Abstract
Sepsis is associated with acute peritonitis, which can be induced by lipopolysaccharide exposure and feces. Generally, lipopolysaccharide induces mono-microbial peritonitis, whereas feces cause poly-microbial peritonitis; the latter is a more complicated and closer to the clinical diseases. Although several reports have discussed the mechanism of immune response in peritonitis-induced sepsis, however, the role of natural killer (NK) cells in sepsis, especially the relationship between NK cells and stabilization of the vascular endothelial barrier, is still unclear. Accordingly, in this study, we assessed the roles of NK cells in an acute sepsis model in mice. NK cells were injected via the tail vein into mice with acute sepsis, and nitric oxide (NO), anti-inflammatory cytokine, and angiogenic factors were tested to explore the effects of NK cells on sepsis. The survival rate of septic model mice infused with NK cells was significantly improved compared with the control group. Interestingly, the levels of NO, interleukin-10, and vascular endothelial growth factor (VEGF) decreased in NK cells therapy group. After the injection of NK cells, CD31 positive endothelial cells significantly increased in the kidneys and liver, although the expression of VEGF, ANGPT-1, and ET-1 was downregulated. Consistent with our hypothesis, the transfusion of NK cells into mice with sepsis blocked inflammation and increased endothelium integrity. Overall, these findings suggest that NK cells may block sepsis by modulating the VEGF pathway.
Introduction
Sepsis is a systemic inflammatory response caused by systemic or localized infections, resulting in devastating proinflammatory effects on the microcirculation 1 . Sustained inflammation during sepsis leads to disseminated microvascular thrombosis and organ ischemia. Furthermore, sepsis can manifest as disseminated intravascular coagulation (DIC), which is fatal 2 . A report by the National Heart, Lung, and Blood Institute proposed a unifying concept for sepsis as a severe endothelial dysfunction syndrome that results in multiorgan failure in response to intravascular or extravascular microbial agents 1 . Endothelial dysfunction leads to a widespread increase in vascular permeability 3 . Microcirculatory perfusion is responsible for fine-tuning of the oxygen supply to the organs 4 and microcirculatory alterations may play important roles in the pathogenesis of sepsis-related organ dysfunction 5 . These alterations have been reported in experimental models of sepsis in many organs, including the liver and small bowel mucosa 6 , and their occurrence has been evaluated 7 . Thus, increasing microvascular permeability and leukocyte activation, along with reduced vascular resistance and blood flow caused by sepsis, may result in tissue damage, multiple organ failure, and even death without appropriate treatment 8 .
Despite continual developments in treatment methods for sepsis, the effects of novel treatments are not sufficient. The most common treatment methods are antibacterial and antiendotoxin treatments 9 , antitissue factor and anticoagulation therapy 10 , anticytokine therapy 11 , and recombinant human activated protein C treatment 12 . Sepsis is characterized by inflammatory cytokine release, disruption of the vascular endothelial barrier, and alterations in fibrinolysis homeostasis 13 , often resulting in death. Although immune cells can launch a response to the alterations observed in sepsis, these responses have not been thoroughly defined. Therefore, further studies are needed to elucidate the immune mechanisms associated with sepsis to facilitate the development of novel and effective therapeutic strategies for the management of severe sepsis.
Natural killer (NK) cells are essential components of the natural immune system. These cells function to maintain homeostasis by killing tumor cells and virus-infected cells in the absence of specific immunization directly or by secreting various cytokines 14 . Moreover, NK cells provide continual and rapid responses to cancer or infections 15 and can regulate the immune state by affecting a variety of immune responses and by interacting directly with other immune cells. NK cells also can positively regulate adaptive immune responses by promoting the functions of T cells or dendritic cells (DCs) 16 and can negatively regulate these same immune responses via other mechanisms 17 . In addition, NK cells could secrete cytokines, such as vascular endothelial growth factor (VEGF), which is important for endothelial function and angiogenesis 18 ; however, the roles of NK cells in sepsis are still unclear.
Accordingly, in this study, we hypothesized that NK cells may have substantial effects in sepsis. Therefore, we investigated whether NK cells could decrease the inflammatory response or play protective roles in a mouse model of sepsis. As the key contributors to the pathogenesis of sepsis, proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, and IL-6 significantly increased by 18 h after sepsis induction 19 , so we injected NK cells at 0 and 19 h to reduce mortality and repair the injury in time. Furthermore, we carried out a preliminary study on the mechanisms of NK cell treatment with the goal of exploring the effects and mechanisms of NK cells in sepsis.
Method
Animals Studies
Our animal studies were performed with 8- to 10-week-old male C3H mice from HFK Bio (Beijing, China). Mice were housed under specific pathogen-free conditions (Animal laboratory code: SYXK_LU20200017). Mice had an average weight of 22 g (range: 20–25 g) prior to the start of the experiments.
Animal Experiments Statement and Ethics Declarations
All animal experiments were approved by the Ethical Committee on Animal Experiment of Shandong University Qilu Hospital (Approval No. DWLL-2019-023). Cord blood was collected from normal full-term pregnant women in department of obstetrics, and the use of cord blood was approved by the Ethics Committee of Qilu Hospital of Shandong University (Approval No. KYLL-2017(KS)-0143). This study was carried out in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines 2.0 and relevant regulations.
Preparation of Fecal Solution
The method of inducing peritonitis and the dose of feces refer to the published literature20–22. In brief, fresh feces were collected randomly from male C3H mice, weighed, and mixed with a calculated volume of normal saline solution to give fecal concentrations of 90 mg/ml. The mixture was thoroughly shaken before being filtered and centrifuged at 300 × g for 5 min. The supernatant was collected and stored at −80°C until further use.
NK Cells Culture and Expanded NK Cell Immunophenotype
NK cells expansion in genetically engineered K562 cells was performed from umbilical cord blood mononuclear cells (UB-MNCs). Irradiated K562 feeder cells were received from Hangzhou Zhongying Bio-Medical Technology (Zhejiang, China), which coexpress membrane-bound IL-21 and CD137L (4-1BBL) 23 . On day 0, UB-MNCs and 2 × 107 genetically engineered K562 cells (K562-mb215-41BBL, Zhongying Bio-Medical Technology) were resuspended in 40 ml NK cell medium [containing 90% RPMI-1640 HyClone, Logan, UT, USA; 10% fetal bovine serum, Gibco, Carlsbad, CA, USA; and 100 µg/ml IL-2, R&D, Minneapolis, MN, USA]. Complete medium changes were performed every 3 days, and the cell concentration was sustained at less than 2.0 × 106 cells/ml. On day 7, K562-mb215-41BBL cells (8 × 107) were added again. The cells were observed and counted every 3 days, and flow cytometry was conducted every 5 days.
Flow cytometry was performed to characterize NK cells. The following cell surface epitopes of NK cells were detected using mouse antihuman CD3 conjugated with fluorescein isothiocyanate (FITC) (Cat. No. ab34275; Abcam, Cambridge, UK), mouse antihuman CD56 conjugated with phycoerythrin (PE) (Cat. No. ab191100; Abcam), mouse antihuman NKG2D conjugated with PE (Cat. No. ab178446; Abcam), and mouse antihuman NKp30 conjugated with PE (Cat. No. FAB1849P; R&D). All antibodies were diluted 100 times and used according to the manufacturer’s protocol. Flow cytometry was performed using the Guava easyCyte 6HT (EMD Millipore, Billerica, MA, USA).
Mouse Peritonitis-Sepsis Model and Therapy Design
Male C3H mice were randomly divided into three groups: normal group (n = 12), in which 0.5 ml of 1 mg/ml saline was injected intraperitoneally (i.p.) in each mouse; control group (n = 12), in which 0.5 ml of 1.5 mg/ml fecal solution was injected i.p. in each mouse; and test group (n = 12), in which 0.5 ml of 1.5 mg/ml fecal solution was injected i.p. in each mouse. The experimental design was shown in Fig. 2A. The general conditions and survival states of mice were observed and recorded hourly. In our mouse model, bacterial load was fairly uniform, and the results were not affected by clinical procedures, including anesthetic and surgical techniques.
At 0 and 19 h after infection, mice received 0.2 ml of 1 mg/ml normal saline (normal and control groups) or 108 NK cells/kg in 0.2 ml (test group) injected via the tail vein. This NK cell injection dose is based on published data24,25. Their general condition and survival states were observed and recorded hourly.
Evaluation of Bacterial Clearance
Bacterial load was determined in blood and peritoneal lavage. In brief, mice were anesthetized with excess pentobarbital sodium at 24 h postinfection. The blood samples were collected in heparinized tubes through retro orbital sinus, mixed, and placed on ice bath. Mice were then injected 2 mL sterile saline i.p. and abdomen was gently massaged. Later the skin over the abdomen was cleansed with 70% alcohol and cut open to expose the peritoneal cavity to collect the peritoneal lavage fluid (PLF). PLFs and blood (100 µl) were placed on ice and serially diluted with sterile saline; 10 µl of each diluted sample was placed on Blood Agar Plates (Huankai Microbial, Guangdong, China) and incubated at 37°C for 24 h. The numbers of bacterial colonies were then counted and expressed as colony-forming units (CFUs) per milliliter of blood and peritoneal lavage.
Nitric Oxide (NO) Detection in Serum and Tissue Homogenates
At 19-h postinfection, the general conditions of the mice were observed. Upon euthanasia, cardiac blood was collected, and organs were removed. Mice were euthanized 24 h after infection if not prior, and excess pentobarbital sodium (Solarbio, Shanghai, China) was injected i.p. Cardiac blood was collected and centrifuged to obtain serum for the detection of NO levels. Peripheral blood (PB) was collected to measure routine blood biochemistry. The liver, brain, skin, spleen, bone marrow (BM), and kidneys were removed, and a portion of each tissue was stored at −80°C for later use; the other portion was homogenized for the detection of NO concentrations.
NO content was measured using an NO detection kit (KeyGen Biotech, Nanjing, China) using the nitrate reductase method. Absorbance was determined at 550 nm using a spectrophotometer (DNM-9602; Beijing Pulang New Technology Co., Ltd, Beijing, China). Overall nitrite levels were calculated as an indicator of total NO production.
Immunohistochemistry
BM was collected and stored at −80°C until subsequent use. Parts of the skin, liver, brain, kidneys, and spleen were stored at −80°C as well. The other portions were fixed with 4% formaldehyde (Solarbio), embedded in paraffin, and sectioned into 4-µm-thick sections. Immunohistochemistry was performed using an SP Rabbit & Mouse HRP Kit (3, 3’-diaminobenzidine, DAB; CWBIO, Beijing, China) according to the manufacturer’s instructions. Anti-CD31 primary antibodies (Cat. No. ab28364; Abcam; 1:100) were used to detect blood vessels within the tissues. The positive region of interest was measured in three independent sections from each sample (Olympus DP73; Tokyo, Japan).
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted with TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. RNA concentrations were measured by determining A260/280 absorption on a spectrophotometer (Merinton, Ann Arbor, MI, USA). RNA was then reverse transcribed into complementary DNA (cDNA) with a ReverTra Ace qRT-PCR RT Master Mix kit with genomic DNA (gDNA) Remover (TOYOBO, Osaka, Japan). The primers used for each gene are listed in Table 1 (BioSune, Shanghai, China). qRT-PCR was performed on an Applied Biosystems 7500 RT-PCR System (Applied Biosystems, Foster City, CA, USA) and detection using SYBR Green Realtime PCR Master Mix (TOYOBO) in 20 µl of reaction mixture: Stage 1: 95°C 30 s reps 1 and Stage 2: 95°C 5 s, 53°C 10 s, 72°C 15 s reps 40. Data were analyzed using Sequence Detection Software 1.4 (Applied Biosystems). Samples were assayed in triplicate, and mean values were calculated; gene expression level was determined by normalization to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) expression, and expression in the normal group was set to 1. The mean relative gene expression was determined and calculated using the 2–ΔΔCt method, and a melt curve was performed to check the integrity of the amplifications.
Primers Used in qRT-PCR.

qRT-PCR: quantitative reverse transcription polymerase chain reaction.
Western Blot Analysis
Western blot analysis was carried out to analyze the expression of VEGFA (vascular endothelial growth factor A) and VEGFR2 (vascular endothelial growth factor receptor 2) in spleen, liver, BM, and skin. In brief, the total protein of the petrous part of the temporal bone samples was extracted using Radio-Immunoprecipitation Assay (RIPA) protein extraction reagent (Beyotime, Shanghai, China) supplemented with phenylmethylsulfonyl fluoride (Beyotime) at 4°C for 20 min. Samples were then centrifuged at 4°C (15 min, 14,000 × g). The protein concentrations were measured using a bicinchoninic acid (BCA) protein detection kit (Thermo, USA). The lysates were separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and then transferred onto a polyvinylidene fluoride membrane. After blocking with 5% bovine serum albumin, the membrane was incubated with primary antibodies raised against VEGFA (Cat. No. ab46154; Abcam; 1:1000), VEGFR2 (Cat. No. ab11939; Abcam; 1:500), and GAPDH (loading control; ZSGB-BIO, Beijing, China). Goat antirabbit antibodies conjugated to horseradish peroxidase were used as secondary antibodies (1:500; SA00001-2, Proteintech, Rosemont, IL, USA), and proteins were visualized using enhanced chemiluminescence (Tanon, Tanon-5200, Shanghai, China). The band intensity was quantified using ImageJ software. Three independent experiments were performed.
Statistical Analysis
All data are presented as the mean ± standard error of the mean (SEM) of three separate experiments. An unpaired Student’s t-test was used to compare two groups, and one-way analysis of variance (ANOVA) followed by Bonferroni’s multiple comparison test was conducted to determine statistically significant differences among more than two groups. Statistical analyses were performed using SPSS Statistics 21.0 (SPSS, Inc., Chicago, IL, USA). A two-sided probability level less than 0.05 (P < 0.05) was regarded as statistically significant.
Results
Expansion of NK Cells
The overall experimental design and experimental grouping are shown in Fig. 1A. NK cells can be expanded upon activation by IL-2; therefore, we used genetically engineered K562 cells as feeder cells and IL-2 to enhance NK cell expansion in vitro; we obtained almost 4.3 × 108 cells with a high purity of up to 92%. The total number of expanded cells and the purity of CD3−CD56+ NK cells will reach a peak on day 15 and then drop slightly (Fig. 1B, C). An example of the immunophenotype of NK cells pre- and postexpansion and after infusion is shown in Fig. 1D, E. Expanded NK cells exhibited very high CD56 expression. Cell surface expression of the activating receptors NKp30 and NKG2D was also increased on expanded NK. These data demonstrate that NK cells are markedly activated after expansion. Therefore, NK cells expanded to day 15 were used in the following animal experiments.

Experimental design roadmap and ex vivo NK cell expansion. (A) Schematic of the treatments used at different time points in the three groups: Normal group, Control group, and Test group. (B) Expansion curve of total cell number of cord blood mononuclear cells during NK cell culture. (C) Changes in the percentage of CD3−CD56+ NK cells during amplification. Values are the means ± SEM from three independent experiments. (D) Representative flow cytometric analysis of CD3−CD56+ NK cells on days 5, 10, 15, and 20. (E) Representative flow cytometric analysis showing expression of the activating receptors NKp30 and NKG2D on unexpanded and expanded NK cells. NK: natural killer.
Injection of NK Cells Improved the General Condition and Survival Rates of Sepsis Model Mice
After intraperitoneal injection of fresh fecal solution, we observed that mice in the control and test groups showed less activity than those in the normal group. Mice in the control and test groups also showed adverse responses to touching of their abdomens; this response was not observed in the normal group. At 18, 20, and 21 h after infection, one mouse in the control group was euthanized; no mice in any of the other groups were euthanized (Fig. 2A).

Injection of NK cells decreased NO concentrations and weakened the inflammatory response. (A) Percent survival in the three groups. (B) NK cells decreased NO in different tissues 24 h after sepsis induction. (C) NK cells decreased numbers of WBC and NEU in PB. (D) Percentage of neutrophils in PB. (E) Numbers of WBC and NEU in PLF. (E) Percentage of neutrophils in PLF. (G) Effect of NK cells therapy on bacterial count in PB and PLF in sepsis mice. The data are expressed as the mean ± SEM. NK: natural killer; NO: nitric oxide; WBC: white blood cell; NEU: neutrophils; PB: peripheral blood; PLF: peritoneal lavage fluid. *P < 0.05; **P < 0.01.
Injection of NK Cells Weakened the Inflammatory Response and Decreased NO Concentrations
The highest NO levels were observed in the control group, whereas the lowest NO levels were observed in the normal group in blood and tissue homogenates 24 h after sepsis induction. Compared with the control group, injection of NK cells in the test group decreased NO concentrations, although not to the level of the normal group. Therefore, treatment with NK cells significantly suppressed the increases in NO levels, to some extent, following induction of sepsis (P < 0.05; Fig. 2B).
After fecal solution intraperitoneal injection, the numbers of leukocytes and neutrophils in PB and PLF were significantly higher than those in the normal group (Fig. 2C–F, P < 0.05 vs normal group). The number of neutrophils in PB was significantly reduced by tail vein infusion of NK cells (Fig. 2C, D, P < 0.05 vs normal group). Although NK cell infusion also reduced the number of leukocytes in PB, and the number of leukocytes and neutrophils in PLF, there was no significant difference compared with the control group (Fig. 2C, E, F, P > 0.05 vs control group).
To determine whether the protective effect of NK cells in the sepsis model was due to antimicrobial activity, bacterial CFUs were estimated in PB and PLF. NK cells showed significant reduction in bacterial count in PB (P < 0.05 vs control group) while no significant reduction in PLF (Fig. 2G, P > 0.05 vs control group).
NK Cells Improved Microcirculation in Kidneys and Liver
There are abundant blood vessels in the liver, kidneys, and skin. We used immunohistochemical technique to label CD31 positive vascular endothelial cells, which appear as brown during DAB staining. Feces-induced sepsis resulted in a significant reduction in the number of capillaries with brown positive signal in the kidney and liver (P < 0.01 vs normal group; Figs. 3 and 4). After the injection of NK cells, the number of brown positive capillaries significantly increased in the kidneys and liver (P < 0.01 vs control group; Figs. 3 and 4). However, no significant differences were observed in the skin and spleen following NK cell injection (data not shown).

Infusion of NK cells significantly increased the density of CD31 positive blood vessels in the kidney. The brown ring is considered a positive signal. The nuclei were counterstained with hematoxylin. After injection with NK cells, the positive signal increased. (A) Normal group. (B) Control group. (C) Test group. (D) Statistical analysis of quantified data from the three groups. The data are expressed as the mean ± SEM. Bar = 100 µm. NK: natural killer. **P < 0.01.
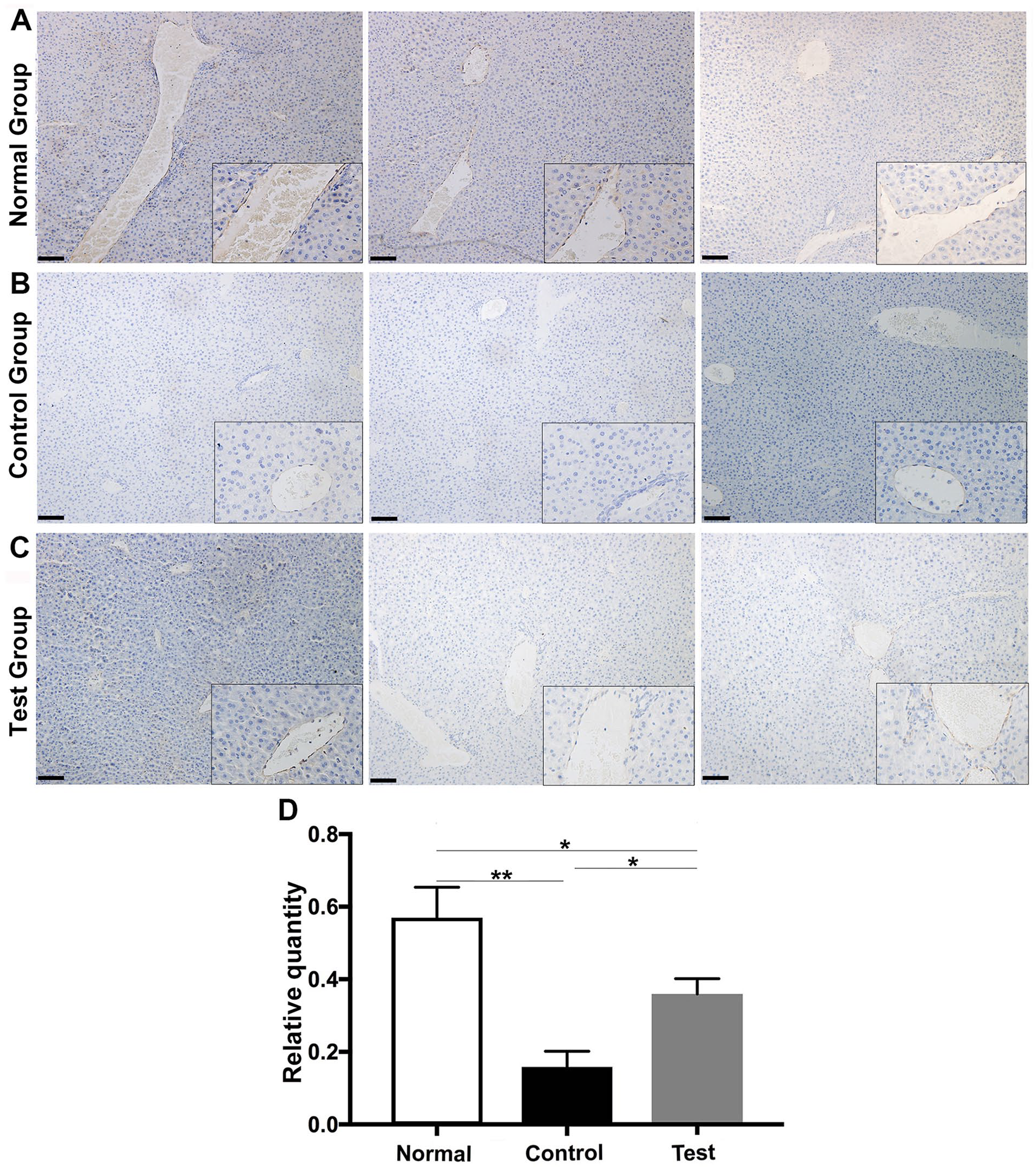
Infusion of NK cells significantly increased the density of CD31 positive blood vessels in the liver. The brown ring is considered a positive signal. The nuclei were counterstained with hematoxylin. After injection with NK cells, the positive signal increased. (A) Normal group. (B) Control group. (C) Test group. (D) Statistical analysis of quantified data from the three groups. The data are expressed as the mean ± SEM. Bar = 100 µm. NK: natural killer. *P < 0.05; **P < 0.01.
Injection of NK Cells Decreased the mRNA of IL-10, Angiopoietin-1 (ANGPT-1), Endothelin (ET)-1, and VEGF
Using RT-PCR method, we analyzed the mRNA of four cytokines in different tissues. Compared with that in the normal group, induction of sepsis in the control group resulted in increased IL-10, VEGF, ANGPT-1, and ET-1 expression levels; this effect was blocked by NK cell injection in the test group. Thus, injection of NK cells improved the condition and integrity of blood vessels at the gene level (Fig. 5).

NK cells altered IL-10, VEGF, ANGPT-1, and ET-1 expression. (A) IL-10 expression. (B) VEGF expression. (C) ANGPT-1 expression. (D) ET-1 expression. The data are expressed as the mean ± SEM. NK: natural killer; VEGF: vascular endothelial growth factor; ANGPT-1: angiopoietin-1; ET-1: endothelin-1; BM: bone marrow. *P < 0.05; **P < 0.01.
Injection of NK Cells Changed VEGFA and VEGFR2 Expression
In all the spleen, liver, BM, and kidney tissues we tested, VEGFA protein levels were increased in the control group compared with those in the normal group (P < 0.01 in spleen, liver, and BM, P < 0.05 in skin; Fig. 6). The expression of VEGFR2 also increased after sepsis modeling (P < 0.01 in spleen and P < 0.05 in liver). In addition, treatment with NK cells in the test group resulted in decreased levels of VEGFA proteins compared with those in the control group (P < 0.01 in liver and BM; Fig. 6A and E, B and F). For VEGFR2, it was significantly downregulated in spleen and liver after NK cell infusion, compared with those in the control group (P < 0.01 in spleen and P < 0.05 in liver). In BM and skin, the expression of VEGFR2 increased after NK cell infusion, compared with those in the control group (Fig. 6C and G, D and H). These findings suggested that feces-induced sepsis resulted in disorders of angiogenesis, whereas injection with NK cells alleviated these alterations to some extent in spleen and liver.

Western blot analysis of VEGFA and VEGFR2 expression. VEGFA and VEGFR2 protein expression changed significantly after sepsis model or NK cells infusion in (A, E) spleen, (B, F) liver, (C, G) BM, and (D, H) skin. These experiments were performed three times with similar results. VEGFA: vascular endothelial growth factor A; VEGFR2: vascular endothelial growth factor receptor 2; NK: natural killer; BM: bone marrow; Nor: normal group; Ctl: control group; Test: test group; GAPDH: glyceraldehyde-3-phosphate dehydrogenase. *P < 0.05; **P < 0.01.
Discussion
Sepsis is a fatal disease that is frequently encountered by clinicians worldwide. When organ dysfunction occurs, the mortality rate increases 26 . Thus, effective treatments for sepsis are urgently needed. Sepsis is induced by microbial components or toxins during infection, and most animal models of sepsis are poly-microbial models that mimic intra-abdominal sepsis and typically include feces-induced peritonitis 20 . In this study, male C3H mice were injected with fresh fecal solution, imitating sepsis induced by acute peritonitis 21 . This mouse model of sepsis was originally developed by Huet and colleagues 22 and remains a reliable and internally consistent approach. In our current study, three mice in the control group were euthanized within 24 h after infection and showed signs of abdominal pain. In addition, IL-10, NO, and ET-1 expression levels were upregulated, which was consistent with clinical manifestations of sepsis. These results suggested the occurrence of severe inflammation, disruption of endothelial integrity, and dysregulation of blood vessels and supported that the sepsis mouse model was successfully constructed 27 .
Injection of NK cells in our sepsis model improved survival rates in mice and reduced abdominal pain. Importantly, NK cells regulate immune responses, such as cytotoxicity and cytokine production, which affect DCs, macrophages, neutrophils, T cells, and B cells 28 . NK cell cytotoxic function in human sepsis is greatly decreased 29 . It is apparent that NK cells are major effectors of the final outcome in sepsis through modulation of interferon-gamma (IFN-γ) production 30 . NK cell activation provides protection from pneumonia through excess production of IFN-γ which inhibits bacterial growth and has a major impact on sepsis outcome 31 . When treated with recombinant IFN-γ, patients with severe sepsis demonstrate reversal of sepsis-induced monocyte dysfunction and overall improved sepsis survival 32 . Usually, human NK cells are reinfused into severe combined immune deficient (SCID) mice for tumor treatment experiments24,25,33. Although the immune molecules of human and mouse do not match, the infusion of human NK cells into sepsis mice may increase the intensity of mutual immune response. However, many factors secreted by human NK cells, including IFN-γ, granomycin, and TNF, can also partially simulate the role of mouse NK cells. In our study, NK cells partially protected mice from feces-induced sepsis via downregulation of NO. This suggests that transfusion with NK cells could improve outcomes in patients with sepsis. Infusion of NK cells also reduced IL-10 in septic mice, although there was no significant difference. IL-10, a characteristic cytokine marker of sepsis 34 , can counterbalance the proinflammatory response and is substantially upregulated during sepsis. Thus, detection of IL-10 levels may be useful for evaluating the severity of sepsis 35 . In this study, IL-10 levels were decreased after transfusion with NK cells, indicating that NK cells enhanced the body’s immune response, which may have protected the animals from death due to serious infection. In states of sepsis, critical illness, and states of inflammation, IL-10 potentiates effector T cell suppression that prolongs recovery and may dispose to increased complications.
The endothelial cell lining (ECL) of the vasculature is a unique cellular system that can regulate hemostasis and vasomotor control 36 . In sepsis, severe endothelial cell dysfunction occurs, resulting in dysregulation of the ECL and disruption of vascular activity 37 . The function of the ECL is determined largely by the glycocalyx, which is associated with sepsis 38 . Shedding of the glycocalyx is associated with sepsis-induced organ failure and may result in endothelial release of NO or ET 37 , thereby contributing to the loss of vascular reactivity, which may lead to DIC. Notably, NO metabolism plays important roles in the regulatory function of the ECL and influences vascular function. In a previous study, NO and ET expression levels were found to increase in endotoxic shock 39 . Consistent with these findings, in the current study, we also found that NO and ET levels increased plasma and tissue homogenates from septic mice. The maintenance of endothelial barrier integrity may be important in the development of novel therapies for the treatment of sepsis 40 . The inexplicable result in this experiment is that NK cells reduced the expression of these important proangiogenic factors, including ET-1, VEGF, and ANGPT-1, which is opposite to the increase of vascular density in immunohistochemistry item. It is also mentioned that the decrease of VEGF may be protective. It may be more important to maintain the stability of existing blood vessels than angiogenesis 41 . The barrier properties of endothelial cells are critical for the maintenance of water and protein balance between the intravascular and extravascular compartments. In septic mice, plasma VEGFA could destabilize the vascular endothelial barrier integrity41,40. VEGF levels are significantly elevated in septic patients and correlated with disease severity and vascular leak 42 . In addition, VEGF blockade was proven to be protective in models of experimental sepsis and is currently being tested for its therapeutic potential 43 . The upregulation of VEGF may increase vascular permeability 44 , and reducing the level of VEGFA is beneficial to protect septic mice 41 . Therefore, the decrease of VEGFA, ANGPT-1, and ET-1 in peripheral serum after infusion of NK cells is helpful to reduce the mortality of septic mice and maintain the integrity of vascular endothelial system.
Moreover, VEGFA and VEGFR2 protein levels were significantly lower in the test group than in the control group in the spleen, liver, and BM; the opposite results were observed in the skin. These findings suggested that there may be an interaction between NK cells and the VEGFA pathway to modulate angiogenesis in sepsis. However, more samples and detailed analyses are required to confirm these findings and elucidate the mechanism. Because NK cells can induce vascular growth, remodel the vasculature, and secrete angiogenic factors 45 , these cells may protect the integrity of the endothelium and promote angiogenesis. Indeed, we found that blood vessel formation increased in the kidneys and liver following treatment with NK cells; however, no differences were observed in the spleen and skin. The proangiogenic effects of NK cells have been reported previously 46 ; however, little is known regarding the angiogenic functions of NK cells in an inflammatory environment, such as sepsis. Thus, our findings indicated that NK cells promoted angiogenesis in the kidney and liver. Further studies are needed to increase the observation time, to assess the roles of NK cells in the spleen and skin.
In conclusion, we described the systematic response of mice with feces-induced sepsis to NK cell injection. After NK cell treatment, the levels of NO, IL-10 and VEGF in sepsis mice decreased, the bacterial load and the numbers of NEU also reduced. A major cause for these improvements might be the observed stabilization of the vascular endothelial barrier, which rendered the treated mice more tolerant to systemic infections. Mechanistically, the functions of NK cells in sepsis may be related to the VEGFA pathway and the integrity of the endothelium may play an important role. Further studies are needed to define the contribution of immune and vascular effects of NK cells for sepsis therapy.
Footnotes
Author Contributions
Jing Lan performed all the experiments and participated in writing the manuscript. Hong Zhang, Linghong Liu, Hui Zhao, Qing Shi, and Dong Li assisted with the experiments. Dong Li and Xiuli Ju conceived the project, analyzed the data, and actively participated in manuscript writing.
Ethical Approval
This study was approved by the Ethics Committee of Qilu Hospital of Shandong University, Jiangsu Province, China (Approval No. KYLL-2017(KS)-0143).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Ethical Committee on Animal Experiment of Shandong University Qilu Hospital (Approval No. DWLL-2019-023) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from each umbilical cord blood donor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science & Technology of Shandong Province (ZR2020MH063), Rongxiang Regenerative Medicine Foundation of Shandong University (No. 2019SDRX-18), and Quancheng Industrial Leader Project (2017018).
