Abstract
We report the cases of 2 patients admitted to our hospital at a 17-year interval, both with 90% total body surface area (TBSA) burns. These two young patients were in good health before their accident, but major differences in time of intensive care and hospitalization were observed: 162 versus 76 days in intensive care unit and 18 versus 9.5 months for hospitalization, respectively. We have analyzed the different parameters side-by-side during their medical care and we have identified that the overall improved outcomes are mainly due to a better adapted fluid reanimation in combination with the evolution of the surgical management to encompass allogenic cellular therapy (Biological Bandages). Indeed, autologous cell therapy using keratinocytes has been used for over 30 years in our hospital with the same technical specifications; however, we have integrated the Biological Bandages and routinely used them for burn patients to replace cadaver skin since the past 15 years. Thus, patient 1 versus patient 2 had, respectively, 83% versus 80% TBSA for autologous cells, and 0% versus 189% for allogenic cells. Notably, it was possible that patient 2 was able to recover ∼6% TBSA with the use of Biological Bandages, by stimulating intermediate burn zones toward a spontaneous healing without requiring further skin grafting (on abdomen and thighs). The body zones where Biological Bandages were not applied, such as the buttocks, progressed to deeper-stage burns. Despite inherent differences to patients at their admission and the complexity of severe burn care, the results of these two case reports suggest that integration of innovative allogenic cell therapies in the surgical care of burn patients could have major implications in the final outcome.
Keywords
Introduction
With over 11 million consultations in hospitals and 260,000 deaths annually, burns remain among the common causes of trauma and death worldwide 1,2 . For severe burn patients having over 40% to 50% total body surface area (TBSA) burns, the situation is life-threatening as immediate skin grafting and wound closure become challenging. With extended and massive burns, the remaining surface of healthy skin available for autologous grafting is insufficient to achieve early closure, while repeated autografting delays skin cover. As a consequence, burn patients are disposed to fluid and electrolyte loss, hemodynamic instability, and sepsis 3 . Indeed, skin is a tissue that provides a physical barrier against ambient pathogens and subsequent to a burn injury this barrier is destroyed leaving the body prone to infection. Hence, the prevalence of infection in burn units is more than 60% and sepsis is the major etiology of mortality for burn patients 4,5 . These critical conditions have advocated for the development of alternative surgical strategies in the past years for an early closure of burn wounds.
In the literature, an interesting approach has been suggested in order to cope with the limited amount of available skin for an early cover of large burns, which consisted of permanent homografting from monozygotic twins 6 . However, for obvious reasons this approach has its limitations for a universal application. In recent years, besides conventional use of temporary skin covers such as cadaver or porcine skin 7 , several dermal substitutes and cell-based approaches have been developed to assist the surgical procedures for an early wound coverage 8,9 . Among these approaches, we can mention the use of cultured epithelial autografts (CEA) and cultured dermal–epithelial autografts (CDEA), both present in the form of cell sheets that can be grafted alone or in combination with regenerative matrices 10,11 . These latter have also been reported to be used in clinics in combination with various allogenic cell sources 12 or in the form of Biological Bandages embedded with human progenitor fibroblasts and used as first covers 13,14 , many approaches that illustrate the effort to advance the care of the severely burned patient in a multidisciplinary insight.
Considering the surgical strategy for patients with deep and more than 40% of burned skin, surgical treatment consists in early debridement to avoid deep infection as long as skin covering with autologous skin grafts (if available) and skin substitutes are accomplished. As soon as the patient has been stabilized in terms of ventilation and fluid reanimation, surgery can begin. It is important to keep in mind that culture of CEA takes 18 days and CDEA 6 weeks in order to cover a portion of the patient at a time. During this waiting period, time goes on with multiple risks for the patients such as fluid and protein loss, loss of electrolytes as well as magnesium, copper, and zinc, and finally a high risk of infection due to the skin barrier destruction exists. Deep infection will increase the instability of the patient and lower his chance of survival. Early debridement, accomplished several times along with repeated showers to clean the wounds are important surgical steps. These events are followed by skin coverage with different temporary substitutes available and all together will be the key of success.
Here, we present the case of two massively burned patients with comparable TBSA (92% and 90% TBSA, respectively), who were admitted to the Burn Center of our hospital at a 17-year interval. Rare are the surviving cases for such massively burned patients (>90% TBSA burns), hence the relevance to investigate for the few surviving cases the elements that could improve their care. Interestingly, we observed major differences between these two patients with massive burns, namely for (i) time of intensive care (162 vs 76 days) and (ii) hospitalization (18 vs 9.5 months). We aimed, therefore, to determine the evolution in the care management, which would explain such a significant difference between these two patients. Burn management in Switzerland belongs to the “highly specialized medicine,” involving multidisciplinary team, among which are intensivists, reconstructive surgeons, and biologists (for the cell therapies). However, the medical care for severe burn patients has not evolved significantly for the past years at our hospital, following the same management pathway (Fig. 1); consequently, we have given special focuses in the present case report, on the evolution of the surgical care between these two patients, including newly implemented cell therapies (i.e., the Biological Bandages).

Flow chart illustrating the medical and surgical pathways for a severely burned patient admitted to the CHUV Burn Center. Burn care management did not change over the past 20 years, except for the introduction of PBBs as temporary covers with or instead of cadaver skin.
Materials and Methods
We collected data on patients by looking at the surgical reports for type and number of surgeries, cell-based techniques and substitutes used, as well as the medical parameters (complications, infection, fluid resuscitation, respiratory system, blood products, and kidneys function).
Results

Evolution of TBSA burn between time of admission and the final TBSA evaluation that required surgery, for patient 1 (A) and patient 2 (B), respectively. Hatched areas for patient 2 correspond to the body zones were considered as second-degree intermediate, and where PBBs were applied in order to help the wounds to evolve favorably toward spontaneous healing. It is worth mentioning that a third-degree burn and second deep degree burns are treated similarly from a surgical point of view (pink zones), i.e., both need debridement and skin grafting as the deep burns cannot heal spontaneously. Please note that for both patients, temporary covers (either cadaveric or PBBs) were applied at day 7, while nowadays we aim to cover after the first debridement around days 4 to 5 (as stated in the surgical pathway in Fig. 1).
The first days after the patient admission were marked by a severe hypovolemic shock requiring a resuscitation with 45 l of fluid within the first 24 h, which led to an abdominal compartment syndrome (ACS) followed by emergency decompressive laparotomy. He was also treated with large escharotomies on the whole body; the escharotomy on the abdominal wall was closed at day 3. Over the first month, the patient developed many episodes of systemic infection and septic shock (n = 11) with secondary acute respiratory distress syndrome (ARDS). Mechanical ventilation was maintained for 4.5 months and an open lung biopsy showed interstitial lung disease with bronchiolitis obliterans organizing pneumonia (BOOP). Deep burns of the hand necessitated secondary reconstruction, but distal phalangeal amputations were inevitable. The equivalent of 198% TBSA was debrided and we used temporary cover with 100% TBSA cadaver skin (glycerol preserved). Several reconstructive surgeries and skin grafts were performed on the body and face (nose, eyelids, ears, and lips). Overall, he had the equivalent of 101% TBSA grafted, including 75% TBSA of CEA, 18% of CDEA, and the rest with autologous skin grafting: total of 30 surgical interventions.
After spending 162 days in the intensive care unit (ICU), he was then transferred to the plastic surgery floor, where he received intensive physio- and ergo-therapy, and was finally discharged after a total of 18-month hospitalization.
For resuscitation, he received 16 l of fluid within the first 24 h. Multiple escharotomies were performed on the trunk and the four limbs; closure of the escharotomies on the trunk occurred on days 17 and 24.
At day 8, the patient developed an acute renal failure KDIGO III with a normal intrabdominal pressure and no finding on the computed tomographic scan. Continuous renal replacement therapy was needed until day 46, followed by intermittent dialysis until day 63. Although there was no inhalation injury on bronchoscopy, the patient remained intubated for 30 days. He had five infection events (three cutaneous and two pneumonia). His wounds were debrided, followed by skin grafting without requiring any amputation. The same surgical debridement technique was used as for patient 1, using a Weck’s blade, a Watson’s blade, and curettage. As often as possible, a tourniquet for the limbs was employed. Wounds were injected with saline adrenalized solution before debridement, to minimize blood loss (as a standard procedure). As for patient 1, timing of debridement started at day 3 post traumatism, with a maximum of 15% to 20% TBSA debridement per surgery, and the mean interval between debridement was 48 h until the burned tissues were fully accomplished. The equivalent of 110% TBSA was debrided, 189% TBSA was covered with Biological Bandages based on progenitor cells and used as temporary cover, and 7% TBSA was covered with glycerol-preserved cadaver skin. Overall, the patient had 85% TBSA grafted (80% TBSA of CEA and the rest with autologous skin graft), with a total of 15 surgical interventions. The final evaluation of deep burns was evaluated at 75% TBSA versus 81% TBSA at admission (Fig. 2B), because several body zones of second intermediate burns (chest, abdomen, back, thighs, and legs) healed by themselves. These zones received the application of Biological Bandages (Fig. 3), which when compared to past experience were thought to stimulate these intermediate-degree burns toward a spontaneous healing of particular body regions (Fig. 4).

Summary over time of the main surgical interventions (and their respective zone) for both patients, including days of debridement, grafting types and zone, the type of temporary cover (cadaveric or Biological Bandages), as well as the use of skin substitute or PRP. The table is organized by rows of 5 days, each day represented by a single square: gray squares are days without any surgical interventions, and split squares correspond to several surgical interventions per day, with each intervention identified by its color code. Acronyms of the grafting types: CDEA: cultured dermal–epithelial autograft; CEA: cultured epithelial autograft; PRP: platelet-rich plasma; STSG: split-thickness skin autograft.
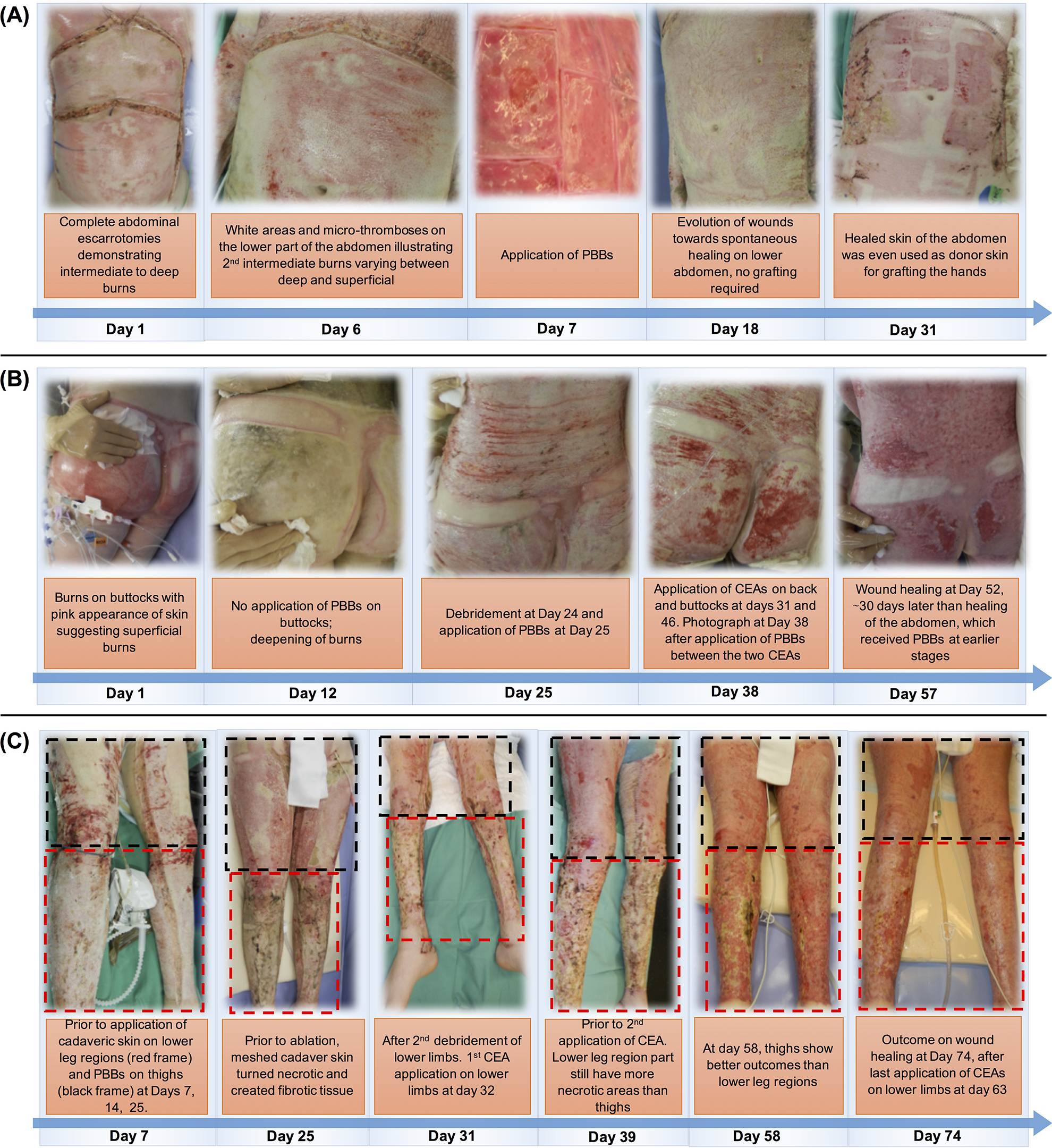
Photographs of the three body zones of patient 2 illustrating the beneficial effect of a treatment with PBBs as temporary covers, allowing a favorable evolution of intermediate degree wounds. (A) The abdomen that had deep burns, also shown by the escarotomy performed at the admission, received Biological Bandages at day 7, which contributed to a spontaneous healing; no grafting was required for the abdomen, but in the contrary this zone was used as a donor site at day 31 for split-thickness skin autografting. (B) The burns on the buttocks, which initially appeared to be less deep than the burns on the abdomen, did not receive Biological Bandages, and the burns on this zone rapidly became deeper and even became necrotic at day 12. The buttocks required surgical grafting, and wound closure happened at day 57, during which there was a delay of almost 30 days when compared to the abdomen. (C) The lower limbs of patient 2 received two types of temporary covers, the lower leg regions had cadaveric skin (red frame), and Biological Bandages were applied on the thighs (black frame). We can see that the cadaveric skin turned necrotic and created fibrotic tissues, while the Biological Bandages did not require stapling and were easy to remove with water leaving a clean wound bed, which in turned allowed a better CEA graft adhesion at day 74.
After spending 76 days in the ICU, he was transferred to the Plastic Surgery floor, then to a rehabilitation center, where he received intensive physio- and ergo-therapy. He was finally discharged after a total of 9.5-month hospitalization.
The data for both patients regarding their burn injury and the received medical treatments are summarized in Table 1. A summary over time of the main surgical interventions and the corresponding body regions is shown for both patients in Fig. 3.
Comparisons for Both Patients of Their Trauma, Medical and Surgical Care and Outcomes.

(*) marks the range between the evaluation of deep burns at the time of admission and the final evaluation that actually required surgery.
ARDS: acute respiratory distress syndrome; BOOP: bronchiolitis obliterans organizing pneumonia; CDEA: cultured dermal–epithelial autograft; CEA: cultured epithelial autografts; FFP: frozen fresh plasma; PC: platelet concentrate; PRP: platelet-rich plasma; RBC: red blood count; TBSA: total body surface area.
Discussion
As mentioned above, major differences in stay durations in ICU (162 vs 76 days) and hospitalization (18 vs 9.5 months) were observed for both cases, which can also be observed for the time to wound closure, 7.5 versus 4 months for patient 1 and 2, respectively. In order to identify the factors that may explain these improvements for the second patient, we focused our observations on three aspects: (i) the dissimilarities in the initial burn trauma between these two patients, (ii) differences in the medical management, and (iii) overall surgical care and cell therapies used.
The Burn Trauma
While the surface of the burn injury was similar for both patients at the time of their admission at the burn center (92% vs 90% TBSA, respectively), the final evaluation of the wound severity that required surgical grafting was higher for patient 1, as the surface of deep injuries requiring grafting were 87% versus 75% TBSA, respectively. This 12% TBSA difference could be explained by the use of allogenic Biological Bandages as First Cover for patient 2. Indeed, it is known that subsequent to a burn injury the severity of the trauma can evolve over time in the following days; the use of Biological Bandages could have contributed to a favorable evolution of at least 6% TBSA of intermediate zones to superficial degree (Figs. 2 and 4), notably by reducing local edema simultaneously with the release of bioactive components 13 , reducing thus the wound surface requiring to be grafted (as further detailed in the section “The Surgical Care”). We would not expect that the remaining 6% TBSA difference in deep burns between both patients would have a significant impact for overall outcome. Indeed, in the case of massive burns (above 70% TBSA), the systemic inflammatory response implies an influx of cytokines and inflammatory reactions systematically, making these two cases comparable overall.
Patient 1 suffered from inhalation injury implying a tracheotomy at day 7, and respiratory complications ARDS (4×), pneumonia (4×), and BOOP. This inhalation syndrome and subsequent complications required longer ventilation assistance for patient 1 (4.5 vs 1 month, respectively), which clearly could have accounted for a further 3.5-month prolonged hospitalization for patient 1. Nevertheless, patient 2, on the other hand, developed other complications such as renal failure, which required dialysis and this was not required for patient 1. Other common complications for burn patients such as the number of cutaneous infections were similar for both patients but patient 1 presented a higher number of general infectious events. Overall, dissimilarities in the initial traumatism and subsequent complications can justify to some extent a hospitalization difference between both patients; however, these complications do not necessarily fully substantiate the 8.5-month difference. Most importantly, the altered time to wound closure and subsequent surgical needs could be easily compared between the two patients, as the last grafting performed on patient 1 was at d ay 111 versus day 63 for patient 2 (Fig. 3).
The Medical Management
While the medical management was conducted following the same pathway for both patients, patient 1 received a more aggressive fluid resuscitation, 45 versus 16 l, corresponding to a 2× and 0.5× Parkland, respectively. An over-resuscitation can cause major complications such as ACS, as observed for patient 1, but also might complicated the intermediate second-degree burns with the induced edema and lymphatic and microvasculature damage 15 . However, the patient abdomen was closed 48 h later with no subsequent abdominal or digestive consequences, which suggests that ACS was not necessarily a major factor accounting for prolonged stay and greater septic morbidity.
Bacomo and co-authors reported a mortality rate of 97% for burned patients with >60% TBSA and ACS, which advocated for a less aggressive resuscitation for patient 2 using the modified Brooke formula: crystalloids 2 ml/kg/%TBSA burn for the first 24 h (half in the administered in first 8 h and half in the remaining 16 h). Thus, the modified Brooke’s formula limits the damage, as a resuscitation volume exceeding 250 ml/kg in the first 24 h is an independent predictor of death 16 . It has been suggested that the most important consideration is to monitor the actual state of resuscitation of the patient with bedside assessment, for a main goal of 0.5-1 cc/kg/h urine output and blood work follow-up (lactate, pH) 17 . Limited fluid resuscitation is currently applied in clinical routine at our burn center with close monitoring of effective volemia.
The Surgical Care
As previously mentioned, the main change in the surgical care between the two patients was the implementation of an allogenic cellular therapy, notably by the use of Biological Bandages (0% vs 189% TBSA) instead of cadaver skin (100% vs 7% TBSA) as a temporary substitute. The Biological Bandages consist of fetal progenitor fibroblasts from a clinical cell bank and seeded on an equine collagen scaffold. These bandages are applied on the cutaneous wound and are changed on average every two days up to four times (usually after patient shower) for local release of growth factors and cytokines favoring the healing 8,14 . Indeed, the immunomodulatory property of the fetal progenitor cells and their potential to induce minimal scars allow a faster healing of the superficial and intermediate zone in a burn wound, in particular by reducing local edema and risk of capillary thrombosis 13,18 . Moreover, the Biological Bandages can prepare the wound bed in deep burn zones for better outcomes of a subsequent grafting; while on the contrary, cadaver skin might induce immune reactions, has the risk to transfer disease, and usually leaves necrotic tissues after application 13 .
In order to illustrate the beneficial effect of the Biological Bandages, allowing a favorable evolution of intermediate-degree wounds, we have focused on three body regions of patient 2, namely the abdomen, the buttocks, and the legs (Fig. 4). The abdomen comprised deep burns, shown by white zones and requiring escarotomy at the admission. At day 7, the abdomen received Biological Bandages, which contributed to a spontaneous healing and no grafting was required; on the contrary, this zone was used as a donor site at day 31 for split-thickness skin autografting (Fig. 4A). Whereas, the burns on the buttocks, which initially appeared to be less deep than the burns on the abdomen, did not receive Biological Bandages, and the burns on this zone rapidly became deeper and even became necrotic at day 12 (Fig. 4B). The buttocks required surgical grafting, and wound closure occurred at day 57, which corresponds to a delay of almost 30 days when compared to the abdomen. Remarkably, the lower limbs received two types of temporary covers, the lower leg regions had cadaveric skin (red frame, Fig. 4C), and Biological Bandages were applied on the thighs (black frame, Fig. 4C). We can see that the cadaveric skin turned necrotic and created fibrotic tissues, while the Biological Bandages did not require stapling and were easy to remove during the shower (hydrotherapy) leaving a clean wound bed, which in turn allowed a better CEA graft adhesion at day 74.
The more favorable and rapid evolution of patient 2, to which Biological Bandages might have contributed, can be observed by a reduced time to wound closure (7.4 vs 4 months, respectively), a reduced number of required surgeries (30 vs 15), a reduced debridement surface (198% vs 110% TBSA), and a reduced number of hydrotherapies under general anesthesia (81 vs 14), while the mean time per surgery was the same for both patients (Table 1).
First clinical trials using the Biological Bandages were conducted in 2000-2005 providing safety studies 14,18,19 , and therefore the specific cell therapy continued as hospital exemption. One of the major changes over the last two decades is the evolution of regulatory in this field, which implied that the Biological Bandages would have to be regulated as an advanced therapy medicinal product (ATMP). Our hospital is now in progress to provide full documentation for clinical trials using ATMP’s with good manufacturing practices (GMP) processing of all cell therapies. Other clinics that want to use the Biological Bandages are able to do so if they fulfill all of the registered programs of transplantation and clinical trial registers, together with GMP cell production facilities. Indeed, clinical studies are ongoing in Taiwan and in Japan to assess and quantify the safety and the efficacy of therapies based on progenitor cells 20 and these are derived from the same GMP-cell bank established for the Biological Bandages reported herein.
While in this study we give special emphasis on the allogenic Biological Bandages and their effects in favoring the healing of second-intermediate-severity wounds, it goes without saying that none of the two patients would have survived without the use of the autologous cell-based therapies, namely CEA/CDEA, as these patients did not have sufficient skin for autografting without a culture step in order to close their wounds. Thus, regarding the use of other cell therapy techniques, both patients were treated with cultured autografts with comparable surface covered, namely 93% versus 90% TBSA CEA/CDEA, corresponding to grafted surfaces of 18,750 versus 18,000 cm2, respectively. It is worth to mention that the 93% TBSA of patient 1 covered by cultured autografts is composed of 75% CEA and 18% CDEA, while patient 2 was not grafted with CDEA (only CEA). This is mainly due to the fact that CDEA requires 6 to 8 weeks to be prepared, and the favorable evolution of patient 2 was rapid enough and there was not sufficient time available to prepare a grafting with CDEA.
Also, patient 2 experienced less infectious events: 11 diagnosed infections (cutaneous, central catheter, pneumonia, urosepsis) versus 5 (cutaneous and pneumonia), respectively. While infections are the most common complications in burn units and an infection source might be multifactorial, less infectious events in patient 2 could be explained partially by more rapid a coverage and wound closure, to which Biological Bandages have contributed together with a more adapted reanimation.
Finally, permissive anemia was allowed for both patients (hemoglobin between 70 and 80 g/l) 21 , but patient 1 received more red blood count (78 vs 59), frozen fresh plasma (18 vs 2), and platelet concentrate (2 vs 1), probably because of the higher number of surgeries. However, patient 2 received a limited amount of platelet rich plasma (0 vs 6 ml, respectively), which could have also contributed to the healing of specific burned zones 8 .
Long-term outcomes are shown in Fig. 5, after 20 years for patient 1 and after 3 years for patient 2. Overall, the skin of patient 2 showed more inflammation than patient 1 as illustrated by the red scars. However, this higher inflammation is totally normal given the shorter time period at the evaluation (3 vs 20 years), and should evolve favorably for patient 2 over time. Also, the skin of patient 1 is more wrinkled than patient 2, because a grafted skin on a burn tends to form wrinkles over time more rapidly than a normal skin as it has lost its original elasticity. Unfortunately, we do not have the photographs for patient 1 at 3-year follow-up (or similar time period), which renders the comparison for long-term outcomes difficult. Nevertheless, we can observe that the abdomen of patient 2 that received the Biological Bandages and healed spontaneously shows no sign of inflammation and scar (Fig. 5A). Likewise, the latero-anterior region of the thighs, which also received Biological Bandages, seems to have less inflammation than the lower region of the legs, which received cadaver skin (Fig. 5B). However, this observation cannot be made for the posterior side of the legs, where there is inflammation everywhere, even in regions that received Biological Bandages (Fig. 5D). The back of patient 2, which also received Biological bandages, seems to be less inflammatory than the rest of the body (Fig. 5C), but this could be explained by the fact that this region is often subject to compression (when the patient sleeps or is laying on his back), which results to a better adhesion of the grafts and less subsequent inflammation. Finally, no adverse effect due to use of Biological Bandages was observed at 3-year follow-up.

Long-term outcome, after 20 years for patient 1 and 3 years for patient 2, respectively. Patient 1 is on the left of each panel and patient 2 on the right. (A) Photographs of the chest and abdomen and anterior side of the arms. (B) Photographs of the anterior side of the legs. (C) Photographs of the back and posterior side of the arms. (D) Photographs of the posterior side of the legs.
Conclusions
Large and deep burns remain a challenge for medical teams requiring a multidisciplinary approach for patient care. For the two extreme cases reported here, we can observe a significant progress between these patients, which occurred at a 20-year interval. Despite some inherent differences to patients at their admission (e.g., inhalation syndrome), we have identified that the overall improved outcomes are mainly due to a better adapted fluid reanimation in combination with the evolution of the surgical management to encompass allogenic cellular therapy, which advocates for a pluridisciplinary management in the burn care of massive burn patients and a strategy must be therefore discussed with all the participants concerned in the burn unit.
The difference in length of stay at the ICU and at the hospital was the motivation to investigate and compare these two cases, and as mentioned above, several factors could have an impact for this difference; however, from a surgical perspective we now have tangible evidence toward improved wound closure when early interventions with allogenic cells used as first cover are scheduled. In particular, we have identified the Biological Bandages as a major change instead of cadaver skin for patient 2, which could have contributed significantly to a more rapid wound healing. Over the past 30 years we have developed significant expertise in cellular therapy at our burn center, which was the first center in Europe to be accredited for burn management. Nevertheless, further developments and constant improvements of cell therapy are necessary to further advance the surgical care of severely burned patients, which in turn considerably improves the quality of life of these patients.
Finally, we would like to point out that both patients have returned to daily life activities including driving, cooking, and running; patient 1 has participated in several marathons and can perform other extreme sport activities such as climbing, hiking, and biking; patient 2 has begun his own company. Therefore, this is a message of hope that advocates for further research and development of surgical techniques including cell therapies to improve the future of severely burned patients.
Footnotes
Acknowledgments
We would like to thank Ms Murielle Michetti for assistance in the creation of the figures. The content of this article was expressly written by the authors listed and no ghostwriters were used.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with animal subjects, and human investigation was in conformity with the Swiss Act on Research involving Human Beings.
Statement of Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Philippe Abdel-Sayed acknowledges funding from the Marie Sklodowska-Curie Action (grant number 833594—PHAS).
