Abstract
The scale of the cosmetic market is increasing every day. There are many safety risks to cosmetics, but they benefit people at the same time. The skin can become red, swollen, itchy, chronically toxic, and senescent due to the misuse of cosmetics, triggering skin injuries, with contact dermatitis being the most common. Therefore, there is an urgent need for a system that can scientifically and rationally detect the composition and perform a toxicological assessment of cosmetic products. Traditional detection methods rely on instrumentation and method selection, which are less sensitive and more complex to perform. Engineered skin tissue has emerged with the advent of tissue engineering technology as an emerging bioengineering technology. The ideal engineered skin tissue is the basis for building good in vitro structures and physiological functions in this field. This review introduces the existing cosmetic testing and toxicological evaluation methods, the current development status, and the types and characteristics of engineered skin tissue. The application of engineered skin tissue in the field of cosmetic composition detection and toxicological evaluation, as well as the different types of tissue engineering scaffold materials and three-dimensional (3D) organoid preparation approaches, is highlighted in this review to provide methods and ideas for constructing the next engineered skin tissue for cosmetic raw material component analysis and toxicological evaluation.
Keywords
Introduction
People are gradually paying more attention to their appearance as their living standards are improving. Currently, cosmetic products are no longer just providing basic finishing and concealing functions such as rouge and lipstick but have positive effects in many aspects, such as moisturizing, whitening, sunscreen, spot removal, and antiaging effects. Cosmetic safety issues, such as irritation, corrosion, and heavy metals, have emerged while using a wide variety of cosmetics. This issue is increasingly of great concern to the majority of consumers. Therefore, cosmetic toxicology testing is required to ensure safe use and a favorable experience for consumers before cosmetics enter the market. This testing can rule out the presence of harmful substances in cosmetics that can cause harm to the human body (such as corrosion and irritation and other nonreactions), especially in conditions of disease and harm 1 .
Traditional cosmetic composition testing mostly relies on instrumental analysis methods such as UV spectrophotometry, high-performance liquid chromatography (HPLC), capillary electrophoresis, animal experiments as in vivo efficacy evaluation methods, and two-dimensional (2D) cell models as in vitro testing methods2–6. Although the test techniques are easy to operate and inexpensive, they also have many drawbacks, such as the high consumption of organic reagents in the pretreatment process, low detection throughput, and a long analysis cycle7–10. In fact, traditional animal models and in vitro 2D cellular models are unable to accurately describe the toxic effects of cosmetic ingredients, but they are used to predict the actual response in vivo. Because of the obvious interspecies differences between humans and animals, a physiologically relevant tissue microenvironment recapitulating human skin is lacking 11 . For example, 2D skin multitype cells are cultured in the laboratory and they tend to adhere to flat surfaces. This traditional culture method depletes growth media and metabolic wastes, which may result in cell death, nutrient depletion, and damage to the environment in which they are grown, thus preventing complete cell function12–16. It also does not adequately consider the influence of the three-dimensional (3D) microenvironment under natural physiological conditions on its internal response and affects dose effectiveness and dose toxicity prediction data 17 . The inability to provide living tissue structure, mechanical/biochemical signals, and cell behavior has informative limitations18,19.
The study of animal-friendly and high-throughput rapid detection techniques is gradually becoming a research direction of interest for researchers, with the development of animal replacement tests and the gradual deepening of the concept of green chemistry. The cosmetic industry, guided by ethical considerations and regulatory requirements, has developed and evaluated alternative methods suitable for regulatory implementation, such as in vitro assays and electronic information methods for testing the toxicological endpoints and efficacy of cosmetic products and components20–22. Artificial skin is constructed by cells and scaffolding materials, which have certain physiological functions and can better restore the ecological environment of skin tissue, and the test results more accurately reflect the real quality of cosmetics. The application of an engineered skin tissue technology can provide better statistical test results, which overcomes the problems of immune rejection and difficult access to skin of autologous or allogeneic origin 23 . In vitro reconstructed 3D models are used as an alternative means of tissue engineering with material scaffolds that provide an extracellular matrix (ECM) that enables cells to proliferate, differentiate, mechanically respond, and communicate among other functions24–27. The human skin is highly simulated in terms of gene expression, tissue structure, and metabolic activities28–30.
The emergence of artificial skin is significant, based on the current status of the application of engineered skin tissues in the field of cosmetic ingredient testing. With the increasing cosmetic market, there is an urgent need for a system that can scientifically and rationally detect cosmetic components and toxicology. Engineered skin can simulate natural skin in vitro and has high-throughput detection capability. So it is an ideal model for cosmetic component and toxicology detection. This review focuses on the characteristics, testing capabilities, and advantages of engineered skin tissue. Different types of scaffolds and preparation methods of engineered skin tissue were introduced. This research aims to guide researchers in the selection of appropriate skin models based on different kinds of cosmetic tests and in the toxicological evaluation of the latest products.
Comparison of the Existing Cosmetic Composition Detection Methods
There are many kinds of cosmetics and complex ingredients. Particularly for makeup (blush, mascara, and lip gloss), their complex components can have an effect that enhances or suppresses the detected signal. This will seriously affect the accuracy and precision of quantitative analyses. The existing detection technologies are spectrophotometry, HPLC, Raman spectroscopy, and so on.
Cosmetic Component Detection
To a large extent, the selection of analytical techniques depends on the nature of the sample, the analytical instruments available in the laboratory, the number of samples available, and the sensitivity of the method needed. The availability of various analytical techniques and instruments, as well as the abundance of sample preparation techniques and sample types, makes the selection of the correct analytical method crucial for method development and validation. The correct and efficient analysis of certain components of cosmetic products depends on the choice of instruments and methods (Fig. 1 and Table 1).
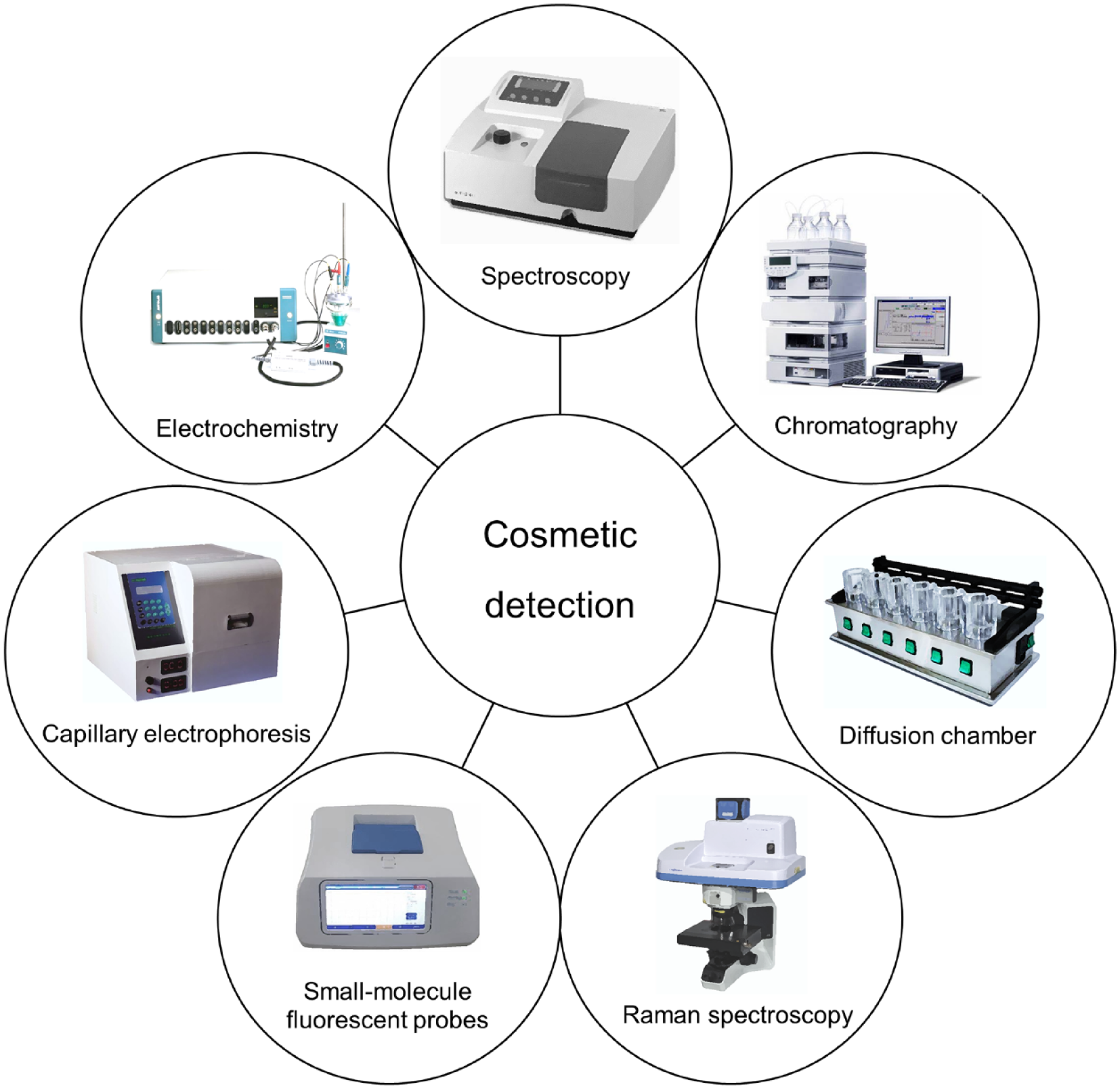
Traditional cosmetic components detection. With the continuous development of science and technology, the existing cosmetic ingredient testing techniques are becoming more and more sensitive and efficient. Spectroscopy, chromatography, diffusion chamber, Raman spectroscopy, small-molecule fluorescent probes, capillary electrophoresis, electrochemistry, and other methods are very commonly used.
Comparison of the Advantages of Existing Cosmetic Composition Detection Methods.

The 3D skin model based on normal keratin-forming cells can provide an effective way to study and claim the barrier-promoting effects of cosmetic ingredients and formulations. The results of genetic, protein, metabolic, and permeation barriers need to be integrated to comprehensively evaluate the barrier-enhancing efficacy of cosmetic products using keratin-encapsulated structural protein molecules of the skin barrier as key indicators45,46. When cosmetics are exposed to 3D skin models, the barrier-enhancing potential of the cosmetics is determined by the upregulation of the expression of bioindicators that contribute to the enhancement of skin barrier function. For example, in a cosmetic barrier-enhancing efficacy evaluation experiment, the protein levels of EpiKutis, a 3D epidermal model under different treatment conditions, were analyzed using immunofluorescence techniques. Compared with the control group, both filaggrin (FLG) and loricrin (LOR) were significantly increased after treatment, which demonstrated that the samples could promote the expression of FLG and LOR simultaneously and enhance skin barrier function.
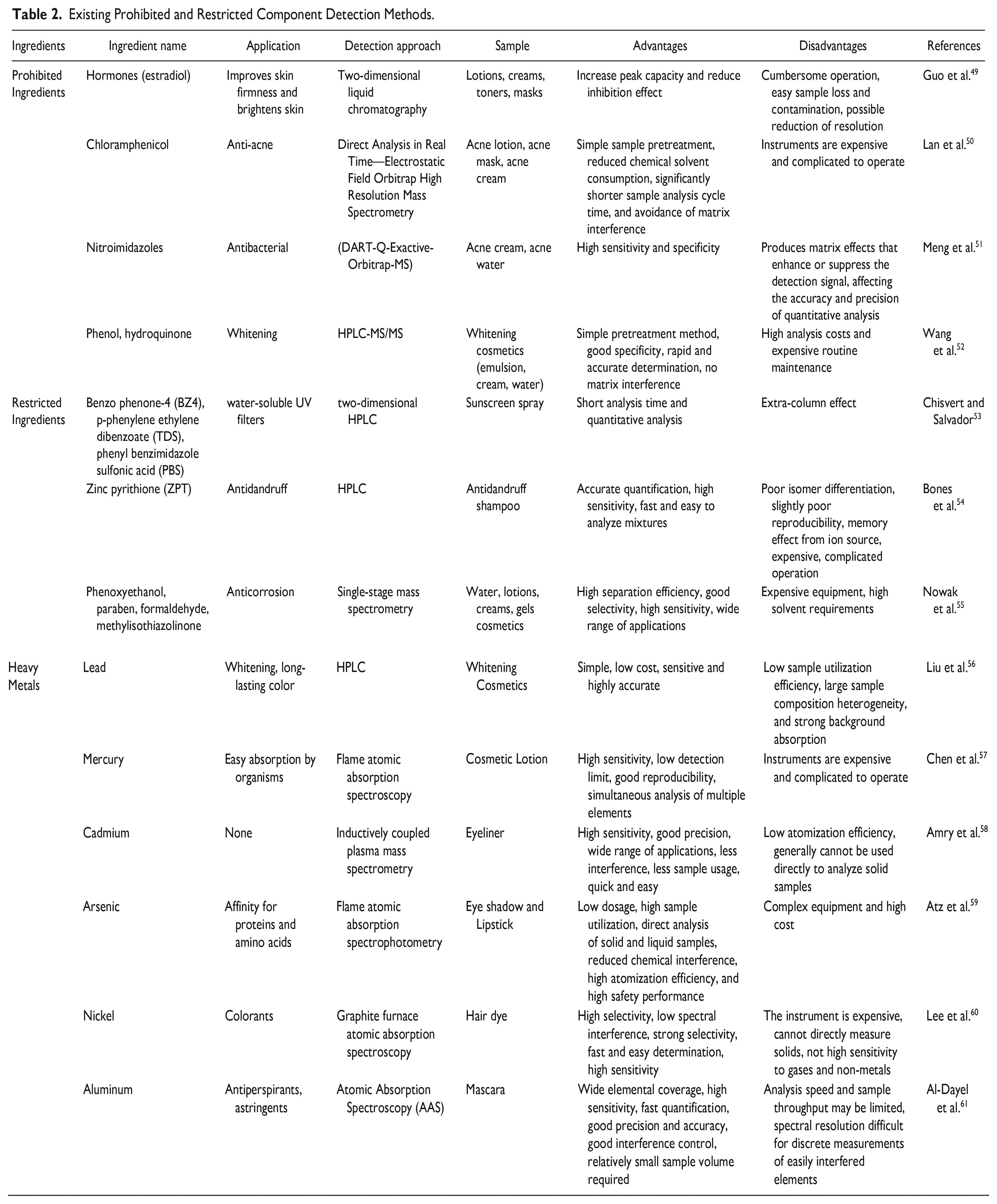
For some cosmetic producers in pursuit of fast-acting product efficacy, the production process illegally adds an excess of prohibited ingredients, causing a threat to consumer health and reducing the trustworthiness of the product. The EU and China explicitly included sex hormones in the category of banned substances in cosmetics in 2000 and 2015, respectively 47 . Heavy metals, such as lead, mercury, cadmium, arsenic, and nickel, as well as aluminum, classified as light metal, have been detected in makeup, face and body care products, hair care cosmetics, herbal cosmetics, and other types of cosmetics. In addition, essential elements such as copper, iron, chromium, and cobalt are also present in cosmetics but are harmful only when they occur in excess 48 . The available methods for the detection of prohibited and restricted ingredients are listed in Table 2.
Existing Prohibited and Restricted Component Detection Methods.
Skin Toxicology Evaluation
In general, toxicological tests for safety evaluation should cover all newly developed cosmetic products. In vivo animal experiments for skin irritation/corrosivity tests, acute eye irritation/corrosivity tests, skin allergy tests, and skin phototoxicity tests should be determined according to the actual situation when selecting test items 62 . The toxicological safety evaluation methods of cosmetics mainly use guinea pigs, rabbits, and other test animals. The experimental methods can be viewed as cruel, such as corrosion and eye drops, which can cause great harm and suffering of the animals. Moreover, the inability of species and individual differences to respond to scientific human outcomes has caused animal experiments to fail to meet assessment needs 63 . In the “3R principle” of substitution, reduction, and optimization proposed by scientists, substitution is the first principle to be considered when conducting animal experiments 64 .
Engineered skin tissue has achieved promising results in recent years, and products have been released one after another, which has led to the application of engineered skin tissue not only in treatment research but also in other fields, such as cosmetic safety testing, and has laid the foundation for skin testing models instead of animals for skin testing. Engineered skin tissue has become an alternative to several skin toxicology methods for cosmetic safety evaluation and provides a model for conducting toxicology studies on cosmetic ingredients or finished products and for developing technical guidelines for alternative methods of safety evaluation that are closer to the human environment (see the “Skin Toxicology Evaluation” section for details).
Emerging Technology: Engineered Skin Tissue
Tissue engineering technology features the study of cell–cell and cell–extracellular microenvironments and the interaction pattern between materials, with the core of material molecular biology and the goal of exerting seed cell function. Engineered skin tissue precisely mimics the microenvironment of specific cells in vivo and the intertissue microstructure and is closer to the structure and function of human skin in terms of cell phenotype, migration, proliferation capacity, surface receptor expression, ECM synthesis, cell signaling pathway conduction, cell metabolism, and other characteristics.
Preparation Process
The main process to produce engineered skin tissue is divided into three stages 65 : (a) seed cell induction, (b) scaffold material preparation, and (c) reconstruction of epidermal or subdermal structures (Fig. 2). Seed cells play a crucial role in this process. These cells determine the final morphology and function of the entire engineered skin tissue, and each step has a decisive role in the quality of the product. The search for a scaffold material that can maintain the properties of the dermis while having good biological properties is also one of the focal issues of scholars at home and abroad. Therefore, how to construct a new biological skin that can retain the normal phenotype and physiological functions of the human body has become the direction of efforts in recent years, and the use of natural active material through in vitro culture or in vivo implantation to achieve this goal has attracted widespread attention. A variety of new technologies are emerging, providing more options for clinical applications, especially 3D printing, which is the most prominent, in recent years66–68.
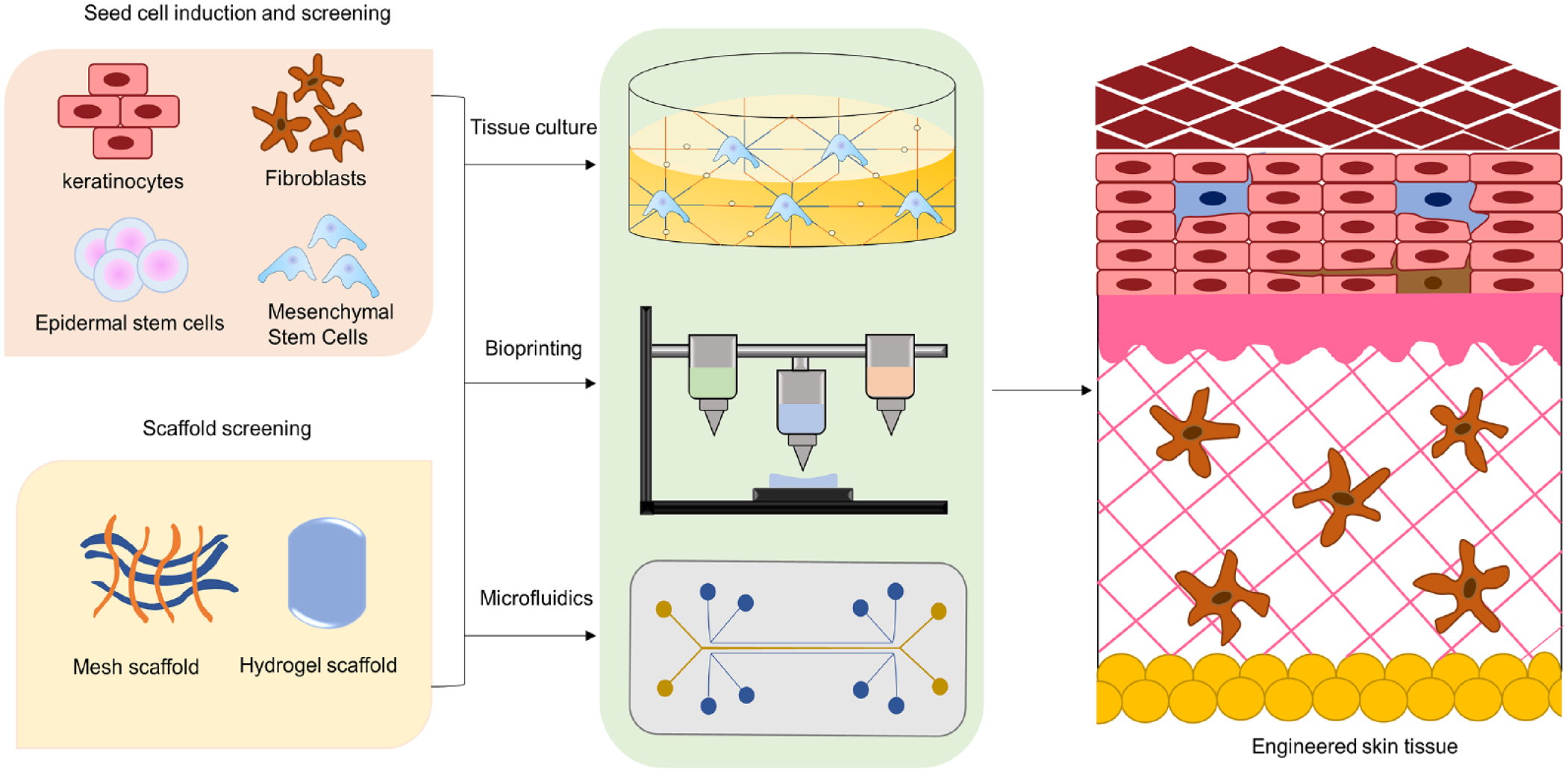
Process of engineered skin tissue preparation. Engineered skin tissue preparation requires three processes: seed cell induction, scaffold material preparation and epidermal or subcutaneous structure reconstruction. In this process, the seed cells determine the final form and function of the whole tissue engineered skin. Meanwhile, a scaffold material that can maintain dermal properties and has good biological properties is also one of the focuses of current research. In addition to tissue culture, new technologies such as bioprinting and microfluidics also provide more options for clinical applications.
Various Artificial Skin Products
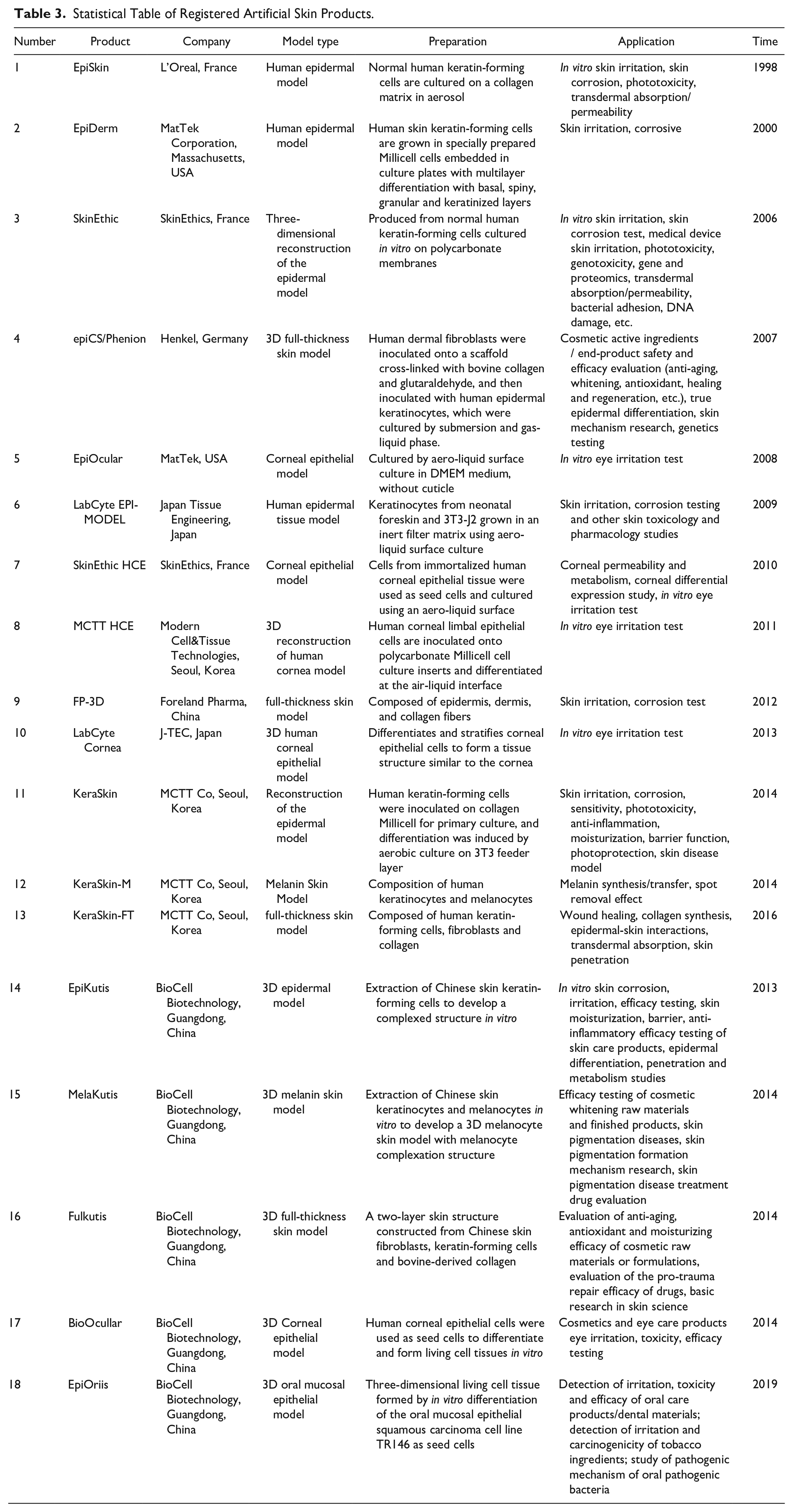
Engineered skin tissue is a relatively mature research field at present. Commonly used engineered skin tissue can be divided into three categories: (a) epidermal cells without dermal components, (b) dermal component only, and (c) true epidermal bilayer structures. Commercial products, such as Epical, CellSpray, AlloDerm, OASIS Wound Matrix, PermaDerm, StrataGraft, and Anbody Skin, are approved by the Food and Drug Administration (FDA) and are being marketed69–73. The existing registered artificial skin products are listed in Table 3. However, these products have many drawbacks. The abovementioned artificial skins are mainly based on keratin-forming cells and fibroblasts, with the common significant defect being that they lack hair follicles, nerves, sebaceous glands, sweat glands, blood vessels, and so on. Subcutaneous structures have an important role in lesion healing and transdermal absorption. With the development of science and technology, it has gradually been realized that using stem cells for self-renewal is an effective means to solve this problem. With the introduction of tissue engineering into regenerative medicine, the study of cell growth factors and stem cells has provided new ideas for engineered skin tissue construction 74 .
Statistical Table of Registered Artificial Skin Products.
Special Properties of Engineered Skin Tissue
Engineered skin tissue construction involves a combination of seed cell screening, scaffold materials, biocompatibility considerations, and other elements of the process. Optimization of the tissue culture environment for seed cells, appropriate scaffold materials to provide proper mechanical support, and good biocompatibility are the key points and research directions that should be explored in the future for engineered skin tissue cosmetic composition testing and toxicological evaluation of trauma damage and accelerated repair. In addition, the development of trauma dressing materials is also a new field.
Physical and chemical properties
Excellent seed cell feedstock and biocompatible scaffold materials are the basis for the successful construction of engineered skin tissue. They are easy to handle, have good stretch and toughness, can flexibly cover the surface of irregular wounds, have low storage requirements, and are resistant to stress 75 . These engineered skin tissues adhere rapidly to the wound surface, have the desired physical and mechanical properties, and undergo controlled degradation with little batch-to-batch variability 76 . The physicochemical properties of scaffolds depend on multiple factors, such as the chemical composition, intermolecular interactions, polymerization method, relative molecular mass, pH, and porosity 77 . Scaffolds with optimal pore size have been shown to exhibit good cell physiological properties in terms of cell spreading and migration, and sufficient porosity is used to transport nutrients and waste products 78 . Natural ECMs are typically highly porous structures where water and soluble factors fill and diffuse through the voids, and the porous structure provides a large surface area for cell attachment and growth, allowing efficient molecular translocation and the formation of local bioreactors for biochemical reactions 79 . When the diffusion coefficient is increased, the morphology, adhesion, and activity of cells grown in hydrogel scaffolds with a high-density network of small pores are improved 80 . Many studies have shown that hydrogels modified by cross-linking chemical techniques have rapid degradation and high mechanical stability 81 . In addition to cross-linking techniques that support tissue growth, pH plays an important role in skin tissue architecture, maintaining a stable physiologically relevant environment for cellular components. Sharpe et al. 82 demonstrated that the optimal pH to support the proliferation of epidermal keratin-forming cells and dermal fibroblasts is 7.2–8.3. Therefore, it is also important to assess and monitor the pH of the artificial skin during the design of the scaffold.
Morphological structure and mechanical properties
The overall ultrastructural appearance of the epidermis in the skin model is highly similar to that of natural tissues and contains epidermal layers, including the basal, spiny, granular, and stratum corneum 83 . The biocompatibility of the scaffold material with the cells depends mainly on its mechanical properties, and mechanical stability is necessary to support cell attachment and tissue growth in engineered skin tissue. Cells in vivo are spatially characterized by certain morphological structures, and the regulation of cell growth and differentiation functions is also carried out spatially by signaling pathways 84 .
Biocompatibility
Scaffold materials are biocompatible with cells and support living cells in performing their biological functions without affecting the physiological and vital activities of the host cells in an engineered skin tissue model 85 . In addition to biocompatibility and functionality, there are additional properties that promote cell attachment and proliferation 86 . The artificial skin structure contains all cell populations of the epidermis and/or dermis for a physiological environment that more closely resembles natural skin tissue. These cells interact with the surrounding ECM and regulate intercellular and intracellular communication through signaling mechanisms that include growth factors, enzymes, and related bioactive substances 87 .
Advantages and Disadvantages of Engineered Skin Tissue in Cosmetic Component Detection
Induced Pluripotent Stem Cells Used as Seed Cells
Pluripotent stem cells have a strong expansion capacity, making them the most used seed cells in regenerative medicine and tissue engineering. In addition, it is involved in life processes such as immunomodulation, inhibition of inflammation, tissue repair and secretion of cell growth factors 88 . Induced pluripotent stem cells (iPSCs) are one of the seed cells that have been used to create artificial skin. iPSC technology has been utilized, and it has great potential for application in disease research, individualized therapy, regenerative medicine, and model preparation. Among them, iPSCs have created an increasing number of skin transformation models, advancing the process of skin tissue engineering research and model functional improvement.
During the COVID-19 pandemic, multiple human induced pluripotent stem cell (hiPSC)-derived organoids have been used to mimic multiorgan severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection as well as for the study and treatment of various endogenous and exogenous injuries or infectious diseases in humans. Ma et al. 89 established a hiPSC-derived skin organoid with hair follicles and a nervous system that is sensitive to external damage. This skin organoid was used to study the effects of SARS-CoV-2 infection on cell types and the pathological features associated with skin infection. The generation of hairy skin organoids from two male hiPSC lines was recently reported 90 . This technique opens up exciting new possibilities for developmental research in multiple skin diseases (e.g., epidermolysis bullosa), modeling provision and regenerative medicine.
Skin organoids have also been combined with 3D bioprinting technology to provide more promising tools for future research. Skylar-Scott et al. compacted iPSC-derived organoids via centrifugation to form a living tissue matrix, and a sacrificial gelatine ink was printed within the matrix that serves as a perfusable channel for custom-shaped vascularization in the pattern of a single or branching conduit after the removal of the inks. A living matrix composed of IPSC-derived cardiomyocytes and primary cardiac fibroblasts creates a powerful, perfusion-ready heart tissue that can fuse and beat synchronously within 7 days using this approach 91 . Researchers are also actively exploring the possibilities and optimal methods for combining these two techniques.
Remodeling the Skin Microenvironment
Engineered skin tissue has an extremely fine composition and spatial arrangement of cells and ECM within the skin to maximize the reproduction of the real skin microenvironment in which the cells perform precise functions in their own life cycle. The skin microenvironment is composed of keratin-forming cells, melanocytes, and fibroblasts, which undergo cellular interactions and contact external environmental stressors 92 . The skin microenvironment is altered in the presence of inflammation, aging, and traumatic injury. Senescent cells exhibit an altered secretome, and senescence-associated secretory phenotype (SASP) secretion of proinflammatory factors greatly alters the skin microenvironment 93 . Subsequently, the transition to cellular senescence triggers inflammation and subsequent expression of a clinical skin senescence phenotype 94 . In the early stages of skin injury, the organism undergoes an immune response to clear the pathogen, and immune cells come into play, which drives the healing process into the inflammatory, proliferative, and tissue remodeling phases of wound healing 95 . During remodeling, resting fibroblasts at the injury site are activated and differentiate into myofibroblasts with contractile properties and α-smooth muscle actin (α-SMA) expression. At this point, myofibroblasts synthesize and secrete excess ECM, which becomes a major component of the sarcomere and is deposited on the wound surface as a scaffold for other repair cells 96 . Each phase has a specific “time window” in which multiple cells, biosignalling molecules, and ECM act in fine-tuned synergy to determine the rate and quality of wound healing and remodeling back to a healthy skin microenvironment 97 .
Traditional skin disease research is mainly based on 2D and 3D skin cell models, which lack the wound microenvironment and heterogeneity and do not truly reflect the disease development mechanism and drug response 98 . Artificial skin fabricated by tissue engineering can simulate the near-real human ecological environment and create a tissue regeneration microenvironment. Studies have shown that downregulation of Nkx2.5 expression in fibroblasts derived from lung tissue could inhibit myofibroblast formation 99 . Researchers found in a rabbit ear model that the enhanced nuclear translocation of NFE2L2 was accompanied by the inhibition of Smad2/3 phosphorylation, further preventing myofibroblast formation 100 . Therefore, myofibroblast formation is closely associated with activators or inhibitors in the wound microenvironment. An optimal model for high-throughput, large-scale, rapid, and low-cost drug screening is needed for the development and screening of drugs for the treatment of skin trauma regeneration, skin tumors, immune and metabolic diseases, and inflammatory diseases before clinical trials can be conducted and disease treatment performed 101 . Engineered skin tissue models are the optimal choice for efficient composition testing and toxicological analysis of cosmetics and for establishing precise drug screening systems for the targeted treatment of individual symptoms. Engineered skin tissue has the future potential to be widely applicable in the field of skin disease research and treatment at the laboratory stage and before clinical research can be conducted.
Reproducing the Molecular Mechanisms
Engineered skin tissue represents the physiological condition and health of the organism to the greatest extent possible, simulating the positive effects and adverse reactions that occur in the skin after the use of cosmetics. These different skin states are progressive processes that are the result of a combination of multiple factors. New research has revealed that altered signaling pathways play a key role in the detection of cosmetic ingredients and toxicological evaluation processes using artificial skin. Signaling pathways and regulatory factors that modulate melanin biosynthesis are crucial in melanin models used for the efficacy evaluation of whitening cosmetics 102 .
These pathways included the α-melanocyte-stimulating hormone (α-MSH)-induced signaling pathway and the PI3K/Akt, MAPK, and Wnt/β-catenin signaling pathways. Among these pathways, p38 MAPK is one of the most important pathways of MAPK signal transduction. The p38 pathway is a key regulator of proinflammatory cytokine biosynthesis at the transcriptional and translational levels, making different components of this pathway potential targets for the treatment of autoimmune and inflammatory diseases. The upstream and downstream molecules involved in the regulation of p38 are abundant and can be phosphorylated after being stimulated by hypoxia, ultraviolet radiation, and other factors. After being activated by MKK3/4/6 phosphorylation, p38 is transferred to the nucleus and phosphorylates ATF-2, Elk-1, p53, and other transcription factors to activate transcription. It plays a key role in the regulation of the inflammatory response and wound healing 103 .
The Wnt/β-catenin signaling pathway is critical for skin tissue regeneration and is regulated through the Wnt/β-catenin signaling pathway. The Wnt/β-catenin signaling pathway is activated to cause overexpression of β-catenin in hair follicle stem cells when the skin is damaged. β-Catenin enters the nucleus and forms a transcriptional complex with LEF1 to activate downstream target genes such as C-myc, which promotes the proliferation of hair follicle stem cells and rapid recruitment to the wound surface for repair 104 . The Wnt/β-catenin signaling pathway is also an important signal for aging-related diseases and is closely related to the onset, progression, and regression of many diseases 105 . The Wnt/β-catenin signaling pathway acts on targets that are involved in cell survival, proliferation, and differentiation, as well as maintaining tissue homeostasis 106 .
The main signaling pathways that regulate skin photoaging include the transforming growth factor (TGF)β1/Smad signaling pathway, nuclear factor kappa-B (NF-κB) signaling pathway, Nrf2/ARE signaling pathway, and MAPK signaling pathway. TGFβ1 activates TGFβRII by phosphorylation by binding to membrane TGFβ receptor II (TGFβRII), which activates TGFβRI. Phosphorylation of activated TGFβRI activates Smad2 and Smad3, which bind to Smad4 to form complexes that are translocated into the nucleus, thereby regulating target gene transcription. Many studies have shown that TGFβ1 can promote fibroblast proliferation, stimulate fibroblasts to synthesize collagen and other ECM components, and inhibit the activity of proteases and stromal enzymes, thus causing ECM deposition, and it is the strongest known fibrotic cytokine 107 . UV activates the IκB kinase complex (IKK) through the activation of growth factors, antigen receptors, MAPKs, and other pathways to degrade IκB and promote the release of NF-κB into the nucleus to perform its role 108 . NF-κB can promote the expression of the matrix metalloproteinase (MMP)-1, MMP-3, and MMP-9 genes. In addition, NF-κB can interact with the MAPK, Jun, and p38 signaling pathways to promote the activity of MMPs, degrade collagen and other matrix components in the dermis, and lead to skin photoaging 109 .
Simulation of Natural Skin
Maximum simulation of human skin is the most significant advantage of engineered skin tissue. In vitro models have been an important research tool in the field of dermatology for nearly two decades. The original in vitro skin was a 2D epidermal model system consisting of differentiated epidermal cells. The 3D epidermal model EpiKutis emerged with the development of tissue engineering technology. It uses keratinocytes isolated from skin tissues as seed cells to develop a 3D epidermal model with a complex layered structure in vitro, which has a highly similar complex layered structure and physiological and metabolic functions to natural skin and is used in in vitro assays for corrosion, irritation, and barrier repair. FulKutis, a 3D full-layer skin model with a slightly more complex structure, has subsequently emerged. FulKutis is a two-layer skin model containing epidermal and dermal structures constructed by in vitro recombination technology using fibroblasts and keratin-forming cells as seed cells and bovine-derived collagen as scaffolding material, which accurately reflects the antiaging and antioxidant effects of cosmetics after they have been applied to the body and enables the detection of multidimensional data (genetic level, protein level, cellular level, etc.). The 3D melanin model MelaKutis uses a finely regulated serum-free medium to develop keratinocytes and melanocytes in vitro into a 3D melanin skin model with a melanocyte lamellar structure, which is highly similar to natural skin in terms of melanin particle distribution, epidermal lamellar structure, and physiological and metabolic functions and can be widely used in the field of in vitro whitening efficacy testing.
To date, the evaluation of cosmetic skin toxicology is still mainly based on animal experiments. Especially in China, animals for experiments are relatively inexpensive and plentiful. The increased awareness of animal protection and the development of the 3R principle have given rise to numerous ethical issues regarding experimental animals, in addition to the fact that experimental animals may experience pain and discomfort when performing irritable tests, such as skin irritation, resulting in predicted results that do not always match human responses. Domestic and international alternatives to animals for skin toxicology evaluation include some engineered skin tissues, but they are difficult to produce on a large scale.
Limitations and Challenges of Skin Models
The intact skin consists of the epidermis, dermis, and subcutaneous tissue, and the three layers of tissue have important roles such as protection, sensation, secretion, and metabolism 110 . The epidermis is mainly composed of keratin-forming cells with no internal vascular network structure and relies on blood vessels in the dermis for nutritional support. The dermis is mainly composed of fibroblasts, which synthesize ECM and effectively maintain the mechanical strength and elasticity of the skin. This layer also contains a large number of vascular network structures and skin appendages that effectively function in nutrient transportation and waste excretion. The subcutaneous tissue is mainly vascularized adipose tissue, which is important for storing fat and maintaining body temperature. Three-dimensional skin models constructed in vitro are full-layer structures of skin cells but lack immune cells, blood vessels, nerve distribution, and sweat glands. Ideally, engineered skin tissues should be reconstructed with the internal vascular structures, appendages (hair, sweat glands, sebaceous glands, etc.), neural structures, and immune system that normal skin contains, in addition to achieving the barrier function of the skin, to mimic real skin.
The simplest in vitro simulation of the skin is the 2D coculture system, which simulates the immune response of the skin. It uses structural and immune cells such as keratinocytes, fibroblasts, lymphocytes, eosinophils, basophils, and mast cells 111 . These 2D coculture systems are inexpensive and simple to maintain. However, they cannot simulate skin structure or represent intercellular or ECM interactions in 3D space 112 . In addition, the use of these biopsies in the laboratory is limited due to skin donor restrictions and variability issues. Therefore, 3D culture systems that include immune components in vitro have attracted a great deal of attention from researchers. The simplest approach is to add relevant cytokines to the 3D skin equivalents. Alternatively, 3D skin is cocultured with immune cells. For example, Langerhans cells have been added to the full-thickness skin equivalent (FTSE) model, which express CD1a and Lag and contain the same Birbeck granules as epidermal Langerhans cells 113 . Other immune cells of the skin dermis, such as dermal dendritic cells, T cells, and macrophages, can also be added to the FTSE model114–116. Disease-associated immune cells have now been used to build models related to inflammatory skin diseases, such as Alzheimer’s disease and psoriasis. These models recapitulate the phenotypes of several diseases and demonstrate their potential for drug screening and elucidation of pathogenesis. Recently, 3D skin structures containing immune cells and neurons have been developed as the importance of neuro–immune–skin interactions in the inflammatory response has emerged. A microfluidic culture device called a “skin chip” has also been developed, which mimics the structure and function of human skin by perfusion to simulate the migration of immune cells in the vascular system. This 3D skin model with an immune component can be used to test drugs for inflammatory skin diseases.
Vascular growth factors play an important role in all stages of wound healing, such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), basic fibroblast growth factor (bFGF), TGF, angiopoietin, and many other growth factors 117 . Although these growth factors are effective in promoting angiogenesis, they are all at low concentrations under physiological conditions and are unable to satisfy the need for good vascularization to avoid tissue hypoxia and cell necrosis 118 . Therefore, promoting angiogenesis through direct supplementation of various growth factors in engineered skin tissue is one of the most direct approaches. To make growth factors work continuously and stably in engineered skin tissue, a large number of studies targeting drug release systems have gradually become a key exploration direction. Common drug release systems include hydrogels, polymeric microstructure and nanostructure, and smart response systems119–121. The effect of promoting neovascularization can be achieved. In addition, stem cells play an important role in the process of neovascularization; they can not only induce the migration and proliferation of vascular endothelial cells by secreting a variety of growth factors (including VEGF-A, FGF, TGF-β, etc.) but also differentiate themselves into vascular endothelial cells to directly participate in the formation of neovascularization 122 . Neovascularization can also be promoted by inoculating cells into engineered skin tissue. Some angiogenic strategies based on active nanoparticles have also been successively investigated. These active nanoparticles can be used to promote neovascularization by inducing the generation of reactive oxygen species (ROS) or by loading some active molecules, such as plasmid DNA, microRNA, GF, and so on 123 .
Ideal engineered skin tissue models replicate the structural integrity and function of natural skin to allow wound repair, temperature control, and sensation to be achieved. During new tissue formation induced by newly constructed engineered skin tissue, nerve regeneration is dependent on newly formed blood vessels. Nerve regeneration is almost completely inhibited due to lack of blood supply124,125. The effect of vascularization on nerve regeneration is significant, and the vascular system provides nutrients for the regeneration of axons and associated cells, which may improve their long-term survival. Vascular endothelial cells secrete molecules that favor nerve regeneration. Schwann cells play a key role in peripheral nerve repair, and their role is activated after the onset of injury. Schwann cells, axonal contents, and inflammatory nerve tissue around the nerve come into contact with the injured axon and release large amounts of nutrients and growth-inducing molecules to promote axon sprouting and regeneration 126 . Seed cells are the core functional performers in tissue engineering, and in the treatment of stem cells, the goal of serving as seed cells for tissue engineering is achieved by inducing differentiation of different kinds of stem cells exhibiting Schwann cell phenotypes. Blood vessels also serve as trajectories for Schwann cell migration and guide axon growth 127 . Gene editing, on the other hand, is an important and direct intervention that can act on stem cells to make them express desired properties. The use of stem cells to enhance vascularization can help improve the survival of engineered skin tissue cutaneous nerves.
The development of engineered skin tissue has opened a new way for the repair of large skin defects in the last decade. However, all kinds of artificial skin cannot reconstruct the functional skin appendages such as sweat glands, which reduces the skin’s ability to adapt to the external environment. Most studies have constructed a new generation of engineered skin tissue containing sweat glands by culturing sweat gland epithelial cells, epidermal stem cells, and other cells in 3D stereoscopic culture with sweat gland–like structure formation 128 . However, further research is needed to explore how to construct functional engineered skin tissue by using stem cells with sweat gland phenotype as seed cells. In the near future, the emergence of sophisticated artificial intelligence (AI) platforms such as generative AI will reshape the research landscape. Incorporating appropriate mechanical properties, pigmentation, vascular networks, hair follicles and glands, subcutaneous layers, and innervation will make future skin models more clinically relevant129–132. Consequently, this will reduce the reliance on animal models and provide more clinically relevant models for drug and cosmetic development as well as for elucidating the mechanisms behind many dermatologic diseases and disorders 133 .
Application of Engineered Skin Tissue
Engineered skin tissue is widely used in cosmetic testing. It is generally used for safety testing, including skin corrosion, skin irritation, skin phototoxicity, and genotoxicity, and for efficacy testing, including skin barrier, moisturization, anti-inflammation, UV protection, and transdermal absorption. The 3D epidermal model-based efficacy assay can integrate multidimensional data at the gene level, protein level, and tissue level to achieve a comprehensive efficacy assessment (Fig. 3).
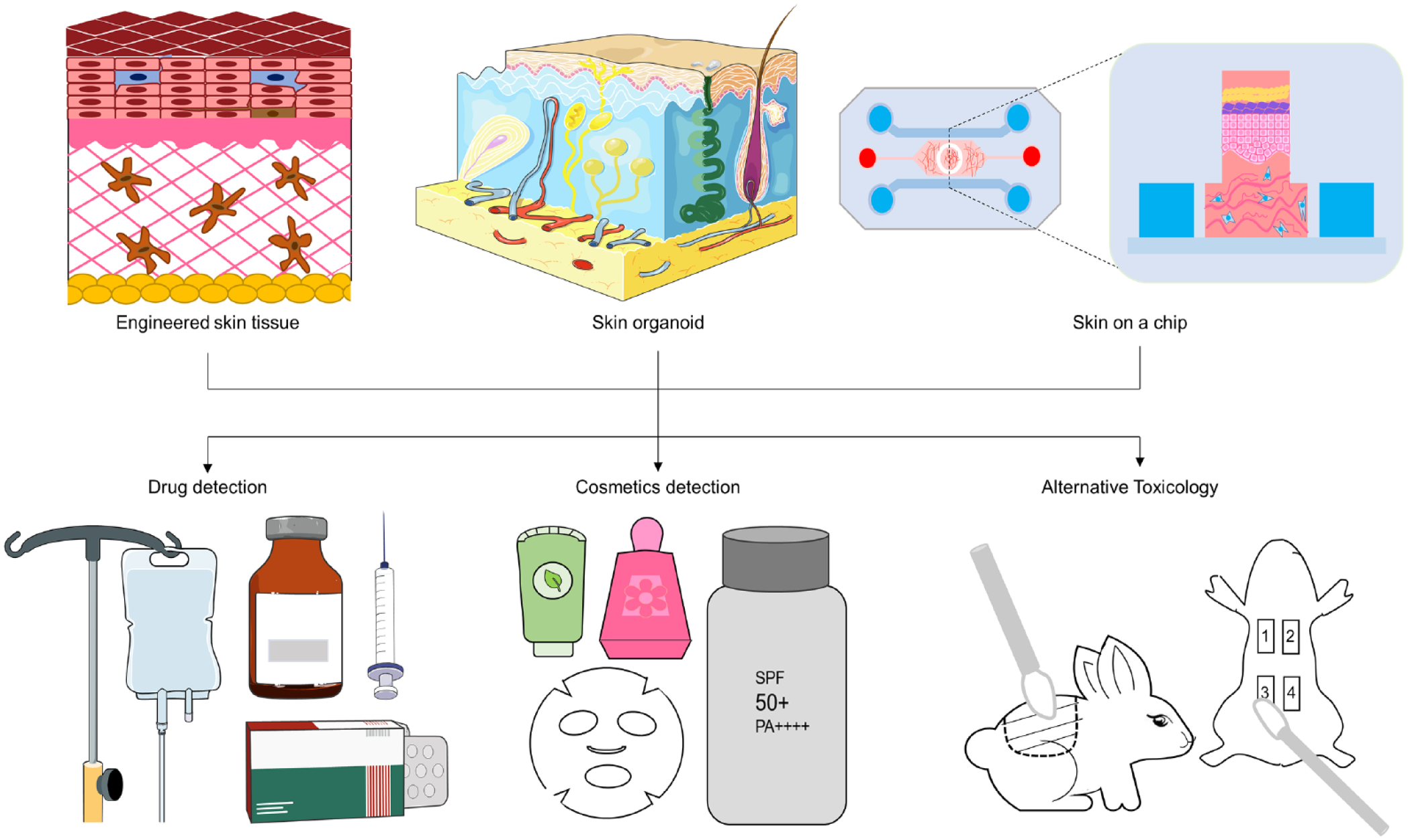
Application of in vitro skin modeling. Engineered skin tissue models, 3D skin models, and skin microarrays have been widely used in animal substitution experiments for safety testing of food, drugs, and cosmetics, such as skin corrosion, skin irritation, skin phototoxicity, genotoxicity, and so on. Meanwhile, these models can also be used for in vitro testing of skin barrier, moisturizing, anti-inflammatory, UV protection, and other effects.
Cosmetic Component Detection
The key to cosmetic efficacy testing models is to simulate in vitro the efficacy response of human skin tissue to the test substance. Three-dimensional skin models are structurally and physiologically metabolically consistent with human skin, enabling in vitro scientific testing of cosmetic ingredients and formulations.
The 3D reconstituted skin model is slightly different from human skin in terms of lipid composition and does not contain blood vessels, hair follicles, sweat glands, and so on. The transdermal pathways of substances in the 3D reconstituted skin model are transcellular and intercellular. There is still no accurate in vitro test method to determine whether many efficacious ingredients in cosmetic products can penetrate through the skin to reach their targets and the exact amount of penetration in recent experiments. The 3D reconstituted skin model is slightly different from human skin in terms of lipid composition, and it has a poorer barrier function than human skin 134 . Three-dimensional reconstituted skin models for transdermal absorption studies are currently available, such as EpiDerm, SkinEthic, and EpiSkin, and can be used to some extent as a substitute for human and animal skin for transdermal absorption studies of substances when the subject is an aqueous solution 135 . Therefore, the advent of engineered skin tissue in in vitro experiments has attracted much attention from the cosmetic research community. Gabbanini et al. 136 used SkinEthic, an engineered skin tissue, to perform transdermal experiments on eight ingredients commonly found in cosmetics, namely, camphor, carvone, 1,8-cineole, linalool, menthol, alpha-thujone, menthone, t-anethole, and then analyzed the results kinetically and compared them with the gas chromatography (GC)/HPLC–mass spectrometry (MS) instrument results, and the results were reliable. Zhen Liu 137 used a recombinant human skin model with a layer of target cells (L5178Y) in the lower layer of the skin model to determine the transdermal absorption of TiO2 nanomaterials and their mutagenicity by an indirect experiment. Pfuhler et al. 138 also used an EpiDerm skin model and cosmetics after a period of contact to analyze the damage to cellular DNA, thus applying it to the detection of the genotoxicity of cosmetics.
The effect of cosmetics on cell proliferation and differentiation and antioxidant capacity can be investigated by applying cosmetics to engineered skin tissue models and measuring relevant indicators such as the free radical scavenging capacity, antioxidant capacity, and collagen content. Grazul-Bilska et al. 139 applied moisturizing cream to the EpiDerm model and measured the oxygen radical absorption capacity. Santa-María et al. 140 used engineered skin tissue as a model and found that water-soluble enzymes in rice bran raw material reduced lipid peroxidation values. Antiaging efficacy evaluation on engineered skin tissue models better reflects the real state of the organism, as opposed to experiments conducted directly with skin cells.
In vitro reconstruction of human skin models has been widely used for skin irritation and skin phototoxicity studies, while in vitro construction of epidermal or whole skin models containing melanocytes can also be used to evaluate the efficacy of skin lightening cosmetics. For example, SkinEthic, a subsidiary of L’Oréal, has developed model containing melanocytes in addition to single-layer epidermal models, while others, such as MatTek’s EpiDerm series, also have similar models. In vitro skin models with epidermal barriers similar to human skin and multiple types of skin cells can provide comprehensive information for overall safety and efficacy evaluation by understanding the skin absorption of whitening agents, assessing skin irritation, studying melanin inhibition, and analyzing multiple skin cell interactions 141 .
On the other hand, 3D epidermal models are excellent models for testing sun protection function, and indices such as tissue viability, burn cells, and associated inflammatory factors based on 3D epidermal models can demonstrate the damage caused by UV irradiation to the models and the sun protection effect of the tested substances. Bernerd et al. 142 identified wavelength-specific biological damage in in vitro reconstructed human skin and evaluated two broad-spectrum sunscreen compound formulations that provided protection after topical application. Duval et al. 143 used whole-layer reconstructed skin, including differentiated epidermal and living dermal equivalents, and identified UVB-induced and UVA-induced biomarkers at the keratin-forming cell and fibroblast levels. Typical sunburn response markers as well as skin damage associated with the photoaging process can be reproduced in this model.
Skin Toxicology Evaluation
Engineered skin tissue is used as an alternative in vitro model for predicting the safety and toxicity of cosmetic products for the detection of skin absorption, acute toxicity, corrosion, irritation, and skin sensitization. In 2019, the European Centre for the Validation of Alternative Methods (ECVAM) presents a series of validated cell-based in vitro models for alternative animal experiments to predict the safety and toxicity of cosmetic ingredients. These models have proven to be valuable and effective tools to overcome the limitations of in vivo studies in animals. For example, 3D human skin models are used as the optimal standard for assessing skin irritation and skin absorption. Three-dimensional epidermal models are also used in testing studies of cosmetic products for phototoxicity, genotoxicity, and skin penetration. They tend to recapitulate the in vivo characteristics of human skin in terms of epidermal morphology, differentiation, and barrier function 144 .
Skin corrosiveness/irritation is one of the more important toxicological endpoints in the safety evaluation of cosmetics. The Organisation for Economic Co-operation and Development (OECD) operational guidelines have two types of surrogate tests for skin irritation/corrosiveness: the recombinant human epidermal model test and the in vitro skin corrosive membrane barrier test. The rationale of the first type is based on the assumption that corrosive/irritating chemicals can penetrate through the stratum corneum by diffusion or erosion and exert toxic effects on the cells in the underlying layers. The model is a multilayered, highly differentiated, 3D epidermal model constructed using tissue engineering techniques and contains structures similar to normal skin, such as the basal layer, spiny cell layer, granular layer, stratum corneum, and lipids. However, no blood vessels are formed, and therefore, cellular activity is chosen as the assay endpoint for the assessment of cell and tissue damage. TG431 was applied topically to the 3D RHE model, and after a specified time, the cellular activity of the subject RHE tissue was measured using the Methylthiazolyldiphenyl-tetrazolium bromide (MTT) reduction method. Then, the corrosive subject was identified and classified by comparison with a threshold value 145 . TG439 further exploited the characteristics of the RHE model by incubating rinsed RHE tissues in fresh medium for a sufficiently long time before performing cellular activity measurements. Thus, both allow RHE tissues to recover from weaker cytotoxic effects and provide sufficient time to express clear cytotoxic effects 146 . The in vitro skin corrosive membrane barrier test is a method based entirely on an artificial, nonbiological skin model. Its test system consists of an artificial polymeric biofilm and a chemical detection system (CDS). The principle is based on the assumption that the artificial polymeric biofilm has a similar mechanism of corrosive action to that of living skin, and the subject is applied locally to the surface of the polymeric biofilm. After a specific action time, the barrier function of the polymeric biofilm is detected by the CDS to determine whether the barrier function of the polymeric biofilm is disrupted and whether the object being tested penetrates the polymeric biofilm, to identify and classify the corrosiveness of the molecule 147 . The ECVAM guidelines “Technical standards for human skin models for in vitro skin irritation tests,” published in May 2007, and the “Validation methods for two in vitro skin irritation tests” statement, published in November 2008, have indicated that engineered skin tissue, such as the engineered skin tissue EpiSkin, EpiDerm, and SkinEthic, can be used as an alternative model for in vitro skin irritation tests 148 .
Normal human corneal epithelial tissue consists of five to six layers of differentiated ectodermal cells that protect the cornea and constitute its visual properties 149 . The 3D corneal epithelial model is a layered epithelial tissue that simulates corneal epithelial and conjunctival injury and can be used for acute ocular irritation/corrosion testing. The epithelial cells used to construct the corneal model were derived from primary cultures of neonatal perithelial keratinocytes, which closely resemble the corneal epithelium. This method uses an in vitro reconstituted 3D corneal model to test the ability of chemicals to alter tissue viability and thus predict the potential ocular irritation of chemicals. In addition, there are eye irritation experiments using the EpiOcular corneal model, SkinEthic eye irritation test (EIT), and LabCyte corneal model.
Artificial epidermis/skin models have been used in skin allergy replacement experiments. The full-layer skin culture in vitro has a two-layer structure of epidermis and dermis, and keratinocytes are cultured in the dermis that has been stripped of collagen or the upper layer or at the gas–liquid interface that mimics skin structure and metabolism. Currently, cells used in sensitization studies include dendritic cells, and keratinocytes, of which the first two include Langerhans cells, human peripheral blood mononuclear cells, CD34+ hematopoietic stem cells, and dendritic cell lines (THP-1, U937, KG-1, and MUTZ-3) 150 . Among them, THP-1 cell culture has been widely recognized for sensitization studies. Coqutte et al. 151 used stimulants and sensitizers in an in vitro whole skin culture model to determine the effect of sensitizers on interleukin (IL)-1α and IL-8 at the protein and mRNA levels. This system does not fully reflect the sensitization process due to the lack of immune cells that trigger the sensitization reaction in artificial skin and therefore still needs to be improved.
The current in vitro assay system for in vitro phototoxicity testing has major drawbacks. The monolayer cultured fibroblast system is structurally homogeneous compared with 3D skin. Moreover, some chemicals with low solubility can only be detected at low concentrations. In addition, the system cannot detect complex mixtures or formulated products. Reconstructing skin models for phototoxicity testing is a meaningful potential alternative. Researchers have developed an in vitro phototoxicity test method using a human-derived reconstructed skin model, EpiSkin. Previous studies have concluded that this method is capable of assessing the phototoxicity of chemical end products and complex formulations 152 . Both epidermal skin models containing keratinocyte and fibroblast layers and full-layer skin models containing keratinocyte and fibroblast layers are human 3D skin models. In contrast to cells, human 3D skin models can simulate the topical application of materials onto skin. Because the 3D skin model has a stratum corneum barrier, the absorption and penetration results of compounds and molecules produced by light are closer to the in vivo situation.
Skin Organoids in Primary Culture
In vitro models have been an important research tool in the field of dermatology for two decades. Most of the existing in vitro skin research models are based on 2D skin cell and 3D skin cell models cultured in vitro, and there is still a large gap between the interlayer interactions and the real physiological state. Skin organoids based on stem cell culture technology are similar to the in vivo developmental process compared with the traditional skin allograft model, are capable of self-organization and directed differentiation based on cell categories, and can generate skin appendages such as hair follicles and sebaceous glands that are incomparable to skin models. Using 3D skin organoids, such as human umbilical cord blood stem cells and pluripotent stem cells, that have undergone differentiation for in vitro environment simulation culture can improve the limitations of existing 2D and 3D model cultures, which have a single cell type, lack cell—cell interactions with the ECM, and cannot self-organize and differentiate in vitro, to thus narrow the differences with in vivo multicellular tissues and their physiological functions153–155. For example, skin organoids are significantly more resistant to a given dose of hydrogen peroxide or silver nitrate and have better resistance to oxidative stress than monolayer cultured keratinocytes and fibroblasts. Sun et al. used 3D skin organoids containing human dermal fibroblasts, keratinocytes, and small vessel endothelial cells to study the cells’ ability to respond to a given dose of hydrogen peroxide or silver nitrate stress. It was found that the skin organoids were significantly more resistant and had better resistance to oxidative stress 156 . However, at the same time, there are some limitations of skin organoids; for example, the growth cycle and consistency have not yet reached consistent standards. In recent years, with the implementation of animal replacement tests in Europe and the United States, in vitro-constructed skin organoids have been used to simulate the human skin environment to the greatest extent possible in vitro for more intuitive and effective research and treatment of skin diseases and have been widely used to study the pathophysiology of skin diseases and systemic repair damage in vitro.
Animal experiments that have long been applied to scientific research have many limitations, such as long experimental cycles, individual variability, difficulty in modeling, and interspecies differences 157 . Moreover, drugs with severe toxicity in animal testing are not tested in humans, so some drugs that are safe and effective in humans may be missed in the screening process 158 . New in vitro alternative assays need to be developed, such as the in vitro culture of various types of skin cells, tissues, and organoids, combined with computer simulations to predict toxicity, refine and complement drug preexperiments, and serve as a prescreening prior to animal experiments, with better consistency at a higher throughput and on a large scale. Therefore, the construction of organoids with certain structures and functions can be used to achieve large-scale, high-throughput experimental models under specific conditions as a way to complement and improve traditional animal experiments 159 .
Skin organoids are primary cultures that maintain the structural features of cells, intercellular relationships, and ECM in vitro. While the epidermis of the skin is derived from the ectoderm, the dermis has a different embryonic origin, and regardless of the dermal origin, all skin types require the interaction of epithelial (epidermal) and mesenchymal (dermal) cells to eventually develop and form appendages160–162. The epidermis of the skin produces hairs and glands, with keratinocytes as the main cell type, which contribute to thermoregulation and barrier formation. The dermis of the skin encompasses other structures, such as blood vessels and nerves, with dermal fibroblasts as the main resident cells, which produce ECM and contribute to hair follicle initiation and circulation. Therefore, the construction of in vitro models of skin and its appendage organoids that mimic the skin tissue system by using cells of different origins in targeted culture or induced differentiation according to the physiological structural features of the skin can help in understanding the complex interactions of different cell types and molecular signaling pathways during development and in vivo homeostasis. In the embryonic development of skin, signaling pathways such as Wnt, bone morphogenetic protein (BMP), and FGF play crucial roles, with Wnt signaling having emerged as a major pathway controlling skin patterns and controlling differentiated skin cell function 163 . For example, Wnt pathway-dependent hair follicle regeneration after skin trauma in adult mice was identified in earlier studies 164 . However, how these pathways regulate the genetic network interactions of epidermal stratification during embryonic development remains unclear 165 . With the widespread research and application of organoids, the application of skin organoids is expanding from the construction of laboratory research models to the fields of postinjury healing, skin cancer, immunometabolic disease and inflammatory disease treatment, and drug testing.
Bionic Skin-on-a-Chip
Organ-on-a-chip technology and microfluidic chips developed in recent years can be used to design human organs with clear structure and function, simulate organ and organ system activities and biomechanical and physiological metabolic reactions, and precisely control the cell and microenvironment, enabling the study of human physiology of specific organs in specific environments. Endothelial cells are applied to cultured skin equivalents by fixing them to external pumps and tubes to form perfusable vascular channels. Sriram et al. developed a well-differentiated and keratinized human skin equivalent based on a fibrin-based dermal matrix combined with a bionic “organ-on-a-chip” system. This model enables low-cost, high-throughput in situ permeability and toxicity testing 166 . Lee et al. 167 developed a microfluidic 3D skin chip using polydimethylsiloxane (PDMS) and hydrogel to model the oxygen and glucose transport reactions in the skin chip. Ataç et al. 168 developed a multiorgan chip to culture skin and hair follicles in vitro. This hair chip was constructed by replicating the main features of hair follicle morphogenesis and the hair cycle to build up a fully functional hair follicle model.
The application of skin-on-a-chip technology containing microcell culture devices allows 360° cell spreading, migration, proliferation, differentiation, and interactions to be witnessed while simulating multiple physiological metabolic functions of the skin, such as lipid metabolism, metabolite analysis, skin permeability, and in vitro skin irritation assessment models169,170. Skin-on-a-chip models have also been established by microfluidic systems to simulate skin problems such as edema, psoriasis, skin cancer, and wounds, but most of the reported skin microarrays rely on skin cells without epidermal keratinization and without other nonskin cell types, which are completely different from the dynamic and complex natural skin environment. As a result, current skin chips are not yet able to perform complex assessments, such as sensitization and toxicity to internal organs. The utilization of emerging artificial skin chips to design increasingly complex microenvironments in the future to better support organ-level functions provides a state-of-the-art in vitro platform for human cellular evaluation of food, drug, and cosmetic safety and efficacy.
Conclusion and Perspectives
Currently, the use of animal models for testing food, drugs, and cosmetics is being massively reduced or even banned worldwide, and it is necessary to establish reliable human skin models in an in vitro environment to simulate physiological events in skin tissues. Both fine toxicological evaluation of drug and cosmetic compounds by human skin models and the best reference for product efficacy for pharmaceutical and cosmetic industries are possible.
We summarize the existing methods for testing cosmetic ingredients and for toxicological evaluation, enumerating and discussing the limitations of traditional testing methods and their current applications. The focus is also on 3D skin models, which can significantly improve toxicology, even if they are not considered ideal by current standards, which shows the promise of skin models for toxicological testing.
The ideal tissue-engineered skin should be as close as possible to normal skin in terms of morphology and function, and the key lies in finding the right seed cells. It is believed that with the development of a multidisciplinary combination of bioengineering, life science, and material science, the bottlenecks that restrict engineered skin tissue will be future directions of research. At present, engineered skin tissue model testing can only be performed in the laboratory using sophisticated instruments and complex procedures. Its development requires sustained efforts by all researchers to make significant progress.
Many skin models do not include either the microenvironment (e.g., multiple cell types, skin appendages) or the various dynamic functions of our skin barrier. As researchers begin to drive innovation to fill this gap, the inherent limitations of the “current model” will become increasingly apparent. Leading industry stakeholders are incorporating Langerhans cells, melanocytes, and diseased cell types to create immunologically active, hyperpigmented or diseased skin models. This trend is also reflected in research on biotechnological developments such as 3D bioprinting and microfluidics. In the future, technologies such as nanocomposites, advanced bioprinting strategies, and microfluidic engineering will be applied to enhance the relevance and reproducibility of in vitro skin models for dermatological science and cosmetic testing applications, enabling the translation of engineered functional structures to the clinic for the treatment of various dermatological conditions (e.g., tissue defects, psoriasis, and sweating) and personalized drug discovery and therapy 171 .
Advanced material preparation techniques can better simulate the ECM and extracellular microenvironment. Three-dimensional polymer scaffolds allow surface-inoculated cells to grow inwards. They offer inherent advantages, including biocompatibility, conductivity of cell adhesion, and sensitivity to cell-secreted enzymes and cell-mediated remodeling. However, they impose restrictions on the cells and do not reproduce the fibrillar properties of natural ECM proteins. Therefore, generating highly biomimetic designs that address batch-to-batch variability and using high-throughput analytical methods can advance and deepen our understanding of cell–cell/material/extracellular microenvironment interactions. These methods and techniques will continue to receive attention to advance the development of skin models.
Footnotes
Acknowledgements
Not applicable.
Authors’ Contribution
Not applicable.
Availability of Data and Material
Not applicable.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
