Abstract
We reported in 2018 that among several extracellular matrices, fibronectin, type I collagen, type IV collagen, laminin I, fibrinogen, and bovine serum albumin, fibronectin is particularly useful for adhesion of porcine pancreatic tissue. Subsequently, we developed a technology that enables the chemical coating of the constituent motifs of fibronectin onto cell culture dishes. In this experiment, we used islets (purity ≥ 90%), duct epithelial cells (purity ≥ 60%), and acinar cells (purity ≥ 99%) isolated from human pancreas according to the Edmonton protocol published in 2000 and achieved adhesion to the constituent motifs of fibronectin. A solution including cGMP Prodo Islet Media was used as the assay solution. In islets, adhesion was enhanced with the constitutive motifs of fibronectin compared with uncoated islets. In the functional evaluation of islets, insulin mRNA expression and insulin secretion were enhanced by the constitutive motif of fibronectin compared with non-coated islets. The stimulation index was comparable between non-coated islets and fibronectin motifs. In duct epithelial cells, adhesion was mildly promoted by the fibronectin component compared with non-coated component, while in acinar cells, adhesion was inhibited by the fibronectin component compared with the non-coated component. These data suggest that the constitutive motifs of fibronectin are useful for the adhesion of islets and duct epithelial cells.
Keywords
Introduction
Pancreatic islet transplantation is a curative treatment for insulin-dependent diabetes mellitus (type 1 diabetes) and aims to stabilize blood glucose and wean the patient from insulin therapy by transplanting islet tissue (especially insulin-producing β cells)1–4. However, fewer than half of islets can be retrieved from the pancreas with the current technology. To solve this problem, we have been developing purification solutions using porcine islets, which are similar to human pancreatic organs 5 . However, it is usually necessary to transplant islets from two or more donors, with multiple transplants required.
In addition, as islet transplantation involves the transplantation of another person’s islet tissue, rejection (an immune response to eliminate foreign tissue) occurs, just as in organ transplantation. Several types of immunosuppressive drugs must therefore be taken for a long time to suppress the rejection reaction 6 . Technical limitations, such as difficulty maintaining transplanted islets for a long period of time, thus plague these procedures.
In recent years, the development of biomaterials related to islet transplantation, such as immune isolation membranes 7 , encapsulation of cells8,9, and organoid microphysiological systems 10 , has been actively pursued to improve these issues. The application of biomaterials with a high functionality as scaffold materials for islets has shown a marked effect on the prognosis of islet transplantation.
Extensive research over the past 20 years on islet culture has involved the development of techniques to extend culture time and to maintain or improve the survival and function. In addition, international shipments of human islets for research have been made possible via the development of media suitable for long-term culture of islets. Pioneering cell therapy techniques have been actively implemented in the development of culture media and organ preservation solutions. First, the islet survival is promoted by medium supplementation with fetal bovine serum (FBS) 11 or human serum albumin (HSA) 12 for islets removed from the pancreas, and islet preservation at 4°C has been demonstrated to be superior to 22°C or 37°C culture13–15. In a study on the composition of culture media for islet transplantation 16 , RPMI-1640 was used as the basic medium, and glucose 17 , vitamins (Vitamin E 18 , Vitamin D3) 19 , ions (Selenium20,21, Magnesium 22 , Zinc)23,24, lipids (Linoleic acid) 25 , and hormones (Insulin 26 , HGF 27 , Ghrelin28,29, L-Glutamine 30 , Cysteine 31 , Transferrin 32 , Albumin 32 , Heparin 33 , Pyruvate) 34 were selected as medium additives 16 . In addition, because the composition and temperature of the preservation solution for organs and islets has a strong influence on the survival rate and functional maintenance of islets for islet transplantation, research on the preservation solution used for islets has been active. University of Wisconsin (UW) solution35,36, developed in the 1990s in the United States, is a widely used organ preservation fluid. Tryptophan ketoglutarate (HTK) solution 37 , ET-Kyoto solution 38 , M-Kyoto solution 39 , and HN-1 solution 40 were later developed in Japan. The comparison and improvement of the composition of these organ preservation solutions have been reported40,41,42,43.
The pancreas consists of exocrine glands and endocrine glands (islets of β cells), and the main cells comprising the exocrine glands are pancreatic ductal epithelial cells and acinar cells. We previously reported the compositional analysis of proteins expressed in porcine pancreatic exocrine tissue and islets using liquid chromatography with tandem mass spectrometry (LC-MS/MS) 44 .
In islet isolation and culture, the extracellular tissue of the islet is destroyed, and the internal blood vessels and nerves are blocked. The loss of extracellular matrix (ECM) during the isolation process is one cause of post-transplant islet dysfunction. Therefore, attempts have been made to reconstruct cell-ECM using laminin, which is a major component of basement membrane, and fibronectin, which is found abundantly beneath endothelial cells and epithelial ducts in the interstitial matrix 45 , to reconstruct important signaling between cells and ECM. In assays using insulin secretion as an indicator, such ECM reportedly inhibits islet apoptosis and promotes insulin secretion46–48.
We previously performed an assay using porcine islets and reported that fibronectin promotes cell adhesion and the survival of islets 49 . In the present study, we investigated the effect of fibronectin on cell adhesion and the survival of three types of cells derived from the pancreas using a polymer scaffold (fibronectin motif) containing the RGD sequence motif, which consists of three consecutive motifs of R (arginine), G (glycine), and D (aspartate) in the amino acid sequences of fibronectin and vitronectin.
The fibronectin motif is composed of three consecutive motifs. This motif is a chemical coating material for plastic materials with a wide range of applications aimed for good manufacturing practice (GMP)–compliant cell production. We operate our clinical cell manufacturing facilities under GMP control. The process control of clinical cell manufacturing has recently shifted from manual human labor to mechanization, similar to historical trends, such as when the islet isolation method announced in 2000 was automated and several relevant new pieces of equipment introduced.
Experts who think that islet culture is unnecessary have the following three opinions: (1) The islet yield is reduced because of culture. (2) There is a clinical need to transplant islets in a non-cultured state. (3) Even in cases where transplantation cannot be performed immediately after islet isolation, such as because of patient pretreatment, islet cryopreservation using UW solution is superior to islet culture with regard to post-transplantation results within 48 h 13 . There have been no pioneering technological developments in the field of pancreatic islet culture technology because of factors associated with issues (1) to (3) mentioned above. However, in recent years, molecular biological techniques, such as chimeric antigen receptor T cell (CAR-T cell) therapy50,51, have enjoyed increased opportunities for clinical application beyond the scope of conventional research. With the further development and improvement of these techniques, new therapeutic methods using pancreatic islets may be developed. However, as pancreatic cells after islet isolation cannot be cultured for a long time, gene transfer to pancreatic cells in vitro is difficult 52 .
In the present study, we focused on the scaffold material and examined the adhesiveness of pancreatic islets, duct epithelial cells, and acinar cells after isolation. As it is prohibited to use human pancreatic tissues collected from cadavers for research in Japan, human pancreatic tissues were donated through the United Network for Organ Sharing (UNOS) and used for research purposes in this study.
Materials and Methods
Preparation of Dot Pattern Cell Culture Plates
CDS and CDSD plates were provided by Sekisui Chemical Co., Ltd. (Osaka, Japan). Six-well tissue culture (TC)-treated culture plates (Corning Incorporated, Corning, NY, USA) were coated with a synthetic polymer scaffold containing a fibronectin motif. Dot patterns were printed with a synthetic polymer ink diluted in ethanol.
Pancreas Procurement and Islet Isolation
The islets (purity ≥ 90%), duct epithelial cells (purity ≥ 60%), and acinar cells (purity ≥ 99%) isolated from human pancreas were provided by Prodo Laboratories, Inc. (Aliso Viejo, CA, USA). cGMP Prodo Islet Media provided by Prodo Laboratories was used for the culture of islets, duct epithelial cells, and acinar cells. Donor information was as follows: UNOS ID# AIFA337, AIFL413, AIGE232, AIHS080, AJET409, AJE4120.
Cell Adhesion Assays
To evaluate the adhesive properties of islets to fibronectin motif chemically treated plates, islets [total of 250 islet equivalents (IEQ)/well] were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). To evaluate the adhesive properties of duct epithelial cells to fibronectin motif chemically treated plates, duct epithelial cells (total of 25 aggregates/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). To evaluate the adhesive properties of acinar cells to fibronectin motif chemically treated plates, the acinar cells (total of 1.5–3 × 106 cells/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). DNA was extracted/purified carried out using Dr P Kit-Isolation of RNA, DNA, and Protein from the same piece of tissue simultaneously (BioChain Institute Inc., Newark, CA, USA) from the adherent islets, duct epithelial cells, and acinar cells, and DNA concentrations were measured using NanoDrop One/Onec (Thermo Fisher Scientific, Tokyo, Japan).
Islet Function Assays
Islets (total of 250 IEQ/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). A total of 250 IEQ were incubated with either 2.8 or 25 mM glucose (FUJIFILM Wako Pure Chemical Corporation, Tokyo, Japan) in RPMI-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA) for 2 h at 37°C and 5% CO2. The supernatants were collected, and the insulin levels were determined using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (Insulin ELISA; Mercodia, Uppsala, Sweden). The stimulation index was calculated by determining the ratio of insulin released from the islets in high glucose to the insulin released in low glucose. The data were expressed as the mean ± the standard error of the mean (SE).
Dynamic Perifusion Assays
Islets (total of 500 IEQ/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). Before setting the six-well plate in the assay device, the wells were washed three times with RPMI-1640 medium. The top cover of the six-well plate was removed, and the iP-TEC (Sanplatec Corporation, Osaka, Japan) and iP-TEC holder (Sanplatec Corporation) were attached. The flow rate (1 mL/min) of the dynamic perifusion assay device (Tokai Hit., Co., Ltd., Shizuoka, Japan) was set. The medium (low glucose [2.8 mM]) was perfused for 10 min before the start of the assay. After the start of the assay, (1) low-glucose (2.8 mM) perifusion for 5 min, (2) high-glucose (25 mM) perifusion for 10 min, (3) low-glucose (2.8 mM) perifusion for 20 min, (4) high-glucose (25 mM) perifusion for 10 min, and (5) low-glucose (2.8 mM) perifusion for 25 min were performed. Sampling of the medium discharged from the perifusion device was performed at 2.5-min intervals. When switching between media with different glucose concentrations in the flow path of the perifusion device, an acclimation cycle of 5 min was performed. All steps during the assay were performed in a clean bench under a flow rate of 1 mL/min at 37°C.
ATP Assays
The ATP levels were determined using an ATP Assay Kit-Luminescence (Dojindo, Kumamoto, Japan). The luminescence was measured with a micro plate reader at a wavelength of 450 nm using SpectraMax iD3 (Molecular Devices, San Jose, CA, USA).
Enzyme-linked immunosorbent assays
The insulin levels were determined using a commercially available ELISA kit (Insulin ELISA; Mercodia). The absorbance was measured with a micro plate reader at a wavelength of 450 nm using SpectraMax iD3 (Molecular Devices).
Real-Time PCR and Reverse-Transcription PCR Analyses
RNA was prepared using a SuperPREP II Cell Lysis & RT (reverse-transcription) Kit for quantitative polymerase chain reaction (PCR; TOYOBO CO., LTD. Osaka, Japan) according to the manufacturer’s instructions. Real-time PCR was performed using a StepOnePlus system (Life Technologies, Carlsbad, CA, USA). The Luna Universal qPCR Master Mix (New England Biolabs Inc., Ipswich, MA, USA) was used according to the manufacturer’s instructions. For the design of primers other than those cited in other papers, the gene names were retrieved from the US National Library of Medicine NIH website (https://www.ncbi.nlm.nih.gov/pubmed/). The primers were designed using the Primer 3 Plus application (http://www.bioinformatics.nl/cgi-bin/primer3plus/primer3plus.cgi). The primers used for PCR were as follows: human Insulin (BT006808.1) 176 bp (forward) AACGAGGCTTCTTCTACACACC (reverse) GTAGAGGGAGCAGATGCTGGTA human integrin α1(NM_181501.1) 234 bp (forward) CAATGAGACAGTCCCTGAAGTTATT (reverse) GAGTTGATACTGAAAGGATCCTCAA human integrin α2(NM_002203.3) 176 bp (forward) TTCTACTGGAGAAAGCACTCACTTT (reverse) CTTTATCCACATCAACTGAACACAG human integrin α3(NM_002204.3) 201 bp (forward) ATAGCAACACAGACTACCTGGAGAC (reverse) TGTACCCAATATAGAGGTTTCCTTG human integrin α4(NM_000885.5) 203 bp (forward) CTTTGAGATAGAAGAACTGCAAAGG (reverse) GAGGCAAGCACATATGGTAAGTAGT human integrin α5(NM_002205.4) 229 bp (forward) CTGCTACCTCTCCACAGATAACTTC (reverse) GATCAGGTACTCGGGGTAATAAGAT human integrin α6(NM_000210.3) 195 bp (forward) GTTTTGTTTCCTCCCCTATCTGTAT (reverse) GCTCCCCATATAACTTAACATTGTG human integrin α7(NM_001144996.1) 220 bp (forward) CTCTGCTGAGAAGAAGAAAAACATC (reverse) CAAGTTCTTTATGGAGGACTTCACT human integrin α8(NM_003638.2) 213 bp (forward) AAGGTGAACTAAGGTGAAATGACTG (reverse) CTCACCCTCACTCACAAGATTATTT human integrin α9(NM_002207.2) 235 bp (forward) AGTTCTCAGCTTCATTGTTACTGCT (reverse) GTAGACCTGAAGGGTGATATTGATG human integrin αE(NM_002208.4) 153 bp (forward) CTGGGATCACTCTGAGGAGTTACTA (reverse) GAATGGTACTTCTCATCTTTCAGGA human integrin αL(NM_001114380.1) 187 bp (forward) CTGCAGATGATGTTTAATACACTGG (reverse) TGACATAGAGTGTGGAGTCTTCTTG human integrin αM(NM_000632.3) 152 bp (forward) GTGTCCTCAAGAGGATAGTGACATT (reverse) CAGAGTACTGCATCAAAGAGAACAA human integrin αV(NM_001144999.2) 191 bp (forward) AGATTAGACAGAGGAAAGAGTGCAA (reverse) ACATTAGTGGTAACCAATGTGGAGT human integrin α2b(NM_000419.4) 206 bp (forward) CGGCTATTATTTCTTAGGTCTCCTG (reverse) CGACGACATATTCTGTAGTGTTGAG human integrin β1(NM_002211.3) 179 bp (forward) CTGAAGACTATCCCATTGACCTCTA (reverse) GCTAATGTAAGGCATCACAGTCTTT human integrin β2(NM_000211.4) 182 bp (forward) GATGGTGAAGACCTACGAGAAACT (reverse) AGAAGGAGTCGTAGGTGACTTTCAG human integrin β3(NM_000212.2) 156 bp (forward) GCACTTAATGATAAGCAGTCATCCT (reverse) CACACTTCCACATACTGACATTCTC human integrin β4(NM_000213.4)220 bp (forward) CTTTGCTGTCACCAACTACTCCTAT (reverse) AGTCCTCGTCTTCTGGAACATCT human integrin β5(NM_002213.4) 224 bp (forward) GGAGTCTGTAAAACCAGCATACAGT (reverse) AGTACTAAACAAACACGGACAGGAG human integrin β6(NM_000888.4) 206 bp (forward) GATCCATATACCTTTGATCACTTGG (reverse) CATTGCAACCTGTATATTGCTGTAG human integrin β7(NM_000889.2)190 bp (forward) AGCAGGACAGTAATCCTCTCTACAA (reverse) GAATTAGTCCCCTACCAAGGTCTTA human integrin β8(NM_002214.2) 151 bp (forward) TTTGAGACTAGTGTCGTTGTAGCAC (reverse) AAACTCTCTCACAGCATTGTTCTCT human cadherin 1 (CDH1) (NM_004360.4) 191 bp (forward) GCCACATCTTGACTAGGTATTGTCT (reverse) GCAGCACTTTAGGCACTATTCTAAG human cadherin 2 (CDH2) (NM_001792.4) 219 bp (forward) AGTGTTCCCAAGACAATTCAGTAAG (reverse) GGGTTGATAATGAAGATACCAGTTG human cadherin 3 (CDH3) (NM_001317195.1) 210 bp (forward) AGTCCTACCAGGTACTTCTGTGATG (reverse) GTCAGTGTGTACTCAGGGACTTTTT human cadherin 4 (CDH4) (NM_001252339.2) 163 bp (forward) AGTATGATGTACTCACACCTGCAAA (reverse) ACACTGAAAACATCAACTCTCTTCC human cadherin 5 (CDH5) (NM_001795.4) 224 bp (forward) AAGTCTACCCCTGGTATAACCTGAC (reverse) TCGTGGTGTTATGTCCTTGTCTATT human cadherin 6 (CDH6) (NM_004932.3) 204 bp (forward) CGTCTTCAGTAGTTCCAAGCTAAAG (reverse) AGACATGAACGATACTGAGGAAAAC human cadherin 7 (CDH7) (NM_001317214.1)208 bp (forward) ATGCAGATATTCTACTGGATGGTGT (reverse) AAGATACAAAAGTTAGCTGGGTGTG human cadherin 8 (CDH8) (NM_001796.4) 213 bp (forward) CTGAAATTAGGAACCACAGTCAGAT (reverse) CCATTTCTGGAAGGAGACTGTATAA human cadherin 9 (CDH9) (NM_016279.3) 170 bp (forward) TATGTTCCATACAGTTGACACCATC (reverse) TGTCAGTACCTGTGTACTCTTCCAA human cadherin 10 (CDH10) (NM_006727.4) 165 bp (forward) TATTGATGGTGACGGTACTGATATG (reverse) CCTAGGTAATAAAAACGGGGATCTA human cadherin 11 (CDH11) (NM_001308392.1) 222 bp (forward) TGTAGTATTGTTTGTGACCCTGAGA (reverse) CCAGGTCTAGGCATGTACTGATACT human cadherin 12 (CDH12) (NM_001317227.1) 189 bp (forward) TGCAATAAGGAGCCTAGATAGAGAA (reverse) ACAGGAGACATTTCTGGAACAGTAG human cadherin 15 (CDH15) (NM_004933.2) 160 bp (forward) CGTCTTCTCTATCGACAAGTTCAC (reverse) ATTCTGATCCACAACTACAATCTCC human cadherin 18 (CDH18) (NM_001167667.1) 222 bp (forward) ACTCAACAAAAAGCCTAGACAGAGA (reverse) CAGTAGCTGTCACCTGTAGAACAGA human cadherin 19 (CDH19) (NM_001271028.1) 235 bp (forward) AGTGCCTGACAAAATATAACTGCTC (reverse) AGTAACTGAGGAGGAGAGAGGAAAG human cadherin 20 (CDH20) (NM_031891.3) 162 bp (forward) GACAGAGAAGCCAAAGAATACTACG (reverse) CAACACACTCATCTGGTAATGTTTC human cadherin 22 (CDH22) (NM_021248.2)246 bp (forward) AGGCTGTTCACTTACTAAGCACCTA (reverse) CATTTGACAGATGAGGAAACTGAG human cadherin 24 (CDH24) (NM_022478.3) 152 bp (forward) CTCCAGGAATCTTTGTCTCTATCTG (reverse) GGAGACTATGTGTGAGCTTCAGAAC human β-actin (NM_001101.3) 224 bp (forward) GTGACATTAAGGAGAAGCTGTGCTA (reverse) CTTCATGATGGAGTTGAAGGTAGTT
Statistical Analyses
Statistical analyses were performed using Student’s t-test to compare two samples. Comparisons between multiple groups (more than two groups) were performed using a one-way analysis of variance with the StatPlus software program (AnalystSoft, Walnut, CA, USA). Statistical significance was set at P < 0.05 for all tests.
Results
Adhesive Properties of Islets to Fibronectin Motif
Six-well plates chemically coated with a fibronectin motif in a dot shape were custom-made. The CDSD-500 has a dot size of 500 μm, a total of 638 dots, and a total adhesive area of 1.2 cm2. The CDSD-1500 has a dot size of 1,500 μm, a total of 69 dots, and a total adhesive area of 1.2 cm2. For comparison, a plate (CDS plate) in which the surface treatment of the dots was performed on the entire bottom surface of the well was used. To evaluate the adhesive properties of islets to the fibronectin motif chemically treated plate, the islets (total of 250 IEQ/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). Photographs of islet cells taken by optical microscope at 1 week after seeding on each plate are shown (Fig. 1A). DNA was extracted/purified from the adherent islets, and the DNA concentration was measured. As a result, islet cell adhesion to the CDS, CDSD-500, and CDSD-1500 plates was more strongly promoted than that of the control non-coated plate. There was no significant difference in the number of adherent cells in islets between CDSD-500 and CDSD-1500, which have the same area of fibronectin motif chemical treatment (Fig. 1B). In addition, there was no significant difference in the number of adherent cells among islets that adhered to the CDS plate, which had about 7.5 times the area of fibronectin motif chemical treatment compared with the CDSD-500 and CDSD-1500 plates (Fig. 1B).

Adhesion of pancreatic islets to fibronectin motif. (A) The islets (total of 250 IEQ/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). The CDSD-500 has a dot size of 500 μm. The CDSD-1500 has a dot size of 1,500 μm. For comparison, a plate (CDS plate) in which the surface treatment of the dots was performed on the entire bottom surface of the well was used. Photographs of islet cells taken by optical microscope at 1 week after seeding on each plate are shown. Scale bar = 500 μm. (B) DNA was extracted/purified from the adherent islets, and the DNA concentration was measured (n = 4; control, n = 1). The data are presented as the mean ± SE (standard error of the mean). *P < 0.05. (C) The expression of integrins α1, α2, α3, α5, α6, αV, β1, β3, β5, β6, and β8 in islets (n = 1). β-actin housekeeping genes were used as denominators for comparing samples. (D) The expression of cadherins 1, 2, 3, 5, 6, 7, 10, 11, 12, 18, 19, and 22 in islets (n = 1). β-actin housekeeping genes were used as denominators for comparing samples.
Expression of Integrin and Cadherin in Islets
As various integrins are known to bind to fibronectin, we evaluated the expression of integrin chains in islets using real-time PCR. The islets expressed integrins α1, α2, α3, α5, α6, αV, β1, β3, β5, β6, and β8 (Fig. 1C); cadherins 1, 2, 3, 5, 6, 7, 10, 11, 12, 18, 19, and 22 (Fig. 1C); and α5β1, αVβ1, αVβ3, and αVβ6, integrin chains of fibronectin receptor. Therefore, islets demonstrated strong adhesiveness to fibronectin.
Assessing the Islet Function
The islet function was assessed by monitoring the insulin secretory response of the purified islets during glucose stimulation using the procedure described by Shapiro et al. 53 As the amount of human pancreatic islets available was 3,000 IEQ, the assay was performed using 250 IEQ per well in this experiment. In brief, 250 IEQ were incubated with either 2.8 or 25 mM glucose (FUJIFILM Wako, Osaka, Japan) in RPMI-1640 medium (Thermo Fisher Scientific, Tokyo, Japan) for 2 h at 37°C and 5% CO2. The supernatant was collected, and the insulin levels were determined using a commercially available ELISA kit (Insulin ELISA; Mercodia). The stimulation index was calculated by determining the ratio of insulin released from the islets under high-glucose conditions to the insulin released under low-glucose conditions. The mRNA was extracted from the cells after collecting the culture supernatant, and the insulin mRNA expression was determined using real-time PCR. Insulin mRNA expression by islets was comparable in low- and high-glucose media under three conditions: non-coated, CDSD-500, and CDSD1500 (Fig. 2A). Furthermore, compared with the control, non-coated islets cultured on fibronectin chemically treated plates secreted 2.5 (CDSD-500) to 5.0 (CDSD1500) times as much insulin under high-glucose conditions (Fig. 2B). The stimulation index was not significantly different between the control non-coated and CDSD-500 and CDSD-1500 plates. This result indicates that the fibronectin motif promotes insulin mRNA synthesis and increases insulin secretion. Furthermore, fibronectin does not affect glucose sensitivity or glucose-responsive insulin secretion in islets (Fig. 2C). Islets (total of 500 IEQ/well) were seeded onto six-well plates (non-coated plate: control) chemically coated with a fibronectin motif (CDSD500 or CDSD1500). Pancreatic islets were sampled from various wells, and ATP concentrations were measured. As a result, islets adhered to non-coated wells had very low ATP concentrations, whereas islets fully coated with fibronectin motifs (CDS) and islets with dot patterns of fibronectin motifs (CDSD-1500) showed extremely high ATP concentrations. This result indicates that the fibronectin motif increased islet cell activity. However, as the size of islets ranges from 50 to 500 µm in diameter, islets in the dot pattern with the fibronectin motif (CDSD-500) may have partially protruded from the dot pattern and become cell-adhered. Therefore, there was considered to be no significant difference in ATP concentrations between the dot pattern with fibronectin motifs (CDSD-500) and islets adhering to non-coated cells (Fig. 2D).
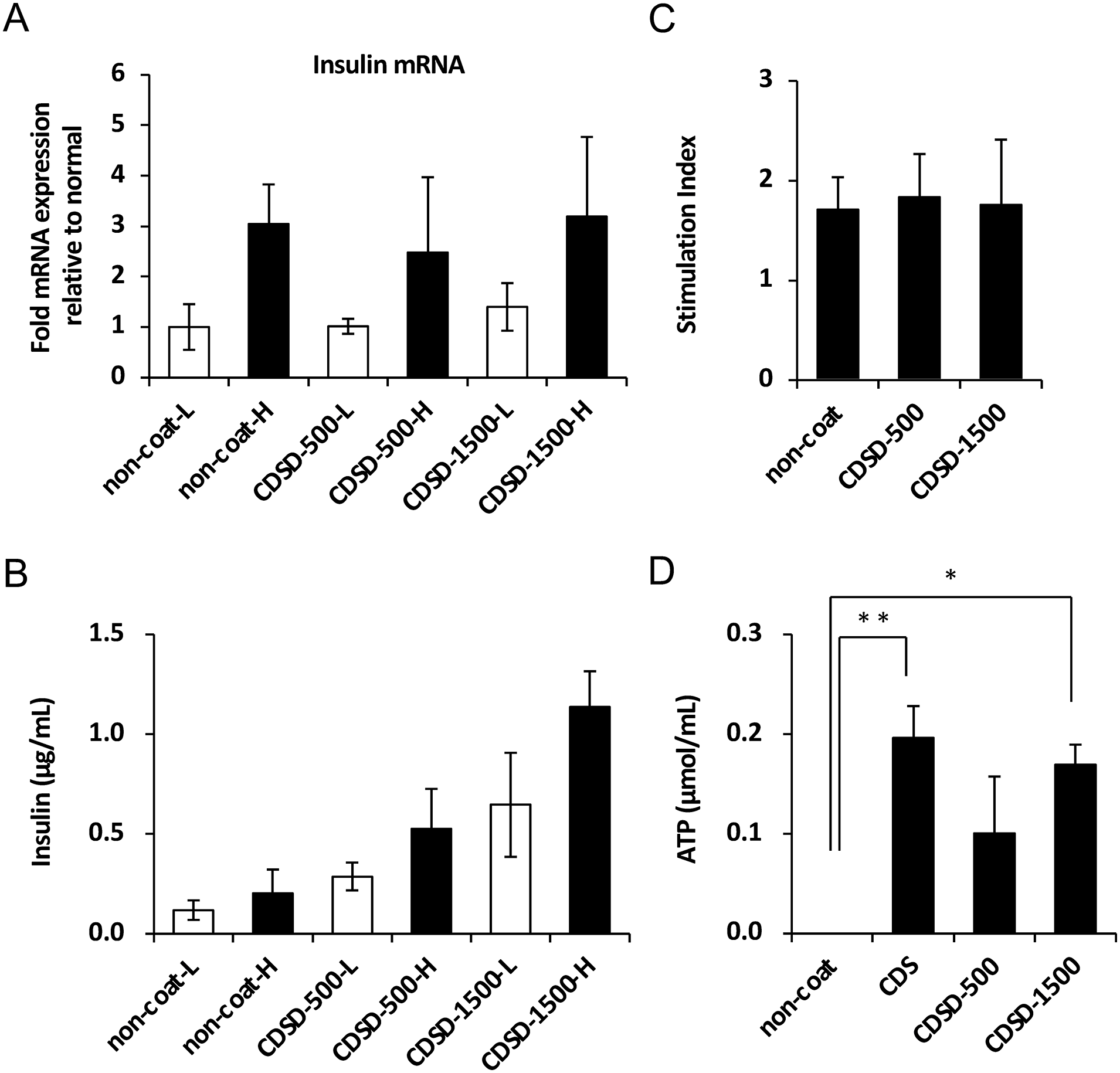
The assessment of the islet function. (A) The insulin mRNA expression from islets in high- or low-glucose media was measured using real-time polymerase chain reaction (n = 4). (B) The insulin levels from islets in high- or low-glucose media for 2 h at 37°C and 5% CO2 were determined using a commercially available enzyme-linked immunosorbent assay (ELISA) (n = 4). (C) The stimulation index was calculated by determining the ratio of insulin released from islets in high-glucose media to that released in low-glucose media (n = 4). The data are presented as the mean ± SE (standard error of the mean). *P < 0.05. (D) ATP assay/well experiment (n = 3). The data are presented as the mean ± SE. *P < 0.05; **P < 0.01.
Development of a Perifusion Assay Device and Perifusion Assay of Pancreatic Islets
We attempted a perifusion assay to analyze the ability of the fibronectin motif to secrete insulin to the islets in greater detail. As the dot pattern plate of the fibronectin motif was prepared using six wells, it was necessary to perform the perifusion assay using six wells. However, the islet perifusion assay system reported previously was a system in which islets were enclosed in a micro column54,55. Therefore, we designed a six-well perifusion assay system. As an overview of the system, we first prepared a medium supply source that could switch between low- and high-glucose medium (R1, R2, and pinch valve in Fig. 3A). Next, six precision miniature peristaltic pumps were prepared to create precise positive pressure to supply an equal volume of medium to each well of a six-well plate (pump in Fig. 3A) and a motor control device that could set the flow rate from 0 to 6.0 mL/min in 1,000 steps (KSX-Type1-A22R; Tokai Hit.). In addition, a perifusion attachment, a holder (iP-TEC; Sanplatec Corporation), and a silicon top cover for six wells were prepared to create positive pressure in the wells. The six wells were placed on a hot plate (TPiDE-OT-A22D; Tokai Hit.) set at 37°C. For sampling of cell culture medium, a manual sampling device (OT-A22S Tokai Hit.) was prepared to collect medium draining from each well into each well of a 24-well plate. The direct perifusion assay system for six-well plates was fabricated by incorporating these devices (Fig. 3B). The direct perifusion assay device was actually perfused with low- and high-glucose media at a flow rate of 1 mL/min to six wells, and the glucose concentration of the sample collected in 24 wells coming out of the flow channel was measured (Fig. 3C). The total measurement time was 97.5 min, and the step of perfusing low-glucose medium followed by high-glucose medium was repeated twice (the orange area in the figure indicates the perifusion time of the high-glucose medium).

Functional evaluation of pancreatic islets using the dynamic perifusion assay. (A) Schematic illustration of the flow path of the perifusion assay device. (B) Visual illustration of the perifusion assay device. (C) Real-time monitoring values of medium glucose concentrations in the perifusion assay (n = 6). Times in blue in the graph indicate perifusion times for low glucose. Times in orange indicate perifusion times for high glucose. (D) The islets (total of 500 IEQ/well) were seeded onto six-well plates (non-coated plate: control). Real-time monitoring values of medium insulin concentrations in the perifusion assay of cells (n = 6). Times in blue in the graph indicate perifusion times for low glucose. Times in orange indicate perifusion times for high glucose. (E) The islets (total of 500 IEQ/well) were seeded onto six-well plates chemically coated with a fibronectin motif (CDSD500). Real-time monitoring values of medium insulin concentrations in the perifusion assay (n = 6). Times in blue in the graph indicate perifusion times for low glucose. Times in orange indicate perifusion times for high glucose.
Next, the assay was performed using islets actually cultured in non-coated plates (Fig. 3D) and CDSD500 plates (Fig. 3E), and the insulin concentration of the medium sampled into 24 wells was measured using an ELISA. An examination of these measurements showed a mismatch in insulin concentrations between the high- and low-glucose concentrations of the perfused medium in the six wells. In the experiment by Bentsi-Barnes et al., who first reported the direct islet perifusion assay 54 , the islet seeding density of 200 IEQ/well was set at 0.73 cm3 perifusion columns, and the perifusion rate was reported to be 130 μL/min. Using the attachment, the volume of medium covering the bottom of the wells is 1.9 cm3, which equates to 2 mL, as the volume of medium covering the bottom of the wells is 2 mm in height, so 2 mL is required. Therefore, we set the perifusion rate at 1 mL/min. In the present experiment, the islet seeding density of 500 IEQ/well was established for six-well plates. This means that the cell count was 2.5 times higher (500/200) and the perifusion rate 7.69 times higher (1,000/130) than in the experiment of Bentsi-Barnes et al. The measurable insulin concentration was calculated to be 0.33-fold (2.5/7.69), which was considered to meet the detection criteria of the ELISA. Therefore, we believe that the cell counts and perifusion speeds we selected are acceptable based on the previous literature.
Adhesive Properties of Duct Epithelial Cells to Fibronectin Motif
We used 200 to 300 aggregated duct epithelial cells from one donor as aggregates for our experiments. Non-coated plates were used as the control for the assay. Fibronectin chemically coated plates were CDSD-500, CDSD-1500, and a fully coated CDS plate was used for the comparison. To evaluate the adhesive properties of duct epithelial cells to fibronectin motif chemically treated plates, the duct epithelial cells (total of 25 aggregates/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). Photographs of duct epithelial cells taken by optical microscope at 1 week after seeding on each plate are shown below (Fig. 4A). DNA was extracted/purified from the adherent duct epithelial cells, and the DNA concentration was measured. As a result, duct epithelial cell adhesion to the CDS, CDSD-500, and CDSD-1500 plates was more strongly promoted than that of the control non-coated plate. There was no significant difference in the number of adherent duct epithelial cells between CDSD-500 and CDSD-1500, which have the same area of fibronectin motif chemical treatment (Fig. 4B). In addition, there was no significant difference in the number of adherent cells among the duct epithelial cells that adhered to the CDS plate, which had about 7.5 times the area of fibronectin motif chemical treatment compared with the CDSD-500 and CDSD-1500 plates (Fig. 4B).

Adhesion of duct epithelial cells to fibronectin motif. (A) The duct epithelial cells (total of 25 aggregates/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). The CDSD-500 has a dot size of 500 μm. The CDSD-1500 has a dot size of 1,500 μm. For comparison, a plate (CDS plate) in which the surface treatment of the dots was performed on the entire bottom surface of the well was used. Photographs of duct epithelial cells taken by optical microscope at 1 week after seeding on each plate are shown. Scale bar = 500 μm. (B) DNA was extracted/purified from the adherent duct epithelial cells, and the DNA concentration was measured (n = 2). The data are presented as the mean ± SE (standard error of the mean). *P < 0.05. (C) The expression of integrins α1, α2, α6, αV, β1, β5, β6, and β8 in duct epithelial cells (n = 1). β-actin housekeeping genes were used as denominators for comparing samples. (D) The expression of cadherin 1, 2, 3, 6, 7, 10, 11, and 12 in duct epithelial cells (n = 1). β-actin housekeeping genes were used as denominators for comparing samples.
Expression of Integrin and Cadherin in Duct Epithelial Cells
As various integrins are known to bind to fibronectin, we evaluated the expression of integrin chains in duct epithelial cells using real-time PCR. The duct epithelial cells expressed integrins α1, α2, α6, αV, β1, β5, β6, and β8 (Fig. 4C); cadherins 1, 2, 3, 6, 7, 10, 11, and 12 (Fig. 4C); and α5β1, αVβ1, and αVβ6, integrin chains of fibronectin receptor. Therefore, duct epithelial cells demonstrated adhesiveness to fibronectin.
Adhesive Properties of Acinar Cells to the Fibronectin Motif
We used about 2–4 × 107 cells of acinar cells for our experiments. Non-coated plates were used as a control for the assay. Fibronectin chemically coated plates were CDSD-500, CDSD-1500, and fully coated SDC for the comparison. To evaluate the adhesive properties of acinar cells to fibronectin motif chemically treated plate, the acinar cells (total of 1.5–3 × 106 cells/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). Photographs of acinar cells taken by optical microscope at 1 week after seeding on each plate are shown in Fig. 5A. DNA was extracted/purified from the adherent acinar cells, and the DNA concentration was measured. As a result, acinar cell adhesion to the CDS, CDSD-500, and CDSD-1500 plates was weaker than that to control uncoated plates. There was no significant difference in the number of adherent cells in the acinar cells between CDSD-500 and CDSD-1500, which have the same area of fibronectin motif chemical treatment (Fig. 5B). In addition, there was no significant difference in the number of adherent cells among the acinar cells that adhered to the CDS plate, which had about 7.5 times the area of fibronectin motif chemical treatment compared with the amount in the CDSD-500 and CDSD-1500 plates (Fig. 5B).

Adhesion of acinar cells to fibronectin motif. (A) The acinar cells (total of 1.5–3 × 106 cells/well) were seeded onto six-well plates chemically coated with a fibronectin motif (non-coated plate: control). The CDSD-500 has a dot size of 500 μm. The CDSD-1500 has a dot size of 1,500 μm. For comparison, a plate (CDS plate) in which the surface treatment of the dots was performed on the entire bottom surface of the well was used. Photographs of acinar cells taken by optical microscope at 1 week after seeding on each plate are shown. Scale bar = 500 μm. (B) DNA was extracted/purified from the adherent acinar cells, and the DNA concentration was measured (n = 2). The data are presented as the mean ± SE (standard error of the mean). *P < 0.05. (C) The expression of integrins α4, α6, α8, αV, β3, β5, β6, β7, and β8 in acinar cells (n = 1). β-actin housekeeping genes were used as denominators for comparing samples. (D) The expression of cadherin 1, 4, 6, 7, 9, 11, 12, 19, 22, and 24 in acinar cells (n = 1). β-actin housekeeping genes were used as denominators for comparing samples.
Expression of Integrin and Cadherin in Acinar Cells
As various integrins are known to bind to fibronectin, we evaluated the expression of integrin chains in acinar cells using real-time PCR. The acinar cells expressed integrins α4, α6, α8, αV, β3, β5, β6, β7, and β8 (Fig. 5C); cadherins 1, 4, 6, 7, 9, 11, 12, 19, 22, and 24 (Fig. 5C); and αVβ3, α4β7, and αVβ6, integrin chains of fibronectin receptor. Therefore, acinar cells demonstrated adhesiveness to fibronectin.
Discussion
The RGD sequence Arg-Gly-Asp-Ser 56 in the fibronectin type III domain 57 has been reported to have many binding integrins, such as α5β1, αvβ3, αvβ5, αvβ6, αvβ1, α3β1, α5β1, α8β1, and αIIbβ3 58 . In addition to the RGD sequence, there is an REDV sequence (Arg-Glu-Asp-Val) 59 with α4β1- and α4β7-binding integrins and an LDV sequence (Leu-Asp-Val) 60 , which has integrins for α4β1 and α4β7. However, if the number of binding integrin types is small, the cell types that can be adhered to are likely to be limited. Against this background, we developed a technique to chemically coat the RGD sequence on the resin surface of cell culture vessels. The receptors for fibronectin are α4β1, α5β1, α8β1, αvβ1, αvβ3, αvβ6, α4β7, αDβ2, and αIIbβ3 61 . Therefore, fibronectin motifs in which the RGD sequence is the main constituent and the concentration of the RGD sequence is artificially prepared have different cell adhesion activities compared with fibronectin proteins. We are also developing an approach to clinical cell production. Therefore, we conducted experiments using chemically coated plates of fibronectin motifs, a fully synthetic scaffold material, using cells from human islets and other pancreatic tissues.
We previously reported that porcine pancreatic islets expressing integrins αVβ1, αVβ3, and α5β1, which are receptors for fibronectin, promote cell adhesion to fibronectin 49 . First, the RGD sequence bound to integrins in human pancreatic islets and promoted cell adhesion (Fig. 1A). We then checked whether or not the fibronectin motif consisting only of the RGD sequence had the same effect on promoting insulin expression as fibronectin. The results showed that the RGD sequence promoted the expression of high-glucose-responsive insulin mRNA and insulin secretion in pancreatic islets. However, the insulin synthesis/secretion activity under low-glucose conditions was also higher than that without coating, and the stimulation index was not higher than that without coating (Fig. 2C). These results indicate that islet viability is higher with the RGD sequence than without coating and that the RGD sequence is not effective at promoting insulin expression/secretion specifically to islets. This result can be attributed to the fact that the fibronectin motif does not unnecessarily activate the intrinsic signaling pathway through integrins. Next, to examine whether or not islets have adhesion preference, we prepared plates coated with fibronectin motifs in a dot pattern. As a result, islets adhered to cells on or adjacent to the fibronectin motif. This result indicated that the fibronectin motif activates the viability of the islet, and the islet then migrates to the fibronectin motif and adheres to it. In this study, we performed experiments with low islet seeding (250 IEQ/well), as it was difficult to obtain human islets that can be used for research in Japan. Therefore, the CDSD plates with dot patterns had a similar amount of cell adhesion to islets compared with the CDS plate with a fibronectin motif coated on the whole well (Fig. 1B).
mRNA was extracted/isolated from cell pellets of pancreatic islets, duct epithelial cells, and acinar cells, and an integrin/cadherin mRNA expression analysis was performed using real-time PCR. In this experiment, we used three types of cell pellets collected in non-culture for the mRNA expression analysis. The three types of cells constituting pancreatic tissue were highly purified via the islet isolation method developed by Prodo Laboratories (http://www.lidsen.com/journals/transplantation/transplantation-03-01-052). However, the purity of each type of cell was not 100%. Therefore, the reliability of the integrin/cadherin expression patterns of various cells needs to be determined with consideration of the purity of the cells. The results of the cadherin mRNA expression analysis showed that pancreatic islets and duct epithelial cells expressed cadherin 1 mRNA particularly strongly. Cadherin 1, also known as E-cadherin, has recently attracted attention as a biomarker for pancreatitis and pancreatic cancer in pathology 62 . It has also been reported that E-cadherin plays a role in promoting islet cell proliferation 63 and maintaining physiological functions64–66. We believe that the results of our mRNA expression analysis of various integrins and cadherins using highly purified human pancreatic islets, duct epithelial cells, and acinar cells are highly reliable.
In this experiment, to artificially control integrin-mediated cell adhesion, we developed fibronectin motifs and conducted cell adhesion assays using cells in islets and other pancreatic tissues and evaluated the islet function. As a result, fibronectin motifs promoted cell adhesion and the survival activity of pancreatic islets but not the insulin synthesis/secretion ability, which is a factor used to assess the cell function of pancreatic islets. In recent years, it has been reported that not only integrins but also cadherins play a major role in the function of pancreatic islets. Therefore, we are currently developing an artificial synthetic scaffold material focusing on the cadherin motif and are planning to publish a paper on the functional evaluation of pancreatic islets using the new material as well as its practical use as a material for clinical cell production.
Footnotes
Author Contributions
Author roles: study design: YoN, HI, KT, YuN, SH, MT; study conduct: YoN, HI, KT; data collection: YoN, HI, KT; data analysis: YoN; data interpretation: YoN, HI, KT, YuN, SH, MT; drafting manuscript: YoN, MT; revising manuscript content: YoN, YuN, KI, NK, SH, MT; approving final version of manuscript: YoN, HI, KT, YuN, KI, NK, SH, MT. YoN takes responsibility for the integrity of all data analyses.
Ethical Approval
Ethical approval is not applicable for this article. Human samples were provided by Prodo Laboratories, Inc. (Aliso Viejo, CA, USA) in accordance with strict ethical standards, including informed consent.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article. Human samples were provided by Prodo Laboratories, Inc. (Aliso Viejo, CA, USA) in accordance with strict ethical standards, including informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by AMED under Grant Number JP22bm0104001. This study was conducted in collaboration with companies SEKISUI CHEMICAL CO., LTD., Tokai Hit., Co., Ltd., and CiRA Foundation. SEKISUI CHEMICAL’s role was to provide culture equipment and support research. Tokai Hit.’s role was to develop a perifusion assay device.
