Abstract
Patients with multiple system atrophy (MSA), a progressive neurodegenerative disorder of adult onset, were found less than 9 years of life expectancy after onset. The disorders include bradykinesia and rigidity commonly seen in Parkinsonism disease and additional signs such as autonomic dysfunction, ataxia, or dementia. In clinical treatments, MSA poorly responds to levodopa, the drug used to remedy Parkinsonism disease. The exact cause of MSA is still unknown, and exploring a therapeutic solution to MSA remains critical. A transgenic mouse model was established to study the feasibility of human adipose-derived stem cell (ADSC) therapy in vivo. The human ADSCs were transplanted into the striatum of transgenic mice via intracerebral injection. As compared with sham control, we reported significantly enhanced rotarod performance of transgenic mice treated with ADSC at an effective dose, 2 × 105 ADSCs/mouse. Our ex vivo feasibility study supported that intracerebral transplantation of ADSC might alleviate striatal degeneration in MSA transgenic mouse model by improving the nigrostriatal pathway for dopamine, activating autophagy for α-synuclein clearance, decreasing inflammatory signal, and further cell apoptosis, improving myelination and cell survival at caudate-putamen.
Introduction
Clinically, multiple system atrophy (MSA) includes a variable combination of dysautonomia, parkinsonian, and cerebellar symptoms. Two main forms of MSA are recognized: MSA-P (parkinsonian form, formerly known as striatonigral degeneration) and MSA-C (cerebellar form, formerly known as olivopontocerebellar atrophy) 1 –3 . The prevalence of MSA is reported as 1.9 to 4.4 cases per 100,000 habitants, and the median life expectancy after disease onset is less than 9 years 4 . No treatment is currently available to delay disease progression. Among all the MSA patients, around 80% of the patients will eventually develop parkinsonian features 5,6 .
MSA-P (parkinsonian form) represents a symptom of lacking dopamine. Clinically, levodopa is the first-line treatment for MSA, but dopaminergic responsiveness is poor and transitory, providing only 20% to–30% improvement of symptoms over 2 to 3 years 6 . Due to the degeneration of both striatum and substantia nigra, providing levodopa is insufficient; more researches are required to better understand this rare disease and, more importantly, explore effective therapeutic options for MSA patients.
The pathogenesis of MSA, including α-synuclein aggregation 7 , neural inflammation 8 , mitochondrial dysfunction, and oxidative stress, as well as altering the regulation of cell death and trophic support, was reported 7,9 –11 . Among these pathogeneses, α-synuclein is a key protein in MSA neuropathology. Several factors, such as oxidative stress and post-translational modifications, can induce α-synuclein aggregation 12,13 . The accumulation of α-synuclein, an important feature of MSA, was not only observed in neurons but also in oligodendrocytes. The presence of α-synuclein glial cytoplasmic inclusions (GCIs) is required to make MSA diagnosis 14 . It has also been reported that α-synuclein is involved in regulating cellular functions. Extracellular signal-regulated kinase (ERK) phosphorylation can be inhibited by overexpression of α-synuclein 15 , and the inhibition gave a signal to induce dopamine neuron cell death 16 . From another point of view, both microglia and astrocytes have been shown to interact with α-synuclein 17 . Upon activating microglia and astrocytes, inflammatory cytokines and chemokines were produced, leading to the maintenance and enhancement of the inflammatory condition 18 . It was reported that activation of microgliosis could help eliminate deleterious debris and might produce neurotrophic factors to protect neurons 19 .
Recently, the feasibility of stem cell therapy for neural degenerative disease has been widely explored. Among the studied stem cells, with neuronal linage differentiation ability and neurotrophic factor secretion characteristics, mesenchymal stem cells (MSCs) were reported to inhibit apoptosis 20 as well as promote neurogenesis 21,22 , glial scar formation, immunomodulation, angiogenesis, and neuronal and glial cell survival in the central nervous system 8,20,23 . It has also been reported that MSCs showed highly therapeutic potential in tissue injuries caused by oxidative stress or in pathologies linked to the shortening and premature aging of telomere induced by stress 24 . These features suggest that MSCs have the potential for clinical application.
MSCs, traditionally reported to be derived from the bone marrow 25 , can be derived from various tissues, including adipose tissues 26,27 , umbilical cords 28 , and dental tissues 29 . Among these sources, adipose tissue is ubiquitous and adipose-derived stem cells (ADSCs) can be acquired by a minimal invasive procedure 30 .
Research studies were conducted to investigate α-synuclein aggregation, a key pathogenesis of MSA 7,31 . The α-synuclein is a presynaptic protein involved in vesicular and synaptic transport of neurons. Since its origin and the mechanism to form aggregation or GCI in oligodendrocytes are not fully clear 32 –34 , research efforts to clear aggregations are under investigation. A lysosomal pathway involving the formation of autophagosomes could degrade α-synuclein aggregates 35 . LC3-immunoreactivity and BECN1 binding protein were found to be associated with α-synuclein GCI 36,37 , the pathological hallmark of MSA 7,38,39 . A failure of the autophagic pathway or impaired autophagy maturation was linked to MSA 40,41 . An autophagy-enhancing strategy could support therapeutic efficacy for MSA 42 .
Abnormal aggregation of α-synuclein has been strongly linked to the neurodegeneration observed in MSA 43 –45 . Myelin-based protein (MBP) transgenic mice, generated by the University of California in San Diego, USA (UCSD), were prepared by applying the MBP promoter to express exceeding α-synuclein in oligodendroglia 9,46 . In 2008, the MBP mouse model was used to investigate the ability of the antibiotic rifampicin in reducing α-synuclein aggregation and the associated neurodegeneration 31 . A few years later, MBP mice were used to evaluate MSC for hypoxia-ischemia due to its effect in forming new neurons 47,48 . Accordingly, the MBP mouse model could be useful for investigating α-synuclein GCI in oligodendrocytes and evaluating ADSC as a therapeutic option for MSA.
The dose response of ADSC transplantation and a possible mechanism for ADSC therapy were explored in this study using the MBP1 transgenic mouse model. To date, there are few clinical trials for MSA associated with cell therapy; our study was expected to provide a new reference on the effective dose of ADSC for future MSA clinical trials.
Materials and Methods
Establishing Animal Model
MBP1 transgenic mice, developed by Dr Masliah of UCSD USA, showed overexpression of human α-synuclein driven by oligodendrocyte-specific promoter MBP. Human α-synuclein mRNA was reported to express in the white matter tracts, cerebellum, brainstem, and the frontal cortex. Dr Masliah and his team have confirmed the GCIs in the transgenic mice similar to that in MSA patients and the human α-synuclein is oligodendroglial originated 9 . MBP Line 1 (MBP1) mice were transferred to Taiwan, bred with B6D2F1, and maintained at National Animal Research Laboratory (NARL) in Taiwan. B6D2F1 mice, the background mice for MBP1 mice, were supplied by BioLASCO Taiwan Co. Ltd. and served as a normal group. Prior to experiments, all tested mice were housed at the animal facilities at National Dong Hwa University. All of the animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of National Dong Hwa University (approved number: 106007) and conducted in accordance with the animal experiment guidelines of National Dong Hwa University. MBP1 mice at 12-week old with body weight 20 to 25 g were used for our study.
The level of neurodegeneration was assessed using latency to fall from the rod. MBP1 mice treated with levodopa were used to verify the MSA-P animal model.
Immunofluorescent (IF) analysis was used to examine the colocalization of α-synuclein aggregation in the striatum. Brain tissues were collected and fixed with 100% paraformaldehyde for 14 days. The tissues were then processed into paraffin-embedded samples. Coronal slides with 4 µm thickness were permeabilized with 0.3% Triton-X100 in phosphate-buffered saline plus 0.1% TWEEN-20 (PBST) for 30 min, washed with PBST, and incubated for 120 min with 1% fetal bovine serum (FBS) in PBST at room temperature for 120 min. These specimens were then incubated with a mix containing α-synuclein (human) monoclonal antibody (15G7) (ALX-804-258-LC05 supplied by ENZO, Farmingdale, NY, USA; 1:100) and human mitochondria (MAB1273 supplied by Merck; 1:100) or Olig2 (Anti-Olig2 supplied by Merck Millipore; 1:200) in PBST containing 1% bovine serum albumin (BSA) overnight at 4°C. Histofine Mouse stain kit supplied by NICHIREI Biosciences Inc. applied to as primary antibody was hosted from the mouse. Sections were then rinsed three times in PBST and incubated for 4 h with Alexa Fluor 488, 555, or 594 supplied by Invitrogen (Waltham, MA, USA; 1:100) and Hoechst33342 (1:100; Invitrogen) simultaneously to label the positions of cell nuclei. Fluorescent-stained sections were analyzed on microscopy at magnifications of 400.
Investigating the Effect of ADSC
Human ADSCs, provided by Taiwan Mitochondrion Applied Technology Co., Ltd., Hsin-Chu, Taiwan, were cultured and expanded in a flask seeded with 5000/cm2 cell density. Medium was changed every two days. The medium used for ADSC culture was Keratinocyte-SFM (KSFM, supplied by Life Technology, Carlsbad, CA, USA, 500 ml) medium containing 5% HyClone FBS defined (Thermo Fisher, Waltham, MA, USA), bovine pituitary extract (Life Technology), epidermal growth factor (Life Technology), 2.5 mM N-acetyl-
Cell transplantation was carried out via intracerebral (IC) injection using a stereotaxic apparatus. Three cell dose levels (2 × 104, 2 × 105, 2 × 106 ADSCs/mouse) were evaluated to study the efficacy of ADSC. For each mouse in the treatment group, 10 µl of ADSCs of a tested cell concentration was delivered into the striatum in each side of the brain. The injection points were located at anteroposterior (AP) −0.7, mid-lateral (ML) ±2.75, dorsoventral (DV) −2.5 (2.75 mm left/right away from the center, 3 mm down then up 0.5 mm to 2.5 mm deep) For sham control, the same protocol was followed except replacing ADSC with PBS.
Post-IC injection accelerated rotarod behavior tests were conducted on a weekly basis to assess the effect of ADSC transplantation on motor coordination. For each test run, a tested mouse was put on a 35 mm diameter rod set with an initial rod speed at 5 rpm, a final rod speed at 15 rpm, 60-s ramp-up time from initial speed to final speed, and forward rolling run for a total of 200 s. Latency to fall from a rod was recorded. For each mouse, 6 to 7 observations were made, and repeated measures were collected.
All statistical analyses were carried out using R. To account for the reliability of an estimate of the mean, our results were presented as the mean ± SE of the mean. A P value <0.05 was considered statistically significant. Before statistical analysis, if the relative SE higher than 25%, data were processed by replacing outliers with the average of all measurements within a group or among groups. One-way analysis of variance (ANOVA) with blocks was examined to confirm tested mice were the same at week 0. One-way ANOVA was examined to compare the rotarod measurement among different groups. When the null hypothesis (all means are the same) was rejected, Tukey–Kramer test was also checked to compare different pairs of means to see which groups are significantly different from each other.
Studying the Mechanism
To elucidate the role of ADSCs in alleviating neurodegeneration of MSA mice, we sacrificed tested mice at the end of the 4-week test period. Their brains were removed and fixed in 35% formaldehyde solution (Sigma-Aldrich USA) for 14 days. Paraffin-embedded tissues and coronal sections of the striatum were prepared for immunohistochemical (IHC) and IF analyses. Mice brains planned for Western immunoblotting were immediately stored at –80°C freezer before separating the striatum for protein sample preparation.
We analyzed the expression of D1 receptor protein and antidopamine-regulated and cyclic adenosine monophosphate (cAMP)-regulated phosphoprotein (DARPP32) to study the change of dopaminoceptive neurons at striatum. Anti-glial cell-derived neurotrophic factor (GDNF) expression was used to verify if ADSC could secrete neurotrophic factors and support the repair of neurons. GDNF levels were measured following test protocols provided by GDNF ELISA kits (ab100525 and ab171178, supplied by Abcam USA). This provided a quantitative comparison to further examine the effect of ADSC on GDNF secretion. Glial fibrillary acidic protein (GFAP) and Iba-1 (allograft inflammantory factor 1, AIF-1, also known as Iba-1) were used to mark astrocytes and microglia, respectively. Anti-tumor necrosis factor-alpha (TNF-α) expression was used to evaluate neuroinflammation. IHC staining was used to detect D1, DARPP32, GDNF, and TNF-α, GFAP, Iba-1 in the striatum. Coronal section samples were fixed by heating for 1 h and rehydrated by xylene and alcohol. Antigen was recovered in the pressure cooker (high pressure and temperature) for 15 min. Samples were treated with hydrogen peroxide for 5 min and blocked with 2% BSA for 1 h at room temperature. All antibodies were diluted in 2% BSA and incubated overnight at 4°C. After washing with PBS, samples were incubated with Biolinhylated immunoglobulins for 20 min and streptavidin peroxidase for 20 min at room temperature. Staining was presented by a 3,3′-diaminobenzidine solution. The primary antibodies and secondary antibodies used in IHC analyses were listed in Table 1.
Primary Antibodies and Corresponding Secondary Antibodies Used for IHC/IF Analyses.
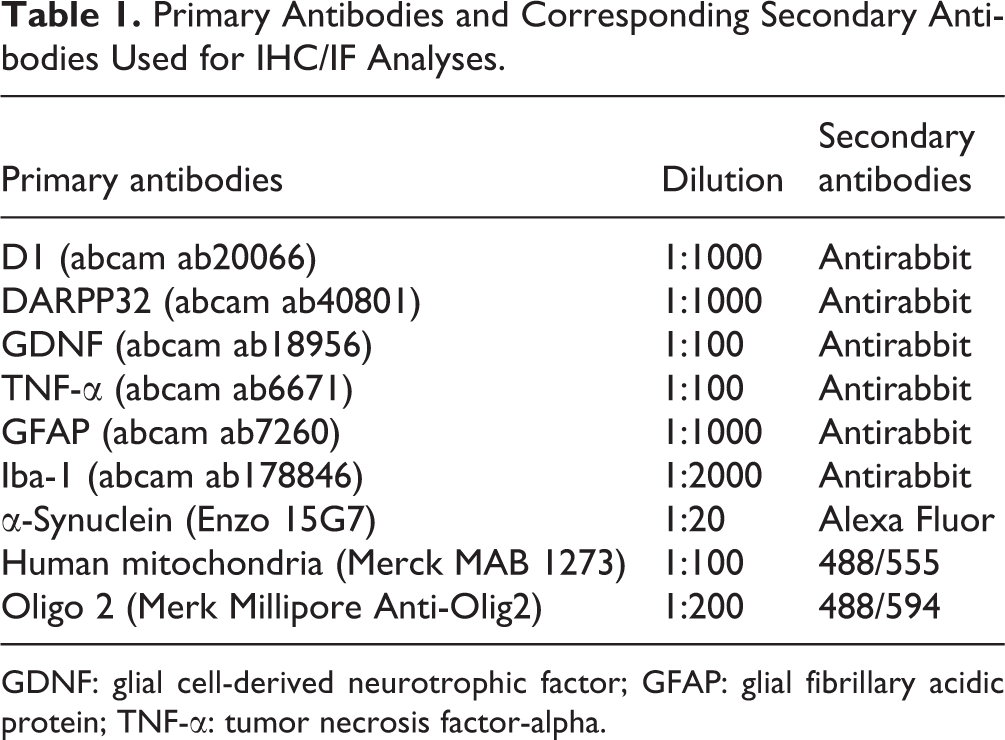
GDNF: glial cell-derived neurotrophic factor; GFAP: glial fibrillary acidic protein; TNF-α: tumor necrosis factor-alpha.
To prepare protein samples for Western immunoblotting, the striatum tissue was separated from each brain and lysed in a protein extraction buffer containing PRO-PRER Protein Extraction Solution (iNtRON Biotechnology, Seongnam, Gyeonggi, South Korea), EDTA (Sigma-Aldrich), and phosphatase inhibitor kit (524629, EMD-Millipore, Burlington, MA, USA). Tissue was put on ice while being homogenized for 5 min. It was then sat on ice for 20-min extraction and followed by centrifuging under 13,000 rpm for 15 min using MPW 150-R set at 4°C. Supernate was collected as a protein sample and kept at −80°C freezer for storage or −20°C freezer for immediate use. Bradford protein assay with BSA standard was used to determine protein concentration using the Multiskan Go spectrophotometer (Thermo Scientific, Waltham, MA, USA). Electrophoresis was performed by loading 20 to 90 µg of protein into each well to run through a 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel. Protein was then transferred from gel to polyvinylidene difluoride membrane under 400 mA for 90 min. Upon the completion of the transfer, the membrane was washed using PBST, blocked with 5% skim milk in PBST for 1 h, incubated in primary antibodies (Table 2) at room temperature for 1 h or 4°C for overnight, washed using PBST, and incubated in appropriate secondary antibodies (Table 2) at room temperature for 2 h. Protein bands were detected using horseradish peroxide (HRP) on Invitrogen (Thermo Fisher) at 595 nm.
Primary Antibodies and Corresponding Secondary Antibodies Used for Western Immunoblotting Analyses.

ERK: extracellular signal-regulated kinase; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; GDNF: glial cell-derived neurotrophic factor; HRP: horseradish peroxidase; PARP: poly (ADP-ribose) polymerase; TNF-α: tumor necrosis factor-alpha.
Results
MSA Symptoms Revealed by the Over-expression of α-Synuclein Driven by MBP Promotor
In our established transgenic mouse model for MSA, the over-expression of α-synuclein in the transgenic mouse was confirmed at both gene level (Fig. 1A) and protein level (Fig. 1B). The brown stains in the IHC analysis indicated α-synuclein at caudate-putamen (Fig. 1C). Glia, labeled by Olig2, and α-synuclein-positive GCIs was found to colocalize at caudate-putamen, as revealed by IF analysis (Fig. 1D). In addition to this key criterion for definite MSA diagnosis, we also examined the neurodegenerative change in striatonigral structure. As the rotarod performance shown in Fig. 1E, the motor function of untreated transgenic mice was degenerated. Treating levodopa was found unable to alter this change.

Establishing 12-week old MSA transgenic (Tg) mouse model. (A) Human α-synuclein detected in 12-week old Tg mouse using PCR. (B) Human α-syn protein detected in 12-week old Tg mouse using Western blotting. (C) Typical accumulation of α-synuclein detected at caudate-putamen of 12-week old Tg mouse using immunohistochemistry. (D) Typical co-localization of α-synuclein accumulation detected at oligodendrocytes in caudate-putamen of 12-week old Tg mouse using immunofluorescent analysis. (E) Behavior performance of 12-week old B6D2F1 (normal mice) and 12-week old untreated MBP1 (sham mice) using the rotarod test. All measurements were repeated in four separate biological replicates, and statistical analysis on mean values (± SEM) was reported. *P < 0.05, **P < 0.01, ***P < 0.001.
Beneficial Effects on the MSA Transgenic Mouse Model Revealed by ADSC Transplantation
To explore the potential of ADSC therapy for MSA, we conducted a feasibility study using the established transgenic mouse model for MSA. We treated 12-week old MSA transgenic mice with human ADSC at three dose levels. Rotarod tests were performed before and after cell transplantation to monitor the change in motor function. Rotarod behavior was measured weekly and analyzed statistically. In Fig. 2, we revealed that the mice treated with ADSC at a medium level or high level had significant improvement in rotarod performance as compared with the untreated mice, while little improvement was seen in the mice treated with ADSC at a low level. Moreover, our statistical analysis also indicated that the beneficial effect of ADSC was not significant by increasing the ADSC dose from the medium level to high level. Considering dose dependence, the effective dose of ADSC for efficacy study was then determined to be the medium level (2 × 105 cells). Every tested transgenic mouse in the ADSC group was treated with 1 × 105 cells on each side of the striatum via IC. Latency to fall from rod was recorded on a weekly basis, and the comparison among all groups (normal, ADSC, and sham) was reported with statistical significance, as shown in Fig. 3. Our in vivo study clearly suggested that ADSC at an effective dose could restore the rotarod behavior to a normal-like level. The normal-like performance sustained well during our 4-week test period.

Determining the effective dose of ADSC using rotarod tests. Latency to fall was measured weekly after IC injection. Compare three dose levels of ADSC. All measurements were repeated in four separate biological replicates, and statistical analysis on mean values (± SEM) was reported. *P < 0.05, **P < 0.01, ***P < 0.001. ADSC: adipose-derived stem cell; IC: intracerebral.
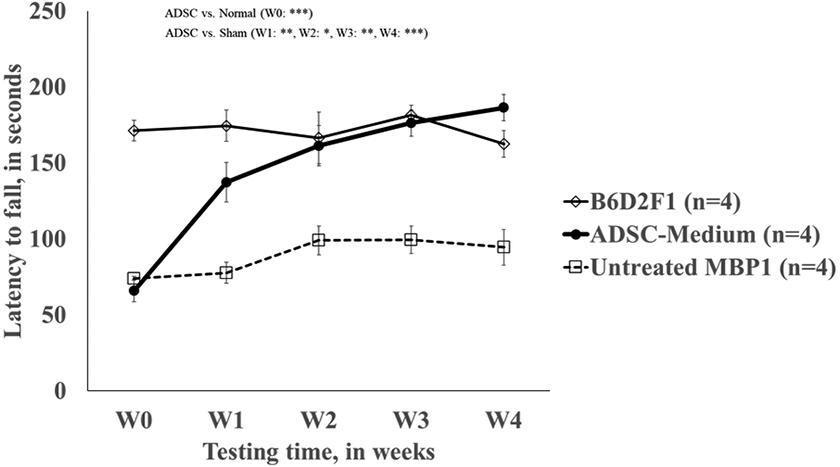
Evaluating the therapeutic effect of ADSC using rotarod tests. Latency to fall was measured weekly after IC injection. Compare 12-week old untreated MBP1 (sham mice, n = 4), 12-week old ADSC-treated MBP1 (n = 4), and 12-week old B6D2F1 (normal, n = 4). All measurements were repeated in four separate biological replicates, and statistical analysis on mean values (± SEM) was reported. *P < 0.05; **P < 0.01, ***P < 0.001. ADSC: adipose-derived stem cell; IC: intracerebral.
Nigrostriatal Pathway for Dopamine Signal Improved by ADSC Transplantation
In the brain, dopamine was produced in the substantia nigra and transported toward the striatum to form the nigrostriatal pathway. This pathway is responsible for the control of movement, as part of a system called basal ganglia motor loop. In MSA, due to striatonigral degeneration, impaired function of the nigrostriatal pathway for dopamine resulted in symptomatic motor deficits. Post-ADSC transplantation, cell tracing and migration were examined by labeling ADSC with transfected GFP. Photo images during the first week were taken on live mice to track grafted cells (Fig. 4A). At the end of the 4-week test period, the brain sample was analyzed by IF. Few ADSCs were detected in the striatum, and some ADSCs seemed to locate in the lateral ventricle (Fig. 4A). We found major recovery for the nigrostriatal pathway in the striatum post-ADSC transplantation in our animal model, resulting in a high expression of the D1 receptor protein (Fig. 4B) and DARPP-32 (Fig. 4C) in caudate-putamen.

Tracking ADSC after ADSC transplantation using photo imaging as well as immunofluorescent analysis and assessing the effect of ADSC on receptive neurons in the striatum using immunohistochemistry. (A) Check migration of ADSC on the live mouse using a photo imaging device and examine the survival of ADSC at the end of the 4-week test period. (B) Compare D1 expression at caudate-putamen of 12-week old wild type (normal mouse), 24-week old untreated MBP1 (sham mouse), and 12-week old ADSC-treated MBP1 (Tg mouse treated with ADSC). (C) Compare DARPP32 expression at caudate-putamen of 12-week old untreated MBP1 (sham mouse) and 12-week old ADSC-treated MBP1 (Tg mouse treated with ADSC). ADSC: adipose-derived stem cell.
Potential of ADSC Transplantation in Activating Autophagy Protein Clearance Pathway to Decrease the Levels of Unaggregated Form of α-Synuclein
Upon the completion of the 4-week test period after cell transplantation, tested mice were sacrificed for ex vivo study. Mice brains were sectioned coronally and analyzed for α-synuclein expression in the striatum using IF. The α-synuclein expression of the ADSC-treated mouse showed reduced aggregation and fewer counts in the IF analysis (Fig. 5A). Furthermore, the Western immunoblots in Fig. 5B suggested that the soluble α-synuclein was highly reduced, and the quantitative analysis also supported a reduction in insoluble α-synuclein. To possibly connect the dots between the reduction of α-synuclein and autophagy protein clearance pathway, the expression of BECN1 and LC3B-II was checked and confirmed to increase after ADSC transplantation, as shown in Fig. 6.

Examining the effect of ADSC in reducing α-synuclein in the striatum of 12-week old mice using immunofluorescence and Western immunoblot. (A) Immunofluorescent stains of α-synuclein and Hoechst at caudate putamen at the end of the 4-week test period after ADSC transplantation. (B) Western blots of soluble α-synuclein and insoluble α-synuclein in the striatum at the end of the 4-week test period after ADSC transplantation. ADSC: adipose-derived stem cell.
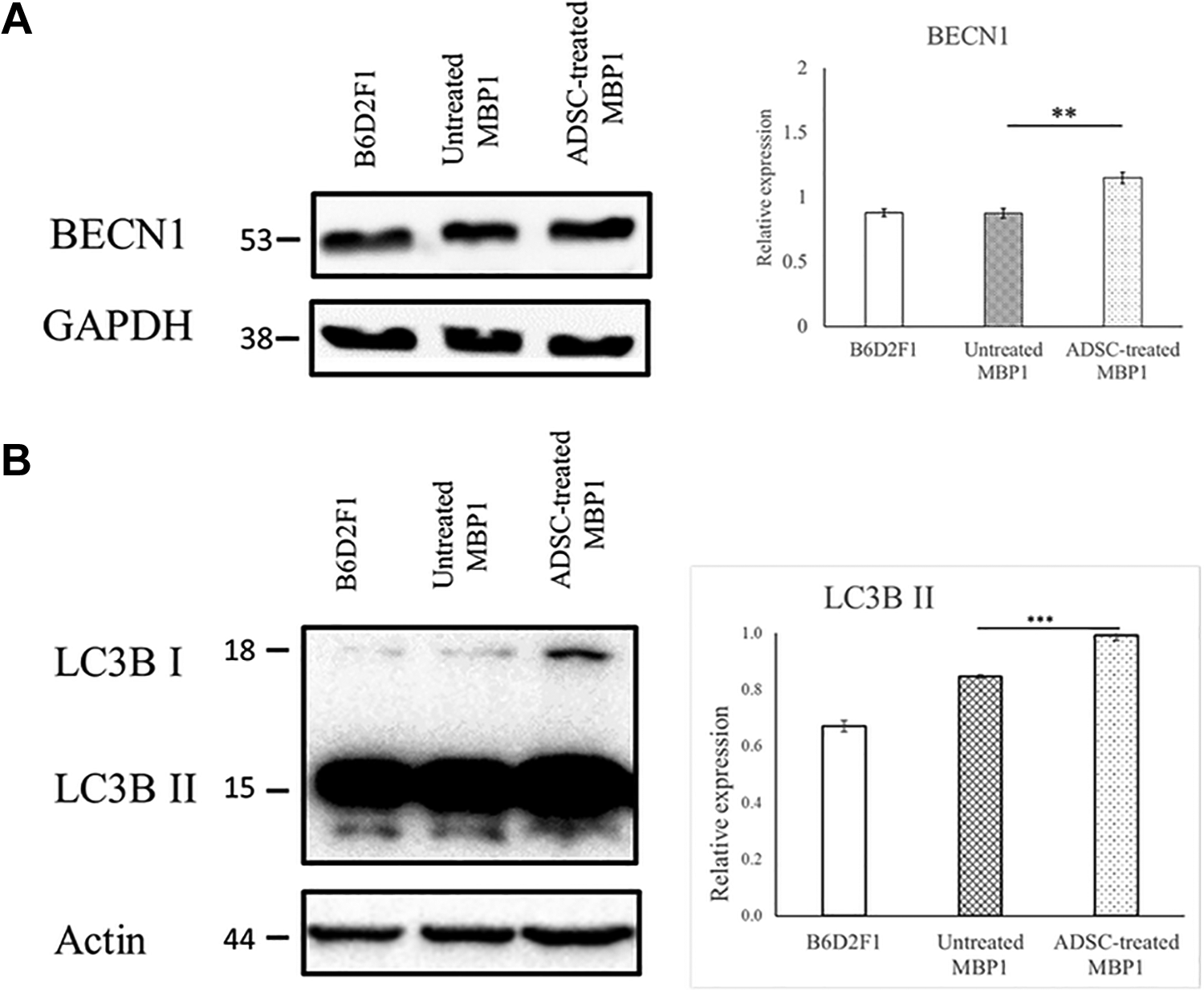
Examining autophagy pathway-related proteins to explore the effect of ADSC on a 12-week old mouse. (A) Western immunoblots of BECN1 for B6D2F1 (normal mouse), untreated MBP1 (sham mouse), and ADSC-treated MBP1 (mouse treated with ADSC) at the end of the 4-week test period after ADSC transplantation. (B) Western immunoblots of LC3B for B6D2F1 (normal mouse), untreated MBP1 (sham mouse), and ADSC-treated MBP1 (mouse treated with ADSC) at the end of the 4-week test period after ADSC transplantation. ADSC: adipose-derived stem cell.
Potential of ADSC Transplantation in Decreasing Inflammatory Signal and Further Cell Apoptosis
The IHC analysis in striatum indicated an increase of GDNF (Fig. 7A) and a reduction of TNF-α (Fig. 7B) after ADSC transplantation. GFAP positive and Iba-1 positive were stronger after ADSC treatment (Fig. 7C). The expression of ERK, a protein associated with the GDNF pathway to support cell survival, was also found to increase after ADSC transplantation, as shown in Fig. 8. In Fig. 9, the expression of apoptosis proteins, including poly (ADP-ribose) polymerase (PARP) and caspase 3, was decreased for ADSC-treated mouse.

Assessing pathological improvement using immunohistochemistry and Western immunoblotting. (A) GDNF detected at striatum at the end of the 4-week test period after ADSC transplantation using immunohistochemical stains, Western blots, and ELISA GDNF kits. (B) TNF-α detected at striatum at the end of the 4-week test period after ADSC transplantation using immunohistochemical stains and Western blots. (C) Iba-1 and GFAP were detected at striatum at the end of the 4-week test period after ADSC transplantation using immunohistochemistry stains. ADSC: adipose-derived stem cell; GDNF: glial cell-derived neurotrophic factor; GFAP: glial fibrillary acidic protein; TNF-α: tumor necrosis factor-alpha.

Studying a protein associated with the GDNF pathway to explore the possible effect of ADSC. Western immunoblots of p-ERK1/2 and total ERK for B6D2F1 (normal mouse), untreated MBP1 (sham mouse), and ADSC-treated MBP1 (mouse treated with ADSC) at the end of the 4-week test period after ADSC transplantation. ADSC: adipose-derived stem cell; ERK: extracellular signal-regulated kinase; GDNF: glial cell-derived neurotrophic factor.

Studying proteins related to the apoptosis pathway to explore the possible effect of ADSC. Western immunoblots of PARP and cleaved Caspase 3 for B6D2F1 (normal mouse), untreated MBP1 (sham mouse), and ADSC-treated MBP1 (mouse treated with ADSC) at the end of the 4-week test period after ADSC transplantation. ADSC: adipose-derived stem cell; PARP: poly(ADP-ribose) polymerase.
Discussion
Our animal behavior test result demonstrated that cell therapy with 2 × 105 ADSCs/mouse dose level could significantly recover the motor coordination in the transgenic mouse model for MSA in vivo. Based on the rotarod performance, the restoration occurred instantly, in the first week after cell transplantation. The recovery was sustained for 4 weeks. Cell tracing indicated that few ADSCs remained detectable in the striatum and some in the lateral ventricle at the end of the 4-week test period after ADSC transplantation. Activated microglia and astrocytes, often found in neurodegenerative diseases like Parkinson’s and Alzheimer’s, were confirmed in our model for MSA. Activation of microgliosis after ADSC treatment might have played a role to contribute to the release of GDNF and resulting in better cell survival. Considering the GDNF expression was boosted up to normal range at the end of the 4-week test period, as shown in the ELISA result in Fig. 7A, we expect the benefit of ADSC would last for at least 4 weeks. These findings make us confident to believe that the ADSC level in our test was effective for cell therapy to treat MSA mouse. The effective dose, as low as 2 × 105 ADSCs/mouse, might provide a safer reference for the future clinical trial of ADSC for MSA patients.
At the end of the 4-week test period, based on the morphology check using IF, we detected low α-synuclein accumulation in ADSC-treated mice ex vivo. A further analysis of α-synuclein using immunoblots showed that both soluble α-synuclein and insoluble α-synuclein were reduced after ADSC treatment. These results suggested that ADSC transplantation has a potential to clear α-synuclein GCI in the striatum by reducing α-synuclein in the form of soluble α-synuclein oligomers albeit low insoluble α-synuclein aggregates. Soluble α-synuclein oligomers were reported to be more toxic as compared with insoluble α-synuclein aggregates 49,50 . The reduced level of soluble α-synuclein oligomers after ADSC treatment could have alleviated neurodegeneration in striatonigral structure and possibly restored motor function of striatal neurons. To reveal the role of ADSC in reducing α-synuclein GCI, we examined the autophagy pathway and microgliosis. Autophagy pathway-related proteins, such as BECN1 and LC3B, were used to indicate the activation of autophagy 42,51 . High expression of BECN1 and LC3B-II detected in ADSC-treated mice ex vivo suggested that transplanted ADSC might have an effect to initiate autophagy and elongate phagophore. This result provided a possible mechanism for ADSC to clear α-synuclein CGI in the form of MSA via the autophagy pathway.
Apoptosis promoted by neuroinflammation could lead to cell death of receptive neurons in the striatum. As reported by Stefanova et al in 2007, neuroinflammation induced by α-synuclein inclusions could contribute to the degeneration of midbrain dopaminergic neurons 52 . After ADSC treatment, we detected reduced TNF-α, a proinflammatory cytokine. We also found the low activity of Caspase 3 and low expression of PARP in ADSC-treated mice, indicating apoptosis was not promoted. The reduced neuroinflammation might have alleviated neurodegeneration while ADSC clearing α-synuclein GCI in the striatum.
Tassigny et al reported in 2015 that the GDNF pathway could support neuronal survival by upregulating ERK 53 . The high expression of GDNF, ERK, D1, and DARPP32 after ADSC transplantation found in our ex vivo study suggested the GDNF secreted from ADSC might have improved the survival of dopaminoceptive cells and medium spiny neurons while ADSC clearing α-synuclein GCI in the striatum. In the same report, Tassigny et al proposed in 2015, GDNF was involved in cell apoptosis by regulating the activity of Caspase 3. GDNF released by activating microgliosis after ADSC treatment could have inhibited apoptosis by downregulating the activity of Caspase 3.
In the treatment of Parkinson’s disease using MSC reported that MSC secretome could protect the neurons and increases cell survival and neurogenesis 54 . The high expression of GDNF, low activity of Caspase 3, and low expression of PARP detected in ADSC-treated mice suggested that GDNF secreted from the transplanted ADSC might have an effect to supplement glial-derived neurotrophic support for myelination of striatal neurons, resulting in reduced cell apoptosis for MSA.
There have been debating in adult neurogenesis in brains 55 –57 . Regeneration of neurons in brains, however, was expected only in the area where a bundle of neuronal stem cells located, such as the hippocampus or lateral ventricle 58 . In our study, the ADSC was delivered into caudate-putamen, a location very near to the lateral ventricle. At the end of the 4-week treatment, some ADSCs were found in the lateral ventricle. It should be interesting to better understand if the ADSC transplanted in caudate-putamen has an effect on activating stem cells in the lateral ventricle and promoting the neurogenesis in the dopamine pathway. Future work will be required to fully characterize the role of ADSCs in the regeneration of striatal neurons. Accordingly, we proposed a possible model outlining the effects of ADSCs in treating MSA in Fig. 10.
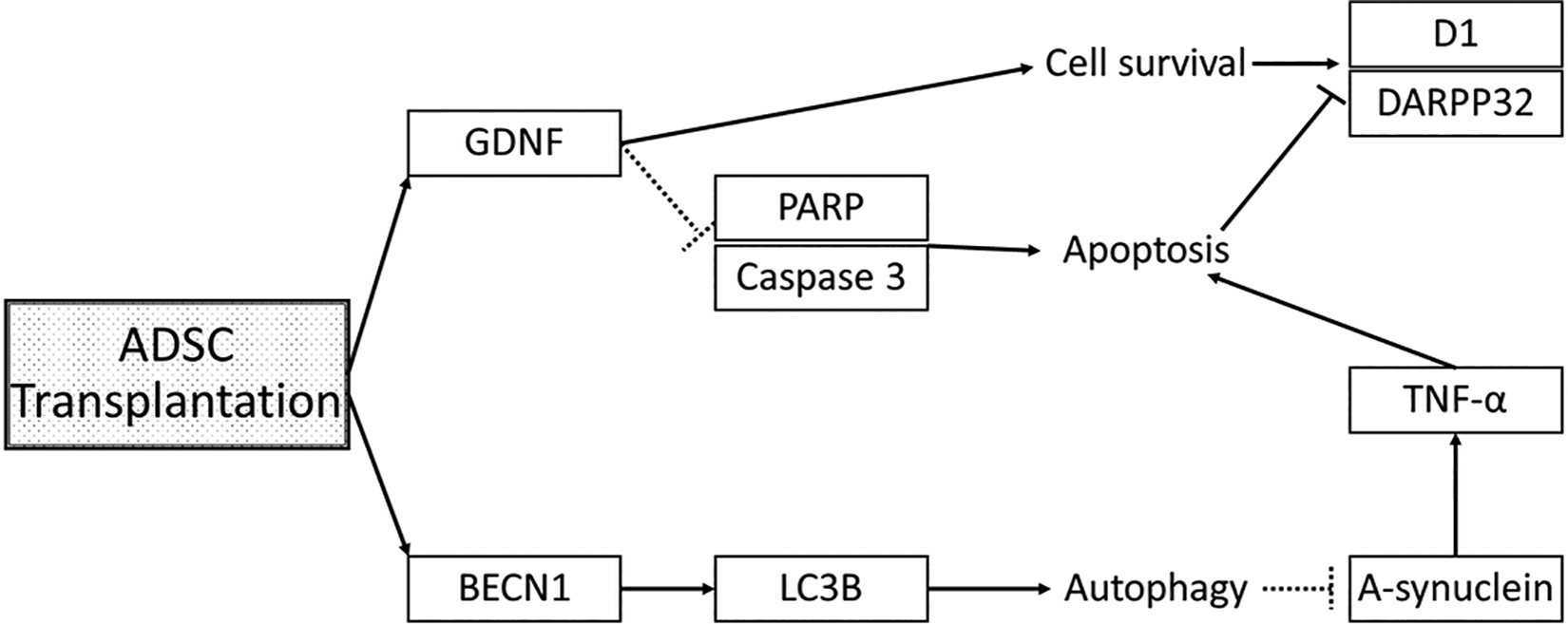
Proposing a possible model for adipose-derived stem cells in treating multiple system atrophy.
Conclusions
In summary, our work represents the first successful human ADSC feasibility study in alleviating the neurodegeneration in a transgenic mouse model for MSA. The graft changed disease progression through the survival of D1 and DARPP32. The benefits observed in these mice, such as improved rotarod performance, have sustained for 4 weeks. Our work concludes 2 × 105 ADSCs/mouse, which is an effective dose to restore the motor function of dopaminoceptive neurons and striatal medium spiny neurons possibly by improving the nigrostriatal pathway for dopamine, activating autophagy to clear α-synuclein GCI in the form of MSA, decreasing inflammatory signal and further cell apoptosis, and activating microgliosis to protect myelination and cell survival. Our work provides a new reference for future MSA clinical trials.
Footnotes
Acknowledgments
We acknowledge Dr Masliah of UCSD for the contribution in providing two pairs of MBP1 mice for this study. We thank the National Laboratory Animal Center (NLAC), NARLabs, Taiwan, for technical support in repository services. We thank Taiwan Mitochondrion Applied Technology Co., Ltd., for providing human ADSCs.
Ethical Approval
The source of the human ADSCs and its use in this study was approved by China Medical University & Hospital Research Ethics Committee (IRB approval number: CMUH104-REC1-007). The design and execution of animal experiments was approved by the IACUC 106007.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the animal experiment guidelines of National Dong Hwa University, Taiwan, and approved by the Institutional Animal Care and Use Committee (IACUC) of Dong Hwa University, Taiwan (Approval number: 106007).
Statement of Informed Consent
ADSC sample used in this article was provided by a healthy donor with written informed consent in compliance with IRB approval protocol (CMUH104-REC1-007).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
