Abstract
In the immunocompromised setting, recipients of solid-organ or hematopoietic stem-cell transplants carry an increased risk of post-transplant lymphoproliferative disorder (PTLD). Burkitt lymphoma (BL) PTLD is a rare form of monomorphic B-cell PTLD, which lacks a standard best treatment. Here, we report the successful treatment of refractory BL-PTLD with autologous anti-CD19 chimeric antigen receptor (CAR) T-cell therapy. A male patient was diagnosed with BL-PTLD, with an increasing Epstein-Barr virus (EBV) viral load, at 21 months after undergoing living liver transplantation from his mother due to neonatal biliary atresia. After 10 cycles rituximab +/− intensive chemotherapy and surgical tumor resection, the tumors significantly advanced. Next-generation sequencing (NGS) was performed on formalin-fixed paraffin-embedded tumor tissue, revealing one mutation in exon 5, TP53: p.A159 V, which may be associated with chemo-resistance. Thus, treatment was started with autologous anti-CD19 CAR T-cell therapy. We administered 9.0 × 106/kg autologous anti-CD19 CAR T-cells, after conditioning with cyclophosphamide and fludarabine. Unexpectedly, the patient experienced only mild (Grade II) cytokine release syndrome (CRS) without neurotoxicity. Finally, he went into complete remission (CR), and has achieved 16-month event-free survival to date. In addition, liver function has remained stably within the normal range without any immunosuppressive therapy. The literature includes only five previously reported BL cases treated with CAR T-cell therapy. In conclusion, the present case suggests that autologous anti-CD19 CAR T-cell therapy may represent a new therapeutic option for some cases of refractory BL-PTLD.
Clinical trial number: ChiCTR2000032211.
Introduction
Post-transplantation lymphoproliferative disorder (PTLD) is a rare but severe complication of solid-organ or allogeneic hematopoietic stem-cell transplantation, which is related to immunosuppression and the Epstein-Barr virus (EBV) 1 . The PTLD incidence after liver transplantation ranges from 1.0–5.5% in the adult population. The majority of cases are diagnosed in the first year post-transplant, although this disorder may occur at any time 2 . Burkitt lymphoma (BL) PTLD is a rare form of monomorphic B-cell PTLD that is associated with poor outcomes 3 . The first-line treatment involves rituximab-based chemotherapy, but other therapeutic strategies for BL-PTLD include reduction of immunosuppression 4 , surgical resection of localized disease, local radiation, immunochemotherapy, and cellular immunotherapy, such as with EBV-specific cytotoxic T-lymphocytes (CTLs) 5 . There are no reported clinical trials for BL-PTLD treatment, and the lack of standard treatment modalities renders BL-PTLD management challenging.
Here, we report the first case of refractory BL-PTLD that was successfully treated using autologous anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, which yielded sustained complete remission (CR) and manageable cytokine release syndrome (CRS). We also review the relevant medical literature.
Materials and Methods
Diagnosis and Clinical History of a Child with BL-PTLD
The male patient was born with pathological jaundice, and was diagnosed with neonatal biliary atresia. He underwent Kasai portoenterostomy, a palliative bilio-intestinal derivation procedure, but still developed decompensated cholestatic cirrhosis. At the age of 13 months, he received living liver transplantation (LT) using a lateral left-segment graft from his mother. Post-transplantation immunosuppression was performed using tacrolimus and prednisone.
At four months after liver transplantation, the patient showed positive EBV status. The virus DNA load gradually increased, reaching 3.9 × 104 copies/ml at 10 months after liver transplantation. At this time, the tacrolimus dose was halved and kept at below 5 ng/ml, but the EBV DNA load remained at a high level (Fig. 1A).
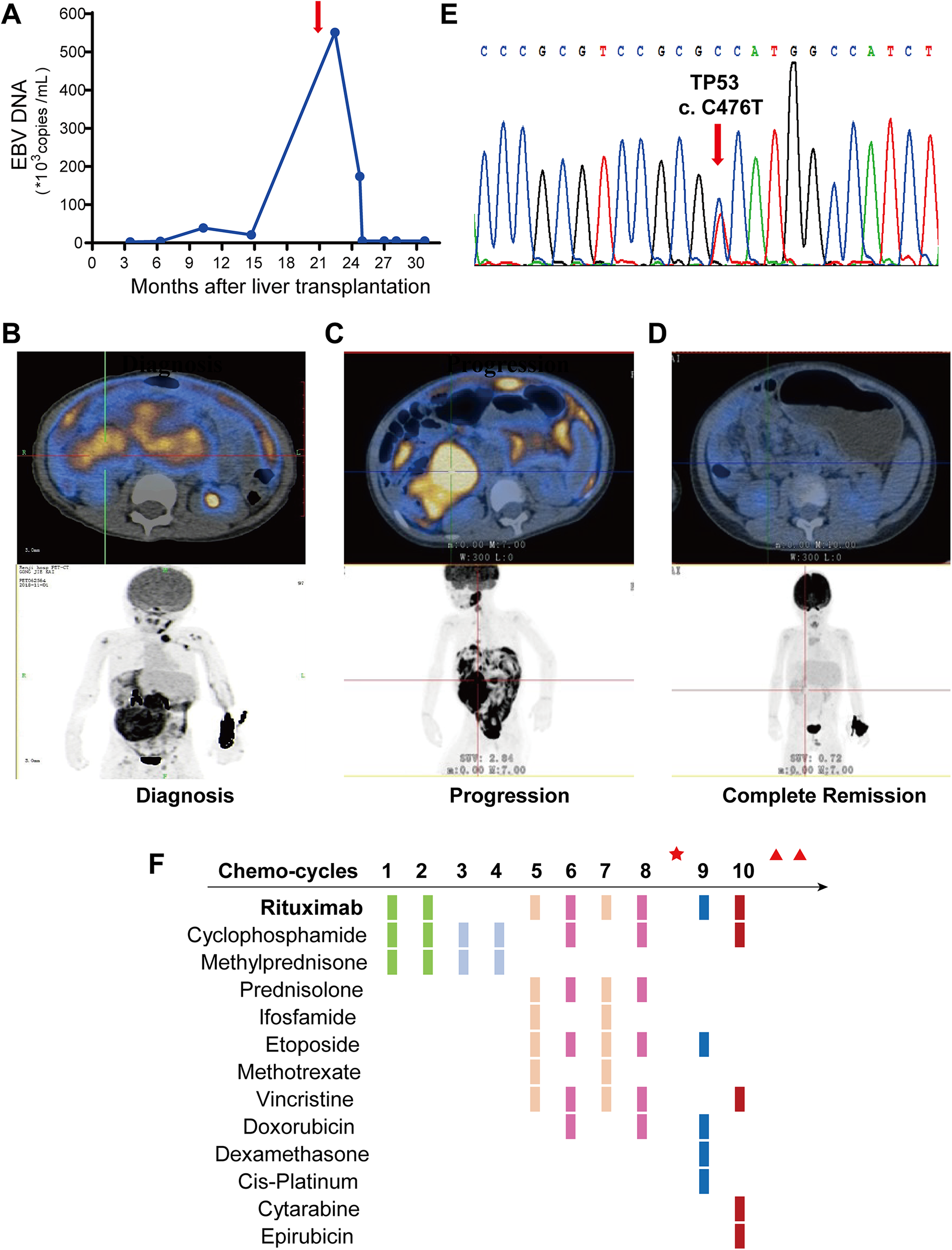
Patient’s clinical features before chimeric antigen receptor (CAR) T-cell therapy.
At 21 months after liver transplantation, the boy presented with lymphadenopathy in the neck, an abdominal mass of ∼7 × 10 cm in size, and repeated abdominal pain and vomiting. Positron emission tomography-computed tomography (PET-CT) revealed multiple nodules in the abdomen and pelvis, multiple enlarged lymph nodes in the left axilla and both sides of the neck, and multiple skeletal lesions that exhibited hypermetabolism (Fig. 1B). Abdominal magnetic resonance imaging (MRI) further revealed multiple abnormal signals in both kidneys, with hydronephrosis in the right kidney. The EBV viral load in peripheral blood was 5.51 × 105 copies.
Lymph-node mass resection was performed on the left side of his neck. Pathological morphology revealed tumor cells that were diffusely spread out and densely proliferated, exhibiting medium cell volume, small cytoplasm, and large nuclei, along with obvious apoptosis and starry-sky appearance. Immunohistochemical analysis indicated that the tumor cells were CD19+, CD20+, CD10+, Bcl6+, Bcl-2 part+, Ki67+ (98%), CD3−, CD30−, and ALK− (Fig. 2A, B). EBV status was positive based on in situ hybridization for EBV-encoded small RNA (EBER) in resected tissue (Fig. 2C). Fluorescence in situ hybridization (FISH) indicated an abnormal C-myc gene fraction (Fig. 2D). Based on these findings, the diagnosis was BL-PTLD according to the 2008 World Health Organization (WHO) classification 6 .
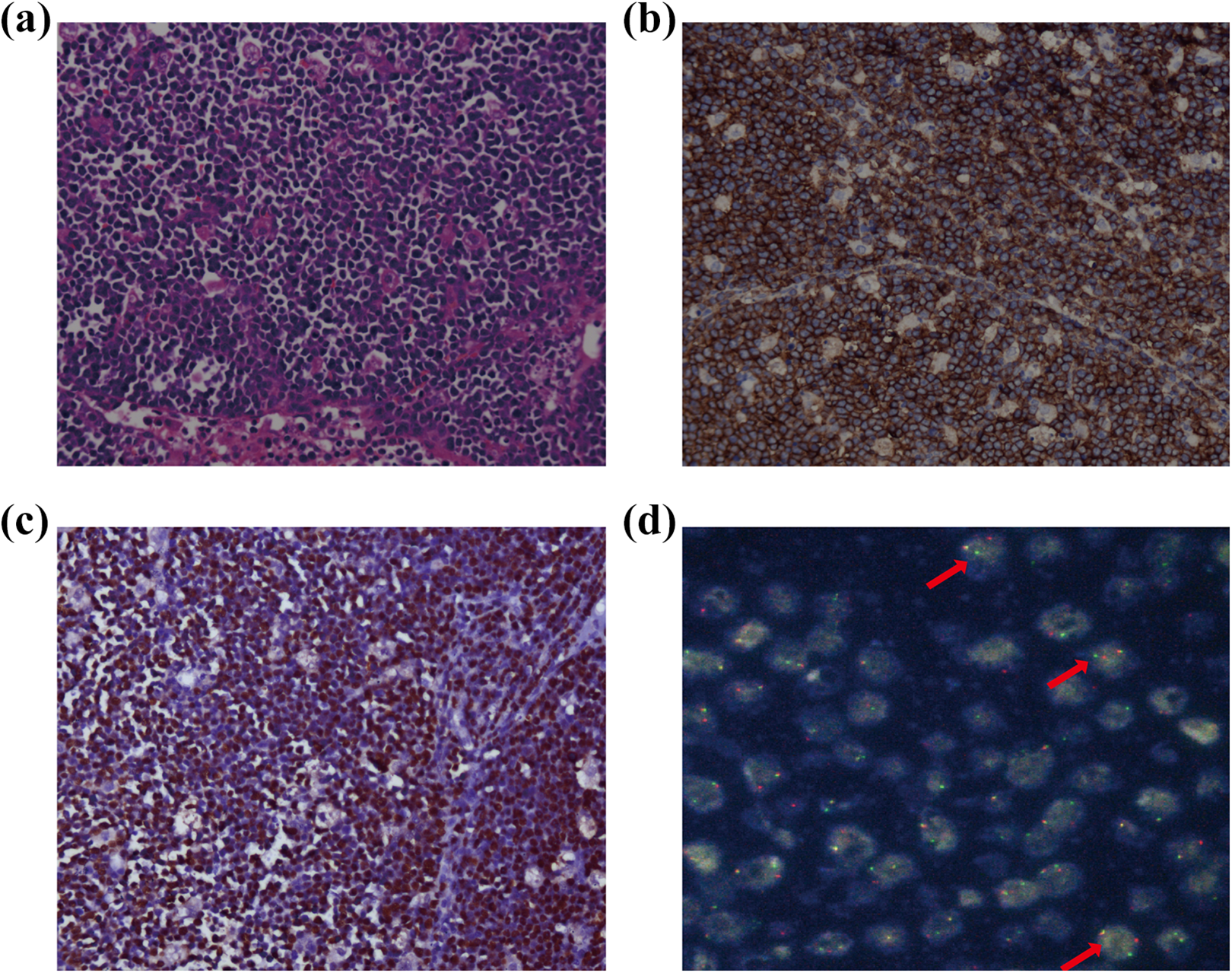
Pathological analysis of the lymph-node mass led to diagnosis of Burkitt lymphoma post-transplant lymphoproliferative disorder (BL-PTLD).
Tacrolimus was stopped upon the diagnosis of PTLD. The patient was administered two courses of rituximab, cyclophosphamide, methylprednisone, and intrathecal prophylaxis (I/T) with methotrexate, dexamethasone, and cytarabine; followed by two courses of cyclophosphamide AND methylprednisone. However, subsequent PET-CT revealed that the tumor had increased in size (Fig. 1C). Thus, four more intensive rituximab-based regimens were added. At this time, the EBV viral load was negative. Next, the boy underwent ileocecal tumor resection and lymph node dissection to reduce the tumor burden. Pathological examination indicated continued existence of tumor cells with necrosis. Surgery was followed by two more chemotherapy courses. Subsequent computed tomography (CT) revealed multiple soft tissue masses in the abdominal cavity, which were significantly more advanced than those previously observed. Next-generation sequencing (NGS) was performed on a formalin-fixed paraffin-embedded tissue sample, revealing one mutation in exon 5, TP53: A159 V (Fig. 1E), which may be associated with chemo-resistance. In an effort to reduce the tumor burden, the patient was administered radiotherapy (total 25 Gy) and tomotherapy (total 16 Gy). Figure 1F presents the specific treatment process.
Results
Administration of Autologous Anti-CD19 CAR T-Cells
Since the patient suffered chemo-resistance disease, we considered administering salvage treatment with CAR T-cells. However, data regarding the efficacy of CAR T-cell therapy in BL are lacking (Table 1) 7,8 . In our experiences of treating over 100 cases of refractory and resistant CD19-positive B-cell acute lymphoblastic leukemia (ALL), CR has been achieved in more than 98% of patients (data not reported). Thus, we decided to administer CAR T-cell therapy. The patient was enrolled in our trial (ChiCTR2000032211) and informed consent was signed by the patient’s guardian. Peripheral blood CD3-positive T lymphocytes were collected from the boy. These T-cells were activated for two days, and then transduced with murinized second-generation FMC63-based anti-CD19 CAR lentivirus, with signals provided by the costimulatory molecules 4-1BB and CD3-zeta. Five days later, a sufficient number of cells were washed and resuspended in saline solution with 2.5% human serum albumin (Fig. 3A). The transfection rate of CAR T-cells was 49.1% (Fig. 3B). To confirm the cytotoxic effect of the CAR T-cells, we incubated anti-CD19 CAR T-cells from the patient, CAR T-cells from a healthy donor, or untransduced T-cells with ALL cell lines (GFP-positive Reh cells) for 24 h at an effector-to-target ratio of 1:4. Both kinds of CAR T-cells killed almost all Reh cells, indicating that the patient’s CAR T-cells had cytotoxic effects comparable with CAR T-cells from a healthy donor (Fig. 3C).
Treatment and Outcomes from Prior Trials of CAR T-Cell Therapy for Burkitt Lymphoma (Literature Review).

a This case was treated with sequential anti-CD19, 22, and 20 autologous CAR T-cell therapies.
HSCT: hematopoietic stem cell transplantation.
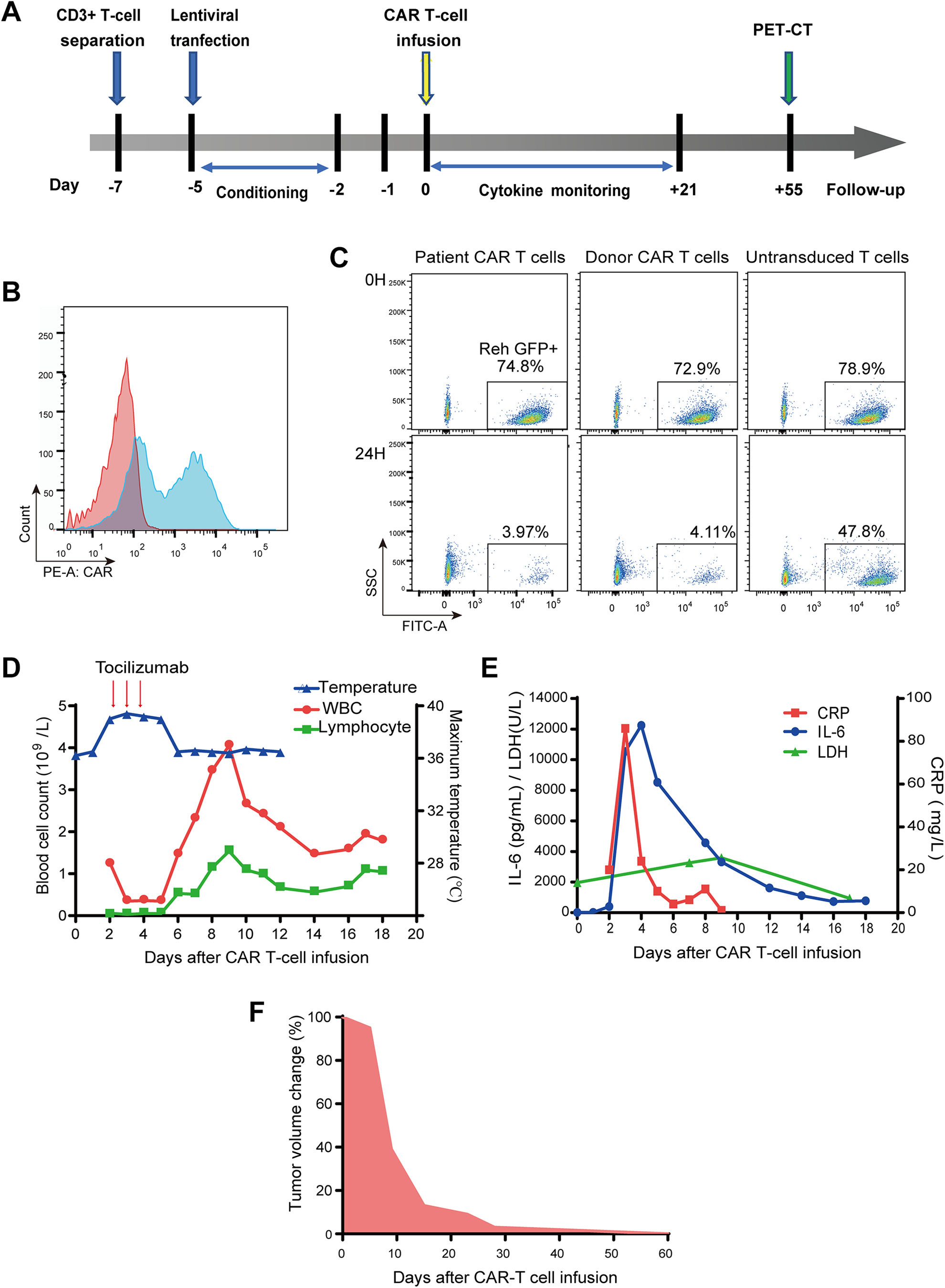
Chimeric antigen receptor (CAR) T-cell therapy efficiency and clinical response.
Starting on day 5, the patient was administered conditioning chemotherapy (cyclophosphamide 500 mg/m 2 /day and fludarabine 40 mg/m2/day) for two days to deplete lymphocytes. Next, on day 0, the child was infused with fresh anti-CD19 CAR T-cells at a total dose of 9.0 × 106 /kg. He was closely monitored for clinical symptoms and physical signs, and grade II CRS was observed without neurotoxicity according to the ASTCT criteria 9 . On days 2–5, the patient experienced a high fever (Fig. 3D). We also detected a significant increase of serum inflammatory cytokines, especially interleukin-6 (IL-6), with a peak IL-6 level of 12245.58 pg/ml on day 4 (Fig. 3E). The trends of changes in C-reactive protein (CRP), lactate dehydrogenase (LDH), and body temperature were consistent with the cytokine levels and the patient’s clinical symptoms (Fig. 3D, E). To control CRS, the patient was administered the anti-interleukin-6 receptor antibody tocilizumab 160 mg/day for 3 days (days 2–4). We did not use glucocorticoid due to concerns about impairing the expansion and antitumor activity of the CAR T-cells. On day 6, the child’s body temperature returned to normal. A series of routine blood panel examinations revealed that the white blood cells and lymphocytes were lower in peripheral blood after lymphodepleting treatment, and then increased on 7–11 days after CAR T-cells infusion (Fig. 3D). The patient then experienced vomiting for six days but recovered after supporting therapy. The patient showed no cough, headache, hypotension, or seizure during treatment, and all physical signs of CRS were manageable.
Exceptional Tumor Response to CAR T-Cells and Stable Graft Function
After CAR T-cell therapy, the tumor size was monitored weekly by abdominal and pelvic B ultrasound. The approximate volume was gradually reduced (Fig. 3F). On day 55, a PET-CT scan was performed, which revealed that the previously observed abdominal, pelvis, and multiple skeletal lesions had essentially disappeared. At this time, no typical malignant tumor-like hypermetabolic lesions were observed anywhere in his whole body. Thus, these imaging results indicated that the patient had achieved CR (Fig. 3D).
Abdominal CT scanning has been repeated every 6 months since then, with no typical tumor lesions discovered. Disease-free survival has persisted for 16 months to date. Other tests have been normal, with the exception of B-cell aplasia, requiring immunoglobulin replacement (0.5g/kg per month) to maintain immunoglobulin levels. The boy has remained free from any serious infection.
Graft function has been monitored monthly by blood test. Until now, the patient does not receive any immunosuppressive therapy, and the liver function has remained stably within the normal range. A liver biopsy was performed 12 months after CAR T-cell therapy, which revealed mild lymphocyte infiltration and hepatic lobule fibrosis, with no sign of rejection. The chimerism assay based on the rs4646006(del) and rs1610937(insert) sites of the donor revealed a 0.005% chimeric rate, which may partially account for the immunotolerance.
Discussion
Current knowledge of PTLD is limited due to its rarity, morphologic heterogeneity, and the lack of prospective trials. Recent data have established rituximab as the standard of treatment, either as monotherapy or followed by systemic chemotherapy 10,11 . A less-aggressive low-dose chemotherapy regimen comprising cyclophosphamide and prednisolone (Cy/Pred) with rituximab yielded a 100% overall response rate (five CR and one PR) for PTLD treatment 12 . However, some BL-PTLD patients still experience disease progression or other treatment failures despite these improvements, and these patients face a dismal prognosis 13 . There remains a need for new and tolerable therapies for such cases. Some researchers explored autologous or third-party EBV-specific CTL to treat rituximab-refractory PTLDs, and the response rates were about 50%∼60% 14 . However, considering the partial human leukocyte antigen (HLA) matching and allogeneic nature of the third-party CTL, we expected them to be short-lived in vivo since the host immune system would target the mismatched HLA molecules. Besides, none of the EBV-specific T-cells products have yet qualified in China. Chemo-resistance is driven by TP53 mutations in many different cancers, including aggressive B-cell lymphomas. In the presently described case, based on the failure of rituximab-based intensive chemotherapy and the finding of TP53 p.A159 V mutation in tumor tissues, we decided to administer autologous anti-CD19 CAR T-cell therapy.
To our knowledge, currently, no trials investigating the use of CAR T-cells for BL-PTLD treatment, many trials have proven the effectiveness of CAR T-cells in B-cell lymphomas 15 . Thus, one could hypothesize that similar results might be obtained in the setting of BL-PTLD. Although there was one trial that reported poor response and death outcomes for three patients with diffuse large B cell lymphomas (DLBCL) PTLD after heart and kidney transplant received CAR T-cell therapy 16 . In one case of an adult patient with refractory BL, a complete response was achieved with anti-CD19 CAR T-cells 17 . Here, we report the first case of refractory BL-PTLD that was successfully treated using autologous anti-CD19 CAR T-cells, which yielded sustained complete remission with an acceptable toxicity profile.
When CAR T-cells engage with target cells, they release a variety of cytokines and chemokines. Macrophages and other innate immune cells also become activated and contribute to this cytokine production 18 . Thus, CRS is the most frequent adverse reaction, which often occurs within two weeks of treatment. Fever is the earliest and most common CRS symptom. Tocilizumab blocks the IL-6 receptor (IL-6 R) and is considered as the first-line treatment for severe CRS. Glucocorticoids exert a wide range of immunosuppressive effects and are commonly used as second-line immunosuppressive drugs. Multivariable analysis of baseline characteristics has identified high tumor burden as an independent predictor of severe CRS 19 . Unexpectedly, our patient experienced only mild CRS.
In our patient, B-cell aplasia occurred as an on-target but off-tumor toxicity, which necessitated immunoglobulin replacement to maintain normal immunoglobulin levels. A prior study reported that B-cell aplasia occurred in all patients who exhibited a response to CAR T-cell therapy and could persist for up to one year after CAR T-cells were no longer detectable by flow cytometry 20 . Notably, B-cell aplasia can be used as a pharmacodynamic measure of the persistence of functional anti-CD19 CAR T-cells 21 .
Resistance or relapse has been reported in patients receiving CD19-directed CAR T-cell therapy. At Boren Hospital in Beijing, China, a child with relapsed refractory BL was administered sequential treatments with anti-CD19, 22, and 20 CAR T-cells. Unfortunately, the patient showed no discernible response and exhibited progressive disease following anti-CD19 CAR T-cell treatment 22 . BL relapse reportedly occurs mostly within the first six months of treatment 23 . The boy in our present case has survived without relapse for 16 months to date. There remains a need for additional clinical data from cases of relapsed/refractory BL-PTLD to determine whether it is preferable to develop CAR T-cells against multiple targets or against CD19 alone and whether CAR T-cell therapy should optimally be used as a solo curative therapy.
Immune system balance is the key for PTLD treatment. Calcineurin inhibitors (tacrolimus and cyclosporin) are the mainstay of liver transplantation immunosuppression, but result in EBV amplification, as well as side effects, such as nephrotoxicity and hypertension 24 . Reduction or withdrawal of immunosuppression is associated with decreases of PTLD incidence and PTLD-related mortality. However, insufficient immunosuppression can lead to high rejection rates 25 . Immunosuppressive therapy should routinely be withdrawn during PTLD treatment; however, the timing of restarting immunosuppressive therapy requires careful monitoring of graft function and immune status. In our present case, the graft has maintained a stable condition without any immunosuppressive therapy, as demonstrated by essentially normal serum biochemistry results. This immunotolerance is encouraging since it confirmed the high specificity of the CAR T-cells, and the achievement of immune system balance. The existence of chimeric cells in the boy at one year after liver transplantation suggests the formation of long-term chimerism, which may partially explain the tolerance 26 . However, a biopsy of the graft suggested mild immunoreaction, which could lead to chronic injury. This finding alerted us to the necessity of restarting immunosuppressive medication and performing further biopsies in future follow-up, even though liver function assay results are normal.
Conclusions
Here we reported the first case of chemotherapy-resistant BL-PTLD in which anti-CD19 CAR T-cell treatment achieved continuous complete remission. These results indicate that CAR T-cell therapies could represent a new safe and effective therapeutic option for some cases of PTLD, as long as close monitoring is provided and adverse reactions are quickly treated. Additional clinical trial data are needed to further validate the long-term safety and efficacy of anti-CD19 CAR T-cell therapy for BL-PTLD.
Footnotes
Acknowledgments
We thank the patient and his parents for agreeing the paper published.
Author Contributions
Tianyi Wang and Mingxuan Feng drafted the manuscript. Chengjuan Luo, Xinyu Wan, Ci Pan, Jingyan Tang, Feng Xue, Minzhi Yin, Dongqing Lu participated in the writing of the paper and data analysis. Qiang Xia, Benshang Li, Jing Chen participated in research design and revised the manuscript multiple times. All authors have read and approved the final manuscript.
Ethical Approval
This study was approved by the Ethics Committee of Shanghai Children’s Medical Center, Shanghai, China (SCMCIRB-K2016067 -1).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Ethics Committee of Shanghai Children’s Medical Center, Shanghai, China (SCMCIRB-K2016067 -1) approved protocols. This study was conducted in accordance with the Helsinki Declaration for human studies.
Statement of Informed Consent
Written informed consent was obtained from the patient’s guardians for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shanghai Collaborative Innovation Center for Translational Medicine (TM201928), and the National Natural Science Foundation of China (81670174, 81670136).
