunodeficient phenotypes were characterized as a nearly complete lack of T cells, B cells, and natural killer (NK) cells 7 . X-SCID rats are very severely immunocompromise and should be carefully bred to avoid infection; thus, performing xenotransplantation in rats with milder immunodeficiency, such as nude rats, which have no mature T cells but with preserved B and NK cell function is desirable 8 . The rejection in xenotransplantation is caused by T cells, B cells, NK cells, macrophages, and dendritic cells 9 ; thus, X-SCID rats theoretically appear to be more susceptible to hiPSCs engraftment than nude rats. However, Samata et al. 10 transplanted hiPSC-derived dopaminergic neurons into the brains of X-SCID and nude rats and revealed no difference in their engraftment. Okuyama et al. 11 transplanted hiPSC-derived airway epithelial cells (hiPSC-AECs) into the tracheal defects of nude rats and confirmed their engraftments. Therefore, the present study transplanted iMSCs into thyroid cartilage defects of nude rats and histologically examined the survival of transplanted cells and cartilage regeneration, and investigated nude rats as an alternative recipient to X-SCID rats.
Materials and Methods
Induction of iMSCs and Formation of C-iMSCs
The iMSC induction and C-iMSC formation were performed following the method described by the previous reports6,12. In brief, hiPSCs (cell line 123A3 13 ) were cultured under feeder-free/xeno-free conditions on iMatrix-511 (Nippi, Tokyo, Japan)-coated cell culture plates in the AK03N medium (StemFit AK03N; Ajinomoto, Tokyo, Japan). NCCs were induced in AK03N medium without supplement C (Ajinomoto) and added with 10 µM of SB431542 (Selleck Chemicals, Houston, TX, USA) and 1 µM of CHIR99021 (Axon Medchem, Groningen, the Netherlands) for 10 days. CD271-positive cells were sorted using BD FACS Aria II cytometer (BD Biosciences, San Jose, CA, USA) after the NCC induction. After sorting, NCCs were cultured on fibronectin-coated plates in AK03N medium without supplement C and added with 10 μM of SB431542, 20 ng/mL of epidermal growth factor (R&D Systems, a Bio-Techne Brand, MN, USA), and 20 ng/mL of fibroblast growth factor-2 (Wako, Osaka, Japan) for 17 days. Then, the cells were differentiated into MSCs by switching to an MSC medium (PRIME-XV MSC Expansion XSFM, Irvine Scientific, CA, USA).
C-iMSCs are formed by seeding iMSCs at a density of 5.0 × 104 cells/well onto 1.5 µg/cm2 of fibronectin (Millipore, CA, USA)-coated temperature-responsive 96-well plates (Upcell, CellSeed, Tokyo, Japan) and cultured in MSC medium. The cell sheets were detached by treatment at room temperature for 15 min and were moved onto low-attachment 96-well plates on Day 5 (Prime surface 96U plate, Sumitomo Bakelite, Tokyo, Japan). Then, the C-iMSCs were formed on Day 7.
Flow Cytometry
The cells were analyzed by flow cytometry analysis using a Human MSC Verification Flow Kit (FMC020, R&D System) and PE-conjugated anti-human CD73 (Mouse, 967542, 1:10; R&D System) after iMSC induction. CD90 and CD73 were used as positive markers for MSC, and CD45, CD34, CD79A, HLA-DR, and CD11b were used as negative markers. The single-cell suspension after iMSC induction in FACS buffer was treated with these antibodies. Then, the cells were analyzed using a BD LSRFortessa Cell Analyzer (BD Biosciences) and FlowJo (Tree Star Inc, OR, USA).
Animals
Transplantation experiments used male nude rats (F344/NJcl-rnu/rnu) purchased from CLEA Japan (Osaka, Japan). The rats were aged 8 to 24 weeks. The animal experimental protocol for this study was approved by the Animal Experimentation Committee of Kyoto University. All animal experiments were performed following the “Guidelines for Animal Experiments of Kyoto University.”
Transplantation
A total of 12 male nude rats were used as transplant recipients. The animals were divided into two groups with different postoperative periods and sacrificed afterward: 4 (n = 6) or 8 weeks (n = 6). Transplantations were performed according to the method described by Yoshimatsu et al. 6 In brief, general anesthesia was induced by an intraperitoneal injection of a mixture of butorphanol tartrate (2.5 mg/kg), medetomidine hydrochloride (0.15 mg/kg), and midazolam (2 mg/kg). Thyroid cartilage was exposed by a midline skin incision and bilateral sternohyoid and sternothyroid muscle separation. Bilateral thyroid cartilage defects were created using a biopsy punch (Kai Industries, Tokyo, Japan) (outer diameter = 1 mm). Three C-iMSCs were transplanted into the defects only on the left side. No transplantations were done in the right defects, which were used as controls. Afterward, sternohyoid and sternothyroid muscles and incised skin were sutured. Fig. 1 shows the schematic study protocol.
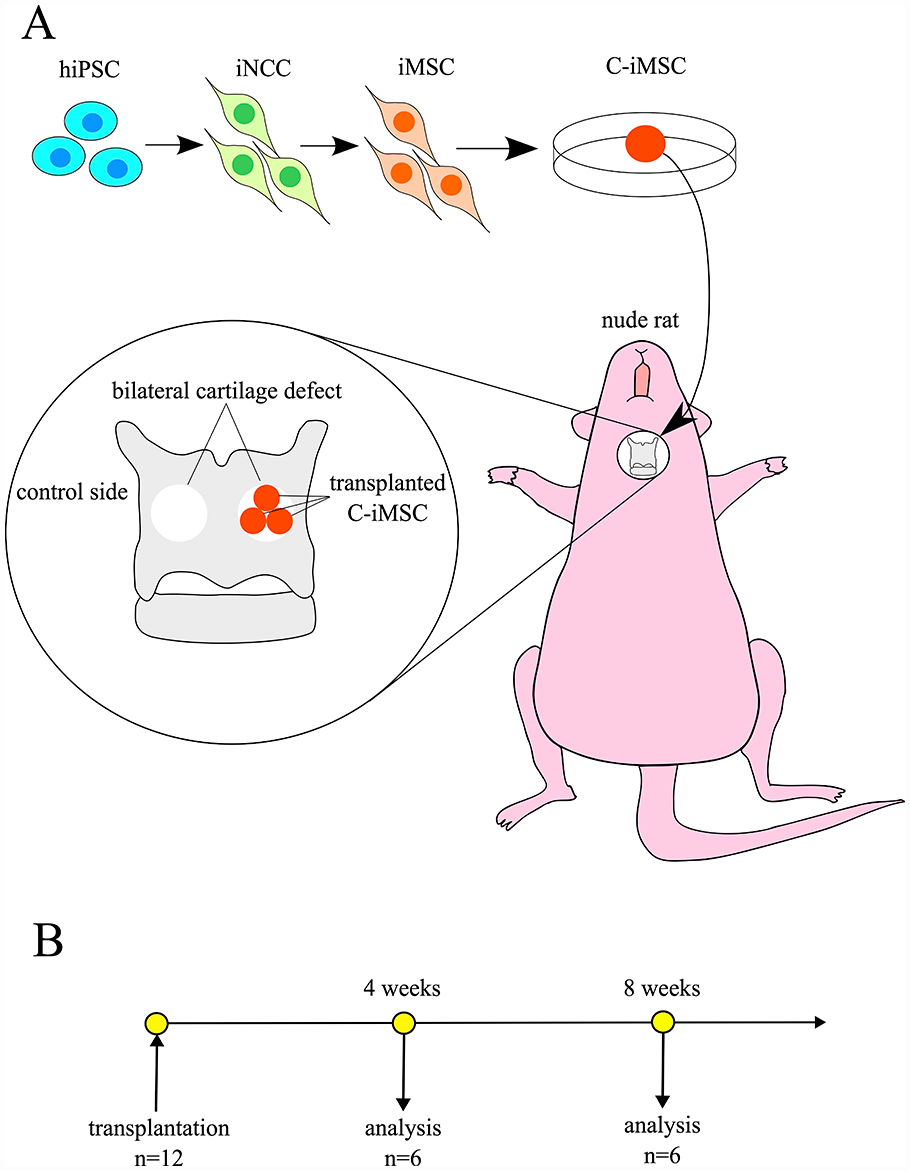
(A) Schematic representation of C-iMSC transplantation into the thyroid cartilage defects of nude rats. C-iMSCs were transplanted into the thyroid cartilage only on the left side. (B) Timeline after the transplantation. C-iMSCs: clumps of iMSC/extracellular matrix complex; hiPSC: human-induced pluripotent stem cells; iMSC: hiPSCs-derived mesenchymal stem cells; iNCC: hiPSC-derived neural crest cells.
Histological Analyses
The animals were euthanized with CO2 4 or 8 weeks after transplantation. The larynx, along with sternohyoid and sternothyroid muscles, was removed and immersed in 4% paraformaldehyde (PFA) overnight. Subsequently, these were immersed in 10%, 20%, and 30% sucrose overnight and embedded in an optimal cutting temperature compound (Sakura Finetek Japan, Tokyo, Japan). Axial cryosections were made at a 10-µm thickness. Hematoxylin and eosin (HE) staining, Alcian blue staining, and safranin-O staining were conducted. Sections were treated with 3% acetic acid for 3 min and stained with Alcian Blue 8GX solution (pH 2.5, 1.061647, Merck KGaA, Darmstadt, Germany) for 30 min for Alcian blue staining. Then, sections were reimmersed in 3% acetic acid for 3 min. Sections were dehydrated and cleared with ethyl alcohol before mounting. The safranin-O staining method was modified from previous reports14,15. Sections were immersed in Weigert’s iron hematoxylin solution for 10 min and stained with 0.05% Fast Green FCF (Sigma-Aldrich, St. Louis, MO, USA) solution for 5 min. Then, sections were treated with 1% acetic acid and stained again in 0.1% safranin-O solution (Sigma-Aldrich) for 5 min, dehydrated, and cleared with ethyl alcohol before mounting.
Immunofluorescence Staining
hiPSC-derived NCCs (iNCCs) were treated with 0.2% Triton X-100 in phosphate-buffered saline (PBS) for 15 min at 4°C for permeabilization following fixation with 4% PFA in PBS for 15 min and rinsing with PBS for immunofluorescence staining of iNCCs. The cells were immersed in Blocking One (Nacalai Tesque, Kyoto, Japan) for 1 h at 4°C to block nonspecific antibody binding and then incubated with anti–transcription factor activating protein-2α (TFAP2A) (Mouse, AB528084: 1:100; DSHB) and diluted with 10% Blocking One in PBS containing 0.05% Tween 20 (PBST) at 4°C overnight. The cells were incubated with a fluorophore-labeled secondary antibody (Alexa Fluor-conjugated secondary antibodies, Invitrogen, Tokyo, Japan) and 4′,6-diamino-2-phenylindole (DAPI; Invitrogen) for nuclei staining for 1 h at room temperature. Images of iNCCs were obtained using a fluorescence microscope (Nikon-eclipse Ti-S), Olympus DP73 camera (Olympus, Tokyo, Japan), and CellSens Standard software (Olympus).
Laryngeal cryosections were digested for 10 min at 37°C with 0.5% pepsin (Sigma-Aldrich) diluted with 0.5 mM of hydrochloric acid for epitope retrieval for anti-type I collagen and anti-type II collagen antibody reactions. Sections were heated in citrate buffer solution (pH 6.0) for 10 min at 98°C, followed by incubation for an additional 30 min at 65°C, for anti-SOX9 staining. Sections were incubated overnight at 4°C with anti-human nuclear antigen (HNA) (Mouse, MAB1281, 1:1000; Millipore), anti-type I collagen (COL1) (Rabbit, ab34710, 1:200; Abcam, Cambridge, UK), anti-type II collagen (COL2) (Mouse, MA5-12789, 1:100; Invitrogen), and anti-SOX9 (Rabbit, AB5535, 1:5000; Millipore) antibodies after blocking with Blocking One (Nacalai Tesque) for 1 h. Sections were incubated with fluorophore-labeled secondary antibodies and DAPI for 1 h at room temperature. Secondary antibodies were diluted with Can Get Signal Solution B (Toyobo, Osaka, Japan) for anti-COL1 and anti-COL2 staining and with 10% Blocking One in PBST for anti-SOX9 staining. All section images were obtained using an OLYMPUS BX 50 fluorescence microscope with an Olympus DP70 camera (Olympus) and an Olympus DP Controller 2002 (Olympus).
Results
Induction of iMSC From hiPSC Through NCC
After 4 days of hiPSC pre-culture (Fig. 2A), iNCCs were induced by culturing in an NCC induction medium and confirmed by immunostaining using an antibody for NCC marker, TFAP2A (Fig. 2B). Then, iNCCs were differentiated into iMSCs by culturing in an MSC medium. Induced iMSCs showed MSC-like morphology (Fig. 2C) and MSC markers expression (Fig. 2D) as similar to that seen in primary cultured MSCs.

(A) Phase-contrast image of 1231A3 line undifferentiated hiPSCs. (B) Immunocytochemistry images of iNCCs using anti-TFAP2A antibody. Scale bar = 50μm. (C) Phase-contrast image of induced iMSCs. Scale bar = 200μm. (D) Flow cytometry analysis of iMSCs. iMSCs show expression of MSC-positive markers, including CD90 and CD73 (red), and no expression of MSC-negative markers, including CD45, CD34, CD79A, HLA-DR, and CD11b (red). The histograms of isotype control are shown in black.
Cartilage-Like Regeneration
Fig. 3 shows the representative case of cartilage-like regeneration after C-iMSC transplantation. Cartilage-like regenerated tissue was observed in the transplanted area on the HE staining (Fig. 3A, D). Safranin-O and Alcian blue are cationic dyes that stain proteoglycans, which are cartilage extracellular matrix (ECM) components16,17. ECM was stained by safranin-O and Alcian blue staining in the cartilage-like regenerated tissue (Fig. 3B, C, E, F), indicating that the ECM of the regenerated tissue is equivalent to normal cartilage.

Histological evaluation of C-iMSC transplanted laryngeal axial sections at 8 weeks postoperatively. The area between the yellow arrowheads indicates the defect area in the thyroid cartilage. (D–F) Higher magnification of the yellow box in the (A–C); transplanted side. (G–I) A higher magnification of the yellow dot box in the (A–C); uninjured area on control side. (A, D, G) HE stain. (B, E, H) Safranin-O staining. (C, F, I) Alcian blue staining. Cartilage-like regenerated tissue was observed in the transplanted area, but not in the control side. (A–C): scale bar = 500 μm, (D–I): 50 μm. HE: hematoxylin and eosin.
Chondrocyte Marker Expression and ECM Production
Fig. 4 shows the representative case of chondrocyte marker expression and ECM production after C-iMSC transplantation. SOX9 is a marker of chondroprogenitors and chondrocytes 18 . Many cells express HNA, DAPI, and SOX9 in the regenerated tissue (Fig. 4B–E). HNA and DAPI co-positivity indicates transplanted cell survival; thus, these findings indicate that surviving transplanted cells differentiated into a chondrogenic lineage. COL2 is an ECM component of the hyaline cartilage; COL1 is not expressed in the hyaline cartilage but in fibrocartilage and fibrous tissue19,20. ECM that surrounds the surviving cells express COL2 (Fig. 4F), but not COL1 (Fig. 4G). Therefore, the ECM of the regenerated tissue is equivalent to normal cartilage with the results of safranin-O, Alcian blue, COL2, and COL1.

Immunofluorescence staining of cartilage-like tissue regenerated tissue using anti-SOX9, HNA, type II collagen (COL2), type I collagen (COL1) antibody, and DAPI staining. (A) The area between the yellow arrowheads indicates the defect area in the thyroid cartilage. (B–G) Higher magnification of the yellow box in (A). (B) SOX9; (C) HNA; (D) DAPI; (E) merge of SOX9 and HNA; (F) merge of COL2 (green), HNA (red), and DAPI (blue); (G) merge of COL1 (white), HNA (red), and DAPI (blue). (A) Scale bar = 200 μm, (B–G): 50 μm.
The Evaluation of Transplanted Cell Engraftment and Cartilage-Like Regeneration
HNA-positive surviving transplanted cells were found in 6 of 6 rats and 5 of 6 rats at 4 and 8 weeks after transplantation, respectively. Regenerated cartilage-like tissue was defined as the tissue that meets all of the following criteria: (1) contains SOX9-expressing surviving transplanted cells, (2) surrounding ECM stained with safranin-O and Alcian blue, and (3) surrounding ECM express COL2, but not COL1. Cartilage-like tissue regeneration was observed in 5 of 6 rats at 4 weeks and 3 of 6 rats at 8 weeks postoperatively. Fig. 5 shows the histology and immunofluorescence staining of all rats with regenerated cartilage-like tissue. All regenerated cartilage-like tissues were in contact with the resection margin of the host thyroid cartilage. The area of cartilage-like regeneration varied in each case. Cartilage-like regeneration was not observed in the control side of all rats.

Histological findings of all rats showing regenerated cartilage-like tissue at 4 (rat A-E) and 8 (rat F-H) weeks postoperatively. The area between the yellow arrowheads indicates the defect area in the thyroid cartilage. The third column (safranin-O staining) indicates a higher magnification of the yellow box in the second column. The fourth and fifth columns indicate immunofluorescence staining, using anti-HNA, anti-SOX9, and anti-COL2 in regenerated tissue. The fourth and fifth columns indicate a higher magnification of the yellow dot box in the third column. With few cartilage-like regenerations in A and D, we indicate the cells positive for safranin-O, double-positive for HNA and SOX9, and double-positive for HNA and COL2 by the white arrow in the third, fourth, and fifth columns, respectively.
Discussion
This study induced iMSCs through NCC and transplanted iMSCs into defects in nude rat thyroid cartilage and confirmed cartilage-like tissue regeneration. Cartilage-like tissue regeneration was observed in 8 of 12 (66.7%) rats (5 of 6 rats at 4 weeks and 3 of 6 rats at 8 weeks), which was comparable to our previous report where 10 of 14 (71.4%) X-SCID rats showed cartilage regeneration 6 . The results showed that iMSCs engrafted on nude rats, as well as on X-SCID rats, and suggested that nude rats could be an alternative to X-SCID rats in thyroid cartilage regeneration experiments using iMSCs. No studies have reported on similar transplantation for cartilage regeneration into X-SCID and nude rats although several reports were published on hiPSC-derived chondrocyte transplantation into the knee joint21–26. The present study is significant because the same method was used for the transplantation of nude rats as those of X-SCID rats and the engraftments were evaluated.
Samata transplanted hiPSC-derived dopaminergic progenitor cells into X-SCID and nude rats 10 , and reported no difference in engraftment efficiency between the two groups. Conversely, Tada transplanted hiPSC-AECs into the middle ear of X-SCID rats and confirmed their engraftments on the middle ear cavity surface. However, transplanted hiPSC-AECs into nude rats did not engraft on the middle ear cavity surface 27 . The present study revealed that the rate of cartilage-like regeneration in nude rats is similar to those of X-SCID rats, although they are not compared head-by-head. Our results and these reports suggest that nude rats may be sufficient recipients depending on the animal species of the recipient, the type of transplanted cells, and the location of transplantation.
There are two possible reasons for the lack of big difference in engraftment between X-SCID and nude rats. One reason is that the transplanted cells were MSCs, which are known to have immunomodulatory properties 28 . MSCs suppress CD4+ T-cell proliferation 29 , induce T regulatory cells, and produce soluble factors, such as prostaglandin E2, transforming growth factor-1, interleukin-10, hepatocyte growth factor, and indoleamine 2,3-dioxygenase, all of which have immunomodulatory effects 30 . In addition, iMSCs have immunomodulatory properties under xenogeneic conditions 31 . Transplanted iMSCs may have suppressed local graft versus host disease in nude rats, resulting in an almost similar engraftment rate to that of X-SCID. Li et al. 30 reviewed 94 reports of MSC xenografts and reported that MSC engrafted and functioned in 88 (93.6%) experiments, with evidence of functional failure in only 6 (6.4%) cases. Paul et al. 32 reported that human bone marrow derived MSC that is implanted into immunocompetent rat hearts survived for 6 weeks, despite the lack of any immunosuppressive therapy. Our study results, together with the results of these studies, suggest that host immunity dosen’t have significant effect on MSC engraftment.
The second reason is that the transplantation site was cartilage. Cartilage’s avascularity supposed that the tissue is immune privileged, whereby a body’s immune system is limited in its ability to detect and reject implanted tissue 33 . The presence of ECM is considered to form a protective barrier around the chondrocytes, which blocks the host immune cell infiltration into the graft 33 . In this study, iMSCs were implanted as C-iMSCs-containing ECM generated from iMSCs; thus, the immune response to iMSCs may be evaded by the ECM.
This study has some limitations. First was the cell quality control of iMSCs. We did not perform the procedures for cell quality control, such as those used in clinical trials 34 , checking the remaining reprogramming vectors, the residual undifferentiated iPSC assay, the in vitro tumorigenicity assay, comparative genomic hybridization, single nucleotide polymorphism analysis, and transcriptome analysis. As for the reprogramming vectors, we used hiPSCs without genome integration of episomal vectors used as reprogramming vectors 13 . Kamiya et al. 12 reported that iMSCs induced from the same cell line by the same protocol used in this study were analyzed and that the global gene expression of iMSCs by messenger RNA sequencing (mRNA-seq) and iMSCs were negative for POU5F1 and NANOG. Al-Akashi et al. 35 reported that the selective elimination of hiPSCs from iMSCs is achieved by dihydroorotate dehydrogenase inhibition using Brequinar. Based on these studies, we try to achieve a clinically applicable level of quality control by selecting pluripotent marker genes for the residual undifferentiated iPSC assay and performing qRT-PCR after induction. Improvements in the protocol, including the in vitro tumorigenicity assay, genome analysis, and transcriptome anasyis, are also desirable for clinical application. Though we did not examine the iMSC differentiation ability in this experiment, in a previous study Kamiya confirmed the differentiation potency of iMSCs into the bone, cartilage, and adipose 12 ; therefore, the iMSCs used in our study were also presumed to have appropriate differentiation potential. In this study, iMSCs were induced via NCC. Because, the thyroid cartilage originates from NCC 36 , we considered it reasonable to use iMSCs induced via NCC for regeneration of thyroid cartilage, rather than using hiPSC-MSCs induced from mesoderm without NCC. However, hiPSC-MSCs derived from mesoderm have been used in many studies37–39. Both types of MSCs derived from hiPSCs can differentiate into three lineages (osteocytes, chondrocytes, and adipocytes) and express surface markers characteristic of MSCs12,37,39. Therefore, the thyroid cartilage regeneration using hiPSC-MSCs derived from mesoderm should also be considered in future.
Another limitation is that this is not a head-by-head comparison between nude and X-SCID rats. However, the induction protocol and implantation of the previous study are identical to those used in this study 6 ; therefore, we have assumed that some comparisons may be permissible. It should be considered that the size of the thyroid cartilage defects in this study was smaller than that typically encountered in clinical medicine. Therefore, applying this technique to large defects is desirable. We plan to transplant iMSC to tracheal defects because tracheal cartilage defects are more likely to be clinically encountered than thyroid cartilage defects. Nakamura et al. 40 developed an artificial trachea that was made of a polypropylene mesh as a framework with a collagen sponge for tracheal reconstruction. This artificial trachea has been successfully applied to humans. However, only adult cases have benefited from this prosthesis to date because the polypropylene frame is non-absorbable and does not expand and is therefore not appropriate for use in growing children41,42. This problem can be overcome by transplanting iMSCs with collagen sponges without polypropylene frame. Our transplantation experiment using iMSC may provide a good platform for future clinical application of cartilage reconstruction.
In summary, we investigated the efficacy of iMSCs in thyroid cartilage regeneration in a nude rat model. Cartilage regeneration was observed in 8 of 12 (66.7%) rats, which was comparable to the previous report on X-SCID rats. The results suggest that nude rats could be an alternative to X-SCID rats in thyroid cartilage regeneration experiments using iMSCs, and this nude rat cartilage transplantation model may help develop cartilage regeneration research concerning fewer problems such as infection due to immunosuppression.
Footnotes
Acknowledgements
We thank the Center for Anatomical, Pathological and Forensic Medical Research, Kyoto University Graduate School of Medicine, for preparing microscope slides (safranin-O staining sections), Dr. Daisuke Kamiya for the advice of C-iMSC generation, and Dr. Naoki Yamada for the advice of iNCC induction. Flow cytometry analyses using BD FACS Aria II and BD LSRFortessa Cell Analyzer were performed at the Medical Research Support Center, Graduate School of Medicine, Kyoto University, which was supported by Platform for Drug Discovery, Informatics, and Structural Life Science from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author Contributions
Keisuke Mizuno: Data collection, data analysis and interpretation, manuscript writing, and final manuscript approval. Hiroe Ohnishi: Conception and design, data collection and assembly, data analysis and interpretation, manuscript writing, and final manuscript approval. Masayoshi Yoshimatsu: Conception and design, data collection and assembly, data analysis and interpretation, manuscript writing, and final manuscript approval. Chengzhu Zhao: Provision of study material, administrative support, and final manuscript approval. Yasuyuki Hayashi: Data collection, administrative support, and final manuscript approval. Fumihiko Kuwata: Data collection, administrative support, and final manuscript approval. Shinji Kaba: Administrative support and final manuscript approval. Hideaki Okuyama: Data collection, administrative support, and final manuscript approval. Yoshitaka Kawai: Administrative support and final manuscript approval.
Nao Hiwatashi: Administrative support and final manuscript approval. Yo Kishimoto: Conception and design, administrative support, and final manuscript approval. Tatsunori Sakamoto: Administrative support and final manuscript approval. Makoto Ikeya: Administrative support and final manuscript approval. Koichi Omori: Conception and design, financial support, administrative support, and final manuscript approval.
Availability of Data and Materials
The materials used in the current study are available from the research institutes or companies described in this article.
Ethical Approval
This study was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee (Med Kyo 23113).
Statement of Human and Animal Rights
The animal experimental protocol for this study was approved by the Animal Experimentation Committee of Kyoto University. All animal experiments were performed following the “Guidelines for Animal Experiments of Kyoto University.” And the experiment using human cell line was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee.
Statement of Informed Consent
The informed consent for establishing hiPSC and using the study for regenerative medicine was obtained from the healthy donors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant-in-Aid for Early-Career Scientists from Japan Society for the Promotion of Science [grant number 19K18803, 22K16898]; Alumni otolaryngology fund from the Department of Otolaryngology Head and Neck Surgery, Graduate School of Medicine, Kyoto University.
