Abstract
Access to functional high-quality pancreatic human islets is critical to advance diabetes research. The Integrated Islet Distribution Program (IIDP), a major source for human islet distribution for over 15 years, conducted a study to evaluate the most advantageous times to ship islets postisolation to maximize islet recovery. For the evaluation, three experienced IIDP Islet Isolation Centers each provided samples from five human islet isolations, shipping 10,000 islet equivalents (IEQ) at four different time periods postislet isolation (no 37°C culture and shipped within 0 to 18 hours; or held in 37°C culture for 18 to 42, 48 to 96, or 144 to 192 hours). A central evaluation center compared samples for islet quantity, quality, and viability for each experimental condition preshipment and postshipment, as well as post 37°C culture 18 to 24 hours after shipment receipt. Additional evaluations included measures of functional potency by static glucose-stimulated insulin release (GSIR), represented as a stimulation index. Comparing the results of the four preshipment holding periods, the greatest IEQ loss postshipment occurred with the shortest preshipment times. Similar patterns emerged when comparing preshipment to postculture losses. In vitro islet function (GSIR) was not adversely impacted by increased tissue culture time. These data indicate that allowing time for islet recovery postisolation, prior to shipping, yields less islet loss during shipment without decreasing islet function.
Introduction
Isolated human islets continue to serve as the gold standard for islet research focused on type 1 and 2 diabetes 1,2 . The high cost of human islet isolation and the requirements for a highly skilled islet isolation team with specialized equipment limit the number of centers that can perform consistent and successful human islet isolations 1 . As such, human islet research has commonly relied on the shipment of islets from expert centers, such as those affiliated with the Integrated Islet Distribution Program (IIDP) 3 –5 . To date, the IIDP has distributed over 261 million human islets to approximately 400 independent researchers across the globe, both in academia and industry. The work from these investigators has resulted in over 700 peer-reviewed publications, documenting the need for this resource 6 .
A number of factors have been shown to significantly impact the quality of islets received after overnight shipment, including encapsulation using alginate 7 , temperature and pressure 8,9 , gas-permeability of the shipping container 10 –12 , and the choice of human serum albumin versus serum in the media 13 . One factor that has not been extensively studied is the optimal length of time that human islets should be held in culture prior to shipping to achieve the best islet recovery upon receipt. Some investigators have requested that IIDP islets be shipped out immediately after isolation; however, culturing the islets prior to shipment has been a standing practice of isolation centers to hopefully descrease loss of islet equivalents (IEQ) during shipment 5,14 .
Tissue culture prior to transplantation has been shown to be comparable to freshly transplanted islets when held between 24 and 72 hours 15 . Those transplant studies, however, were not performed following shipment of islets. All these issues lead to the question: When is the best time to ship islets postculture from isolation centers to recipients, to maintain the most stable IEQ quality and quantity?
The NIDDK-funded IIDP has overseen the distribution of human islets for diabetes research since 2004. The program has emphasized adherence to Standard Operating Procedures (SOPs), including the IIDP shipping protocol that was documented in a publication in 20134. Although prior work summarized qualitative feedback from islet recipients before and after standardization of the shipping procedure in 2007, the IIDP had not attempted to vary or enhance the protocol until conducting a pilot study in 2014 (unpublished), leading to the revision of the IIDP shipping protocol 16 . This revised method required longer pre-shipment culture time (>48 hours in the new SOP vs only 12 hours previously) at 37°C to enhance islet recovery from isolation, use of a different media formulation, and lower temperature during transit to help avoid degradation (6°C from 15°C). Investigator feedback received by the IIDP suggests that there were improvements in islet quality using the new protocol, with an increase in ranking the islets as Excellent/Good from 89% to 92%, statistically significant by chi-square test (P < .0001).
However, it remained unclear whether further increases in preshipment culture time under the new protocol could be beneficial or detrimental to the islets 14 . To address this concern, the IIDP launched a study to measure the effect of preshipment culture time on islet quantity, quality, and function.
Research Design and Methods
Islet preparations were provided by three current IIDP islet isolation centers proficient in providing high-quality islet yields: Scharp/Lacy Research Institute, Aliso Viejo, CA, USA; Southern California Islet Cell Resource Center, City of Hope, Duarte, CA, USA, and the Islet Core Facility at the University of Wisconsin, Madison, WI, USA. The laboratory of Dr. Jonathan R. T. Lakey, Ph.D. at the University of California at Irvine, CA, USA (UCI), performed centralized, post-shipment assays for evaluation of five islet isolations from qualifying donors shipped to them by each of the three isolation centers, for a total of 15 isolations. All pancreata for the isolations were procured from multi-organ donors, following review of social and medical history questionnaire and consent for research use.
To ensure that only acceptable high-quality isolations were included in the study; donor criteria were established for age (21 to 65 years), absence of any history of diabetes, body mass index (BMI ≤ 50), hemoglobin A1c (HbA1c ≤ 6.4), cold ischemia time (CIT ≤ 12 hours, i.e. time from aortic cross-clamp to beginning of islet isolation), and restricted to brain dead donors (No Donation after Circulatory Death). Ethnicity and human leukocyte antigens were recorded but not regulated. In addition, minimum islet isolation parameters were set for number of IEQ, purity, viability, and islet grade postisolation. Donors or isolations outside the specified parameters were excluded from the study.
Each of the 15 isolations that met study criteria was divided into one of four preshipment time periods: (A) held at 6°C (no 37°C culture) until shipped within 18 hours of isolation; (B) cultured at 37°C for 18 to 42 hours; (C) cultured at 37°C for 48 to 96 hours; and (D) cultured at 37°C for 144 to 192 hours, prior to shipment to the evaluation center at UCI. Quadruplicate islet samples were analyzed for quality and quantity under each of three different conditions: (1) immediately prior to shipment at the islet isolation center; (2) postshipment upon receipt at UCI; and (3) postovernight culture after receipt at the UCI laboratory.
Islet Isolation, Culture, and Shipping
Islet isolations were performed at the centers within 12 hours of donor aortic cross-clamp, using automated collagenase digestion with dissociation chamber 17 –19 , and with purification using continuous density gradients and centrifugation using the COBE 2991 Cell Processor 20 . Islets were cultured (for preshipment culture times B-D) at 10,000 IEQ/40 mL in 150 cm2 T-Flasks (Corning Life Sciences, Tewksbury, MA, USA) at 37°C for the prescribed times. Half volume media changes were performed at 18 to 24 hours after isolation, and were repeated every 3 to 4 days until islets were shipped for evaluation. Study islets were recounted in duplicate at the end of the specific culture period and aliquoted to 10,000 IEQ for shipment. Islets were either held and shipped at 6°C within 18 hours or cultured between 18 and 192 hours at 37°C in PIM-Recovery (R) media supplemented with heat-inactivated AB serum (5% v/v, Gemini Bio-Products, West Sacramento, CA, USA), 5 mL of PIM-G (Glutamine/Glutathione, Prodo Laboratories Inc., Aliso Viejo, CA, USA), at a final concentration of 10 µg/mL prepared sterile ciprofloxacin (Corning Life Sciences) followed by cold shipping. PIM-Transport (T) media was used for all islet 6 °C shipments from the islet isolation center to the evaluation center at 10,000 IEQ per 75 mL supplemented as PIM-R media above, with the exception that the AB Serum was added at 2.5% v/v in standard IIDP 60 mL shipping vessels. Islets were packaged using the IIDP standardized Cold Shipping Protocol 21 and Priority Overnight Federal Express shipping, with all shipments traveling through the Memphis, TN, USA Federal Express Distribution Hub prior to delivery at UCI, regardless of the original shipping center, for shipping condition consistency.
Islet Quantity and Quality Assessments
Duplicate samples were removed from preshipment time periods (A to D) at each of the three conditions, preshipment, postshipment, and postovernight culture. Manual counts were performed by two technicians to provide quadruplicate counts and quality rankings for each parameter were evaluated. The islet preparation was stained with dithizone solution (Sigma Chemical, St. Louis, MO, USA) to assist in the evaluations for islet quantity and quality through the standard IIDP protocol for islet equivalence and actual counts, purity, index, and grade 22,23 . Actual islet particles were counted using an optical reticule and guiding grid and then normalized to 150-µm sized IEQ 24,25 . Islet purity was visually determined by comparing the positively dithizone stained islets to the volume of the unstained tissue, reported as percent positive purity. The islet index was determined by dividing the IEQ by the actual count. Islet grade was ranked on a scale of 1 to 10 (with 10 representing the best grade), based on five distinct categories for islet shape, border, integrity, diameter, and amount of single cells, following the IIDP islet ranking as used by the islet isolation centers and their investigators 24 . Aliquots also were removed at each time-point for viability assessment using a double fluorescein diacetate and propidium iodide staining at final concentrations of 0.46 µM and 14.34 µM, respectively, to visualize living and dead islet cells simultaneously 26 . A total of 100 IEQ were inspected and viability per islet was determined visually, and then the average viability for the aliquot was calculated and recorded.
Potency Assessments at UC Irvine Islet Evaluation Center
The evaluation center at UC Irvine performed potency assays of the postculture condition islets, after shipment receipt and overnight (20 to 26 hours) 37°C culture, for the four different preshipment periods (A to D) to assess the function of the experimental islets. UCI conducted static (in triplicate) glucose-stimulated insulin release (GSIR) assays for each shipment category 26 . Islet function was determined by collecting the supernatant and using a commercially available ELISA kit (Human Insulin ELISA kits; Mercodia AB, Uppsala, Sweden) to determine hormone content. The stimulation index (SI) was calculated by determining the ratio of insulin released from islets in high glucose to that released in low glucose. The GSIR assay 27 based on the published Clinical Islet Transplant (CIT) Trial’s SOP 28 was used for the static potency assay.
Statistical Methods
For each of the 15 isolations available to the study, there were 12 different IEQ quantification points, one for each preshipment culture time period (A: no 37°C culture, shipped within 0 to 18 hours, B: 18 to 42 hours of 37°C culture, C: 48 to 96 hours of 37°C culture, and D: 144 to 192 hours of 37°C culture) and for each shipping condition (preshipment, postshipment, and postculture) combination. The raw data combined across the conditions were evaluated for statistical outliers, and those data points greater than 1.5 times the interquartile range (75th percentile to 25th percentile) below the 25th percentile, or similar distance above the 75th percentile, were removed (minimum-maximum number of outliers removed per study variable: 2 to 10, or 1% to 5%).
Following the removal of outliers from the raw data, the change in IEQ was calculated from preshipment to postshipment, preshipment to postculture, and postshipment to postculture. Based on Shapiro-Wilk’s test for goodness of fit, it was determined that the distributions of these changes approximated normality (P values for the three distributions were 0.83, 0.95, and 0.45, respectively). Therefore, parametric repeated measures analysis of variance (ANOVA) was performed to test the study question of whether preshipment culture time influenced change in IEQ, with preshipment culture time period as the repeated measure. (Nonparametric analyses performed with the outliers included yielded similar results.) Further, the analysis was run with the isolation center added as a covariate, and the interaction between culture time and isolation center was tested. For the analyses where the preshipment culture time was found to have a significant effect, Bonferroni-adjusted pairwise comparisons were applied to conservatively adjust for the alpha level. Similar analyses were completed for the difference in islet index, islet score, and islet viability between preshipment and postshipment. The distribution of the difference in islet purity from preshipment to postshipment did not follow a normal distribution, so this quality measure was analyzed using nonparametric repeated analysis. A nonparametric repeated measures analysis also was used to determine if the preshipment culture time had any effect on GSIR. For all analyses, the two-sided alpha level was set at 0.05. Analyses were performed using SAS software v9.4.
Results
Donor and Isolation Data
Table 1 summarizes the donor and isolation parameters from the study. Donor age, BMI, CIT, and total IEQ are noted as mean ± SEM and median with the minimum and maximum (Min
Summary of Donor and Isolation Characteristics for the 15 Isolations Included in this Study.

BMI, body mass index; CIT, cold ischemia time in hours; IEQ, islet equivalents; Min
Table 2 provides the summary data for important parameters evaluated at each of the four preshipment time periods (A-D) for each of the three conditions (preshipment, postshipment, and postculture). Evaluated parameters included actual islet counts, IEQ counts, islet index, islet grade, and percentages of purity and viability, their means ± SEM, medians, and ranges (Minimum
Islet Isolation Results by Shipping Condition and Preculture Times.

Min
Comparison of IEQ Counts
Figure 1
shows the change in IEQ between the preshipment and postshipment counts, at each of the four preshipment culture times. Greatest IEQ loss postshipment occurred with the shorter culture times preshipment: average loss of −2746 IEQ with preshipment culture time of 0 to 18 hours, and −3291 IEQ for 18 to 42 hours preshipment culture time. Longer culture times exhibited minimal IEQ losses: average loss of −1580 IEQ with 48 to 96 hours preshipment culture time, and −662 IEQ with 144 to 192 hours (
Table 3
). Repeated measures ANOVA showed a statistically significant preshipment culture time effect on changes in IEQ from preshipment to postshipment (P = 0.008). Controlling for the isolation center, the preshipment culture time effect remained significant (P = 0.017), and the isolation center effect also was significant (P = 0.02); however, the interaction between isolation center and culture time was not statistically significant (P = 0.28). (Similar results were seen for the comparisons between preshipment and postculture times, as displayed in

Mean, Median, and Range for the Change in Preshipment to Postshipment IEQ Counts, Overall, and by PreShipment Culture Time Periods.

IEQ, islet equivalents; Min
Comparison of the individual culture times, as shown in Table 4 , identified that the average loss was statistically significantly greater for 0 to 18 hours and 18 to 42 hours preshipment culture time compared with 144 to 192 hours (P = 0.001† and P = 0.036δ respectively, adjusting for multiple comparisons, also shown in Figure 1). The average loss did not differ significantly between 48 to 96 hours and 144 to 192 hours of preshipment culture times (adjusted P value of 0.30§).
Pairwise Comparisons of Mean Change in IEQ Count, Preshipment to Postshipment for the Various Preshipment Culture Time Periods of the Study.
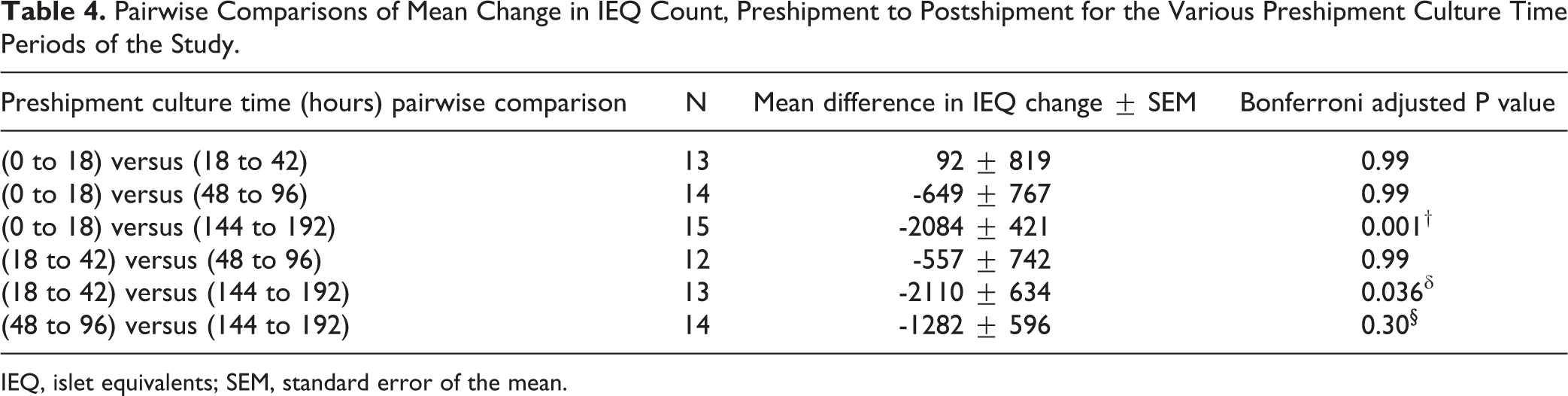
IEQ, islet equivalents; SEM, standard error of the mean.
In a further analysis controlling for isolation center, the preshipment culture time effect remained significant (P = 0.017), and the isolation center effect also was significant (P = 0.02). The interaction between isolation center and culture time was not statistically significant (P = 0.34), indicating that the pattern of culture time differences was similar across centers.
Similar patterns emerged when comparing preshipment to postculture losses, with the 0 to 18 hour and 18 to 42 hour preshipment culture times exhibiting significantly higher losses than the 144 to 192 hour culture time (
Again, average IEQ loss was not significantly different between 48 to 96 hours and 144 to 192 hours preshipment culture time-points (adjusted P = 0.42). When introducing the isolation center into the model, preshipment culture time remained a significant contributor to islet loss (P = 0.005). While the influence of the isolation center was significant (P = 0.049), the interaction between culture time and isolation center was not significant (P = 0.33). When comparing the change in IEQ from postshipment to postculture, the preshipment culture time did not have a significant effect (P = 0.26,
Potency Results
Islet potency by GSIR was not impacted by the increased culture time, with all groups yielding mean SI values ranging between 3.3 and 4.9 with SEMs between ±0.5 and 0.8, acceptable results for human islet potency 29,30 . We conclude that preshipment culture time did not have a statistically significant effect on GSIR (P = 0.07, Table 5).
Glucose Stimulated Insulin Release (GSIR) assay Results from the Different Preshipment Culture Times.

Min
Discussion
There continues to be an overwhelming need for high-quality human islets for a myriad of research studies attempting to understand and treat diabetes. Human islets are a staple for diabetes investigators working on a wide range of experiments, from developing new diagnostic tools 31 , including new chip technology 32 , to prototypes for encapsulation studies 33,34 , alternate transplant sites 35,36 , selection of biomaterials for implants 37 , and basic science for the understanding of islet physiology 38,39 . As the need for human islets increases with the rise in the number of studies requiring them, islet distribution organizations need to be cognizant of variables that influence islet recovery and function. After optimizing the shipping conditions, the IIDP performed this study to determine the influence of preshipment culture time on islet loss and functionality.
The goal of this study was to determine how preshipping culture time would affect the quantity, quality, and functionality of isolated human islets, potentially leading to changes in the current IIDP policy on islet shipping. By using several of the IIDP islet isolation centers for the provision of isolated islets, the normal variability of donors, isolation techniques, and shipping mechanisms would mimic the routine IIDP islet distributions to IIDP recipients. Four preshipment time periods were established: no 37°C culture (A); a short time (B: 18 to 42 hours of 37°C culture) that is allowable in the IIDP Shipping SOP when timing for shipments are affected by delivery delays due to weekends and holidays; the recommended time for the preshipment culture of the islets per IIDP SOP of 48 to 96 hours of 37°C culture (C); and an extended time of 144 to 192 hours of 37°C preshipment islet culture (D), to accommodate normal Federal Express delivery times to academic and commercial islet recipients.
Our study supported the variability of the donor pool that is important in the IIDP strategy and history. As noted in Table 1 and
To ensure isolations that would provide quality islets for this evaluation, a minimum criterion of 200,000 IEQ was needed per isolation to qualify for this study. The mean and median total islet yield easily surpassed this minimum, at 345,173 and 344,434 IEQ, respectively, with a range of 234,700 to 600,816 IEQ. In comparison, IIDP overall data from 548 isolations from which the total islet yield was collected, showed a mean of 297,089 IEQ, a median of 277,193 IEQ, and a much broader range of 35,000 to 1,032,212 IEQ. Although the study was an in-depth investigation of a relatively small sample of donors and islet isolations, the characteristics were similar to the overall pool of IIDP donor summary data for both donor and isolation factors.
To minimize loss from occurring during or after islet shipment to IIDP recipients, this study examined the possibility of maintaining the islets in the culture at the isolation centers, stabilizing the losses preshipment while sustaining islet viability and functionality. In addition, some postshipment islet loss may have been mitigated by media changes performed during islet culture that reduce the concentration of pro-inflammatory cytokines in this study. As in the standardized IIDP culture protocol, media changes were performed at 18 to 24 hours postisolation and every 72 hours after that until islet shipment 21 .
Results here show that increased preshipment culture times are associated with decreased islet loss during and after shipment while maintaining islet quality and function. We suggest that islet survival reaches an equilibrium that occurs after isolation and during subsequent culture due to some of the biological effects previously mentioned. Although losses in islet numbers occur due to tissue culture, we have shown that improvements in islet survival using longer culture periods prior to shipment may offset this loss upon receipt after shipment.
The current IIDP shipping protocol recommends shipment of human islets after 48 hours of 37°C tissue culture. Based on the findings in this study, we have confirmed that at least 2 days of tissue culture (C and D) prior to shipping islets does provide more stable numbers upon receipt than shipping with shorter preshipment culture times. The restrictions of weekend shipping and deliveries, paired with the unpredictable times when human islet donors become available, have caused some islet recipients to reject islet offers of shipment, fearing the effect of a prolonged time of the islets in culture. Our findings should help alleviate the concern of those awaiting islets to allow the isolation centers to maintain the islets until after the weekends or holidays, without major fear of hampering the viability or function of the held islets. We recognize that the results from 15 isolations in this study represent a small sample size. However comparing each preshipment culture time at each of the three conditions for each isolation, a consistent pattern emerged across the islet isolation centers, indicating that extended pre-shipment culture may help ensure the expected quantity and quality of the islets upon receipt by IIDP investigators. These results may prompt further investigation into official changes in our IIDP SOPs and practices.
Conclusion
In summary, we determined that an increased duration of 37°C culture time for human islets prior to overnight shipping may reduce islet loss experienced by investigator recipients. This delay in shipment does not appear to negatively affect the function of islets upon receipt or after subsequent tissue culture. Results from this study may prompt additional investigations which in turn could affect the current protocols and practices of the IIDP.
Supplemental Material
Supplemental Material, sj-jpg-1-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-jpg-1-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-2-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-jpg-2-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-3-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-jpg-3-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-4-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-jpg-4-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-5-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-jpg-5-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-pdf-1-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-pdf-1-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Supplemental Material
Supplemental Material, sj-pdf-2-cll-10.1177_0963689720974582 - Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery
Supplemental Material, sj-pdf-2-cll-10.1177_0963689720974582 for Optimal Time to Ship Human Islets Post Tissue Culture to Maximize Islet Loss and Function Recovery by Barbara J. Olack, Michael Alexander, Carol J. Swanson, Julie Kilburn, Nicole Corrales, Antonio Flores, Jennifer Heng, Jayagowri Arulmoli, Keiko Omori, Peter J. Chlebeck, Laura Zitur, Mayra Salgado, Jonathan R.T. Lakey and Joyce C. Niland in Cell Transplantation
Footnotes
Acknowledgments
The authors would like to acknowledge the three IIDP Human Islet Isolations Centers that participated in this study: Scharp-Lacy Research Institute, David W. Scharp, M.D., Director; Southern California Islet Cell Resource Center, City of Hope, Fouad R. Kandeel, M.D., Ph.D., Director; Human Islet Isolation and Transplant Facility, University of Wisconsin School of Medicine and Public Health, Luis Fernandez, M.D., Director. The authors also would like to thank the IIDP Database Administrator, Jenny Chuang, City of Hope, Duarte, CA, USA, for her assistance in providing data summaries from the comprehensive IIDP database.
Author Contributions
BJO designed and ran the study, organized the participants, wrote the study standard Operating Procedures (SOPs), assisted in the data collection, provided data interpretation, and wrote the manuscript. MA gave input to the study design, assisted in the writing of the SOPs, organized, and performed the research experiments at UCI, assisted in the manuscript writing. CJS helped to design the experiments, assisted in the writing of the SOPs, collected and organized the data, and edited the manuscript. JK performed all statistical analyses for the study and assisted in the statistical writing for the manuscript. NC, AF, and JH performed the research studies and produced the data at UCI. JA, KO, LZ, MS, and PRC assisted with the design of the study, assisted in the writing of the SOPs, and performed the experiments at each of the IIDP isolation facilities. JRTL assisted with the design of the study, provided oversight for the majority of the experiments performed at UCI, provided interpretation of experiments, and edited the manuscript. JCN assisted with the study design, guided the statistical analyses, edited the final manuscript, and funded the study through her IIDP grant. As the guarantor of this work, she had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analyses.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Human pancreatic islets were provided by the NIDDK-funded Integrated Islet Distribution Program (IIDP) at City of Hope, NIH Grant # 2UC4DK098085. In addition, payment for the evaluations done by the Clinical Islet Program at the University of California, Irvine, was provided by the same NIH Grant # 2UC4DK098085.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
