Abstract
The αβ T-cell-depleted hematopoietic stem cell transplantation (HSCT) leads to lower relapse and better outcome, and may correlate strongly with expansion of donor-derived γδ T cells. γδ T cells play an important role in immune reconstitution and can exert a graft-versus-leukemia effect after HSCT. This review showed the recent literature on immune functions of γδ T cells after HSCT. The discrepancies between studies of γδ T cells in graft-versus-host disease may cause by its heterogeneous and various distinct subsets. And reconstitution of γδ T cells may play a potential immunoregulatory role in the infections after HSCT.
Keywords
Introduction
Hematopoietic stem cell transplantation (HSCT) is an important treatment for hematological malignancies and is wildly used in leukemia for decades 1,2 . According to whether grafts are accepted to T cell depletion or not, HSCT is divided into T-cell-depleted HSCT and non-T-cell-depleted HSCT. T-cell-depleted HSCT leads to favorable overall survival (OS) and disease-free survival and deduction of graft-versus-host disease (GVHD), while non-T-cell-depleted HSCT leads to higher risks of GVHD 3,4 . αβ T cells were depleted in T-cell-depleted HSCT for purposes of lower relapse and better outcome 5 , and αβ T-cell-depleted HSCT showed that event free and OS correlated strongly with expansion of donor-derived γδ T cells 6 . γδ T cells might play an important role in immune reconstitution (IR) and could exert a graft-versus-leukemia effect after HSCT 6 –11 . Moreover, higher numbers of γδ T cells might improve clinical outcome following HSCT 12 . In this report, we review the recent literature on immune functions of γδ T cells after HSCT.
General Characteristics of γδ T Cells
According to the surface receptors, T cells can be divided into different subsets. Based on the difference of T cell receptor (TCR), T cells can be divided into αβ T cells and γδ T cells. A large amount of T cells are αβ T cells, while γδ T cells account for a small proportion, approximately 1.2%–15.4% 13 . Human γδ T cells can be recognized by the expression of TCR Vδ and TCR Vγ, and TCR Vδ1, Vδ2, Vδ3, Vγ2, Vγ3, Vγ4, Vγ5, Vγ8, Vγ9, and Vγ11 are the most commonly used gene fragments for rearrangement of δ and γ chains 14 . In human peripheral blood, a large amount of γδ T cells express TCR Vδ2 chain paired with Vγ9 chain, and γδ T− cells expressing TCR Vδ1 or Vδ3 chain can be paired with various Vγ chains 15,16 . Most of γδ T cells in lymphoid organs and peripheral tissue like skin, lung, and intestine usually express TCR Vδ1 or Vδ3 chain, but not Vδ2. As we all known, γδ T cells are CD3+ lymphocytes, and most of them neither express CD4 nor CD8, but a small percentage of cells are CD8+ γδ T cells 17 .
T cells can be divided into cytotoxic T cells, helper T cells, memory T cells, and regulatory T cells (Tregs) according to their different functions. Nowadays, the research of the functions and effects of different subsets of γδ T cells and their application have been a heated topic. Antitumor activities can be exerted by γδ T cells to fight against solid tumors and hematological malignancies via in vitro and in vivo mechanisms like cytotoxicity effect. γδ T cells can effectively kill tumor and directly identify target molecules expressed by leukemic cells in a major histocompatibility complex (MHC) independent manner 18 . In addition, UL16 binding proteins (ULBP) 4 on tumor cells can be matched with γδ T cells, and the overexpression of ULBP-1 and ULBP-4 will cause the cytotoxicity of Vγ9+ Vδ2+ T cells 19 . Various cytokines such as IFN-γ and TNF-α can be produced by activated γδ T cells and play a significant role in potent cytotoxic activity. However, Iwasaki et al. found that the expression of programmed cell death-1 (PD-1) on γδ T cells may diminish the levels of cytokines production and cytotoxicity against programmed cell death ligand-1 tumor cells 20 . And they also reported that the inhibitory effect of PD-1 in γδ T cells may partially overcome by TCR triggering. Moreover, through the inflammatory CCR2/CCL2 chemokine pathway, the γδ T cells that are tumor infiltrating can be collected into the tumor bed 1 .
Previous studies suggested that Tregs might affect the clinical outcome of HSCT, and Tregs counter-regulation might promote allograft tolerance 21 . Distinct subsets of γδ T cells, especially γδ Tregs, can also play an important role in HSCT 7 . We found that γδ Tregs expressed the novel immune checkpoint receptors T cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif domain, CD226, and DNAX accessory molecule-1 (DNAM-1) in aute myeloid leukemia (AML) patients and the expressions were associated with clinical outcomes 22,23 . γδ Tregs were also found to inhibit the proliferation of CD4+CD25− T cells in patients with multiple myeloma 24 . And in healthy donors, Casetti et al. initially discovered Foxp3+ rich Vδ2 T cells, which were stimulated by transforming growth factor-beta 1 (TGF-β1)/IL-15, suppress the proliferation of peripheral blood mononuclear cells (PBMC) 25 . What’s more, another study also suggested that γδ Tregs from human PBMC could facilitate the induction and immunosuppressive function by decitabine combined with zoledronate (ZOL)/IL-2/IL-15-TGF-β1 26 .
Meanwhile, γδ T cells have the ability to effectively induce the CD8+ T cells to proliferate and kill target cells 27 . And γδ T cells can induce the antitumor cytotoxicity of other cells, especially natural killer (NK) cells 28 . Additionally, subsets of γδ T cells coexpress some receptors of NK cells like natural killer group 2D (NKG2D), DNAM-1, and CD16 13,29,30 . Moreover, NKG2D that is expressed on Vδ1 T cells can be combined with MHC class I chain-related A (MICA), and this combination between MICA and NKG2D was stronger than the one between MICA and TCR 31 . As for B cells, B cells can be induced to proliferate as well as secrete IgM by Vδ3 T cells 32 . In another research about dendritic cells, it suggests that γδ T cells might be able to promote the maturation of dendritic cells 33 . In conclusion, through antigen presentation, inducing other immune cells, and many other pathways, γδ T cells play an essential role in interacting with other immune cells (Fig. 1).
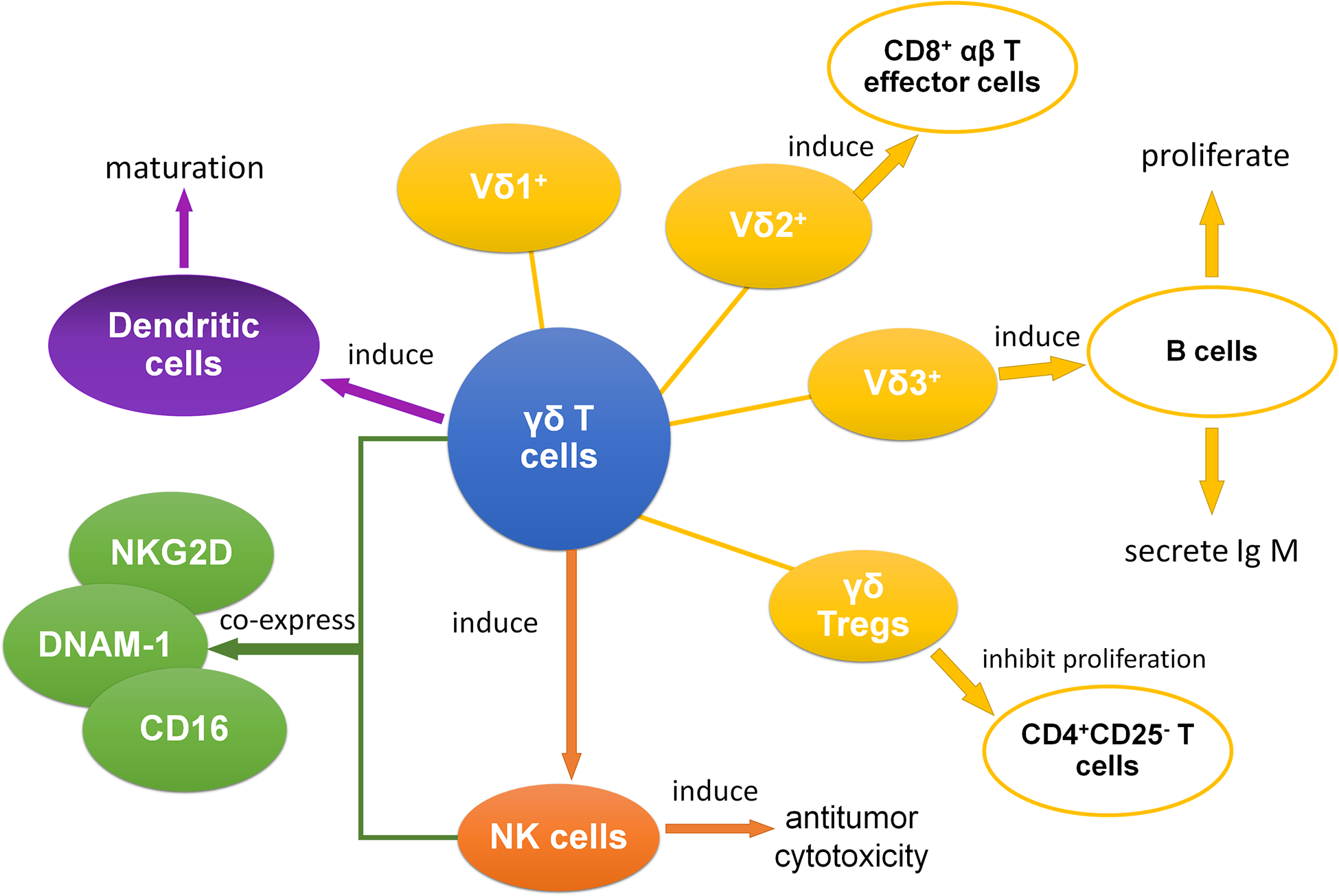
With complexity of cell surface expressed molecules and ability inducing antitumor cytotoxicity of other immune cells, γδ T cells play a crucial role in HSCT. A study established that quick reconstitution of the γδ T cell repertoire after allogeneic HSCT (allo-HSCT), which might help improve OS, retained the overall complexity and proportion of public clonotypes 34 . Increased γδ T cells also have an advantage in improving leukemia-free survival and OS of the patients with acute leukemia after allo-HSCT, which is expected to reduce the recurrence rate considerably and contribute to better effect 6 . γδ T cells also have effect on the tolerance of immunotransplantation. Our previous studies showed that granulocyte colony-stimulating factor (G-CSF) could induce immune tolerance and it might associate with clonality of TCRs on γδ T cells and the increase of γδ Tregs in grafts 7,35 . We demonstrated that γδ Tregs had certain cytotoxic effects on tumor cells after G-CSF treatment 7 . They also found that the proportion of CD27+ Vδ1 Tregs in graft was negatively associated with acute GVHD (aGVHD) 7 . In addition, in liver transplantation, Vδ1 T cells with a unique clone in graft were reported which could recognize a specific ligand and contributed to the establishment of tolerance by suppressing rejection 36 . Therefore, γδ T cells might play a vital role in immune tolerance of transplantation and be beneficial to outcome of HSCT.
IR of γδ T Cells After HSCT
IR is one of the key factors for the success of allo-HSCT and depends on transplant modality, graft treatment, pretreatment, and immunosuppression after transplantation 37 . γδ T cells as effective cells to kill tumors can reconstitute early after transplantation 34 . Donor source and virus reactivation might associate with the reconstitution of γδ T cells and their subsets. It has been shown that in recipients of umbilical cord blood-HCT (UCB-HCT), Vδ2 T cells were almost undetectable 9 . However, Vδ2 T cells have a bimodal response in recipients of HLA-matched sibling or unrelated donor allo-HCT (MSD/MUD-HCT), about 30% individuals have higher Vδ2 repertoires, and others have lower Vδ2 repertoires 9 . Another study on HLA-haploidentical HSCT (haplo-HSCT) also showed that homeostatic donor γδ T cell content was associated with the early γδ T cell recovery following HSCT 38 . Similarly, Perko et al. suggested that donor source affected the IR of γδ T cells 10 . Patients who underwent matched related donor or haplo-HSCT had a significant difference in the reconstitution of γδ T cells compared with matched unrelated donor patients, whereas there was no difference between patient characteristics of age, gender, disease, GVHD prophylaxis, and the recovered γδ T cells 10 . Furthermore, they also found that CD3+, CD4+, and CD8+ T cells also affected the recovery of γδ T cells 10 . In other side, in cytomegalovirus (CMV) reactivation, Vδ1 T cells were increased in both MSD/MUD-HCT and UCB-HCT, but the difference was statistically significant only in recipients of UCB-HCT 9 . Day 30 Vδ1 recovery inversely associated with CMV reactivation in HSCT recipients 38 . And Vδ2 T cells recovery had the negative correlation with Epstein-Barr virus (EBV) reactivation after HSCT 39 . Indeed, more research is required to investigate the IR of γδ T cells after transplantation with a view to develop better application in HSCT.
The Roles of γδ T Cells in GVHD
GVHD is one of the most serious complications of allo-HSCT and is a major obstacle to improve outcomes of patients after transplantation. GVHD is divided into aGVHD and chronic GVHD (cGVHD) according to the time and type of organ involvement. The mechanism of aGVHD is immunocompetent T cells from the donor could recognize host alloantigens and interact with host antigen-presenting cells, then lead to donor T-cell activation and expansion, and cytotoxic effect 40,41 . The aGVHD often implicates skin, liver, and gastrointestinal tract, and combined with the involvement of these three organ systems could give a clinical stage, ranging from 0 to IV 42 . Unlike aGVHD, the mechanism of cGVHD is more complex and involves more organs. It involves three-phase model: early inflammatory response and tissue injury (phase 1), dysregulated B cell and T cell immunity and chronic inflammation (phase 2), and activation of the profibrotic pathway (phase 3) 40,43,44 . Based on National Institutes of Health consensus criteria and the number and severity of affected organs or sites, cGVHD is divided into three grades: mild, moderate, and severe 45 . With the depth understanding of the mechanism of GVHD, it was found that T cells might play an important role in GVHD. In this regard, the role of T cell subsets on GVHD has been extensively investigated. Several researchers reported that αβ T cells were considered to relate to the pathogenesis of GVHD, whereas γδ T cells receptor did not lead to GVHD and had strong antileukemia and antivirus activities 5,46,47 . Furthermore, γδ T cells were reported to play a protective role in GVHD, a heavy obstacle to the effectiveness of HSCT, as the cause of less risk of aGVHD in patients with higher γδ T cell concentration 8 . γδ T cells have potential good effect on GVHD prevention; however, the exact role of γδ T cells in GVHD remains an open question.
In mice models, some research showed that donor γδ T cells were involved in GVHD pathogenesis 48,49 . However, other studies demonstrated that these cells were not associated with the development of GVHD 50,51 . Similar to mice, in human, γδ T cells also play different roles in GVHD (Table 1). Pabst et al. suggested that donor γδ TCR-expressing CD3+ cells counted above the median were related to the cumulative risk of aGVHD II-IV, and γδ T cells in the grafts of patients in GVHD class II to IV were much higher than in the grafts of patients in GVHD class 0 to I 52 . Noteworthy, in their study γδ T cells in human were from unrelated donors and examined as a single-cell population without analyzed the effect of different subsets of γδ T cells. Another study that divided patients with GVHD into limited cGVHD group, extensive cGVHD group, and non-cGVHD group found that the percent of γδ T cells in non-cGVHD group significantly increased compared with other groups, and increasing number of γδ Treg was also reported to be a preferential strategy for controlling cGVHD following HSCT 53 .
aGVHD: acute graft-versus-host disease; cGVHD: chronic graft-versus-host disease; GVHD: graft-versus-host disease.
Recently, Arruda et al. used a high-throughput analysis of TCR Vγ repertoire in AML patients after accepted grafts, which suggested that TCR gamma locus (TRG) clonal distribution were not associated with occurrence or absence of aGVHD 54 . However, our previous study suggested that grafts with higher proportion of CD27+ Vδ1 Tregs was associated with a lower incidence of aGVHD in G-CSF-mobilized allogeneic peripheral blood stem cell transplantation recipients 7 . Similar to our study, a prospective, clinical study from Europe recently suggested that patients with high concentrations of total γδ T cells had lower incidence of aGVHD but did not affect the development of cGVHD after transplantation 8 .
The discrepancies between studies of γδ T cells in GVHD may cause by its heterogeneous and various distinct subsets, and using G-CSF might influence the distribution, expression levels, and clonality of γδ T cell repertoire. Furthermore, the origin of the graft and the heterogeneity of patient characteristics also can infect the clinical outcome. Consequently, it needs further work to evaluate the detailed phenotype of γδ T cells after transplantation, which may provide a great prospect for the treatment of GVHD.
γδ T Cells and Virus Reactivation After Allo-HSCT
γδ T Cells and CMV Reactivation
CMV infection is a common complication following allo-HSCT. Some previous studies reported that CMV reactivation increased transplant-related mortality (TRM) 55,56 . However, others studies demonstrated that CMV reactivation had no significant influence on the OS of patients after allo-HSCT 57 . Moreover, CMV reactivation might decrease the relapse rate of AML patients who accept HSCT 58 . IR following HSCT might be the key of these differences 59 .
CMV reactivation could occur in receptors who accepted the CMV+ grafts from donor or in immunocompromised patients like following HSCT 60,61 . γδ T cells reconstituted rapidly by 30–60 days following transplantation and had promoting influence to IR 34,55 . During CMV reactivation after early HSCT, proliferations of Vδ2 negative T cells but not Vδ2 positive T cells were observed in peripheral blood 62,63 . This result was similar to the finding of Raven et al. who found expansions of heterogeneous Vδ1, Vδ3, and unconventional Vδ2 clones during CMV reactivation after 25–60 days of HSCT 34 . Other studies found that Vδ1 T cells recovery was correlated with CMV reactivation in HSCT recipients by using multiplex PCR system to sequence TRG and TCR delta locus (TRD) CDR3 regions or flow cytometry analysis 9,38 . Several CMV-related γδ T cell clones proliferation in grafts from CMV+ donors were also observed 54 . Therefore, reconstitution of γδ T cells might be involved in the outcome of CMV reactivation after transplantation.
γδ T Cells and EBV Reactivation
EBV infection is another complication about virus reactivation following allo-HSCT. EBV reactivation was related with post-transplantation lympho-proliferation disorder and led to higher TRM and lower OS 64,65 . Vγ9 Vδ2 T cells, which were expanded by pamidronate, with engagement of Fas and TRAIL, were demonstrated to kill EBV-transformed autologous lymphoblastoid B cell lines 66 . Liu et al. found that recovered Vδ2 T cells were inversely related with EBV reactivation after HSCT and had cytotoxic on EBV-infected cells, while immunosuppressants play a negative roll on the anti-EBV capacity of Vδ2 T cells 39,67 . Djaoud et al. also found that Vγ9 Vδ2 T cells were the major innate immunity against EBV and could be expanded after EBV infection 68 . Moreover, their recent study reported that basing on the response to EBV, γδ T cells can be defined to the strong immunity group with large population, which expressing Vγ9 JγP and could produce activated effector cells, and the weak one with small population, which expressing Vγ9 Jγ2 69 .
EBV can be found in the skin or mucosa and may be associated with mucositis 70 . γδ T cells, with distinct subsets, as important innate immune cells for human, present in both blood and tissues and play multiple roles in mucosal inflammation 71 . Though one case report was shown that γδ T cells were found out in peripheral blood but not skin or lung during EBV-associated lymphoproliferative disease 72 . We suggest that γδ T cells might play some certain roles in tissue immunity during EBV-related infection. Regrettably, previous studies about γδ T cells IR after HSCT mainly reported total γδ T cells or Vδ2 T cells in peripheral blood during EBV infection. More studies need to be investigated to find out the effect of distinct γδ T cells IR in tissues during EBV reactivation following HSCT.
γδ T Cells and Other Infection After Allo-HSCT
Hepatitis B Virus
The high risk of hepatitis B virus (HBV) reactivation is associated with GVHD after HSCT and will make negative impact on post-transplant prognosis 73,74 . Currently, some studies reflect the relation between HBV inflection and γδ cells. It shows that in chronic hepatitis B (CHB), Vδ2 T cells decrease in patients’ peripheral blood and relate to less IFN-γ production and cytotoxic activity 75 . In a mouse model of acute HBV infection, liver γδ T cells and IFN-β production increased during the early stages of HBV infection, but there were no much changes in peripheral γδ T cells 76 . And IFN-α can enhance the cytotoxic function of peripheral γδ T cells in CHB 77 . γδ T cells have a potential role in the treatments of HBV infection and the disease it possibly lead to, and it can be more studied in the future.
Human Immunodeficiency Virus
Human immunodeficiency virus (HIV) is a lymphotropic virus, which will lead to acquired and innate immune suppression. Recent study suggested the potential function of allo-HSCT lead to sustained, anti-retroviral-free HIV-1 remission 78,79 . But it is unclear which mechanism might contribute to the HIV remission. HIV infection will cause to the normal ratio of Vδ2:Vδ1 cells by Vδ1 T cells increase and Vδ2 T cells depletion 80,81 . The dysfunction of Vδ2 T cells was probably caused by skewing toward a terminal effector memory phenotype 82 . And abnormal expression of γδ T cells can be a potential surrogate marker of acquired immunodeficiency syndrome progression 83 .
Fungus and Bacteria
Gram-negative bacteria, then followed by gram-positive bacteria and fungus are the main pathogens of bloodstream infection after HSCT 84 . Aspergillus and Candida albicans are common pathogens of invasive fungal diseases following HSCT 85,86 . γδ T cells reconstitution in early HSCT may play a potential immunoregulatory role in bloodstream infection. Vδ1 T cells could respond with proliferation and IFN-γ/IL-17 production to Candida albicans in vitro, whereas Vδ2 T cells could proliferate and produce IFN-γ/IL-17 in response to mycobacteria 87 . In addition, in response to Aspergillus fumigatus antigens, Vγ9Vδ2 T cell clones reactive by phosphate antigens were found to produce tumor necrosis factor in healthy individuals 88 . Similar results were found that Aspergillus fumigatus antigens could induce the expansion of γδ T cells indirectly 89 .
γδ T Cells and Relapse After HSCT
Relapse is a common cause of mortality and treatment failure after HSCT (Table 2). Arruda et al. found that patients with higher γδ T cells present less relapse during IR after HSCT 12 . Moreover previous study has shown higher cumulative incidence of death from relapse in patients with low γδ T cells and Vδ2 T cells 8 . It seems that γδ T cells can be a predict marker of relapse after HSCT. Measurable residual disease, also known as minimal residual disease (MRD), was reported as a predictor for relapse following HSCT 90 . MRD monitoring by using TRD and TRG rearrangement was demonstrated as a useful predictor for the risk of relapse in T-ALL patients with chemotherapy 91 . Furthermore, Galimberti et al. found that MRD eradication did significantly affect TCR γ/δ clones profiles in patients with multiple myeloma follow allogeneic non-myeloablative transplantation 92 . In a case report, Nomura et al. suggested that MRD, which assessed by PCR assay for TRD in the bone marrow, was useful for the prediction of relapse following bone marrow transplantation 93 . More studies need to be investigated to find out the relationship between γδ T cells and MRD, which may explain the mechanism that γδ T cells improved relapse following HSCT.
HCST: hematopoietic stem cell transplantation; OS: overall survival; RFS: relapse-free survival.
γδ T Cells and Immunotherapy
More and more studies showed that γδ T cells have antitumor activity and play an unique role in immunosurveillance, and can be used as a tool for immunotherapy. Ex vivo antigen-driven generating large numbers of autologous Vδ2 T cells and adoptive reinfusion is possible to be a viable strategy for immunotherapy-based intervention 94 . Furthermore, ZOL/IL-2 have significant effect on improving the cytotoxic effect of γδ T expanded in vivo 95 . Phase I clinical trials on γδ T cells, including both expanded in vitro and stimulated in vivo, purpose on adoptive transfer, were completed 96,97 . A few clinical trials (ClinicalTrials. gov Identifier: NCT03533816, NCT03862833) about using γδ T cells for transplantation improvement are ongoing, but the results of these clinical studies have not been reported. Now HSCT following chimeric antigen receptor (CAR)-T therapy were reported favorable outcomes like higher OS and lower relapsed rate 98,99 . CAR-γδ T cells therapy was a novel promote immunotherapy for antitumor 100 and CAR-γδ T cells may be a potential immunological treatment to improve clinical outcomes of HSCT.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant number 81770150), Natural Science Foundation of Guangdong Province (grant number 2020A1515010817), the Guangzhou Science and Technology Project (grant number 201804010425), Medical Scientific Research Foundation of Guangdong Province (grant numbers A2018565 and A2017198), and College Students’ Scientific and Technological Innovation (grant number 202010559081).


