Abstract
Aptamers are single-stranded nucleic acids (DNA, short RNA, or other artificial molecules) produced by the Systematic Evolution of Ligands by Exponential Enrichment (SELEX) technology, which can be tightly and specifically combined with desired targets. As a comparable alternative to antibodies, aptamers have many advantages over traditional antibodies such as a strong chemical stability and rapid bulk production. In addition, aptamers can bind targets in various ways, and are not limited like the antigen–antibody combination. Studies have shown that aptamers have tremendous potential to diagnose and treat clinical diseases. However, only a few aptamer-based drugs have been used because of limitations of the aptamers and SELEX technology. To promote the development and applications of aptamers, we present a review of the methods optimizing the SELEX technology and modifying aptamers to boost the selection success rate and improve aptamer characteristics. In addition, we review the application of aptamers to treat bone diseases.
Introduction
Although aptamers have only 20–100 bases, they can fold into special spatial structures and connect to targets via electrostatic interactions, van der Waals forces, and hydrogen bonding, or fold into specific three-dimensional (3D) architectures such as hairpins, inner loops, and junctions. Different structures allow aptamers to connect to different targets, from ions to whole cells, even living animals1–4. Compared with traditional antibodies, aptamers have the following advantages: (1) high-volume chemical synthesis production, strong chemical stability, low molecular weight, and facile modification 5 ; (2) low immunogenicity and cytotoxicity, penetrating tissues more efficiently 6 ; (3) the potential to identify new biomarkers because aptamers can directly combine with targets 7 ; (4) low dissociation constants (from picomolar to micromolar), rapid bulk production with minimal product differentiation 8 ; (5) plasticity allowing RNA aptamers to capture and fold into structures that are similar to protein structures9,10.
Compared with the antigen–antibody combination, aptamers can be internalized into cells by endocytosis and pinocytosis. In addition, the stable combination, specificity, and diverse targets provide aptamers with broad application prospects in the fields of therapeutics and diagnostics11,12.
The Systematic Evolution of Ligands by Exponential Enrichment (SELEX) technology was first proposed in the 1990s13,14. A target aptamer can be obtained from at least 1,012 complete random sequence oligomers (DNA or RNA libraries) via multiple rounds of screening. The random library contains a central random sequence and a fixed sequence at both ends. Aptamers have since become increasingly important in the field of targeted therapy15,16. With 30 years of rapid development, many kinds of SELEX technologies have been derived such as immunoprecision combined SELEX (ip-selex), capture SELEX, cell SELEX, Icell-SELEX, microfluidic SELEX (m-selex), capability electronics SELEX (ce-selex), atomic force microcopy SELEX (AFM-SELEX), artistically expanded genetic information system SELEX (aegis-selex), animal SELEX, and protein SELEX17–20. In general, the SELEX procedure contains a series of incubation, binding, partitioning, amplification, cloning, and sequencing steps (Fig. 1). A nucleic acid library that contains 1,014–1,015 single-stranded random oligonucleotides is incubated with targets in a specific environment. During the incubation, partial sequences are bound to the targets, whereas other sequences are weakly combined or not bound. The bound oligomers are then separated and amplified by polymerase chain reaction (PCR). After several cycles, ideal sequences with high affinity and specificity are cloned and sequenced 21 .

Basic SELEX process 21 .
Limitations of the SELEX Technology and Improvement Measures
There are some barriers that hinder the development and applications of aptamers such as the low success rate, low aptamer affinity, pharmacokinetics, and serum stability. Therefore, it is important to improve the success rate and parameters of SELEX to promote clinical applications.
Design of the Oligonucleotide Library and Ionic Strength
Because DNA/RNA aptamers only contain four kinds of bases, the proportion of four/five connection structures is less than 1%. In addition, lacking positively charged groups (e.g., lysine and arginine), carboxylate groups (e.g., aspartate and glutamate), and a general purpose acid–base catalyst group (histidine) lead to poor affinity and diversity of aptamers. These factors hinder the progress of aptamers replacing protein antibodies. Therefore, increasing the chemical diversity and complexity of aptamers may help to improve their applicability22,23.
Increasing the composition and sequence length of the starting oligonucleotide pool can enhance the structural diversity and activity of aptamers24–26. Unnatural bases have been used to enhance the affinity and stability of aptamers 27 ; in screening specific aptamers of von Willebrand factor A1-domain (vWF), 7-(2-thienyl) imidazo [4,5-b] pyridine (DS) was randomly inserted into the DNA library as an unnatural base to form a completely random library. Most enriched sequences contained a DS base, indicating that the natural sequence was removed during the enrichment process, and an aptamer with higher affinity can be obtained 28 . However, longer randomized regions tend to misfold, aggregate, or cover the bioactive sequence, and the diversity of these motifs may not increase 29 .
In addition to changing the length of random regions, the introduction of heterogeneous nucleic acids can also improve the aptamer affinity and diversity, such as connecting, locking nucleic acids (LNAs), or threonine ribose, which can improve the affinity and enzymatic stability of the aptamer30,31. Among these measures, LNAs function by locking the furanose ring of LNA nucleotides into an RNA-mimicking N-type conformation 32 . The Slow Off-rate Modified Aptamer, a new kind of aptamer modified by amino side chains at the 5′ uracil position, enables efficient selection of high-affinity aptamers. Of the side chains, aromatic functional groups (such as those similar to phenylalanine and tryptophan side chains) or bicyclic aromatic groups (such as naphthalene) are the most effective33,34. For example, in the introduction of foreign functional groups, aptamers targeting camptothecina have been selected by modifying the DNA pool containing (E)-5-(2-(N-(2-(N6-adeninyl)ethyl))carbamylvinyl)-uracil bases 35 . In addition, to obtain the best aptamer, Andrew et al. 36 reported aptamer affinity maturation by resampling. This new approach has two stages. The first stage is intended to generate an aptamer library using novel software. The second stage is intended to synthesize the aptamer library onto a DNA microarray. The novel aptamer created by this approach exhibited significant adaptation. Generally, SELEX is a process that changes with time; for the initial cycles, longer incubation times and less stringent conditions are conducive to increasing the “selection pressure” 37 .
Metal ions greatly influence the structure, stability, and biological properties of G-quadruplexes, and they can weaken the electronegativity of aptamers, induce the formation of a secondary structure, and improve their affinity. In addition to K+ and Na+, alkaline-earth metal ions, such as Mg2+ and Ca2+, are also involved in the formation of the duplex secondary structure38,39. However, at a higher concentration, ions will attenuate the interaction signals because excess metal ions can lead to competitive binding of aptamers between targets 40 . Cationic species influence the charge stability and binding sensitivity differently. Mg2+ has a smaller ionic radius and greater positive charge than Na+, which can rapidly reduce the electronegativity of aptamers 41 .
Not all ions have a catalytic effect. For example, the sensitivity of the aptamer decreases with increasing concentration of NaCl (X = 0–500 mM) 42 .
Nucleic Acid Separation and Sequencing
It is important to easily collect aptamers to improve screening efficiency. Various methods have been applied for this purpose such as affinity chromatography, magnetic beads-based separation, and surface plasmon resonance43–45. These technologies can obtain aptamers quickly; however, they are vulnerable to various external environments. The micromagnetic separation (MMS) chip, an improved microfluidic separation device, can precisely control the hydrodynamic and magnetophoretic trapping forces. Aptamers with a Kd value of 25–65 nm can be obtained in three rounds using the MMS device. Importantly, this device is not influenced by environmental conditions such as solution composition, ion concentration, pH, and temperature 46 .
Conventional PCR can efficiently amplify target-bound sequences. However, because of the complexity of the oligonucleotide pool, the difference in amplification efficiency may sacrifice the best sequence and the technique is prone to produce by-products 47 . Using a capillary electrophoresis partitioning technique, non-SELEX can be conducted without an amplification step, which means a faster process and fewer by-products. After non-SELEX, high-throughput sequencing (HTS) is performed 48 . HTS is more widely used than low-throughput sequencing approaches to thoroughly analyze the library. HTS can analyze millions of sequences and provide an accurate method to verify aptamers and avoid potential PCR artifacts in the amplification step. Thus, enriched sequences at very low percentage (below 1%) may be detected and a shorter time is needed to obtain aptamers with high affinity49,50. However, an advanced technique needs to reduce millions of sequences to a few candidate aptamers because of the large datasets of HTS. There are currently many calculation methods that can be used to process HTS data. Galaxy Workflows, a semiautomated aptamer HTS data analysis tool, can quickly and effectively collect possible targets and remove unsatisfied aptamers. Importantly, operators can use this tool skillfully and accurately without a thorough understanding 51 . AptaMut and AptaCluster are part of the advanced computational analysis software suite AptaTools. The AptaMut procedure is designed to predict mutated sequences that can help to discover important features related to binding, such as the structural stability or sequence properties. The AptaCluster algorithm allows for efficient clustering of whole pools within hours 52 . Moreover, a novel method has been designed to identify high-binding-affinity aptamers. RNA aptamer Ranker (RaptRanker) can analyze HT-SELEX data by scoring and ranking. With this method, unique sequences and secondary structure features are determined, and all subsequences can be concentrated by similarity. High-binding-affinity aptamers are then selected by calculating the average motif enrichment 53 .
Characteristic Detection of Aptamer Binding Targets
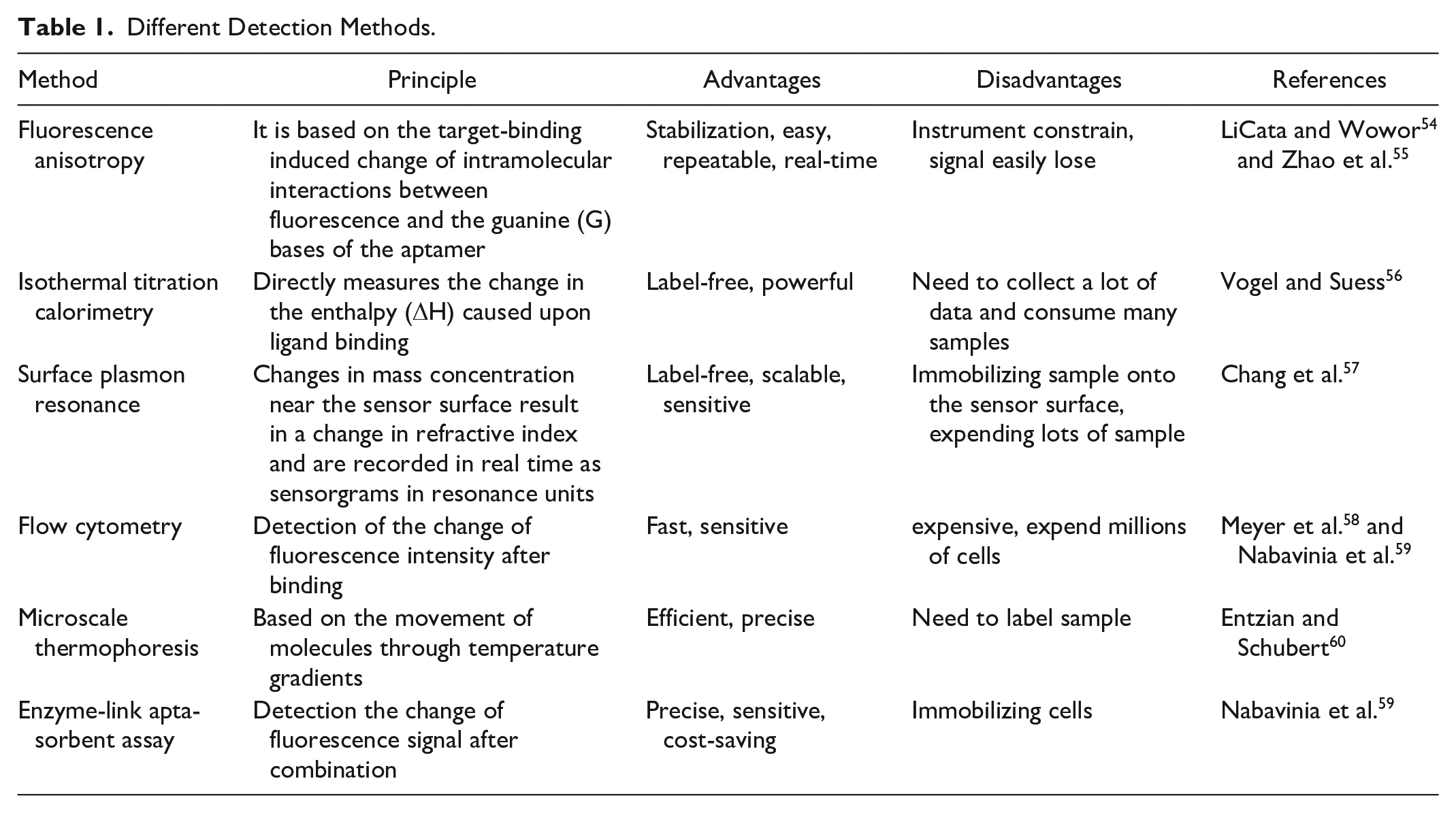
Characteristic detection of aptamers and targets is vital to understanding aptamers and designing subsequent experiments, including affinity, kinetics, and specificity. There are various detection methods with different features (Table 1).
Different Detection Methods.
Aptamer Modifications
Although aptamers have many advantages compared with antibodies, aptamers produced by conventional SELEX have nuclease sensitivity, a fast filtration rate, and low quality that impede their applications 61 . As a result, chemical modifications are needed to address these issues.
Most nucleases attack the phosphodiester bond by polarizing 2′-hydroxy to hydrolyze aptamers. Therefore, the modification of 2′-OH is very important for aptamer stability. Currently, 2′-fluoro (2′-F), 2′-amino (2′-NH2) ribose groups, or 2′-O-methyl on the pyrimidine residues are known to resist nuclease by modifying sugars 62 . Phosphorothioates, known as thio aptamers, have been produced by placing some oxygen atoms on the phosphate backbone with sulfur atoms 63 . An inverted 5′-terminal residue was introduced into aptamers by a phosphodiester bond to form 3′-inverted dT-modified aptamers that can remain longer in serum 64 . In addition to 3′-inverted dT, 3′-biotin can also resist the digestion of 3′-exonuclease and slow the clearance rate of aptamers 65 . An aptamer with better stability and affinity can be obtained by transforming the linear DNA library into a circular library after terminal interconnection 66 . Fully modified nucleic acids (100% 2-O-methyl or 2′-O-methyl A, C, and U in combination with 2′-fluoro G, termed fGmH) have the slowest clearance rate 67 .
Polyethylene glycol (PEG), a non-toxic and non-immunogenic polymer, prolongs the half-life of aptamers in vivo and enhances the activity of aptamers68,69. In the aptamer-based drug, macugen, the PEG chain is introduced at the 5′ end after inserting 2′-fluoro pyrimidine and 2′-O-methyl purines, and its activity and tissue residual time distinctly change
70
. In addition,
Applications to Treat Bone Diseases
Studies have shown that bone formation and bone resorption keep bone in a remodeling state to maintain normal function and structure. Osteoblasts and osteoclasts contribute to bone formation and bone resorption to maintain bone homeostasis. An imbalance between homeostasis results in metabolic bone diseases such as osteoporosis or osteopetrosis73,74. External cytokines can cause diseases by disturbing bone metabolism or affecting the growth of tumor cells such as fibroblast growth factor and vascular endothelial growth factor A75,76. In recent years, aptamers have appeared gradually in the treatment of bone diseases, which provides a new direction for orthopedics (Fig. 2).
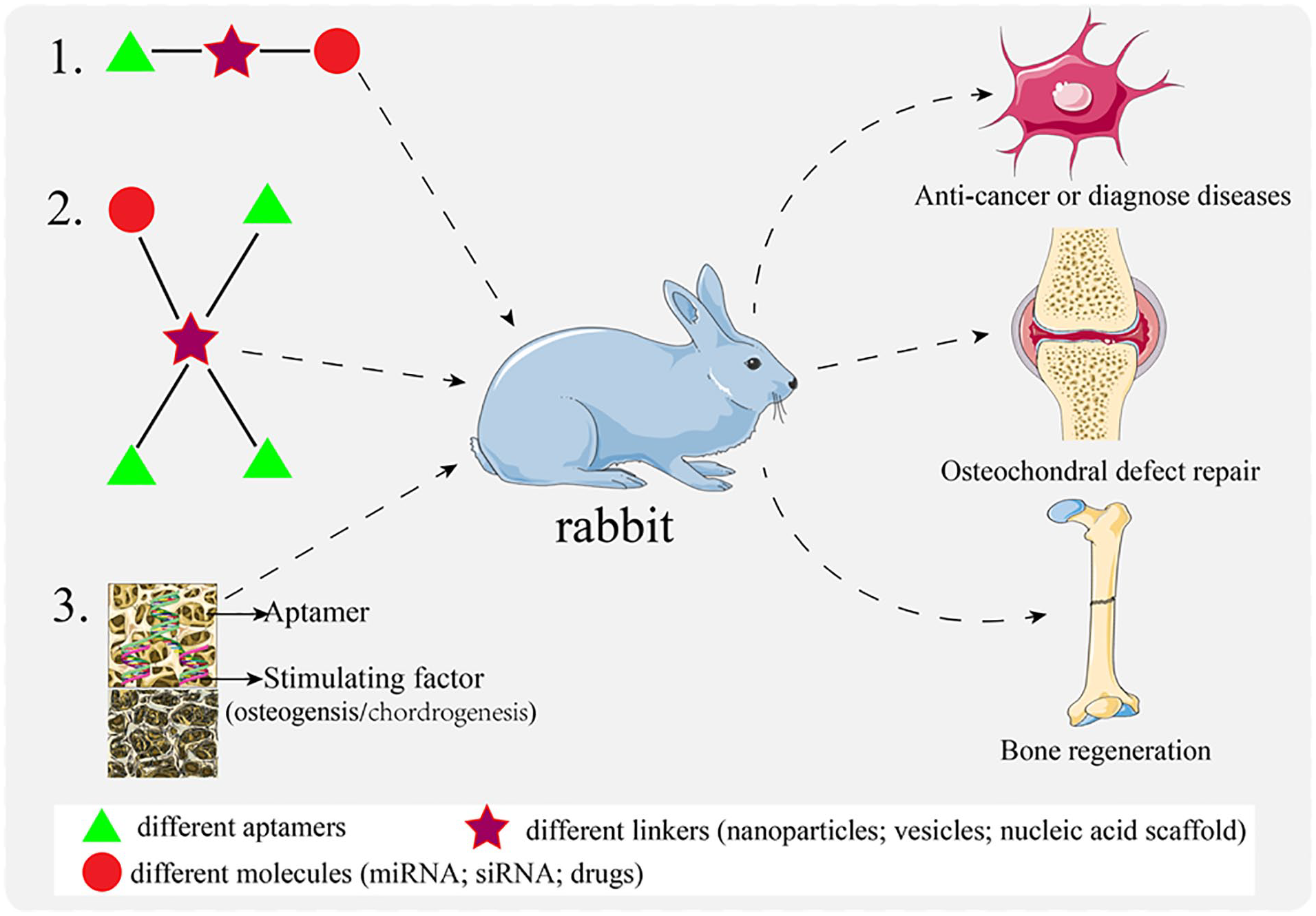
Application of aptamers in the treatment of bone diseases.
Repairing Bone and Cartilage
Bone formation disorders are related to many factors; for instance, as an intracellular negative regulator, casein kinase-2 interacting protein-1 (CKIP-1) does not affect bone resorption. Runt-related transcription factor 2 is considered a “master regulator” to osteogenic differentiation. Together with adapter Schnurri-3, the Nedd4 family HECT (“Homologous to the E6-AP Carboxyl Terminus”) domain E3 ligase WWP1 can ubiquitinate Runx2 and then inhibit bone matrix synthesis. The presence of aptamers Ch6 and C3A specifically interfere with the above processes and promote the mineralization and formation of osteoblasts77,78. A recent study demonstrated the versatility of the aptamer. The CH6 aptamer and C11 peptide were used to modify the G4.0 PAMAM dendrimer; the results showed that this dual-targeting nanocarrier could rapidly accumulate in the bone within 4 and 12 h, and then deliver drugs to sites of osteoblast activity 79 . Similar to a nanocarrier, a sequential release system of PCL/SIS-pBMP2-Apt19s can quickly release Apt19s and release BMP2 in a slow and sustained manner, which promotes bone marrow–derived mesenchymal stem cell (BMSC) homing, osteogenic differentiation, and bone formation 80 .
MicroRNAs are small single-stranded noncoding RNA and some of them are involved in osteogenesis and adipogenesis. MiR-188 regulates BMSC differentiation into adipocytes rather than osteoblasts in aged mice. Overexpression of miR-195 promotes angiogenesis and osteogenesis in mice. Aptamers carrying their agonists or inhibitors into cells can reverse or enhance their function and improve age-related osteoporosis81,82. A target delivery system of
Exosomes directly or indirectly regulate the balance of bone metabolism and promote the osteogenic differentiation of BMSCs. An efficient complex comprising BMSC-specific aptamer and bone marrow stromal cell–derived exosomes was designed to bind and internalize BMSCs, promoting bone generation and accelerating bone healing 85 . In fact, the combined application of exosomes and aptamers is reflected in other areas such as anti-inflammatory, shrinking tumors, and recognizing tumors86–89.
Because of the lack of blood vessels, nerves, and lymphatics, it is difficult to repair cartilage after injury, which often leads to osteoarthritis and osteochondrosis 90 . Pluripotent stem cells have become a research hotspot in repairing cartilage because of their differentiation potential 91 . However, insufficient scaffolding material and spontaneous migration of MSCs lead to limited applications. An approach combining aptamers with biomaterials to form a scaffold is expected to overcome this problem. For example, a bilayer scaffold comprising 3D graphene oxide, sodium alginate, and an aptamer, and a reinforced hydrogel sponge scaffold comprising silk fibroin, hyaluronic acid, and an aptamer have been shown to recruit MSCs to a osteochondral defect and restore cartilage92,93. In addition to promoting cartilage regeneration, it is important to inhibit the progress of inflammation to slow cartilage degeneration. Ra10-6, a specific aptamer of IL-17RA, can inhibit synovial inflammation by reducing the expression of IL-6 mediated by IL-17RA. Specifically, Ra10-6 may prevent or alleviate osteoarthritis 94 . Similarly, Apt21-2 can inhibit the development of arthritis by binding human IL-17A and blocking the interaction between IL-17A and its receptor IL-17R 95 . FGF2, a member of the fibroblast growth factor superfamily that can reduce the production of osteoprotegerin, increases the expression of the receptor activator of nuclear factor kappa B ligand and inhibits bone mineralization 96 . Therefore, many researchers select a specific aptamer of FGF2 to explore bone disease; RBM-007 combined with FGF2 can block the binding of FGF2 to its receptor, alleviating the decrease in bone density 97 . In addition, RBM-007 can restore the proliferation arrest, degradation of cartilaginous extracellular matrix, and premature senescence of chondrocytes by inhibiting FGFR3 signaling98,99. By competing with four cellular receptors of FGF2, APT-F2 can inhibit downstream signaling and cell proliferation induced by FGF2 and restore osteoblast differentiation. In addition, APT-F2 can be modified by PEG to form APT-F2P, which has the ability to improve bone disruption, arthritis, and osteoporosis 100 .
To determine the anti-inflammatory effect of DNA aptamer CCS13, it was injected intra-articularly into injury-associated knee osteoarthritis in a murine model, and µCT imaging was used to observe the changes to the subchondral tissue. CCS13 did not reduce cartilage degeneration or synovitis, and it did not prevent disease progression 101 .
Anti-tumor Characteristics
Bone is a major area of tumor metastasis. Many aptamers have been shown to inhibit tumor metastasis. APT A10-3.2 is a new ligand of PCa cells that expresses prostate-specific membrane antigen, and is conjugated with ATE to carry miR-15a and miR-16-1. ATE-APT efficiently delivered miRNA to PCa cells, revealing tumoricidal efficacy and longer survival time in a bone metastasis mice model 102 . As the specific aptamer of complement C5a, AON-D21 can block the C5a/c5al1 signal axis and effectively reduce bone metastasis and the tumor load of lung cancer 103 . Various studies have demonstrated that active multiple myeloma (MM) highly expressed stromal cell–derived factor-1 (SDF-1/CXCL12) that relates to homing and growth of MM. ola-PEG (the specific aptamer of SDF-1) can neutralize SDF-1 to block SDF-1-dependent signaling and then inhibit MM progression 104 . Recent research indicates that MSCs can migrate from the primary tumor site to the bone marrow to form the premetastatic niche with malignant cells. Migration of MSCs is Osteopontin (OPN)-dependent; after OPN is blocked via the R3 aptamer, MSCs do not migrate to the BM 105 .
As the most invasive malignant tumor, osteosarcoma (OS) tends to occur in children and metastasizes to the lung. It has a high degree of malignancy and mortality, and often induces pathological fractures106,107. To improve the diagnosis of OS, an ssDNA aptamer, called LP-16, was selected to specifically bind OS cells. An in vivo study suggested that LP-16 recognized both the xenograft tumor-bearing mice and OS tissues from patients. LP-16 is the first aptamer that recognizes metastatic OS cells 108 . A new DNA aptamer with high affinity in the nanomolar range against MG-63 OS cells has been identified, and is able to bind to lung and colorectal cancer cell lines 109 . Aptamer-conjugated salinomycin and clustered regularly interspaced short palindromic repeat (CRISPR)-associated Cas9 nuclease (CRISPR/Cas9) have been suggested to reduce the volume and malignancy of an OS tumor110,111.
Separating Cells or Drugs
The conventional cell isolation procedure takes at least 2 weeks. To overcome this problem, aptamers have been used for cell separation or capturing because of their high specificity. A new method based on aptamers was recently established to rapidly and efficiently isolate MSCs from porcine bone marrow. This method suggests that young MSCs have great potential to differentiate into osteoblasts and adipocytes 112 . Similarly, CD31+ cells were collected from a mixture with CD31−293FT cells or peripheral blood mononuclear cells by a specific aptamer for CD31, and their potential for angiogenic and osteogenic behavior was confirmed in vitro113,114. An efficient device similar to double-sided tape was used to interfere with circulating tumor cells (CTCs) using ligand magnetic nanoparticles (MNPs). The device comprises long single-stranded DNA and multiple copies of aptamers that can minimize MNP endocytosis to maintain the purity and activity of isolated CTCs 115 . A multiple aptamer-based DNA logic device has been designed to isolate target cells from similar cell subtypes. The device can collect cells accurately and sensitively from cell mixtures and be augmented to recognize more receptors by operating two dual or triple aptamer-based DNA logic devices 116 .
Sclerostin has become a recognized target for osteoporosis therapy because it is a negative regulator of bone formation. Numerous studies demonstrate sclerostin as a Wnt inhibitor/antagonist blocking the canonical or non-canonical Wnt signaling pathway 117 . Therefore, some aptamers were selected to inhibit the function of sclerostin. In this study, the aptamer showed a temperature-dependent parallel G-quadruplex characteristic and dose-dependent inhibition of sclerostin’s antagonistic effect on Wnt activity 118 . An innovative aptamer-based competitive drug screening platform for osteoporosis was recently designed. Potential inhibitors were used to compete with sclerostin-specific aptamers and six significantly decreased the fluorescence intensity of the aptamer. As a result, these inhibitors were potential osteogenic activators and they suppressed the level of sclerostin 119 .
Conclusion
After three decades of development, aptamers and the SELEX technology have significantly progressed. As a novel material, aptamers have unique advantages and have become a research hotspot. Although there are many shortcomings and barriers in the SELEX process, we have discovered effective methods to solve these problems. As described in this review, these methods will greatly improve the success rate and stability of aptamer screening.
In addition to their structural advantages, aptamers also have some functional advantages. Aptamers work like traditional antibodies and can deliver drugs or molecules as carriers to treat diseases, which means that aptamers have a broader application and potential. Drug delivery is the basis of targeted therapy. siRNA, a classic method of cell intervention, has many disadvantages, such as poor stability and a lack of directional delivery systems, which reduces its therapeutic efficacy. Thus, improving the drug stability and targeting drug delivery have become significant issues. After modification, aptamers have better stability and can form a spatial structure to combine with multiple targets. Therefore, modified aptamers may be connected with siRNA to enhance efficacy. In addition, nanomaterials or liposome composite structures with biocompatibility can act as molecular frameworks. As a result, a multifunctional material comprising a molecular framework, one or multiple aptamers, and one or multiple drugs can directly interfere with multiple organs or tissues and treat multiple diseases at the same time.
Although most aptamer-based drugs are still in the experimental stage, lacking sufficient data from human or animal trials, this review demonstrates their clinical promise.
Footnotes
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [grant numbers 81472103]; the Health Family Planning Research Fund of Wuhan City [grant number WX18M01]; the Wuhan City ‘Huanghe Talent’ Program; and the Wuhan Application Foundation Frontier Project [grant number 2019020701011471].
