Abstract
To address the ongoing shortage of organs available for replacement, xenotransplantation of hearts, corneas, skin, and kidneys has been attempted. However, a major obstacle facing xenotransplants is rejection due to a cycle of immune reactions to the graft. Both adaptive and innate immune systems contribute to this cycle, in which natural killer cells, macrophages, and T-cells play a significant role. While advancements in the field of genetic editing can circumvent some of these obstacles, biomarkers to identify and predict xenograft rejection remain to be standardized. Several T-cell markers, such as CD3, CD4, and CD8, are useful in both the diagnosis and prediction of xenograft rejection. Furthermore, an increase in the levels of various circulating DNA markers and microRNAs is also predictive of xenograft rejection. In this review, we summarize recent findings on the advancements in xenotransplantation, with a focus on pig-to-human, the role of immunity in xenograft rejection, and its biomarkers.
Keywords
Introduction
The improved life expectancy of humans over the past decades has increased the prevalence of a growing number of chronic diseases 1 . The increasing application of organ transplantation, the last resort and definitive treatment for end-stage organ failure, has resulted in a disparity in the supply and demand for such organs 1 . Therefore, xenotransplantation has become an appealing solution to overcome this obstacle 2 . The U.S. Food and Drug Administration defines xenotransplantation as “any procedure that involves the transplantation, implantation or infusion into a human recipient of either (a) live cells, tissues, or organs from a nonhuman animal source, or (b) human body fluids, cells, tissues or organs that have had ex vivo contact with live nonhuman animal cells, tissues, or organs” 3 . Currently, xenotransplant use has been reported mainly for the kidneys, hearts, livers, skin, and corneas 4 .
Pigs are the species of choice to harvest organs for xenotransplantation, as they have anatomically similar organs to humans and are suitable for genetic modification 5 . They are highly bred and often consumed, clearing the way for the ethical decision to use pig organs to treat human diseases. Although the genetic discrepancies between humans and pigs are greater than those of primates, the use of primate organs is not sustainable for ethical reasons and because most primates are considered endangered 5 . Furthermore, primate organs have a substantial chance of carrying viruses that can infect humans 5 . Hence, genetic engineering techniques have been developed to decrease porcine and human genetic dissimilarities 1 , paving the road for the usage of pig organs for xenotransplants. Indeed, recent studies described two successful cases of kidney transplants from pigs in brain-dead patients 6 , and another reported a successful case of a heart transplant from a pig to a human 7 . These breakthroughs marked a great milestone for the field of xenotransplantation.
The main obstacle facing xenotransplants is immunological reactions. Although the mechanism behind hyperacute rejection (HAR) in the xenograft is well defined, the mechanisms of acute cellular rejection are not fully understood 2 . Identifying the mechanisms behind cellular rejection in xenotransplantation may be the key to the longer survival of xenotransplanted organs. Furthermore, unlike allotransplantation, there is a lack of data on standardized predictive and diagnostic markers of xenotransplantation 8 , which could permit close monitoring of xenografts 9 . In this article, we will briefly review the history of xenotransplantation, xenoantigens presenting as obstacles, and genetic modifications to overcome these obstacles. Lastly, we will highlight the role of cellular immunity activated in response to xenotransplantation and describe the immune markers used to predict and detect xenograft rejection.
A Brief History of Xenotransplantation
In the 17th century, the first reported case of xenotransplantation (and blood transfusion) to humans was conducted by Jean-Baptiste Denis who transfused the blood of a lamb into a 15-year-old male suffering from a fever 10 . Denis subsequently continued to transfuse blood from lambs and calves but with variable outcomes, so the French and English Parliaments banned transfusions for several years to come 10 .
In 1838, Sharp-Kissam performed the first corneal transplantation by implanting a pig cornea into the eye of a 35-year-old man 11 . In the 19th century, scientists began using skin xenografts from various animals, such as pigs, sheep, frogs, pigeons, and chickens, as biologic dressings 12 and bovine embryonic skin grafts as skin dressing 13 .
In the 20th century, Voronoff attempted to “rejuvenate” elderly men by conducting several chimpanzee and baboon testicular transplants 14 purportedly thereby elevating energy levels in patients. In the 1960s, Reemtsma carried out 13 chimpanzee-to-human kidney xenotransplants, most of which failed within 4–8 weeks due to rejection or infections, except for one that lasted for 9 months with no signs of rejection on autopsy 15 .
The first cardiac xenotransplantation was performed in 1964 by Hardy with a chimpanzee heart, which was too small and failed within a couple of hours 14 . During the same era, Starzl performed the first reported liver xenotransplants with limited success. However, after the introduction of tacrolimus (a potent immunosuppressor), he performed two baboon-to-human liver xenotransplants, with one patient surviving 70 days14,16. The rising incidence of type-1 diabetes and the similarities between pig and human insulin motivated contemplation of the benefit of islet xenotransplantation 14 . Thus, in 1993, Groth et al. 17 conducted the first pig-to-human islet xenotransplant but identified no clinical benefit.
Xenoantigens and Genetic Modifications
Initial attempts of porcine-to-human xenotransplants were hindered by the production of antibodies against the α-galactose-1,3-galactose (αGal) antigen 18 . Approximately 1% of naturally occurring human antibodies are directed against the αGal epitope and are responsible for HAR of pig organs perfused with human blood 18 . The discovery of the αGal epitope in pigs led to testing its expression in various animal species. In 1988, Galili et al. 19 demonstrated that the anti-Gal antibody binds to various nucleated cells of non-primate mammals, prosimians, and New World monkeys, whereas fibroblasts of humans, apes, and Old-World monkeys indicated no αGal expression.
Advancements in the field of genomic editing have then led to the development of genetically modified pigs to overcome immune rejection 1 , most notably the heterozygous αGal-knockout (GKO) pigs in 2002 and homozygous GKO pigs in 2003 20 . The elimination of αGal increased the survival of pig hearts in baboons for 2–6 months and prevented HAR 21 , but was insufficient to completely evade the immune system 6 , leading to the identification of two additional non-Gal epitopes as targets of antibodies: NeuGc and SDa22,23. These antibodies might have played a key role in the rejection of kidney xenotransplant from Gal-depleted pigs to humans 6 . Adams et al. 24 found that elimination of both Gal and SDa genes extended graft survival to up to 435 days in pig-to-primate transplants. Collectively, Gal, NeuGc, and SDa antibodies constitute more than 95% of antibodies formed against pig cells22,25 and may present as major obstacles to the advancement of clinical xenotransplantation.
Yet, emerging studies in pigs with Gal, NeuGc, and SDa knockout revealed that transplant-induced coagulopathies also hamper the success of xenotransplantation and that overexpression of human coagulation regulatory proteins in animal donors may resolve this issue 1 . Therefore, one of the main goals of genetic modulation has become regulating the coagulation dysfunction in graft recipients, such as thrombomodulin (TBM). Porcine TBM fails to successfully interact with human thrombin, leading to a procoagulation state 26 . Importantly, Miwa et al. 27 found that expression of human TBM in porcine aortic endothelial cells successfully regulated coagulation in human plasma and inhibited antibody-induced complement activation. Moreover, antibody therapy combined with expression of human TBM prevents humoral rejection and coagulation dysregulation and increases graft survival beyond 900 days in pig-to-baboon heart transplants 28 .
Another attractive candidate target for genetic modulation is the endothelial protein-C receptor (EPCR). Although the pig EPCR is compatible with human protein-C 26 , Iwase et al. 29 found a strong positive correlation between the reduction of human platelet aggregation and the expression of human EPCR in pig aortic endothelial cells. Lastly, Wheeler et al. 30 showed that expression of human CD39, which hydrolyzes ATP and ADP and prevents thrombus formation, prevented myocardial ischemia/reperfusion injury in transgenic pigs.
Other genetic modifications are also being studied in attempt to target the cellular xenograft rejection (CXR) pathways. For example, due to the incompatibility of human SIRP-α and porcine CD47 (discussed later in the article), Tena et al. 31 used porcine hematopoietic cells expressing human CD47, which significantly increased engraftment chimerism in human bone marrow. Expression of human CD47 also led to prolonged survival of pig skin grafts on baboons, with one case showing no signs of acute rejection for 53 days 32 . In conclusion, genetic modifications are key for the successful transition of xenotransplantation into clinical settings.
Tolerance Induction in Xenotransplantation
Graft recipients require a combination of intensive immunosuppressive therapy, and various attempts to reduce the dose have failed 33 . Hence, tolerance-inducing strategies are currently under development with the aim of prolonging graft survival times and, eventually, halting immunosuppressive therapy 34 . Currently, donor thymic transplantation is the most effective method of achieving tolerance in xenotransplantation 34 . Studies have demonstrated prolonged swine-to-baboon renal graft survival times of more than 6 months after GKO swine kidney and thymus transplantation35,36. In humans, Montgomery et al. 6 transplanted GKO swine thymus and kidney into two brain-dead patients; however, the follow-up period was too short for the thymus to assert its effects. Nevertheless, the thymuses were able to revascularize and maintained normal architecture.
Mixed bone marrow chimerism (MBMW), which involves the production of both donor and self hematopoietic stem cells by the recipient after non-myeloablative stem cell transplant regimens, has allowed for allogeneic transplants regardless of HLA barriers 34 . Although MBMW is successful in pig-to-mouse models, replicating such results has been difficult in pig-to-primate studies34,37. For example, Liang et al. 38 demonstrated that only 10% of swine-to-baboon MBMWs resulted in successful engraftment, with failure of engraftment associated with increased anti-non-Gal IgG levels post-transplant. Overall, further studies are needed to determine the effectiveness of thymus transplantation and MBMW in inducing tolerance.
Histological and Systemic Outcomes of Xenograft Rejection
Within minutes to hours of graft transplantation, the xenograft is destroyed by HAR, a process mediated by pre-existing αGal antibodies 1 . Binding of these antibodies leads to complement pathway activation, which causes lysis of endothelial cells 1 . Notably, for an unknown reason, the effects of antibody depletion and complement inhibition are generally more effective in heart and kidney transplants than lung and liver transplants39–41. Unlike other rejection types, grafts show no functionality when they undergo HAR 39 . Histologically, this process is characterized by massive hemorrhage and complement, immunoglobulin, and fibrin deposition 39 .
Acute humoral xenograft rejection (AHXR), also known as delayed xenograft rejection, can be initiated by naturally occurring αGal antibodies or antibodies formed after sensitization by the graft 39 . In the latter case, the antibodies may be directed against αGal or non-Gal antigens, such as NeuGc and SDa 39 . Histologically, this process resembles HAR; however, necrosis and transmural granulocyte infiltration of blood vessels may be present 39 .
Lastly, CXR may occur after a significant time lag post-xenotransplantation. Contrary to HAR and AHXR, hemorrhage and fibrin and immunoglobulin depositions are not observed. Complement depositions may be seen but are usually of low intensity 39 . The mechanisms underlying CXR will be described in the next section.
Systemically, three complications characterize xenograft recipients: immune complex diseases, coagulopathies, and infections. Due to the prominent role of antibodies in xenograft rejection, immune complex deposits may be seen in various recipient organs 39 . After porcine-to-baboon xenotransplant, Holzknecht et al. 42 detected deposits of baboon C3 and porcine von Willebrand factor in the spleens and livers of lung recipients. Interestingly, baboons that received porcine hearts and kidneys did not show such depositions. Deposits of rat IgG and IgM have also been found in the glomeruli of recipient rats after a hamster-to-rat liver transplant 43 .
Given the adverse coagulopathy observed in xenotransplant recipients, thrombotic microangiopathy (TMA) might develop as a fatal complication post-transplant resulting in thrombosis within vessels and ischemic injury 1 . Briefly, graft recipients rapidly progress into thrombocytopenia, develop schistocytes, and present with high levels of lactate dehydrogenase 44 . With the progression of TMA, a systemic consumptive coagulopathy may develop leading to recipient death 45 . However, this issue may be resolved with the rapid excision of the xenograft, inhibiting further consumption of coagulation factors and improving recipient survival 45 .
Lastly, potential transmission of pathogens is a major concern in xenotransplantation. Swine pathogens can be generally separated into four categories: pathogens that infect healthy humans, pathogens that infect human-transplant recipients, pathogens resembling those of human-transplant recipients, and swine-specific pathogens 46 . Pathogens of the third category, such as porcine cytomegalovirus (PCMV) and porcine adenovirus, have been associated with syndromic complications in pig and nonhuman primate xenograft recipients 46 . For example, PCMV is responsible for disseminated intravascular coagulation, hematuria, and reduced graft survival times in pig-to-baboon transplants47,48.
Swine-specific pathogens, such as porcine endogenous retroviruses (PERVs), are a growing area of concern due to the potential risk of silent transmission and gene alterations 46 . PERVs integrate themselves within the porcine genome and may be classified as PERV-A, PERV-B, and PERV-C 49 . PERV-A and PERV-B are present in all pig species, while PERV-C is only present in select species 50 . Recombinant PERV-A/C, which is characterized by high titer replication, has shown the capability of infecting human cells 50 . Thus, it is recommended to screen for the presence of PERV-C and only use donor pigs free of the virus 50 . To date, no literature describes PERVs in preclinical pig-to-primate models and clinical transplantations in humans, yet inactivation of the viruses may be completed using genetic modifications if needed 49 . In conclusion, it is essential to further study mechanisms that bypass the fatal complications of TMA and consumptive coagulopathy and develop screening assays for potential infective organisms.
Role of Cellular Immunity in Xenogeneic Rejection
Immune responses after xenotransplantation involve both the innate and immune adaptive systems 1 . Although the main cells involved in allograft rejection are cytotoxic T-lymphocytes, xenograft reactions activate primarily neutrophils, natural killer (NK) cells, and macrophages 51 . Neutrophils rapidly infiltrate both cellular and organ grafts52,53. Upon activation, neutrophils release neutrophil extracellular traps (NETs), network structures that induce damage through the generation of reactive oxidative species (ROS), and release of digestive enzymes2,54,55. Furthermore, macrophages recognize NETs as damage-associated molecular patterns (DAMPs) that cause the release of cytokines and inflammatory markers (Fig. 1A) 54 .
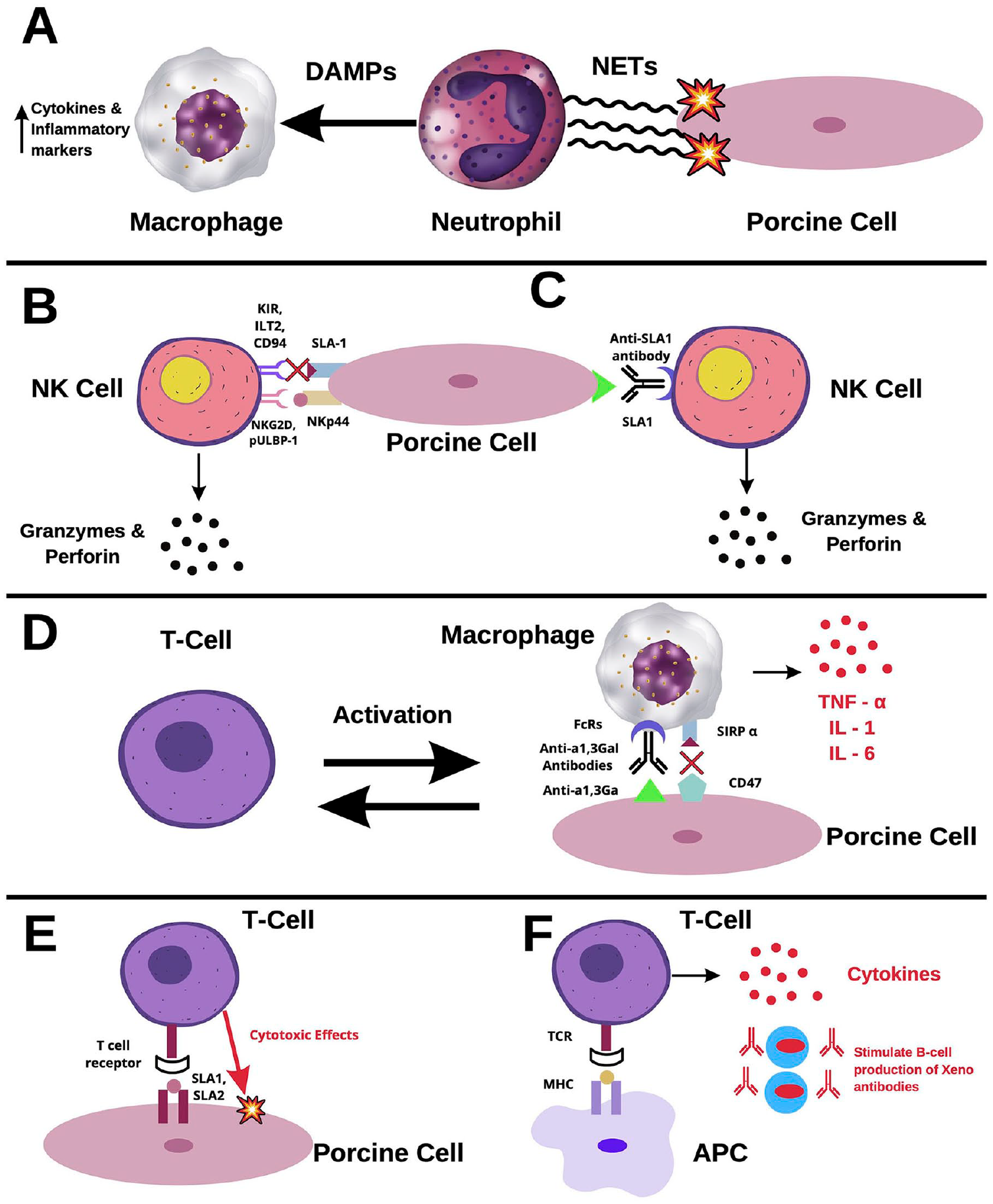
Cellular rejection in xenotransplants. (A) Neutrophil-mediated rejection. Upon activation, neutrophils release tubular networks known as NETs. NETs induce damage to xenograft cells through ROS and are recognized by macrophages as DAMPs. Binding of DAMPs to macrophages triggers the release of cytokines and inflammatory markers. (B) NK cell-mediated rejection via the direct pathway. Stimulating receptors, such as NKG2D and pULBP-1, bind to NKp44 and an unidentified molecule, respectively. Once activated, NK cells release granzymes and perforin, which induce damage in xenograft cells. The inhibiting receptors, KIR, ILT2, and CD94, do not recognize SLA-1 well in porcine cells. Therefore, there is a lack of inhibitory feedback of NK cells in xenografts. (C) NK cell-mediated rejection via the ADCC pathway. Interactions between FcRs and xenoantibodies lead to activation of NK cells. Among the antibodies recognized by NK cells is anti-SLA1. Binding on NK cells to anti-SLA1 induces the release of granzymes and perforin. (D) Macrophage-mediated rejection. Macrophages recognize anti- α1,3Ga antibodies through FcR, inducing the release of inflammatory cytokines, such as TNF-α, IL-1, and IL-6. Macrophages activate circulating T-cells, which further activate more macrophages. CD47 is an important receptor in the pathway of macrophage inhibition. However, it does not readily recognize its ligand, SIRP-α, in porcine cells, causing ineffective inhibition. (E) T-cell-mediated rejection via the direct pathway. SLA1 and SLA2 bind to T-cell receptors, triggering the release of cytokines and mediating direct cytotoxic effects. (F) T-cell-mediated rejection via the indirect pathway. Recipient antigen-presenting cells (APC) express xenogeneic antigens, activating CD4+ T-cells. Activated T-cells induce a cascade of antibody production and B-cell activation. NET: neutrophil extracellular traps; ROS: reactive oxidative species; DAMP: damage-associated molecular patterns; NK: natural killer; NKG2D: natural killer group-2D; pULBP-1: porcine UL16-binding protein-1; KIR: Killer Ig-like Receptor; ILT2: Ig-like transcript-2; ADCC: antibody-dependent cellular cytotoxicity; TNF-α: tumor necrosis factor; IL: interleukin; SIRP-α: signaling regulatory protein; CD94: cluster of differentiation-94; SLA-1: swine leukocyte antigen-1.
Numerous studies have reported infiltration of NK cells within xenografts, implicating them in xenograft rejection51,56. These cells induce rejection by either direct cytotoxicity or antibody-dependent cellular cytotoxicity (ADCC). The direct pathway is tightly regulated by stimulating and inhibiting receptors. NK-stimulating receptors, such as natural killer group-2D (NKG2D) and porcine UL16-binding protein-1 (pULBP-1), bind to the pig ligand NKp44 and an unidentified molecule, respectively57,58, leading to the release of lytic granules such as granzymes and perforin (Fig. 1B) 59 . Contrarily, inhibiting receptors, killer Ig-like receptor (KIR), Ig-like transcript-2 (ILT2), and CD94, do not readily recognize swine leukocyte antigen-1 (SLA1), the porcine major histocompatibility complex-1 molecule, dampening NK inhibition in xenografts 58 . In the ADCC pathway, antibodies deposited on the surface of xenograft cells are recognized by NK cells via interactions with FcRs 1 . Upon activation, NK cells release granzymes and perforin, leading to apoptosis of the targeted cells. Furthermore, NK cells recognize anti-SLA1 antibodies, activating the ADCC pathway (Fig. 1C) 25 .
Macrophages have also been implicated in the rejection of cellular grafts and organ grafts 60 . Peterson et al. 61 have shown that xenogeneic αGal is a direct ligand for human monocytes. In addition, immune complexes of porcine cells with xenogeneic antibodies such as anti- αGal antibodies bind to the Fcγ receptor (FcγR) and produce an activation signal 62 . Once activated, macrophages contribute to a vicious cycle of xenograft destruction, where they are activated by T-cells and, in turn, activate more T-cells 63 . Furthermore, macrophages induce direct cytotoxicity through the production of cytokines, such as tumor necrosis factor (TNF)-α, interleukin-1 (IL-1), and IL-6 (Fig. 1D) 64 . Regarding inhibitory feedback, the signaling regulatory protein (SIRP-α)-CD47 pathway is an important regulator of macrophage activity1,65. The CD47 pathway has been shown to regulate the homeostasis of erythrocytes, platelets, and hematopoietic stem cells 66 . CD47 is recognized by SIRP-a as a “do-not-eat” signal, thus inhibiting phagocytic activity 65 , a signal utilized by cancer cells to evade immune surveillance. However, Wang et al. 67 have reported interspecies incompatibility of CD47 after xenotransplantation, which leads to ineffective inhibition of macrophages.
Like in allograft transplantation, T-cell activation is mediated in xenograft rejection through direct and indirect pathways1,68. Through the direct pathway, interactions between SLA-1 and -2 complexes with T-cell receptors lead to activation of the adaptive immune response against the xenograft (Fig. 1E) 1 . In the indirect pathway, the presentation of xenogeneic antigens by recipient cells leads to the activation of CD4+ T-cells, instigating a cascade of antibody production and B-cell activation (Fig. 1F) 1 . Lastly, cytokines produced through this mechanism significantly enhance the cytotoxicity of NK cells and macrophages 69 .
As mentioned above, B-cells play a role in the rejection of xenografts. In fact, B-cell depletion increased survival time by 8 months after heart transplantation from pigs to baboons, suggesting a significant role of B-cells in xenotransplant rejection, specifically, delayed xenotransplant rejection 70 . B-cells produce the anti-Gal antibody which targets Gal antigens expressed in pig tissues 71 and binds to its antigen, leading to complex formation. Indeed, depletion of the anti-Gal antibody leads to more favorable outcomes, further implicating B-cells in the rejection of xenotransplants71–73. The phenotypic characteristics of anti-Gal antibody-producing subpopulations of B-cells in humans are not clearly identified 72 . One study has shown that splenic B-cells produce anti-Gal antibodies, whereas peritoneal B-cells do not, although they do express anti-Gal receptors 73 . Conclusively, both innate and adaptive immune systems play a significant role in xenotransplant rejection.
Biomarkers of Xenograft Rejection
A lack of standardization among methods used to monitor xenograft rejection drives a crucial need to identify markers that can be used to diagnose and predict rejection 8 . As listed in Table 1, Montgomery et al. 6 observed focal C4d deposition at 54 hours post-pig-to-human kidney transplant but no other significant histologic or immunologic indications of antibody-mediated injury. Zhou et al. 8 also found that CD68+ macrophages and some CD3+ T-cells infiltrated xenografts in pig-to-mouse models on day 3 after transplantation.
Potential Biomarkers of Xenograft Rejection.

NK: natural killer; TLR2: toll-like receptor-2; cpsDNA: circulating pig-specific DNA; cfDNA: cell-free DNA; mRNA: messenger ribonucleic acid; CCL2: C–C motif chemokine ligand-2; CXCL8: C-X-C motif chemokine ligand-8; DNA: deoxyribonucleic acid.
Given that NK cells are a major type of infiltrating cells identified in xenografts51,56,81, Lin et al. 74 used markers such as NK1.1 and DX5 to identify NK cells in pig-to-mouse models. Using a modified ADCC assay, Chen et al. 76 found that toll-like receptor-2 (TLR2) mRNA and protein were also upregulated in porcine iliac artery endothelial cells after exposure to human serum. Furthermore, the levels of porcine pro-inflammatory chemokines CCL2 and CXCL8 also increased through a TLR2-mediated pathway 76 . These findings suggest that blockade of TLR2 may prolong xenograft survival.
Graft biopsies may cause infection, scarring, or induce rejection via immune activation after injury 75 . Therefore, it is important to identify noninvasive markers of rejection for apply in clinical xenotransplantation. Montgomery et al. 6 detected IgM and IgG antibodies directed against non-α-Gal antigens in the serums of pig-to-human kidney transplant patients. Because IgM is confined to the vascular space, its removal via plasmapheresis can, theoretically, be incorporated in future xenotransplantation trials involving humans 6 .
Circulating DNA is released upon cell death or apoptosis, which are considered classic findings in xenotransplantation 8 . The release of circulating pig-specific DNA (cpsDNA) reflects the infiltration of immune cells in the graft and precedes the production of anti-pig IgM/IgG antibodies in pig-to-mouse models 8 . Furthermore, cpsDNA also provided comparable results in monkeys, suggesting potential feasibility in clinical settings 8 . Similarly, cell-free DNA (cfDNA) levels also correlate with tissue injury in xenograft models 77 .
While data regarding organ-specific microRNAs (miRNA) in xenotransplant remain limited, they have shown promising usage as biomarkers of rejection 78 . In a pig model of acute liver failure, recipient plasma levels of various pig-derived miRNAs, including ssc-miR-122, ssc-miR-192, and ssc-miR-124-1, were associated with liver, kidney, and brain injury, respectively 82 . Most miRNAs are conserved among species, limiting their usage in the field of xenotransplantation78,83. However, some miRNAs, such as the pig-specific ssc-miR-199b, could be useful as they may be differentiated from its human counterpart and are expressed in the liver, heart, and lung 78 .
One study also observed increased levels of miR-146a and miR-155 in cardiac xenotransplants and evaluated the effect of immunosuppressive treatment on their expression in cardiac xenotransplant models from mouse to rat. Compared to immunosuppressed animals, Zhao et al. 79 found a significant decrease in miR-146a levels and increase in miR-155 expression, changes that lead to a pro-inflammatory state in recipients. Notably, miR-146a plays a role in inhibiting inflammatory conditions by targeting various NF-κB pathways 84 , and miRNA-155 has also been reported as a promoter of TNF-α expression 85 . Collectively, these findings may provide insight into the potential use of miRNAs as biomarkers and targets of RNA-interfering immunotherapy.
A recent study in nonhuman primates also reported elevated levels of C3 levels in the aqueous humor preceding rejection 80 . Lastly, high CD4+/CD8+ blood cell ratios are correlated with shorter graft survival times in pig-to-non-human islet transplants 86 . However, further studies are required to assess the sensitivity and specificity of any proposed markers.
Conclusion
In light of recent organ shortages, xenotransplantation could provide a much-needed solution for patients requiring organ transplants. Historically, the major obstacle facing xenotransplantation from pig sources was the presence of the αGal epitope. However, genetic modulation allowed development of pig models devoid of this epitope. This advancement has prolonged xenograft survival in humans and illuminated other epitopes, such as NeuGc and SDa, which induce immune rejection. Thus, studies aimed at identifying the immune mechanisms that lead to rejection. NK cells, macrophages, and T-cells have been identified as key players in the pivotal role of the immune system in the rejection of xenografts.
Furthermore, the methods used to identify rejection of xenotransplants are based on those used in allotransplantation due to lack of standardization. T-cell markers, such as CD3, CD4, and CD8, seem promising as predictive and diagnostic rejection markers. Markers of cellular injury, such as cpsDNA and cfDNA, have also been identified as early predictive biomarkers of rejection. Various miRNAs have also been recognized as rejection markers and possible targets for the development of new immunotherapy strategies. Lastly, the detection of non-α-Gal IgG and IgM antibodies has recently been used as a marker for pig-to-human kidney transplant rejection. Given the recent advancements in the field, xenotransplantation might ultimately become a viable clinical option. Nonetheless, further progress is needed to overcome the complications of TMA and consumptive coagulopathy. Furthermore, more studies are required to compare various markers and identify a “gold standard” rejection marker in xenotransplantation.
Footnotes
Ethical Approval
This manuscript is a review article and does not involve any ethical issues. All authors reviewed and approved the final version of the manuscript.
Statement of Human and Animal Rights
This study did not involve any human or animal subjects.
Statement of Informed Consent
This article did not involve any human subjects and, thus, informed consent is not applicable.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Lerman is an advisor to AstraZeneca, CureSpec, Butterfly Biosciences, Beren Therapeutics, and Ribocure Pharmaceuticals. The authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by NIH grant numbers: DK120292, DK122734, HL158691, and AG062104.
