Abstract
Allocation of donated organs for transplantation is a complex process that considers numerous factors such as donor, organ and candidate characteristics and practical issues such as geography. Whole pancreas and isolated islet transplantation are lifesaving for certain individuals with diabetes. Herein, we suggest a revised allocation schema that matches donor characteristics with candidate medical condition while allowing for geographic considerations. It is hoped that adoption of this schema will shorten allocation time, decrease organ waste and optimize the parity between organ donor characteristics and candidate state of health.
Introduction
Type 1 diabetes (T1D) is ‘a’ chronic autoimmune disease resulting in destruction of insulin producing beta cells within the pancreatic Islets of Langerhans. Exogenous insulin provides stabilization of the metabolic imbalance and increases longevity. Yet, for some individuals, insulin therapy is insufficient and is associated with extreme blood glucose variability and life-threatening hypoglycemia. In these instances, islet or whole pancreas transplantation relieves the life-threatening episodes of hypoglycemia and provides a solution to the metabolic derangements of T1D. The technical aspects of islet and whole pancreas transplantation have undergone progressive refinement. Indeed, data suggest that islet transplantation achieves levels of glucose homeostasis on par with whole pancreas transplantation 1 . At present, organ availability is not limiting for whole pancreas or islet transplantation. The existent allocation schema ranks solid organs before islets 2 . However, as the economics of islet transplantation change, competition for organs will increase. To address this, refinements in allocation criteria may be warranted.
Organ Procurement and Transplantation Network (OPTN) pancreas-islet allocation criteria
Factors inherent in donors and candidates predict final outcomes. Ischemic interval length adversely impacts post-transplantation organ function 3 . Appreciation of this resulted in allocation directives dominated by geographic considerations. Yet, time spent in waiting for resolution of higher priority offers to solid organ transplants can result in lower islet yield and poor in vitro islet quality 4 –6 . While important, these considerations are tempered by the reality that allocation generally occurs prior to procurement.
Allocation criteria employ a geographically centered schema to minimize organ ischemic injury. This concept was expressed in the idea of the donation service area (DSA), based on the geographic area served by each organ procurement organization. However, national variation in the size of DSAs resulted in inequity in organ access and loss of potentially transplantable organs. Important concepts and stakeholder groups promoted reconsideration of organs as a national resource not limited by regional peculiarities. In December 2019, the Board of Directors of the United Network of Organ Sharing (UNOS) promulgated revised allocation criteria based on recommendations from the OPTN Pancreas Transplantation Committee (https://optn.transplant.hrsa.gov/governance/public-comment/eliminate-the-use-of-dsa-and-region-in-pancreas-allocation-policy/), which were formally implemented with refinements in March 2021 (https://optn.transplant.hrsa.gov/news/new-kidney-pancreas-allocation-policies-in-effect/). The significant adjustment was elimination of the DSA and regional boundaries. Instead, the updated allocation process prioritizes based on the proximity of a candidate’s hospital in relation to the donor’s hospital. The latter’s position defines a circle of 250 nautical miles (NM). The closer the candidate’s hospital to the donor hospital, the more priority points the candidate would receive. Organs are first offered to all eligible candidates within the 250 NM circle. Organs beyond the fixed circle would be moved to the national pool. This change was applied with different weights to deceased donors 50 years of age or less with a BMI less than or equal to 30 kg/m2 and donors with age and BMI greater than these limits (see new Tables 11-5 and 11-6 of the revised policy). The revised schema re-prioritizes candidates within a given classification but not between classification groups. Unchanged is the weight assigned to combined kidney-pancreas transplantation. However, even in the new schema, islet transplantation continues to be assigned an inferior position in the overall category hierarchy. Specifically, high-quality organs (those from donors less than or equal to 50 years old with BMI less than 30 kg/m2) are first offered to solid organ transplantation candidates. Pancreata that are not accepted are only then offered to islet transplantation candidates.
Improvements in Islet Transplantation Outcomes
Islet transplantation for severe T1D remains an experimental procedure according to the FDA. This has supported health insurance providers in their denial of coverage for the procedure. In the United States, the attendant economics have limited application of islet transplantation to centers that provide complete financial support. While early experience with islet transplantation found limited islet survival and function 7 , current data on glucose homeostasis and prevention of hypoglycemia is comparable, if not superior, to results obtained with state-of-the-art closed loop artificial pancreas systems in individuals with T1D 8,9 . Further, islet transplantation survival data suggests parity with pancreatic transplantation survival 10,11 . Thus, there remains strong justification to continue to employ and expand islet transplantation in select individuals with T1D.
Decreased Pancreatic Transplantation Rates
A decline in whole pancreas transplantation, including alone, or in combination with or after kidney transplantation, has occurred in the United States 12 , although numbers from 2015-2018 indicate a modest increase 13 . These results have been amplified by the complexity and morbidity of the procedure and a loss in training capacity and clinical experience at some transplantation centers. As a result, fewer transplant centers have substantial annual volume or continue to perform the procedure after or without kidney transplantation. Findings of adverse weight gain after combined kidney pancreas transplantation may further temper enthusiasm for whole organ use 14 . Internationally, rates of islet transplantation alone appear stable or are increasing 15,16 . Yet, in North America, islet transplantation rates declined chiefly from continued designation of the therapy as experimental in the United States 17 . This contrasts with new data supporting islet after kidney transplantation 18 . Also, improved management of diabetes in general has made the procedure less acceptable to T1D individuals further decreasing procedure numbers. This trend in pancreatic transplantation emphasizes the need to continue to offer and expand access to islet transplantation for T1D.
Alternative Pancreas and Islet Allocation Criteria
Currently, islet transplantation occurs within the boundaries of clinical trials. Nonetheless, an alternative schema for the allocation of pancreases and islets is presented ( Table 1 ). The schema rates by importance key donor and candidate characteristics and uses a three-point scale assigning 0, 5, or 10 points to a given category. The schema is presented as a compliment to, rather than a replacement of, the current OPTN allocation system. The key candidate factors considered are (i) diabetes disease severity, (ii) risks associated with solid pancreas transplant, (iii) need for associated kidney transplantation, (iv) baseline insulin usage, and (v) calculated panel reactive antibodies (cPRA) status. Diabetes disease severity is reflected by factors such as the presence and severity of hypoglycemia, the degree of variability in blood glucose levels, and time in the target range of blood glucose. Algorithms accounting for these metabolic parameters in estimating disease severity have been developed 19,20 . Concurrent kidney transplantation continues as an allocation priority. Baseline insulin requirement is emphasized as a predictor of the likelihood of the candidate achieving insulin independence, with higher baseline insulin intake presenting an impediment to achieving and/or sustaining insulin independence following islet transplantation. Elevated cPRA, as an indicator of increased allo-rejection potential in islet candidates, is rated to support pancreatic over islet transplantation. The key donor factors scored in the new schema are proximity to the donor hospital, and the age and BMI of the donor, all factors employed in the OPTN scheme. The total points that can be allotted by this schema are 100 for either pancreatic or islet transplants.
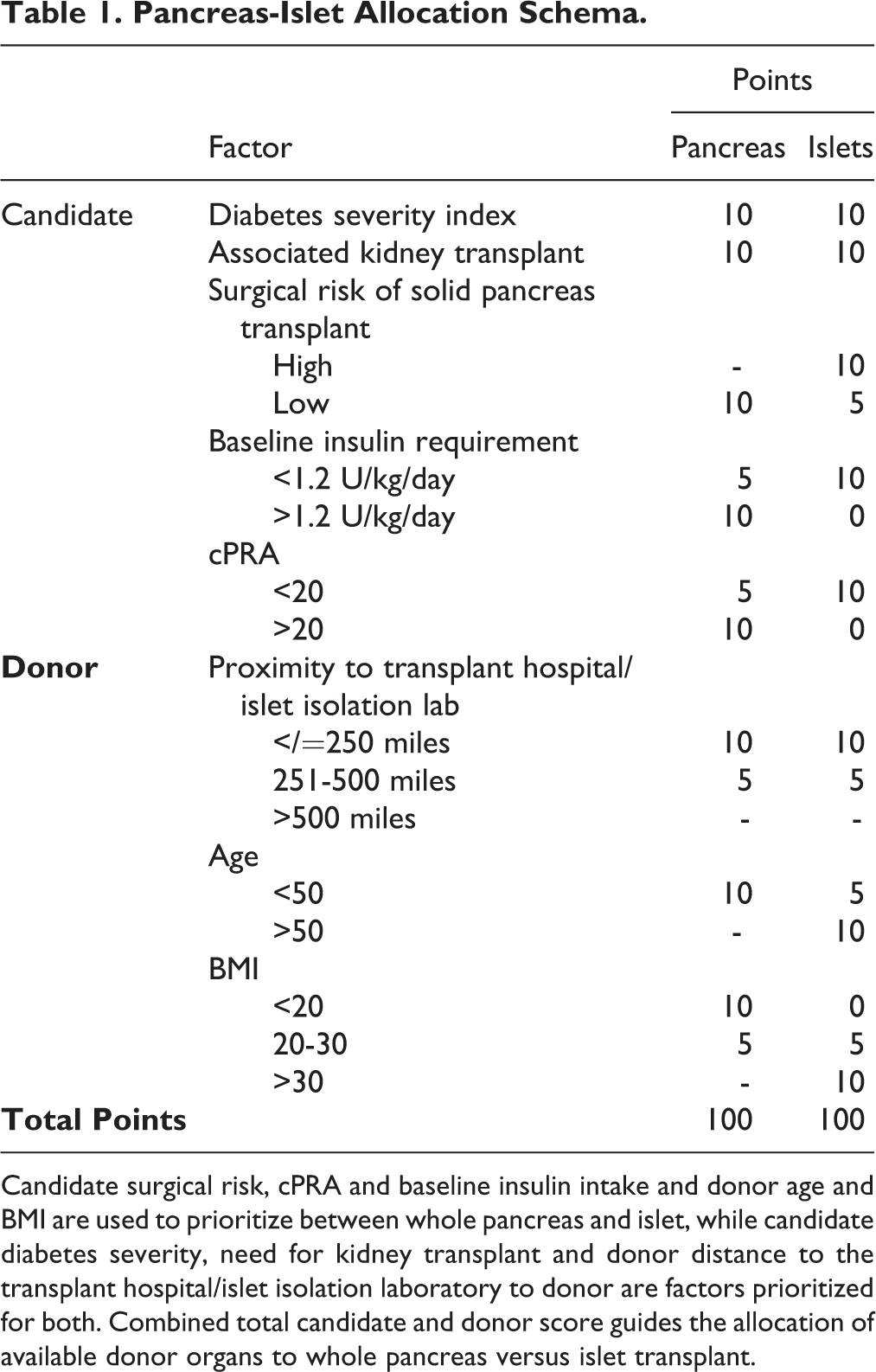
Candidate surgical risk, cPRA and baseline insulin intake and donor age and BMI are used to prioritize between whole pancreas and islet, while candidate diabetes severity, need for kidney transplant and donor distance to the transplant hospital/islet isolation laboratory to donor are factors prioritized for both. Combined total candidate and donor score guides the allocation of available donor organs to whole pancreas versus islet transplant.
The proposed schema is sensitive to existing factors that predict success or failure between both pancreas and islet transplantation and weights these appropriately. For example, elevated cPRA predicts rejection of islets and the schema awards no points to islets if the candidate has cPRA >20%. Low donor BMI is associated with decreased islet yield so again no points are awarded to the islet candidate. The new schema continues to emphasize the importance of proximity weighing this equally for whole organs and islets. However, in contrast to current OPTN/UNOS allocation criteria, in the new schema islet transplantation is not relegated to a lower category ranking but is considered an alternative to pancreas transplantation based on candidate and donor characteristics and proximity of the donor hospital to transplant location.
Conclusions
An allocation schema is presented to compliment the current OPTN/UNOS criteria that does not discriminate unduly between pancreatic and islet transplantation. Application of this schema may provide a means to encourage use of organs that would be otherwise passed on. The schema, although increasing islet transplantation, may also improve outcomes and patient acceptance of this procedure. The hope is that adoption of this model in the United States, and perhaps abroad, will allow for increased optimal and equitable utilization of this resource. It is important to note that the emphasis placed on each characteristic, as indicated by the points assigned, is based upon empiric observations. Additional statistical modeling is warranted to verify this schema.
Footnotes
Authorship Contribution
F.K. and J.S.I. conceived of, designed and wrote the manuscript and prepared the table. M.E.S., G.S., D.C.D., and A.D.R. reviewed the manuscript and provided critical feedback. All authors read and approved the final draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This report does not contain specific human subject information and therefore there was no need for Ethical Committee’s approval.
Statement of Human and Animal Rights
This article did not involve any research with human or animal subjects.
Statement of Informed Consent
This article did not involve human subject research and informed consent was not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wanek Family Project to Cure Type 1 Diabetes and the Arthur Riggs Diabetes & Metabolism Research Institute, City of Hope National Medical Center, Duarte, CA.
