Abstract
Patients fulfilling criteria for euthanasia can choose to donate their organs after circulatory death [donors after euthanasia (DCD V)]. This study assesses the outcome of islet cell isolation from DCD V pancreases. A procedure for DCD V procurement provided 13 pancreases preserved in Institut Georges Lopez-1 preservation solution and following acirculatory warm ischemia time under 10 minutes. Islet cell isolation outcomes are compared with those from reference donors after brain death (DBD, n = 234) and a cohort of donors after controlled circulatory death (DCD III, n = 29) procured under the same conditions. Islet cell isolation from DCD V organs resulted in better in vitro outcome than for selected DCD III or reference DBD organs. A 50% higher average beta cell number before and after culture and a higher average beta cell purity (35% vs 24% and 25%) was observed, which led to more frequent selection for our clinical protocol (77% of isolates vs 50%). The functional capacity of a DCD V islet cell preparation was illustrated by its in vivo effect following intraportal transplantation in a type 1 diabetes patient: injection of 2 million beta cells/kg body weight (1,900 IEQ/kg body weight) at 39% insulin purity resulted in an implant with functional beta cell mass that represented 30% of that in non-diabetic controls. In conclusion, this study describes procurement and preservation conditions for donor organs after euthanasia, which allow preparation of cultured islet cells, that more frequently meet criteria for clinical use than those from DBD or DCD III organs.
Keywords
Introduction
Intraportal (IP) islet cell transplantation allows restoration of a functional beta cell mass (FBM) in type 1 diabetes patients1–3. With metabolic outcome of clinical implants dependent on a minimal number of implanted beta cells, availability of good quality donor organs is considered a main limitation for islet cell transplantation programs 4 . In a number of countries, including Belgium, Luxemburg, The Netherlands, and some regions in Canada, a legal and ethical framework allows organ and tissue donation after euthanasia5,6, thus providing the possibility to increase the organ donor pool 7 . These donors are rarely hospitalized or admitted to the intensive care unit and have not underwent an acute neurological insult with its associated cytokine storm, nor episodes of cardiac dysfunction and decreased organ perfusion8–11. Despite these benefits, organ procurement can only be performed after cessation of circulation, leading to exposure to an initial episode of warm ischemia time (WIT), increasing the risk for organ dysfunction. They are therefore classified as a subtype of donors after circulatory death using a modified Maastricht classification [donors after euthanasia (DCD V)] 12 . Our university hospital is strongly involved in end-of-life care with referrals specific for these procedures. This allowed us to evaluate in vitro and in vivo outcome of islet cell isolations from these donors while selecting conditions that have been shown beneficial for DCD organ procurement and preservation, such as the use of Institut Georges Lopez-1 (IGL-1) preservation solution and limiting acirculatory warm ischemia to 10 min 13 .
Materials and Methods
Euthanasia and Organ Donation
As in some other regions worldwide, a legal and ethical framework exists in Belgium allowing organ and tissue donation after euthanasia. The Belgian law stipulates a number of criteria that should be met before euthanasia can be performed [Wet van 28 mei 2002 Betreffende de Euthanasie (Law concerning Eutanasia); Belgisch Staatsblad/Moniteur Belge, June 22, 2002, https://justice.belgium.be]. This is the case for patients that are terminally ill but also for patients that suffer from untreatable neuropsychiatric disorders or dementia. Patients should voluntarily, well-considered and repeatedly express their request for euthanasia without external pressure. After approval of this request, patients may express the will to donate organs. Decisions and procedures regarding euthanasia and organ donation are completely separated, avoiding social or psychological pressure on the patient or physician 14 but also respecting the patients right to self-determination. Organ allocation is done by Eurotransplant (Leiden, The Netherlands) as an independent allocation organism.
Organ Procurement
Donation after euthanasia involves a particular procedure of donation after circulatory arrest and is classified as DCD V in the modified Maastricht classification 15 . End-of-life therapy can be performed at the hospitalization unit followed by rapid transportation of the donor to the operating theater during the 5-min no-touch period. Heparin (300 U/kg body weight) is administered immediately before administration of end-of-life medication, typically consisting of a sedation with 15 mg midazolam, followed by 3 g thiopental. In our center, as an alternative and after extensive discussion with the patient, sedation can be performed in the presence of the relatives with subsequent transportation to the operating theater where end-of-life therapy is administered. Also followed by a 5-min no-touch period after cessation of circulation before declaration of death by three independent physicians, this approach avoids rushing to the operating theater in the presence of mourning relatives and is preferred by more than 90% of our patients. A rapid sterno-laparotomy is then performed followed by cold flush, topical cooling, and procurement of the pancreas. Because of its beneficial effect on DCD pancreas preservation for islet cell isolation 13 , IGL-1 is used as cold preservation solution in our DCD V procurement. Total WIT is defined as time between end-of-life therapy and abdominal aorta cannulation initiating cold preservation. It consists of an agonal phase (time till circulatory arrest) and a subsequent acirculatory phase 16 . All procedures involved in organ donation after euthanasia were evaluated and approved by the ethical committee of our institution (CME 2013/V7).
Study Design and Cohort Selection
The database of our Beta Cell Bank was used to conduct a retrospective analysis of prospectively collected donor and procurement characteristics and associated quality control data of islet cell isolates. Between January 2015 and December 2020, 13 DCD V organs that matched our set criteria were processed and included for analysis. They were compared with our reference cohort of 234 donors after brain death (DBD) from the same time period and a cohort of 29 DCD III (donors after controlled circulatory death) organs selected for acirculatory WIT <10 min and IGL-1 preservation solution, conditions that have been shown to favor DCD islet cell isolation 13 . Reasons for euthanasia request are shown in Table 1. The study was reviewed and approved by the ethical committee of our institution (B.U.N. 143202042685 and CME 2005/136).
Reason for Request for Euthanasia in 13 DCD V Used for Pancreatic Islet Cell Isolation.

DCD V: donors after euthanasia.
Islet Cell Isolation, Purification, Quality Control, and Culture
Islet cells were isolated using a modification of the automated Ricordi method 17 and purified by continuous gradient with Biocoll (Biochrom, Berlin, Germany) and a cooled COBE 2991 cell processor (Terumo BCT, Lakewood, CO, USA). After isolation and purification, the cell preparations were cultured in cell culture flasks T175 (Sarstedt, Nümbrecht, Germany) at 37°C in a humidified incubator (5% CO2) in a Ham’s F10 based medium (Lonza, Bazel, Switzerland).
Preparations were characterized immediately after purification and after a 1- to 5-day culture period by their beta cell number, insulin content, and insulin purity, as described previously 17 . Beta cell number was calculated from the total nuclear count (NucleoCounter YC-100; ChemoMetec, Allerod, Denmark) and the percentage of insulin positive cells [immunocytochemistry with guinea pig anti-insulin (1/2,000, in-house produced) on 1.5 µm araldite sections; >2 × 103 cells counted; pictures were captured using Nikon Eclipse Ti microscope and analyzed with NIS-Elements AR v5.21 software (Nikon Europe, Amsterdam, The Netherlands; Fig. 1)]. Yield immediately post purification was also expressed as islet equivalent (IEQ), calculated using a volume based method after dithizone staining 19 .

Composition of islet cell preparation. Beta cell number is calculated from the total nuclear count and the percentage of insulin positive cells on immunocytochemistry. This is illustrated by a donor after euthanasia preparation stained for insulin in green (bar indicates 100 µm). Insulin-negative cells include glucagon- or somatostatin-positive cells and non-endocrine cells that consist predominantly of pancreatic duct cells 18 .
Clinical Transplantation and Assessment
Preparations are cultured and consecutively combined into a graft of one or multiple donor preparations. Islet cell grafts were defined by beta cell number, insulin content, insulin purity, and insulin biosynthesis capacity 18 .
In our protocol, non-uremic type 1 diabetes mellitus patients typically receive two IP 20 islet cell transplantations unless they achieve insulin independence or a serum human C-peptide level above 1.0 ng/ml (measured at blood glucose levels below 220 mg/dl) and a glycemia coefficient of variation <25% after a single islet cell infusion 1 .
Immunosuppression consists of an induction with anti-thymocyte globulin (bolus of 9 mg/kg followed by 3 mg/kg during 6 days, unless T-lymphocyte count <50/mm³) for a first transplantation or basiliximab (20 mg on day 0 and day 4) for a second transplantation. Patients receive a single injection of 500 mg methylprednisolone immediately before transplantation and anti-tumor necrosis factor (TNF) therapy during a 10-day period after transplantation (etanercept 50 mg on day 1 and 25 mg on days 3, 7, and 10). Maintenance therapy consists of mycophenolate mofetil and tacrolimus aiming at through levels between 8 and 10 ng/ml in the first year following transplantation, and 6 and 8 ng/ml thereafter.
Metabolic function was determined by glycosylated hemoglobin (HbA1c) concentrations, serum C-peptide levels, fasting blood glucose levels, and its coefficient of variation 21 . Daily insulin dose was adjusted to keep blood glucose levels between 70 and 180 mg/dl. Hyperglycemic clamp tests were performed to measure the achieved FBM expressed as percentage of non-diabetic controls1,22; BETA-2 scores were calculated as described 23 . All islet cell recipients gave written consent for use of their data in our clinical studies.
Statistical Analysis
Individual donor and procurement characteristics reported in the Eurotransplant donor and procurement file as well as on pancreas processing, isolation outcome, and culture characteristics were stored in our Filemaker database (Filemaker, Inc., Santa Clara, CA, USA). Results are presented as median [interquartile range (IQR)] if not otherwise specified. Statistical analysis was performed using SPSS (IBM, Armonk, NY, USA). Nonparametric tests (two-tailed Mann-Whitney U test for continuous variables, chi-square test for categorical variables) were used for analysis, unless otherwise specified. Statistical significance was assumed at P < 0.05.
Results
Donor and Procurement Characteristics of DCD V
Baseline characteristics of 13 DCD V are compared with a reference cohort of 234 DBD and to 29 DCD III selected for preservation solution and acirculatory WIT (Table 2). As most DCD V procedures are planned, time between admission to the hospital and organ recovery as well as time at the intensive care unit was nearly absent. There was no need for vasoactive medication, documented cardiac arrest, or episodes of hypotension in DCD V donors. This translated biochemically in normal serum sodium and creatinine levels. Blood glucose levels were normal in all DCD V. Serum lipase levels were significantly higher in DCD V when compared with DBD with levels above 60 U/l (reference value < 60 U/l) in more than 50% of cases (P = 0.014). Most DCD V procedures were performed at our campus and planned during office hours resulting in a median cold preservation time of 2 h, which is significantly shorter than the median preservation time in DBD or DCD III (P < 0.001), that are imported from other procurement hospitals in more than 50%. Pancreas extraction time was also significantly shorter in DCD V. Total WIT in DCD V ranged between 10 and 22 min, consisting of an agonal WIT between 1 and 14 min and acirculatory WIT below 10 min. While average body mass index (BMI) of DCD V tended to be slightly higher, age and North American Islet Donor Score (NAIDS) were similar between donor types.
Donor and Organ Procurement Characteristics for Pancreases Procured From DCD V Compared With DBD and DCD III.

Data are presented as median (interquartile range); statistical significance (Mann-Whitney U) was assumed at P < 0.05.
DCD V: donors after euthanasia; DBD: donors after brain death; DCD III: donors after controlled circulatory death; WIT: warm ischemia time; NA: not available; NAID: North American Islet Donor Score.
In Vitro Outcome of Islet Cell Isolation From DCD V
Isolation outcome parameters of DCD V are compared with DBD and matched DCD III, and are summarized in Table 3. Pancreas weight, percentage of undigested tissue, and post-digestion cell pellet volume were similar between all groups, while digestion time tended to be shorter for DBD and DCD III. Compared with DBD, DCD V pancreases yielded 50% more beta cells after purification and after 1 to 5 days of culture. When expressed as IEQ, difference in isolation yield after purification was 20%. Subgroup analysis of DBD organs that were preserved using IGL-1 showed higher beta cell yield than organs preserved using histidine-tryptophane-ketoglutarate or University of Wisconsin preservation solution [117 (72–217) vs 96 (44–138) × 106 beta cells after purification and 71 (43–127) vs 55 (30–81) × 106 beta cells after culture, data not shown], but this was still on average 25% lower than what was obtained in DCD V. When compared with DCD III, matched for IGL-1-preservation and acirculatory WIT <10 min, DCD V organs yielded on average two times more beta cells after purification. Beta cell recovery after culture was similar between donor types [DCD V, 61% (52–70); DCD III, 57% (45–73); and DBD, 64% (53–76); P = 0.674 and P = 0.512 vs DCD III and DBD, respectively]. DCD V preparations also contained a significantly higher percentage of insulin positive cells than DBD or DCD III preparations (35% vs 24% and 25%, respectively). Of the 13 donors after euthanasia, 10 islet cell preparations (77%) were used for clinical transplantation, while this was the case for 116 out of 234 DBD preparations (50%, P = 0.055) and 14 out of 29 DCD III preparations (48%, P = 0.083).
Isolation Outcome for Pancreases Procured From DCD V Compared With DBD and DCD III.

Data are presented as median (interquartile range); statistical significance (Mann-Whitney U) was assumed at P < 0.05.
DCD V: donors after euthanasia; DBD: donors after brain death; DCD III: donors after controlled circulatory death; IEQ: islet equivalent; DTZ, dithizone.
Functional Capacity of DCD V Islet Cell Preparation
The functional capacity of a DCD V islet cell preparation following transplantation was demonstrated by one DCD V islet cell preparation that fulfilled our quantitative release criteria for clinical transplantation of 2.0 × 106 beta cells/kg body weight (1,900 IEQ/kg body weight) after overnight culture. It contained 39% insulin positive cells with an insulin biosynthesis capacity of 35 pmol/106 beta cells/2 h (Table 4). It was transplanted in a patient who received an IP transplant 10 years earlier (IP 1 and 2) but with progressive decline in FBM to 6% of non-diabetic controls since 5 years (Table 1). While still on maintenance immune suppressive therapy, she underwent two new IP grafts of which the second IP (IP 4) transplant contained the single donor DCD V islet cell preparation (Table 1). Its in vivo outcome was demonstrated 3 months post-transplant by a hyperglycemic clamp test: this second implant increased FBM by 30% leading to a total FBM of 55% of non-diabetic controls. The obtained FBM was associated with an improved metabolic control as illustrated by HbA1c levels of 5.3%, a decrease in daily insulin requirements to 0.08 U/kg, and a drop in glycemia coefficient of variability from 19.5% to 11.5%. Implant function was also demonstrated in summary tracings of continuous glucose monitoring, with a mean glycemia of 106 ± 29 mg/dl and a time in range (percentage of time between 70 and 180 mg/dl) of 94%. Serum C-peptide levels were maintained above 3 ng/ml (Fig. 2B) during 3-year follow-up.
Recipient, Donor, and Graft Characteristics of a Patient That Received an IP Islet Cell Graft Prepared From a Single DCD V 9 Months After an IP Implant From a DBD.

IP: intraportal; DCD V: donors after euthanasia; DBD: donors after brain death; BMI: body mass index; IEQ: islet equivalent; NA: not available; PT: post-transplantation; NDC: non-diabetic control.
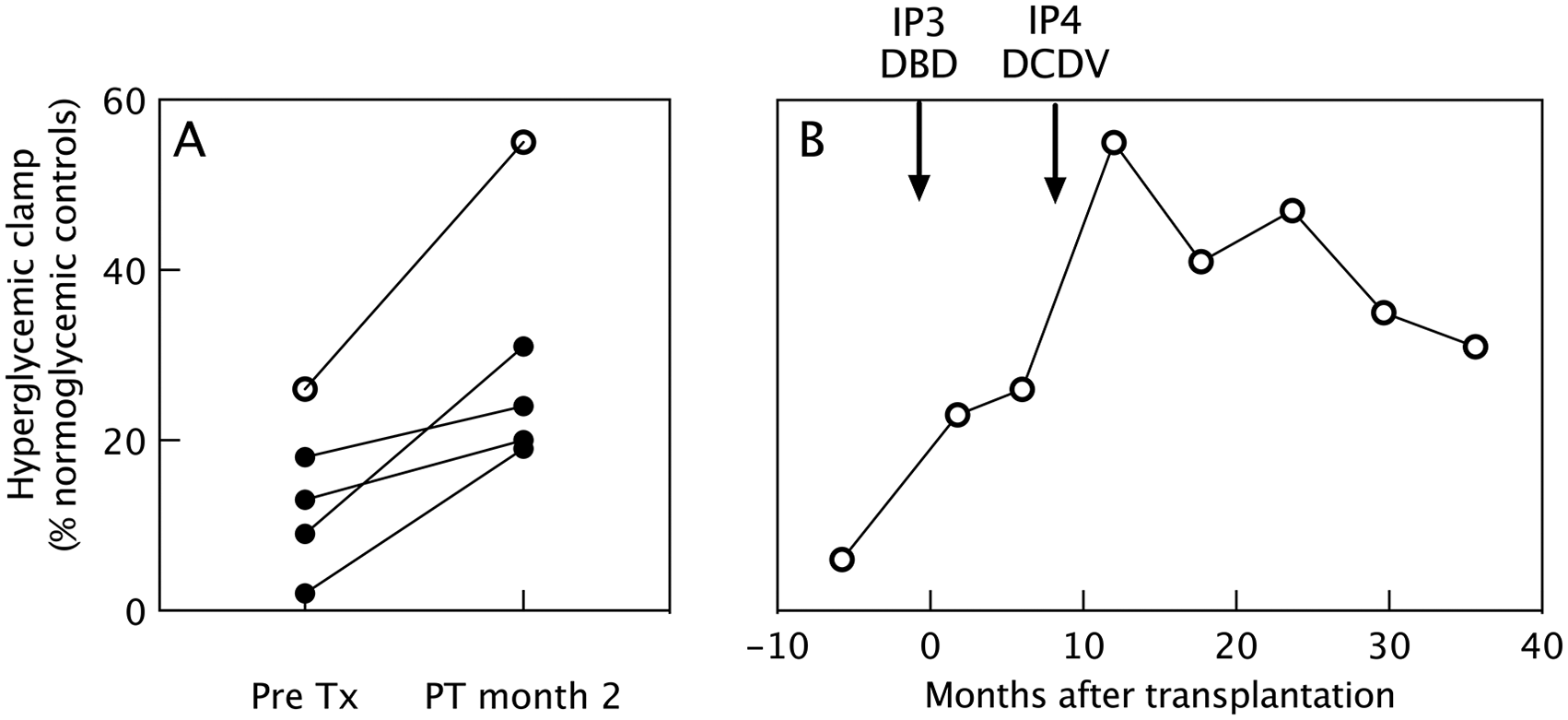
In vivo functional beta cell mass. Functional beta cell mass in intraportal implants as determined by hyperglycemic clamp area under curve (AUC) and expressed as percentage of non-diabetic controls. Comparison of in vivo effect of cultured islet cells prepared from DBD organs (full circles) or from a DCD V organ (open circles). (A) Following transplantation in five different patients. (B) Following consecutive transplantations in the same patient: IP3—single donor DBD, IP4—single donor DCD V: arrows indicate time of injection. DBD: donors after brain death; DCD V: donors after euthanasia; PT: post-transplantation.
We also compared the obtained FBM with the four patients receiving a 16- to 40-h cultured single DBD islet cell graft that contained on average 4.2 × 106 beta cells/kg recipient body weight (range = 2.2–5.1 × 106) at 28% insulin positivity (range = 25%–51%) and with an insulin biosynthesis capacity of 22 pmol/106 beta cells/2 h (range = 15–25 pmol/106 beta cells/2 h). They showed an increase in FBM that ranged between 6% and 22% (median 12%) (Fig. 2A) 2 months post-transplantation.
Discussion
Availability of donor pancreases remains a major limitation for clinical islet cell transplantation 4 . Some centers have therefore expanded their donor pool with DCD III24–28, but this is at the cost of lower isolation yields, which can at least partially be attributed to a period of warm ischemia before cold perfusion13,24,29. In Belgium, an ethical and legal framework is established for organ donation after euthanasia (DCD V), including in case of unbearable suffering due to neuropsychiatric disorders, unbearable pain, or dementia 30 . Like DCD III, this involves organ procurement after circulatory arrest which is associated with initial agonal and acirculatory WIT 12 . While in DCD III the agonal phase is a passive process of therapy withdrawal, in DCD V this is intentionally accelerated by the use of end-of-life medication. Consequently, agonal WIT and acirculatory WIT are shorter in DCD V8,13. Reports from DCD V liver transplantation show that despite shorter WIT, these organs are, as DCD III organs, at increased risk for non-anastomotic biliary strictures31,32.
Organ donation after euthanasia allows optimal conditions during daytime with a fully staffed procurement and islet cell isolation team. These donors have not underwent brain death nor associated cytokine storm 9 , which have a negative impact on isolation yield and in vivo function 10 . DCD V lung recipients showed good early graft function 33 , even when the procedure is initiated outside the hospital 34 . A recent study reported equal short- and long-term outcome as for DCD III and DBD 35 .
DCD V procedures constitute a minority (<10%) of all DCD procedures, but it has been suggested that DCD V organ donation has the potential to double the total number of donor organs available for transplantation 7 . This led us to compare DCD V islet isolation with the golden standard of DBD using in vitro and in vivo outcome parameters. At variance with reports of DCD III13,28, DCD V isolation yield was on average 50% higher than from our DBD cohort when expressed as beta cell number and 20% when expressed as IEQ. DCD V islet cell preparations also contained a significantly higher percentage of insulin positive cells and were more frequently used for clinical transplantation.
The semi-planned nature of euthanasia procedures allowed us to procure DCD V pancreases under conditions (acirculatory WIT ≤ 10 min, IGL-1 cold preservation solution) that have been shown to maintain islet cell isolation yield in DCD III at levels achieved for DBD 13 , further supporting the value of these conditions. When we compared isolation yield in DCD V with a cohort of DCD III from the same time period and matched for these beneficial conditions, beta cell yield after euthanasia was still 50% higher.
Our DCD V also exhibited donor profiles, including for BMI, time in hospital, and sodium and blood glucose levels that favor islet isolation yield36–38. This was not reflected in higher global NAID scores due to negative scoring of a short cold preservation time, which was below 3 h in more than 75% of cases in our DCD V cohort: While previous data emphasize on limiting cold ischemia time of pancreases procured for islet isolation to 8 to 12 h39,40, negative influences of shorter cold ischemia time have been reported 37 .
We did notice higher lipase values in DCD V, which has been reported to be associated with pancreas damage leading to a lower isolation yield36,37. However, in our DCD V cohort, lipase elevation may be related to the use of psychopharmaceutic drugs or opioid analgesics and therefore not influencing islet cell yield. Euthanasia requests and procedures for patients suffering from neuropsychiatric disorders have a predominance of female patients 41 . This was also the case in our DCD V cohort and might positively influence isolation yield 13 .
In our clinical program, islet cell isolates from multiple donors are cultured and combined to meet our quantitative release criteria for clinical transplantation 1 . Multiple injections of multi-donor islet cell isolates are usually needed to achieve metabolic goals, which complicates studying individual donor variables by in vivo outcome in patients1,42,43. Out of 10 DCD V preparations used for clinical transplantation, we identified one that was used as a single donor graft. Its in vivo function was demonstrated by a significant increase in FBM assessed by hyperglycemic clamp before and 3 months after islet cell infusion in this patient that received a previous IP DBD implant. This outcome parameter correlates well with decreasing glycemic variability and insulin needs 21 . Recently, and like others, we aim to continue exogenous insulin at a low dose to support the beta cell implant 44 . While exogenous insulin was not completely withdrawn, FBM and metabolic effect during 3-year follow-up was preserved at a level that has been reported to support a state of insulin independence 21 .
Several factors could have added to this metabolic outcome. The preparation contained a high percentage of insulin and glucagon positive cells, which has been shown in preclinical models to positively correlate with implant function45,46. It is conceivable that a prior period of 10-year immune suppression facilitated survival of the implant. It is likely that the survival of the previous islet cell implant contributed to the metabolic effect achieved by the DCD V implant. These variables also operate in transplants of DBD and DCD III organs, which further underlines the need for multifactorial analysis of well-defined study groups. The present study demonstrates that DCD V organs deserve their place in such analysis. It reports procurement and preservation conditions that more frequently resulted in cultured islet cell preparations meeting the criteria for use in our clinical transplant protocol.
Footnotes
Acknowledgements
The authors thank all procurement teams affiliated with the Eurotransplant network and all collaborators at the Diabetes Research Center of Vrije Universiteit Brussel (VUB) and Universitair Ziekenhuis Brussel (UZ Brussel).
Ethical Approval
Ethical committee approval was obtained from the Comity for Medical Ethics Universitair Ziekenhuis Brussel (UZ Brussel) and Vrije Universiteit Brussel (VUB), and is acknowledged within the text of the submitted manuscript. All procedures involved in organ donation after euthanasia were evaluated and approved (CME 2013/V7). Clinical islet transplantation was evaluated and approved (CME 2005/136). Retrospective study of prospectively collected data on donor, procurement, and isolation characteristics was evaluated and approved (B.U.N. 143202042685).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Comity for Medical Ethics Universitair Ziekenhuis Brussel (UZ Brussel) and Vrije Universiteit Brussel (VUB) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article. Written informed consent could not be obtained from deceased organ donors, but their use was according to Eurotransplant Guidelines and approved by the Comity for Medical Ethics Universitair Ziekenhuis Brussel (UZ Brussel) and Vrije Universiteit Brussel (VUB).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the European Commission (FP7 241883, H2020 681070), the Flemish Government (IWT 130138), the Juvenile Diabetes Research Foundation (17-2013-296, 2-SRA-2019-708-S-B), and the “Wetenschappelijk Fonds Willy Gepts” from Universitair Ziekenhuis Brussel (UZ Brussels).
