Abstract
Mucositis is nearly inevitable following high-dose chemotherapy. Several pro-inflammatory cytokines play a role in pathogenesis of mucositis. Curcumin inhibits inflammatory cytokines through inhibition of nuclear factor kappa-β. We studied the effects of curcumin on the acute toxicities and inflammatory cytokines following melphalan (200 mg/m2) for autologous hematopoietic stem cell transplantation (HSCT) for myeloma. The control group (first 10 enrolled patients who received standard supportive care) was compared with curcumin group (next 30 patients who received chewable curcumin lozenges, 4 g twice daily from 2 days before melphalan till day +28 along with standard supportive care). The toxicities were recorded as per World Health Organization (WHO) criteria and CTCAE v3.0 as applicable. Cytokine profiling was done in both groups at similar time points. In the curcumin group, there was significant decrease in grade 3/4 vomiting (3% vs 40%, P = 0.01) and total parenteral nutrition use (47% vs 90%, P = 0.026). Grade 3/4 mucositis (43% vs 60%) and diarrhea (33% vs 70%) were also less, but not statistically significant. This coincided with 3.2-fold lower area under the concentration time curve (AUC) of IL-8 from day –3 to day 14 in curcumin group compared with control group (P = 0.039). We conclude that curcumin mitigates toxicities of high-dose melphalan, possibly through IL-8 modulation. Randomized studies are warranted to explore benefits of curcumin in HSCT.
Keywords
Introduction
Mucosal injury is an inevitable complication of intensive conditioning regimens used for hematopoietic stem cell transplantation (HSCT) 1 . It is a complex process involving any portion of the gastrointestinal tract resulting in inflammatory or ulcerative lesions. It manifests primarily as oral mucositis (OM), or with such symptoms as nausea, vomiting, and diarrhea as part of gastrointestinal mucositis. The incidence of World Health Organization (WHO) grades I-IV OM with certain myeloablative conditioning regimens is approximately 90%, up to three-quarters of them manifesting with severe mucositis (WHO grade III/IV)1–3. Similarly, gastrointestinal mucositis is reported in up to 20% to 50% patients receiving high-dose chemotherapy conditioning for HSCT 4 .
Proinflammatory cytokines such as interleukin-1 (IL-1), interleukin-6 (IL-6), interleukin-8 (IL-8), interleukin-17 (IL-17), tumor necrosis factor-alpha (TNF-α), transforming growth factor-β (TGF-β), interferon gamma (IFN-γ), and certain prostaglandins (PGE-2) play a central role in its pathogenesis 5 . Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-ĸB), modifies the genetic expression of these cytokines and enzyme6,7. Therefore, inhibition of NF-kB could be beneficial in the reduction of mucosal injury.
Currently, there are no universal protocols that have been accepted as a standard to prevent and treat mucosal injury in the transplant setting. Research is proceeding on several new fronts8–10. Cryotherapy is probably one of the most effective measures in reducing OM 11 , but trials vary considerably with respect to the duration of cryotherapy3,12, and most had small sample sizes. Also, as used in some studies, prolonged cryotherapy for more than 6 h becomes uncomfortable for patients 3 .
Accumulating evidence suggests that curcumin, a polyphenol derivative with low toxicity profile used for its anti-inflammatory actions, inhibits various inflammatory cytokines through inhibition of NF-ĸB13–16. Phase I studies in cancer patients have shown that curcumin can be given at doses ranging from 440 mg to 12 g daily without any dose limiting toxicity17,18. Curcumin typically has low bioavailability; however, special formulations such as solid lipid curcumin particles (SLCP) have been developed for better absorption 19 . The current study was conducted to test the efficacy of SLCP lozenges for the prevention of mucosal injury following high-dose melphalan conditioning in AuHSCT for multiple myeloma.
Patients and Methods
Patients, Setting, and Transplant Procedures
This was a single center prospective study. Patients were eligible if they were more than 18 years old, had performance status 0–2 on the Eastern Co-operative Oncology Group (ECOG) scale, provided written informed consent and were scheduled to undergo AuHSCT with any of the following 3 conditioning regimens—melphalan 200 mg/m2, busulfan with melphalan (Bu-Mel) or carmustine, etoposide, cytarabine, and melphalan (BEAM). Additionally, patients were required to have creatinine clearance >50 ml/min and normal liver function (serum bilirubin < 2 mg/dl and liver enzymes less than 5 times upper limit of normal). Patients who were receiving non-steroidal anti-inflammatory drugs (NSAIDs), aspirin, anti-oxidants, or systemic steroids for >3 months with the last dose being received within 1 week prior to beginning conditioning chemotherapy were excluded. Also patients with active infection at the time of beginning conditioning chemotherapy were excluded. Soon after the study approval, carmustine (BCNU) became unavailable in India, and hence we started using lomustine, cytarabine, cyclophosphamide, and etoposide (LACE) as conditioning regimen for lymphoma transplants. Subsequently, on finding that the acute toxicities were less with LACE regimen, we continued to use LACE for lymphoma transplants during the study period. Also, Bu-Mel was only used for Ewing’s sarcoma transplants and autologous transplants for acute myeloid leukemia, both of which are extremely rarely performed at our center. Hence, although the trial protocol permitted enrolment irrespective of the diagnosis, we eventually could enroll only patients undergoing AuHSCT for multiple myeloma following standard melphalan conditioning (200 mg/m2 on day –1). The study was approved by the institutional review board (protocol number 641) and all patients provided written informed consent. The trial was registered on clinicaltrials.gov (NCT04870060).
Stem cell mobilization was done using cyclophosphamide and granulocyte colony stimulating factor (G-CSF) in all patients. The source of stem cells was peripheral blood for all and all stem cells were cryopreserved. All transplants were carried out in single high efficiency particulate air (HEPA) filtered isolation rooms. As stated above, for all patients the conditioning chemotherapy was melphalan 200 mg/m2 on day –1 of HSCT (Inj Alkeran, GlaxoSmithKline pharma till March 2013 and Inj Megval, Emcure pharmaceuticals subsequently). This was followed by infusion of cryopreserved stem cells on day 0 (to a target of 3 x 106 CD34+ cells/kg). Anti-viral prophylaxis (with acyclovir) and antifungal prophylaxis with voriconazole was used for all patients. No anti-bacterial prophylaxis was used. Strategies for prevention and treatment of OM are explained below (under interventions).
Interventions
The study was conducted in two phases namely, the “Control phase / Control group” and the “treatment phase / Curcumin group.” Patients in the control group received only supportive care for prevention of OM in the form of hexidine mouth wash and clotrimazole lozenges. No additional prophylactic strategies were followed for prevention of OM in both groups. Patients in the curcumin group received four chewable curcumin lozenges (LongvidaTM, Pharmanza Herbals Pvt. Ltd., Gujarat, India) twice daily from 2 days prior to the start of chemotherapy (i.e. day –3) to day +28 post-transplant in addition to supportive care. Each 1 g lozenge contained 100 mg of curcuminoids. The study was conducted in a non-randomized fashion, wherein, the control and curcumin groups were enrolled sequentially. Initially, 10 patients were planned to be enrolled in each group. After the first 20 patients were enrolled, the protocol was amended to include 20 more patients in the curcumin group.
Cytokine Analysis
Blood and saliva samples of patients in the Curcumin group for the measurement of TNF-alpha, IL-1, IL-6, IL-8, IL-17, TGF-β, INF-γ, and PGE2 were collected 1 h prior to the first dose of curcumin (i.e. on day –3), day 0, then thrice weekly (Monday, Wednesday, Friday) till day +14, and then on day +28. The cytokines were analyzed using ELISA kits obtained from BD Biosciences. The assay was carried out in duplicate and the average of the two readings was considered for determining the levels. The area under the concentration-time curve (AUC) for each cytokine from day –3 to day +28 was estimated using the linear trapezoid rule 2 . In the control group, the first blood and saliva sample for cytokine level measurement was done on day –1, 1 h before melphalan infusion and then subsequent measurements were exactly similar to those in the curcumin group.
Outcomes and Measures
Patients were evaluated clinically for OM, diarrhea, vomiting, and use of opioid analgesics for relief of pain daily from the start of chemotherapy (day –1) till (day +28). Nausea was only included as an outcome of interest in the expanded curcumin cohort, but not at the beginning of the study. The toxicity grading was done as per the CTCAE 3.0 scale. Time to neutrophil engraftment (NE) was defined as first of 3 consecutive days when absolute neutrophil count (ANC) was greater than 0.5 x 109/L while time to platelet engraftment was defined as first of 7 consecutive days when platelet count was greater than 20 x 109/L unsupported. TPN was used as per our institutional policy for grade III/IV OM or for significant nausea and vomiting interfering with adequate oral intake or for severe diarrhea. The use and duration of days of TPN use was recorded. Similarly, opioids were used for pain due to grade III/IV OM and pain score was recorded on visual analogue scale. Incidence and duration of use of opiods were recorded.
Comparisons for all the above outcome measures were made between the patients enrolled in the control group (n = 10), who did not receive curcumin and those in the curcumin group phase (n = 30), who received curcumin up to 28 days post-transplant.
Pharmacokinetic Blood Sampling and Analysis
Blood samples for pharmacokinetic studies related to curcumin and curcuminoids such as desmethoxycurcumin (DMC) and bis-desmethoxycurcumin (BDMC) were done 1 h prior to the first dose, 0.5, 1, 1.5, 2, 4, 6, 8, 12 (±15 min) hours post first dose of curcumin lozenges and then weekly trough levels (i.e., before the morning dose) till day +25 and on day +28. Curcumin, DMC, and BDMC were measured by reverse-phase HPLC (UV-visible detection) method developed in-house. Briefly, 1 ml of plasma was incubated with 3 ml methanol at 800C for 10 min. The mixture was centrifuged. The clear supernatant was collected and evaporated to dryness under nitrogen. The residue was reconstituted with 1 ml methanol and filtered. After injecting 50 μL of the filtrate into the HPLC, curcumin levels were detected and quantified at 425 nm.
In addition, the quantity of conjugated curcumin and curcuminoids was determined by enzymatic hydrolysis of the glucuronide and sulfate conjugates. Briefly, aliquots (200 μL) of plasma were mixed with 600 μL Dulbecco’s PBS (Sigma-Aldrich) and 200 μL of 100 units/μL type II h-glucuronidase/sulfatase (total 20,000 units) in Dulbecco’s PBS and incubated at 370C. After 1 h, the incubated plasma was mixed with 1 mL of 0.2% formic acid to acidify the solution, then samples were extracted and analyzed for curcumin content using HPLC as described above. The difference between total curcumin and free curcumin was the concentration of curcumin present as conjugates.
Statistical Considerations
The study was initially designed to test the difference in serum cytokines between the control group and the curcumin treated group in a pilot study of 20 patients. After enrolling 20 patients, it was decided to convert the pilot study into a non-randomized phase 2 clinical trial based on the clinical outcomes observed in the test arm. It was estimated that a sample size of 30 patients in the treatment arm (curcumin treated group) would be required to show a difference of 25% in the incidence of grade III/IV OM between the two arms, assuming the incidence of grade III/IV OM to be 60% in the control and 35% in the treatment arm (power 80%, 2-tailed test, alpha -5%). Thus, a total of 40 patients were enrolled in the study.
The data were checked for normality using appropriate tests. Mann-Whitney U test was used to compare mean cytokine levels between the two groups at various time points. Spearman rho test was used to correlate clinical outcomes (ordered categorical data) and cytokine levels. Fisher’s exact test was applied to compare proportions between control and curcumin groups. No adjustment was made for multiplicity and P < 0.05 was considered statistically significant for all comparisons.
Results
Patients and Settings
Between October 2010 and July 2015, 40 patients were enrolled in the study. As mentioned above, the study was conducted in 2 phases. Initial 10 patients were accrued in the control group and 30 patients were accrued in the experimental phase (curcumin group). The baseline characteristics of the two groups are shown in Table 1. The two groups were comparable with respect to age at diagnosis, age at transplant, gender, diseases stage, disease status at transplant, and infused CD34 cell dose between both the groups.
Baseline Characteristics.

CR: complete response; GFR: glomerular filtration rate; ISS: international staging system; PD: progressive disease; PR; partial response; VGPR: very good partial response.
Clinical Outcomes of Transplantation
Transplant-related complications in curcumin versus control groups
The incidence of grade 3/4 OM was higher in the control arm as compared to the curcumin arm (6/10—60% vs. 13/30—33%) although the difference was not statistically significant. Similarly, the median duration of grade 3/4 mucositis was slightly longer in the control arm compared to the curcumin arm (5.5 days vs. 4.0 days); again, the difference was not statistically significant. Interestingly, protective efficacy of curcumin was observed against grade 3/4 diarrhea (7/10 vs. 10/30; P = 0.066), grade 3/4 vomiting (4/10 vs. 1/30; P = 0.01), and use of TPN (9/10 vs. 14/30; P = 0.026). Other notable but statistically non-significant difference was observed in the requirement of opioid analgesics for the treatment of pain due to mucositis (5/10 vs. 10/30). Seventeen out of 20 patients experienced nausea of some grade but only one of them had grade 3 nausea. These and other clinical outcomes (febrile neutropenia, engraftment kinetics, and duration of hospital stay) are summarized in Table 2.
Post Transplant Toxicity and Outcomes.

NS: non-significant; TPN: total parenteral nutrition.
*p < .05.
Engraftment status
No significant differences in time to neutrophil or platelet engraftment were observed between the two groups. Time to NE ranged from 10 to 16 days with a median of 11 days in both groups, whereas median time to platelet engraftment was 12 (11–17) days in the control arm and 11(9–20) days in the curcumin arm (Table 1).
Cytokines
Comparison of AUC from day –3 to day +14 showed a 3.2-fold lower exposure to serum IL-8 (P = 0.039) and 1.22-fold lower exposure to serum IFN-γ (P = 0.039) in the curcumin group compared with the control group. No significant difference was observed for other serum cytokines between the two groups. None of the salivary cytokines showed significant difference in exposure between the two groups (Table 3).
Cytokine Exposure (AUC d-3 to d14) in Control and Curcumin Groups.

AUC: area under the concentration-time curve; IFN-γ: interferon gamma; IL-1: interleukin -1; IL-6: interleukin -6; IL-8: interleukin-8; IL-17: interleukin-17; NS: non-significant; PGE-2: prostaglandins; TGF-β: transforming growth factor-β; TNF-α: tumor necrosis factor-alpha.
Cytokine levels and clinical outcomes
When all patients were considered (N = 40) serum IL-1, IL-8 and saliva PGE-2 showed significant positive correlation with the severity of clinical outcomes such as vomiting and pain due to mucositis as shown in Table 4. No other cytokine showed statistically significant correlation with any of the clinical outcomes (Supplementary Table 1). When treatment arm alone was considered (N = 30), salivary PGE-2, IL-1, and serum TGF-β and IL-8 showed significant correlation with clinical outcomes such as grade 3/4 OM, grade 3/4 vomiting and pain caused due to mucositis as shown in Table 5. No other cytokine showed statistically significant correlation with any of the clinical outcomes (Supplementary Table 2). A graphical representation of the cytokines showing correlation with clinical outcomes in curcumin group, all patients and both sets of patients is shown in Fig. 1.
Correlation Between Cytokines and Clinical Outcomes in All Patients.
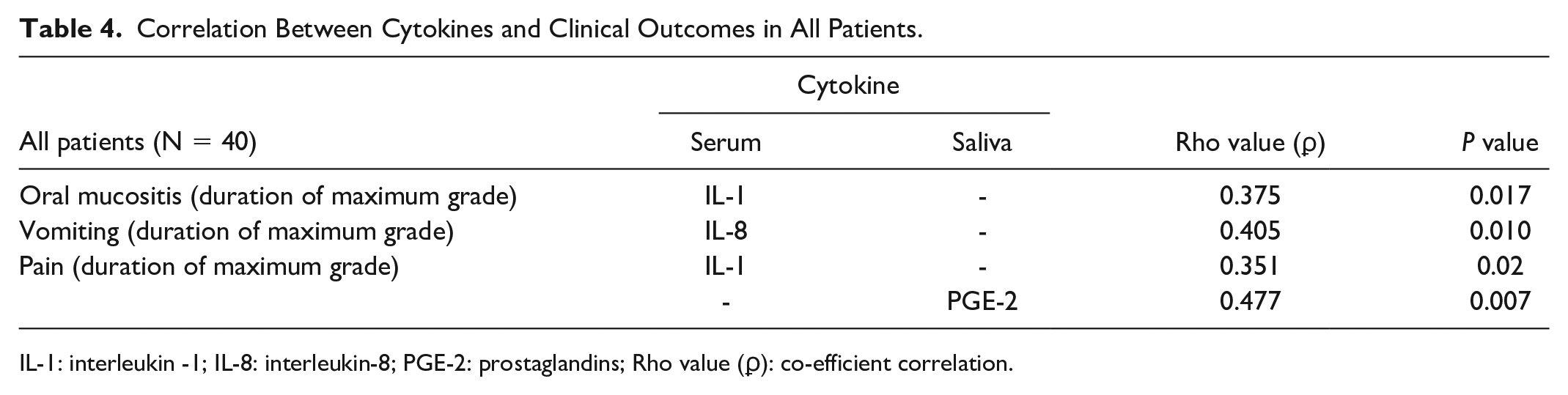
IL-1: interleukin -1; IL-8: interleukin-8; PGE-2: prostaglandins; Rho value (ϼ): co-efficient correlation.
Correlation Between Cytokines and Clinical Outcomes in Curcumin Group.
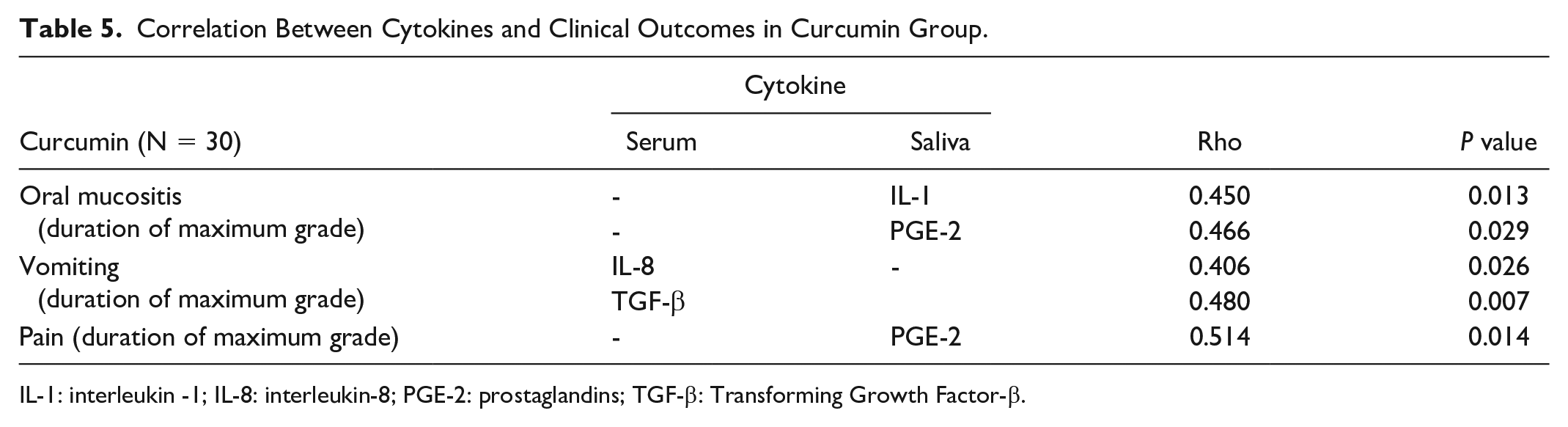
IL-1: interleukin -1; IL-8: interleukin-8; PGE-2: prostaglandins; TGF-β: Transforming Growth Factor-β.
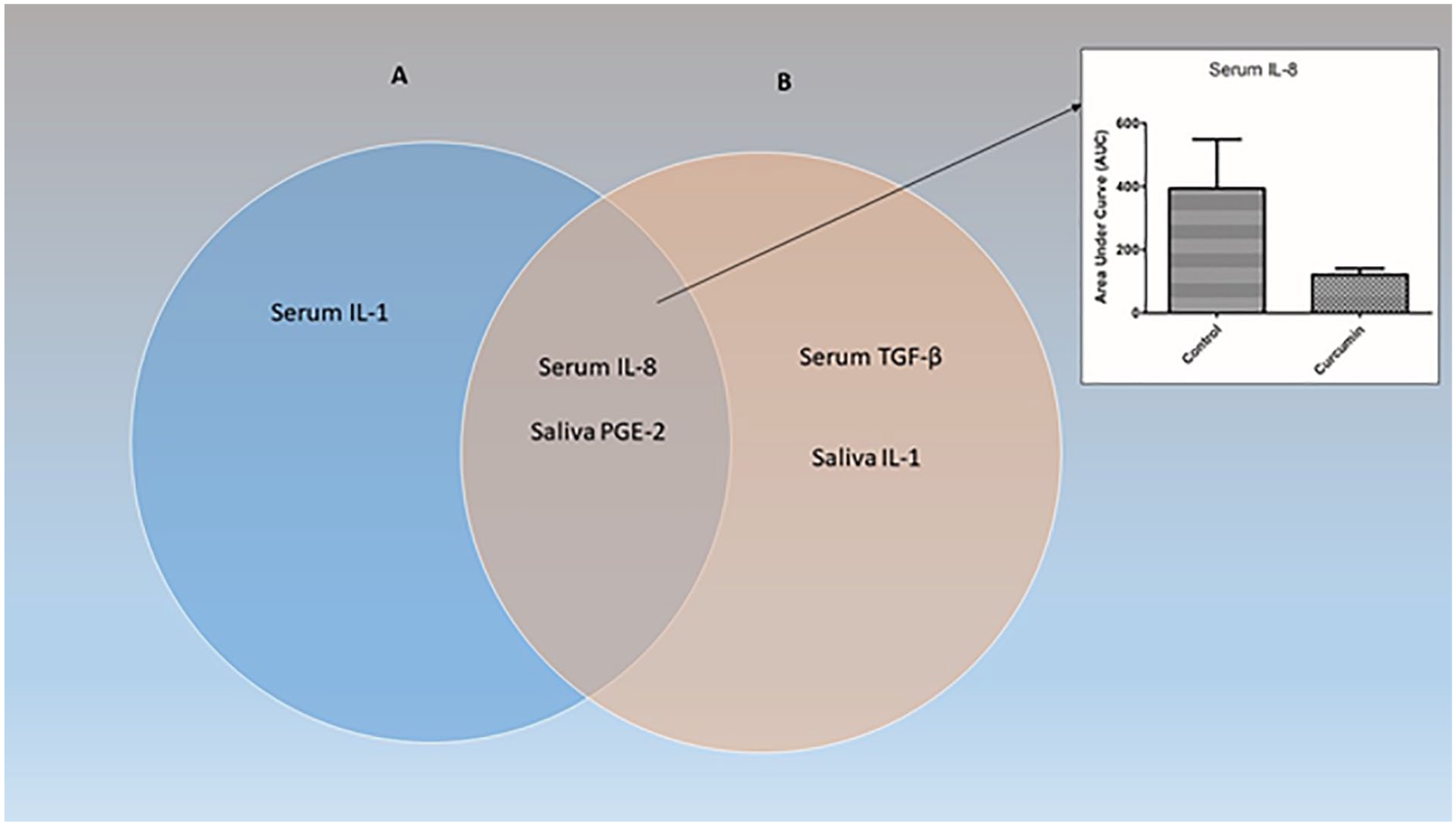
Venn Diagram: The Venn diagram enlists cytokines showing significant correlation with clinical outcomes in all patients (Circle A) and in the Curcumin group (Circle B). Cytokines common to both are listed at the intersection of the two circles. Out of these cytokines, only serum IL-8 showed significantly lower exposure in the Curcumin group compared with the Control group (Inset). IL-1: interleukin -1; IL-8: interleukin-8; PGE-2: prostaglandins; TGF-β: Transforming Growth Factor-β.
Pharmacokinetic Analysis
Drug levels
The pharmacokinetic parameters of the three curcuminoids are shown in Fig. 2 and Supplementary Table 3. The curcuminoids were predominantly (>90%) found as glucuronide conjugates. The AUC of free curcuminoid was found to be 6.0% for curcumin, 3.2% for DMC, and 4.2% for BDMC. Fig. 2 shows the exposure following the first dose of varying concentrations of curcumin, DMC, and bDMC with median and range.

AUC of curcuminoids. Scatter plot showing the area under the concentration-time curve (AUC) of curcumin, DMC and bDMC. The horizontal lines represent the median and range. bDMC: bis-desmethoxycurcumin; DMC: desmethoxycurcumin.
Discussion
Our study demonstrated the utility of oral curcumin in the prevention of some of the complications of high-dose chemotherapy with melphalan including nausea and vomiting, diarrhea, OM, and pain due to OM. Our findings showed that curcumin provides an overall protection against OM in terms of TPN use and the requirement of analgesics, although reduction in the incidence of OM per se was not statistically significant. Curcumin also helped reduce gastrointestinal mucositis by lowering the incidence of grade 3 and 4 vomiting and diarrhea compared with the control arm (although only the reduction in vomiting was statistically significant).
Most of these complications are a result of an inflammatory response to high-dose chemotherapy 20 . Previous studies have shown that proinflammatory cytokines such as IL-1, IL-8, TGF-β, TNF-α, and PGE-2, correlated with clinical outcomes. In the present study, serum IL-8 and serum IFN gamma were the only cytokines that were significantly modulated by curcumin; however, between the two cytokines only serum IL-8 showed a significant positive correlation with clinical outcomes. Hence, the reduction in post-transplant toxicities in the curcumin group could be possibly attributed to its ability to modulate the serum IL-8 levels. Interestingly, there was no significant difference in salivary IL-8 levels between the treatment and control groups. This can be attributed to the fact that curcumin’s ability to modulate cytokines in different organ systems is multifactorial, including the levels achieved in respective target organs. Therefore, it is not surprising that serum IL-8 levels were significantly different between the groups while salivary IL-8 wasn’t.
Curcuma Longa and its three components Curcumin, DMC and bDMC have been known to possess anti-inflammatory effects through the downregulation of NFκB 21 . The relative potency of the three curcuminoids for down-regulating NFκB is C>DMC>bDMC 21 . Interestingly, plasma concentrations of curcuminoids did not correlate with either clinical outcomes or the extent of cytokine modulation suggesting a lack of exposure-effect relationship (data not shown).
Several formulations of curcumin have been successfully tried in the past for the treatment of complications arising from chemotherapy or radiotherapy. There were mostly topical formulations for prevention of OM in head and neck cancer patients receiving chemoradiotherapy or for other indications where systemic chemotherapy was administered22–25. Only one study has demonstrated the utility of a nanomicelle formulation of curcumin in reducing the severity of OM in head and neck cancer patients undergoing radiotherapy following systemic administration 26 . However, this is for the first time that the benefit of curcumin has been demonstrated in patients receiving high-dose melphalan for AuHSCT. It is possible the benefit will hold for other high-dose chemotherapy regimens as well since the underlying mechanism of mucositis is the same for all high-dose regimens. Furthermore, it also proved useful for the prevention of vomiting, which is an important toxicity-related to GI mucosal injury.
In a Cochrane review published by Worthington et al, in 2011, 10 interventions for prevention of OM were analyzed and they concluded that there was some benefit for cryotherapy (ice chips) and palifermin in prevention of OM in transplant setting. According to the Oral Cavity Mucositis Guideline modified from MASCC/ISOO Clinical Practice Guidelines for Oral Mucositis, low-level laser therapy showed promising results as a prevention for OM. Laser therapy at a wavelength of 650 nm, power 40 MW, can be used for prevention of OM in patients undergoing high-dose chemotherapy followed by HSCT 27 . However, these agents are often prohibitively expensive and may not justify the cost, or may be cumbersome to deliver. For instance, palifermin use was associated with an additional cost of $5500 (myeloma) and $14,000 (lymphoma) per day of patient controlled analgesia (PCA) avoided 28 . Curcumin is affordable, safe at very high doses and easily available. Our study provides a strong proof of concept that curcumin lozenges comprising of solid lipid curcumin particles abrogates the GI-related toxicities of high-dose chemotherapy. The strength of the study lies in its comprehensive cytokine profiling. The study has a few limitations that needs to be considered. A possible reporting bias cannot be ruled out because of the non-blinded nature of the study. Also, the sample size being small, no definite conclusions can be drawn regarding the chemoprotective effects of curcumin, although the findings generate strong hypothesis for a confirmatory phase 3 trial. A large randomized study is presently underway at our center to confirm these findings.
To conclude, curcumin lozenges mitigated the clinical toxicities of high-dose melphalan such as vomiting, and the need for TPN. Curcumin treatment resulted in modulation of serum IL-8 which is an important mediator of these toxicities.
Supplemental Material
sj-docx-1-cll-10.1177_09636897221086969 – Supplemental material for Role of Curcumin in Reducing Toxicities Associated With Mucosal Injury Following Melphalan-Based Conditioning in Autologous Transplant Setting
Supplemental material, sj-docx-1-cll-10.1177_09636897221086969 for Role of Curcumin in Reducing Toxicities Associated With Mucosal Injury Following Melphalan-Based Conditioning in Autologous Transplant Setting by Sachin Punatar, Kritika Katti, Deepan Rajamanickam, Pravin Patil, Chetan Dhakan, Bhausaheb Bagal, Anant Gokarn, Avinash Bonda, Lingaraj Nayak, Murari Gurjar, Sadhana Kannan, Shubhada Chiplunkar, Vikram Gota and Navin Khattry in Cell Transplantation
Footnotes
Acknowledgements
We are also thankful to the Australia and Asia-Pacific Oncology Research Development (ACORD) workshop for help in designing the study and developing the protocol.
Ethical Approval
The study was approved by the Institutional Ethics Committee—III of Tata Memorial Centre (Protocol ID 641).
Statement of Human Rights
All procedures in the study were conducted in accordance with the Institutional Ethics Committee’s approved protocol (Protocol ID 641).
Statement of Informed Consent
Written informed consent was obtained from all the study participants. The consent document was approved by the Institutional Ethics Committee—III of Tata Memorial Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by institutional grant from Tata Memorial Centre, Mumbai, India.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
