Abstract
Subcutaneous implants of device-encapsulated stem cell–derived pancreatic endoderm (PE) can establish a functional beta cell mass (FBM) with metabolic control in immune-compromised mice. In a study with human-induced pluripotent stem cell-PE, this outcome was favored by a preformed pouch which allowed lesion-free insertion of devices in a pre-vascularized site. This was not reproduced in nude rats, known to exhibit a higher innate reactivity than mice and therefore relevant as preclinical model: a dense fibrotic capsule formed around subcutis (SC) implants with virtually no FBM formation. Placement in omentum (OM) of nude rats provided a less fibrous, better vascularized environment than SC. It resulted in less donor cell loss (56% recovery at post-transplant-PT week 3 versus 16% in SC) allowing FBM-formation. At PT week 30, 6/13 OM-recipients exhibited glucose-induced plasma hu-C-peptide to 0.1–0.4 ng/ml, versus 0/8 in SC-recipients. These levels are more than 10-fold lower than in a state of metabolic control. This shortcoming is not caused by inadequate glucose responsiveness of the beta cells but by their insufficient number. The size of the formed beta cell mass (0.4
Introduction
Human stem cell–derived pancreatic endoderm (PE) is a potential source for a cell therapy product in type 1 diabetes. Implants in immune-compromised mice were shown to generate glucose-responsive beta cells that protect against hyperglycemia following beta cell depletion in the host pancreas1,2. This was also the case when the PE-cells were inserted in a device and placed subcutaneously3–6. Use of a device may help design local immune protection in further development. It also offers the advantage that implants can be entirely retrieved for safety reasons as well as for quantitative and qualitative analysis allowing correlations with initial cell dose and with the in vivo outcome. Studies in mice indicated the size of the beta cell mass that was associated with metabolic control at posttransplant week 20 and beyond5,7. This effect was, however, not achieved in all recipients of the same initial cell dose. Partial or complete failure can be attributed to larger losses of donor cells following placement in the surgically lesioned subcutaneous tissue. In a study on device encapsulated hu-iPSC-PE implants in the subcutis (SC) of mice, we noticed that the number of failures was lower when devices were inserted in a preformed fibrous pouch which is likely to have reduced tissue damage at the time of implantation and hence exposure of the cells in the device to a potentially cytotoxic environment 7 . We subsequently examined whether a subcutaneous pouch also facilitates survival and function of these implants in nude rats which are known to exhibit a stronger innate reactivity than immune-compromised mice 8 and therefore considered relevant as preclinical model. Since the contrary was observed, we examined whether a better outcome was achieved in the omentum (OM), a site that allows device placement with minimal local tissue damage. Its choice also builds on our experience with this site for implantation of non-encapsulated human islet cells in nude rats 9 and in man 10 . These preparations were shown to engraft in the OM depending on their composition and scaffold9,10. Functional outcome in patients has been marginal in our center 10 and the university of Alberta 11 , but one case of insulin-independence has been reported by the university of Miami 12 ; the centers in Alberta and Miami have now joint a clinical trial with the same study protocol (NCT02821026).
The present study reports on the failure of device-encapsulated hu-iPSC-PE implants in the SC of nude rats, while this product had achieved metabolic control in SCID/beige mice. Placement in the OM did lead to formation of a functional beta cell mass (FBM), however, marginal in size and variable. The combination of in vivo and in situ data indicate processes that underly failure, function, and its variability.
Material and Methods
Transplantation of Device-Encapsulated hu-iPSC-PE in Subcutis or Omentum
Transplantations were conducted with hu-iPSC-PE that had been manufactured by Nestlé Research (Lausanne, Switzerland), as previously described 6 , and shipped cryopreserved to Brussels. After thawing, aggregates were cultured for 72 hours in stage 4 medium composed of Dulbecco’s modified Eagle’s medium high-glucose with Glutamax, 1% penicillin/streptomycin and 1% B27, supplemented by 50 ng/ml Noggin, 50 ng/ml epidermal growth factor, 50 ng/ml keratinocyte growth factor (all R&D Systems, USA), and 30 ng/ml Heregulin-β1 (tebu-bio, France). At the end of culture, preparations contained >90% living cells, the majority nuclear positive for PDX1 (Pancreatic and Duodenal Homeobox 1), 47% ± 6% double positive for PDX1 and NKX6.1 (NK6 Homeobox 1), and 10%–15% single-or double-hormone positive for insulin, glucagon, and somatostatin, percentages that are similar to those in our previous report 7 . Fractions with 5 x 106 hu-iPSC-PE cells were loaded in the same devices, as manufactured and provided by Nestlé Institute Health Sciences—Nestec, Lausanne. They consist of a polypropylene frame that holds an outer 77-µm polyester mesh and a hydrophilic polytetrafluoroethylene (PTFE) 0.45-µm-porous (cell-impermeable) inner membrane together6,13 (Supplemental Fig. 1). One device was implanted in 8- to 10-week-old non-diabetic male Rowett Nude rats (276 ± 21 g body weight, Envigo, USA). After a first series of implants in a preformed subcutaneous pouch (n = 5, placed in the back as described in mice 7 ), devices loaded with the same cell preparation were implanted in SC without pouch (n = 12) or in OM (n = 17). The OM was not incised for placement of a device but was folded over it and glued (Supplemental Fig. 2).
Recipients were followed for up to 30 weeks for health status, body weight, glycemia, and plasma human C-peptide (hu-C-pept) before and following intraperitoneal glucose bolus (3 g/kg body weight). Plasma-hu-C-peptide was measured with Ultrasensitive C-Peptide enzyme-linked immunosorbent assay (ELISA) kit (Mercodia, Sweden) and glycemia with GlucoMen (Menarini Diagnostics, Italy). Protocol and procedures were approved by the Ethical Committee of VUB and carried out according to European Community Council Directive (86/609/EEC). As in our previous work, we use the term “graft” for cells before transplantation and “implant” for the tissue after implantation. Device-encapsulated implants were retrieved for histological (PT-weeks 3 – 30) and functional (PT week 30) analysis.
Histological Analysis of Retrieved hu-iPSC-PE Implants
Retrieved implants were fixed in 4% neutral-buffered formaldehyde, processed, embedded in paraffin, and completely sectioned (5 µm) 7 . Sections were stained with hematoxylin and eosin for general histology. Cell types were identified on basis of reactivity with guinea pig anti-insulin (1/2,000, in house-produced), rabbit anti-glucagon (1/1,000, in house-produced), rat anti-somatostatin (1/100, Abcam, UK), mouse anti-CK19 (1/20, Dako, Denmark), and rabbit anti-PanCK (1/200, Dako, Denmark). Nuclear positivity was determined with DAPI (Sigma-Aldrich, USA), goat anti-PDX1 (1/100, R&D Systems, USA), mouse anti-NKX6.1 (1/100, monoclonal antibody developed by Ole Madsen, Hagedorn Research Institute and obtained from the Developmental Studies Hybridoma Bank, created by the National Institute of Child Health and Human Development (NICHD) of the National Institutes of Health (NIH), and maintained at The University of Iowa, Department of Biology (Iowa City, IA 52242, USA)), and rabbit anti-Ki67 (1/100, Acris, Germany). Host reactivity was analyzed with mouse anti-CD68, rabbit anti-CD34 (both 1/100, Abcam, UK), and mouse anti-αSMA (1/5000, Sigma-Aldrich, USA). Secondary antibodies were Cyanine3- or AlexaFluor-conjugated F(ab’)2 fragments of affinity-purified antibodies allowing multiple labeling (1/500, Jackson ImunoResearch, UK). Images were acquired in Aperio CS2 (Leica Biosystems, Germany) and visualized with Pathomation software (Belgium). Digital images were analyzed with SmartCapture 3 software (DSUK, UK). Total volumes of donor cell types inside devices were determined semi-automatically as previously described 7 . Percentages of Ki67-positive cells and of PDX1/NKX6.1/CK19-positive cells were counted by two technicians independently of each other’s data. The density of recipient cells infiltrating the regions outside the inner membranes was determined as described in Supplemental Fig. 3.
Functional Analysis of Retrieved hu-iPSC-PE Implants
Retrieved devices were opened and their tissue content removed for collagenase dispersion into aggregates (<500 µm diameter) (type XI collagenase, 1 mg/ml, Sigma-Aldrich, USA). After washing, samples were taken for determining cell number (NucleoCounter YC-100, ChemoMetec, Denmark) and composition 7 before loading 0.5 to 1 x 106 cells on a P2-Bio-gel column (Bio-Rad, USA) for assessing insulin secretory responses during perifusion5,7. One-minute fractions were collected and assayed for insulin; data were expressed per 103 beta cells as determined in the sample before loading.
Statistical Analysis
Results are expressed as means ± SD. Statistical analysis was performed using Prism 9.2 (GraphPad Software, USA) for comparison by one-way analysis of variance with Tukey’s post hoc test or Student t-test with or without Welch’s correction (statistical significance at P < 0.05).
Results
Failure of Device-Encapsulated hu-iPSC-PE Implants in Preformed Subcutaneous Pouch
No basal or glucose-stimulated plasma hu-C-peptide was detected in nude rats receiving a subcutaneous implant of device-encapsulated hu-iPSC-PE in a preformed pouch: levels remained under < 0.05 ng/ml (limit of detection [LOD] of the assay) during a 30-week follow-up (n = 5 rats).
Histological analysis of devices retrieved at PT-week 30 showed a low recovery of the initial cell mass (4%–28%); it was mainly composed of CK19-positive cells which formed duct-like structures surrounded by connective tissue (Supplemental Fig. 4); insulin-positive cells were absent. The space between inner and outer membranes was filled by connective tissue with only few capillaries (Supplemental Fig. 4). This connective tissue layer was denser than in the previously studied pouch-devices in mice 7 , which is consistent with a stronger foreign body reactivity in nude rats as also reflected by the abundancy of giant cells directed toward the outer membrane of the device. A preformed pouch did also allow smooth insertion of devices in rats, but resulted in higher collagen deposition in the outer part of the device and thus interfered with interstitial diffusion and capillary migration toward the inner membranes, which together impaired the survival of implanted cells. A preformed pouch was therefore not further used.
Higher Cell Survival and Beta Cell Formation in Device-Encapsulated hu-iPSC-PE-Implants in Omentum Than in Subcutis
Implants of device-encapsulated hu-iPSC-PE in OM also resulted in cell loss, but this was significantly lower than in SC. At PT-week 3, 56% of the initial cell mass was recovered in OM versus 16% in SC (Table 1). The residual cell mass in OM had maintained the same percentage of pancreatic precursor cells (43% PDX1+/NKX6.1+) as in the start preparation, whereas this was lower in SC (21%) (Table 1, Fig. 1). These numbers indicate that approximately 50% of the pancreatic progenitor cells at start were recovered in OM versus less than 10% in SC. OM-implants also exhibited a higher number of Ki67+ cells in the PDX1+NKX6.1+CK19+ epithelial cell layers. The markedly higher loss in pancreatic precursor cells implanted in SC was associated with formation of more connective tissue and less blood vessels in the space peripheral to the inner membranes of the devices (Table 1, Fig. 2). This connective tissue density was higher than that previously observed in SC-devices retrieved from mice 7 where recovery of donor cell mass was higher (average of 48% versus 16% in nude rats). The SC and OM-devices did not differ in density of CD68+ cells at their outer membranes (Table 1), which illustrates the innate reactivity in nude rats.
Cellular Composition of Device-Encapsulated hu-iPSC-PE Implants Retrieved From Omentum and Subcutis at PT-Week 3.

Devices were retrieved from randomly selected recipients (n = 4 per site). Cell volume measurements in the inner chamber indicated the percent recovery of initial cell mass and the relative proportions of cells with pancreatic progenitor markers and of cells in proliferative activity. The tissue between inner and outer membranes was analyzed for the relative proportions of infiltrating activated fibrogenic cells (αSMA+ area), of endothelial cells (CD34+ area) and of macrophages and monocytes (CD68+ area). Values are listed as mean ± SD. Statistical significance of differences between both sites was analyzed by Student’s t-test. hu-iPSC-PE: human induced pluripotent stem cell derived pancreatic endoderm; NKX6.1: NK6 Homeobox 1; OM: omentum; PDX1: Pancreatic and Duodenal Homeobox 1; PT: post-transplant; SC: subcutis.
P < 0.05. **P < 0.01.
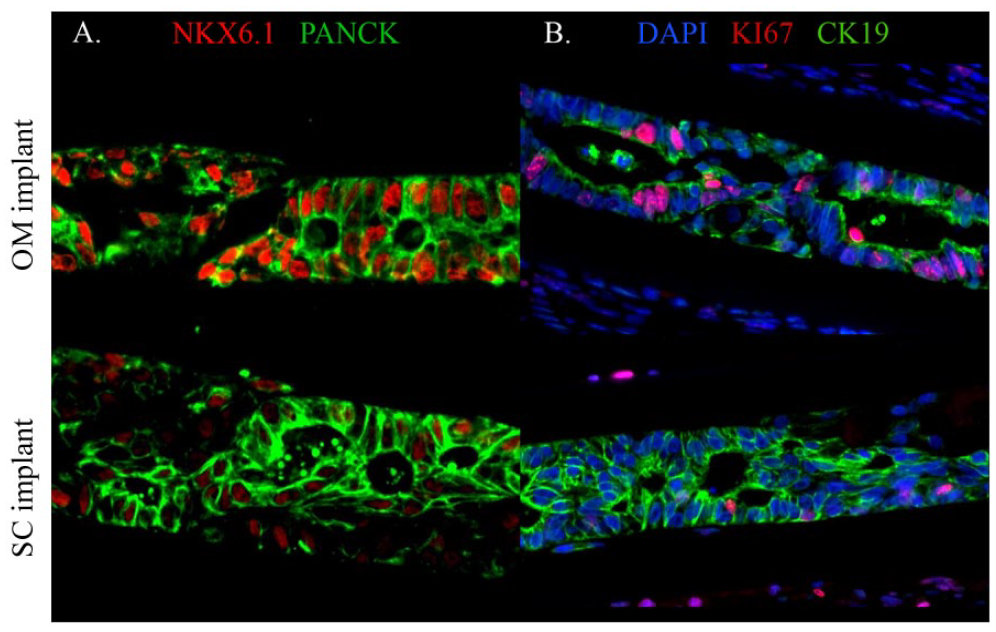
Immunostaining of donor cell-derived tissue in device-encapsulated hu-iPSC-PE implants retrieved at PT-week 3 from omentum (OM) and subcutis (SC). The inner chamber of the devices was mainly composed of cytokeratin-positive cells (stained by Pan-CK- or CK19-antibodies) that formed, or were associated with, duct-like structures. A proportion of CK-positive cells stained positive for the transcription factor NKX6.1; this proportion was higher in OM than in SC where NKX6.1 positivity was also weaker (A). OM-implants also exhibited a higher fraction of CK-positive cells in proliferative activity (stained by Ki67-antibody) (B). Scale bar: 100 µm. hu-iPSC-PE: human-induced pluripotent stem cell derived pancreatic endoderm; NKX6.1: NK6 Homeobox 1; PE: pancreatic endoderm; DAPI: 4′,6-diamidino-2-phenylindole.

Immunostaining of host cells infiltrating the devices in OM and SC implants retrieved at PT-week 3. The space between inner chamber (stars) and the outer mesh (crosses) was stained for the presence of CD68+ cells, of CD34 + cells and of αSMA+ cells (α smooth actin). OM-implants exhibited a higher density of cells positive for the endothelial marker CD34 (A, B) and a lower density of cells positive for the myofibroblast marker αSMA (B). Both sites showed CD68-positive cells associated with the outer mesh (A). Scale bar: 100 µm. hu-iPSC-PE: human induced pluripotent stem cell derived pancreatic endoderm; OM: omentum; PT: post-transplant; SC: subcutis.
At PT-week 30, 11/13 rats with OM-implant exhibited glucose-stimulated plasma hu-C-peptide > LOD (0.05 ng/ml) during an intraperitoneal (IP) glucose test versus 1/8 in the SC-group (Table 2). Their plasma hu-C-peptide positivity became detectable between PT-week 20 and 30. The levels were, however, more than 10-fold lower than in mice that had achieved metabolic control by their subcutaneous device-encapsulated hu-PSC-PE5,7. The lack of metabolic control by the implant is indicated by the failure to lower the 60 min glycemia post-IP under that of control rats: 370 ± 82 mg/dl for the OM-subgroup with highest glucose-stimulated hu-C-peptide (0.1–0.4 ng/ml) versus 376 ± 80 mg/dl in the control group; such lowering was observed in mice with glucose-induce hu-C peptide levels > 6 ng/ml5,7 reflecting control by the formed human beta cells. The transplanted rats remain thus under metabolic control of their pancreatic beta cells which keeps their basal glycemia higher than that in man. This may influence the functional state of the formed human beta cells. It is to be noted that an impact of hyperglycemia has not been demonstrated in mouse recipients of stem cell–derived islet cells 14 .
Assessment of Beta Cell Formation in Device-Encapsulated hu-iPSC-PE Implants in Omentum and Subcutis at PT-Week 30.
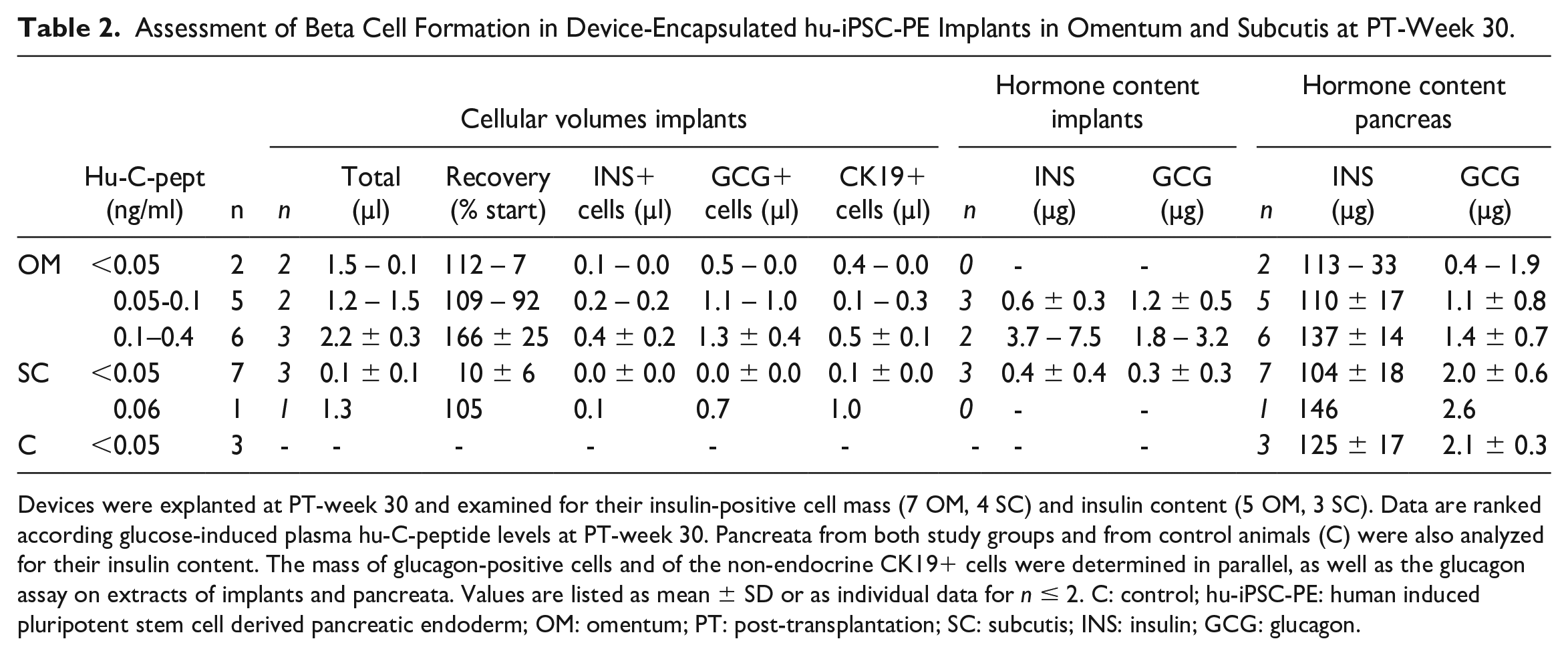
Devices were explanted at PT-week 30 and examined for their insulin-positive cell mass (7 OM, 4 SC) and insulin content (5 OM, 3 SC). Data are ranked according glucose-induced plasma hu-C-peptide levels at PT-week 30. Pancreata from both study groups and from control animals (C) were also analyzed for their insulin content. The mass of glucagon-positive cells and of the non-endocrine CK19+ cells were determined in parallel, as well as the glucagon assay on extracts of implants and pancreata. Values are listed as mean ± SD or as individual data for n ≤ 2. C: control; hu-iPSC-PE: human induced pluripotent stem cell derived pancreatic endoderm; OM: omentum; PT: post-transplantation; SC: subcutis; INS: insulin; GCG: glucagon.
Detection of glucose-induced plasma-hu-C-peptide levels correlated with cell recovery in the device chambers and formation of a beta cell mass (Table 2). Cell volume in retrieved devices was comparable to that at start while it was 10-fold lower for implants with undetectable plasma hu-C-peptide. The subgroup with the higher hu-C-peptide levels (0.1–0.4 ng/ml, 6 OM), exhibited the highest insulin-positive cell mass but its size (mean 0.4 µl) fell in the lower range of the values previously measured for similar implants in the SC of mice 7 . The associated glucagon-positive cell mass was 3-fold higher and reached the values measured in mice 7 . Although only two of these implants were available for measurement of hormone content, it is noteworthy that their insulin content was less than 5% of that in the pancreas, whereas their glucagon content exceeded that of the host pancreas (Table 2).
The organization of the stem cell–generated beta cell implants was similar to that reported in mice. Insulin-positive cells, most of them with PDX1 and NKX6.1 positive nuclei, occurred in small clusters, while the more abundant glucagon-positive cells were more dispersed; some regions consisted mainly of CK19-positive cells that formed duct-like structures (Fig. 3). The percent Ki67-positive cells (2.2%) was lower than at PT-week 3, and was mainly localized in the duct-lining epithelia. The space between inner and outer membrane exhibited loose connective tissue which is at variance with the densely packed strands in SC-devices (Fig. 4).
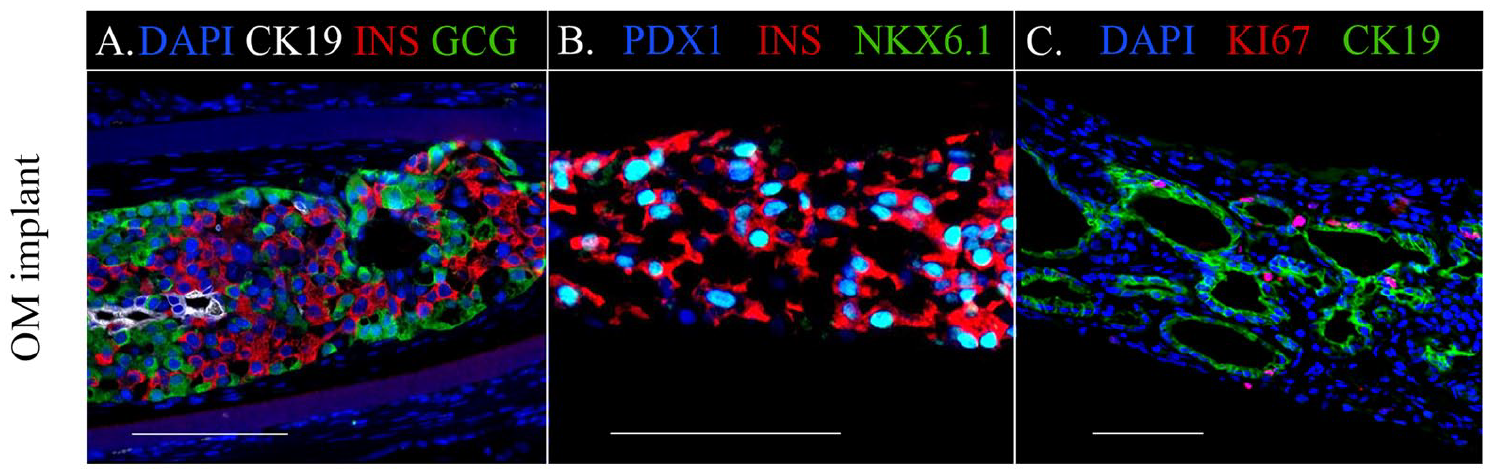
Immunostaining of donor cell-derived tissue in OM-implants of the nude rat subgroup (6/13) with highest plasma hu-C-peptide levels at PT-week 30. The inner chambers of the devices contained insulin-positive cell clusters surrounded by glucagon-positive cells in areas with few CK19-positive cells (A). The majority of insulin-positive cells exhibited nuclear staining for the transcription factors PDX1 and NKX6.1 (B). Most of cells positive for the proliferation marker Ki67 were hormone-negative but positive for the epithelial cell marker CK19; the majority of CK19-posive cells formed an epithelial layer in duct-like structures (C). Scale bar: 100 µm. NKX6.1: NK6 Homeobox 1; OM: omentum; PDX1: Pancreatic and Duodenal Homeobox 1; PT: post-transplantation.

Hematoxylin & Eosin staining of implants retrieved at PT-week 30 from OM and SC. The OM-implant belonged to the subgroup with highest plasma hu-C-peptide levels. Its inner chamber (star) with packed donor cells was surrounded by loosely organized connective tissue with signs of vascularization (A). The SC implant from a recipient without detectable plasma hu-C-peptide, exhibited packed connective tissue at both sides of the inner membranes and extending beyond the outer mesh (B). Crosses indicate the outer polyester mesh while stars indicate the inner chamber bordered by PTFE membranes. Scale bar: 100 µm. OM: omentum; PT: post-transplantation; SC: subcutis.
Glucose Responsiveness of Beta Cells Formed by hu-iPSC-PE-Implants in Omentum
The 6 OM-implants with plasma hu-C-peptide >0.1 ng/ml exhibited glucose stimulated levels following IP-glucose load. These levels were, however, more than 10-fold lower than those associated with metabolic control in mice (>6 ng/ml), which can indicate a shortage in beta cell number and/or beta cell function. We therefore measured insulin secretory responses during perifusion of cell preparations isolated from OM-devices in rats and compared them with those reported for cells which established metabolic control in mice. At start, insulin content of these beta cells (10.3 µg/106 beta cells) was similar to the values measured in the mouse experiment 7 . During perifusion, they exhibited rapid insulin secretory responses to pulses of 10 mM and 20 mM glucose, and addition of glucagon plus IBMX, with similar amplitudes (5 and 8-fold above baseline) as in the previous report (Fig. 5). These data indicate that functional beta cells with adequate qualitative properties have been formed in devices placed on OM but that their mass was insufficient for metabolic control under the present conditions.

Glucose secretory response of hu-iPSC-PE generated beta cells in OM-implants retrieved at PT-week 30 from subgroup (11/13) with detectable plasma hu-C-peptide levels. Tissue was retrieved from the inner chambers and gently dispersed before placement on a perifusion column for analysis of insulin secretory responses. Perifusate was collected over 1 min fractions at baseline (2.5 mM glucose) and during glucose pulses with 5, 10, or 20mM glucose, the latter condition in absence and presence of glucagon (10 nM) with or without IBMX (50 µM). Samples are assayed for insulin and data for 5 experiments expressed as mean + SD. Statistical significance of differences in secretory response was calculated by 1-way ANOVA with Tukey’s multiple comparisons test, comparing pulse periods with baseline (*P < 0.01, ** P < 0.0001) and addition of glucagon plus IBMX with release at 20 mM glucose († P < 0.01). ANOVA: analysis of variance; hu-iPSC-PE: human induced pluripotent stem cell derived pancreatic endoderm; OM: omentum; PT: post-transplantation; GCG: glucagon; IBMX: 3-isobutyl-1-methylxanthine.
Discussion
The present study demonstrates that the innate reactivity of nude rats interferes with survival of device-encapsulated hu-iPSC-PE implanted in the subcutaneous space. This finding is clinically relevant since such reactivity is known to occur in man. It supports use of the nude rat model to further examine cell therapy products that gave promising data in immune-compromised mice. The surgical lesion for placement of the device appeared responsible for formation of dense fibrosis with little vascularization, resulting in a major loss of implanted cells soon after transplantation. Prior creation of a pouch by a retrievable silicon sheet decreased tissue damage when a device was placed at a later time but had resulted in a fibrous capsule with similar consequences instead of forming a border with vascularized loose connective tissue as in mice7,15,16; such capsule formation has been previously reported for polymers that were subcutaneously injected in nude rats 17 .
The OM allowed placement of the device without incision of the implant site thus avoiding the lesioned tissue environment that may have impaired survival of implanted cells in the SC. This site is also known for its vascularizing potential18,19, which may be reflected in the higher density of surrounding endothelial cells. It is, however, not passive in terms of foreign body reactivity: the density in CD68+ cells around the outer membranes is similar to that for SC-devices, possibly marking a sustained host reactivity. Implants in the OM did loose in average 50% of their pancreatic progenitor cells during the first 3 weeks, but their remaining cells appeared sufficient to generate a human beta cell mass with circulating hu-C-peptide in 11/13 recipients over a period of 30 weeks. Cell loss was considerably higher in the SC (>80%) and therefore responsible for the failure to generate a beta cell mass at this site.
The beta cell mass formed in OM-implants was insufficient to achieve the plasma hu-C-peptide levels (>6 ng/ml) and associated metabolic control as previously reported for SC-implants of the same device and initial cell content in mice 7 . This difference can in part be attributed to the 10-fold lower initial cell dose (25 x 106 PE cells per kg BW versus 250 x 106 in mice); this cell dose in rats is in the range of that used in clinical islet cell transplantations10,20,21. When extrapolating the plasma hu-C-peptide levels measured in rat (0.05–0.4 ng/ml) to the 10-fold smaller circulating volume of mice (calculated 0.5–4 ng/ml), they fall in the range of the mouse study which was also found to be wide. The size of the beta cell mass in the OM-devices from rats did fall in the range measured in SC-devices from mice, however, at the lower end where sizes were not associated with metabolic control 7 . The beta cells formed in these OM-devices exhibited similar functional characteristics as those retrieved from mice with metabolic control, in particular insulin content, glucose responsiveness and secretory amplitudes during glucose pulses in perifusion. These data indicate that the failure to establish metabolic control by OM implants in rats is caused by an insufficient number of formed beta cells, not by an inadequate glucose responsiveness of the formed beta cells.
The shortcoming in beta cell mass formation in devices placed in rats is primarily caused by high cell loss shortly after implantation and its insufficient compensation by expansion of residual cells. Since loss of cells in OM was less severe than in SC, cell mass in this site could reach the same size at PT-week 30 as at start; it remained, however, 3-fold lower than that achieved in mice. The major early cell loss was likely caused by the foreign body reaction which created a hypoxic region for the encapsulated cells which, as such, are already deprived of vascularization. We will examine whether it can be prevented by local addition of an inflammasome inhibitor 22 and/or microvessels 23 . Another approach can aim for modifications in the device membranes which are considered to initiate the local reactivity. Along this line ViaCyte has modified the membranes of their encapsulated hu-PSC-PE product to allow capillary ingrowth. This PEC-Direct combination product is currently under clinical investigation24–26. It is, however, not immune-protective since its membranes are open to recipient cells.
The hypoxic environment of the present implants in nude rats may also have been responsible for a relatively lower formation of beta cells than of alpha cells as indicated by the lower ratio of beta cell over alpha cell mass in rats (average of 0.30-Table 2) than in mice 7 (average of 0.57). This ratio may, in more general terms, depend on microenvironmental influences and thus be used to identify adaptations that favor differentiation toward a beta cell mass. Further studies should also investigate the biologic significance of the formed alpha cell mass. The present data show that the glucagon content of the OM-implants with highest beta cell function (6/13 recipients) is higher than that in the corresponding pancreas, and equal to that of the pancreas in control rats. It is conceivable that locally released glucagon sustains beta cell survival and function in the implant as is also the case in isolated rat islet preparations 27 . It is likely that the alpha cell mass in the implant is a source for the higher circulating glucagon levels that we previously reported for mice with device-encapsulated hu-PSC-PE implants4,5,7. It may also be responsible for a down-regulation of pancreatic glucagon reserves, for which the present study lends suggestive data.
A beta cell implant on OM offers other advantages than placement without tissue lesions. It can benefit from its vascularization potential18,19,28–31. Furthermore, the OM can release insulin directly in the portal circulation, as do pancreatic beta cells. The site allows local adaptations such as addition of microenvironmental components that help survival and function of the beta cells including protection against immune reactivity. Implants are accessible for safely taking biopsies, and being outside a vital organ, can be completely resected when needed for safety. These considerations have led to investigating this site for non-encapsulated islet cell implants in animal models and in clinical pilot studies9,11,12,32–34. Clinical studies demonstrated the technical feasibility of implantations and explantations, and its perspective for analyzing composition of implants and their microenvironment. Our observations support further investigations on the OM as implant site for device-encapsulated human stem cell generated cells. The nude rat model can be used to compare their dose-efficacy with that of human islet cell implants.
Supplemental Material
sj-tif-1-cll-10.1177_09636897231167323 – Supplemental material for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats
Supplemental material, sj-tif-1-cll-10.1177_09636897231167323 for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats by Jolien R. Nijns, Ines De Mesmaeker, Krista G. Suenens, Geert M. Stangé, Kaat De Groot, Maria Marques de Lima, Marine R. C. Kraus, Bart Keymeulen, Wim Waelput, Daniel Jacobs-Tulleneers Thevissen and Daniel G. Pipeleers in Cell Transplantation
Supplemental Material
sj-tif-2-cll-10.1177_09636897231167323 – Supplemental material for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats
Supplemental material, sj-tif-2-cll-10.1177_09636897231167323 for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats by Jolien R. Nijns, Ines De Mesmaeker, Krista G. Suenens, Geert M. Stangé, Kaat De Groot, Maria Marques de Lima, Marine R. C. Kraus, Bart Keymeulen, Wim Waelput, Daniel Jacobs-Tulleneers Thevissen and Daniel G. Pipeleers in Cell Transplantation
Supplemental Material
sj-tif-3-cll-10.1177_09636897231167323 – Supplemental material for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats
Supplemental material, sj-tif-3-cll-10.1177_09636897231167323 for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats by Jolien R. Nijns, Ines De Mesmaeker, Krista G. Suenens, Geert M. Stangé, Kaat De Groot, Maria Marques de Lima, Marine R. C. Kraus, Bart Keymeulen, Wim Waelput, Daniel Jacobs-Tulleneers Thevissen and Daniel G. Pipeleers in Cell Transplantation
Supplemental Material
sj-tif-4-cll-10.1177_09636897231167323 – Supplemental material for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats
Supplemental material, sj-tif-4-cll-10.1177_09636897231167323 for Comparison of Omentum and Subcutis as Implant Sites for Device-Encapsulated Human iPSC-Derived Pancreatic Endoderm in Nude Rats by Jolien R. Nijns, Ines De Mesmaeker, Krista G. Suenens, Geert M. Stangé, Kaat De Groot, Maria Marques de Lima, Marine R. C. Kraus, Bart Keymeulen, Wim Waelput, Daniel Jacobs-Tulleneers Thevissen and Daniel G. Pipeleers in Cell Transplantation
Footnotes
Acknowledgements
The authors thank their collaborators at Diabetes Research Center for their continuous contributions to the Beta Cell Therapy program at Vrije Universiteit Brussel and its Universitair Ziekenhuis Brussel.
Authors’ Contributions
J.N.: Conception and design, collection of data, data analysis and interpretation, manuscript writing, final approval of manuscript. I.D.M, K.S, G.S., K.D.G., W.W.: Collection of data, data analysis and interpretation, final approval of manuscript. B.K., D.J.T.T, M.K, M.M.L.: Provision of materials, final approval of manuscript. D.P.: Conception and design, financial support, data analysis and interpretation, manuscript writing, final approval of manuscript.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human subjects. All experimental procedures involving animals were conducted in accordance with the European Community Council Directive (8.6/609/EEC).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research Ethics and Patient Consent
All experimental procedures involving animals were conducted in accordance with the European Community Council Directive (8.6/609/EEC). The study was approved by the ethical committee of Vrije Universiteit Brussel (Belgium). Statement on informed consent: not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work has been supported by grants from the European Commission (H2020 681070) and the Juvenile Diabetes Research Foundation (2-SRA-2019-708-S-B).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
