Abstract
Osteoarthritis (OA) is a degenerative joint disease associated with inflammatory response. Tripartite motif 8 (TRIM8) is a member of TRIM family that has been found to regulate inflammation. The present study was aimed to evaluate the role of TRIM8 in OA chondrocytes. Our results showed that TRIM8 expression was significantly increased in interleukin 1 beta (IL-1β)-stimulated OA chondrocytes. To knock down the TRIM8 expression in chondrocytes, the chondrocytes were transfected with si-TRIM8. Knockdown of TRIM8 attenuated IL-1β-induced production of inflammatory mediators including nitric oxide and prostaglandin E2. The increased expression levels of inducible nitric oxide synthase and cyclooxygenase-2 in IL-1β-induced chondrocytes were suppressed by TRIM8 knockdown. The IL-1β-induced production of proinflammatory cytokines including TNF-α and IL-6 was significantly decreased after transfection with si-TRIM8. Besides, knockdown of TRIM8 mitigated the IL-1β-induced decrease in aggrecan and collagen-II proteins expression and increase in matrix-degrading enzymes in chondrocytes. Furthermore, TRIM8 knockdown prevented IL-1β-induced nuclear factor kappa B (NF-κB) activation in chondrocytes. Taken together, these findings indicated that knockdown of TRIM8 attenuates IL-1β-induced inflammatory response in OA chondrocytes through the inactivation of NF-κB pathway. Thus, targeting TRIM8 might provide therapeutic treatment for OA.
Introduction
Osteoarthritis (OA) is a degenerative joint disease characterized by articular cartilage degradation, subchondral bone sclerosis, and osteophyte formation 1,2 . OA is considered as the most common form of arthritis and remains one of the leading causes of impaired mobility among the elderly population 3 . Many of the cellular and molecular changes that occur in OA indicate that inflammation is an important contributing factor to the development of OA 4 . In addition to synovial cells, articular chondrocytes are also known to contribute to the progression of OA due to the expression of inflammatory mediators and activation of inflammatory pathways 5,6 . Thus, targeting inflammatory pathways could be a promising strategy for the treatment of OA.
There have been many reports of crosstalk between OA and inflammatory pathways 7 . Nuclear factor kappa B (NF-κB) transcription factor family includes homo- and heterodimers of p50 (p105/NF-κB1), p52 (p100/NF-κB2), p65 (RelA), RelB, and c-Rel proteins 8 . These dimers are quiescently sequestered by the inhibitory κB (IκB) proteins in the cytoplasm. Upon stimulation, inflammatory cytokines and factors lead to the phosphorylation of IκBα, which is an important event that results in its subsequent ubiquitination and proteasome-mediated degradation 9 . Finally, the NF-κB pathway is activated and it regulates essential cellular responses. Numerous studies have documented the activation of NF-κB pathway and aggravation of cartilage degeneration during the progression of OA 10 –12 .
Tripartite motif (TRIM) family contains multiple proteins that have various functions in cellular processes such as intracellular signaling, innate immunity, and carcinogenesis 13 . Recent studies have revealed that TRIM family is involved in triggering the assembly of an inflammasome 14 . TRIM21 negatively regulates intestinal mucosal inflammation in patients with inflammatory bowel diseases 15 . TRIM28 bridges endothelial inflammation and angiogenic activity in endothelial cells 16 . TRIM47 is a critical regulator of cerebral ischemia-reperfusion (I/R) injury through regulating apoptosis and inflammation 17 . Tripartite motif 8 (TRIM8) is a member of TRIM protein family and plays a critical role in meditating inflammation 18 . However, the role of TRIM8 in OA remains unclear. In the present study, we investigated the effect of TRIM8 on interleukin 1 beta (IL-1β)-induced inflammatory response in chondrocytes.
Methods and Materials
Primary Human OA Chondrocyte Culture
The knee cartilages obtained from five OA patients were used for the isolation of primary OA chondrocytes using a slight modification of the specific protocol previously published 19 . Briefly, cartilage tissues were minced into small pieces and digested with 0.25% trypsin for 30 min, followed by digestion with 0.2% collagenase II on a shaking plate overnight. The cells were subjected to a sterile filter (70 μm) to separate from the undigested pieces. The obtained chondrocytes were resuspended in Dulbecco’s modified Eagle medium/F-12 supplemented with fetal bovine serum (10% v/v; Hyclone, Logan, UT, USA) and penicillin-streptomycin (1% v/v; Sigma-Aldrich, St Louis, MO, USA), and maintained at 37°C in a humidified atmosphere with 5% CO2. Only passage 1 to 2 was used in our study to avoid the phenotype loss.
Nitric oxide Measurement
Chondrocytes media was collected and centrifuged at 10,000 × g for 1 min. The concentration of nitric oxide (NO) in the supernatant was determined by Griess reagent method using a commercially available NO detection kit (Beyotime Institute of Biotechnology, Beijing, China).
Small-interfering RNA Knockdown Experiment
Small-interfering RNA (siRNA) targeting TRIM8 (si-TRIM8) and negative scrambled siRNA (si-scramble) were purchased from Applied Biosystems (Foster, CA, USA). Chondrocytes were transiently transfected with si-TRIM8 or si-scramble using Oligofectamine and OPTI-MEM medium (Invitrogen, Carlsbad, CA, USA). The protein expression levels of TRIM8 were tested to confirm silencing efficiency.
Quantitative real-time polymerase chain reaction
Chondrocytes were harvested for total RNA isolation and the determination of TRIM8, IL-1β, matrix metalloproteinase 3 (MMP-3), and MMP-13 expression. Isolated total RNA was used for the synthesis of cDNA using the Superscript First-Strand Synthesis System (Invitrogen). PCR amplification and detection were carried out using the system including 4.5 µl diluted cDNA, 0.25 µl forward primer, 0.25 µl reverse primer, and 5 µl SYBR Green Master Mix (Applied Biosystems) on an ABI7300 Real-Time Thermal Cycler (Applied Biosystems). The primers were listed: TRIM8 (F), 5′-CTGCCGTGCAAACACAACTTC-3′, and (R) 5′-TCCACGATGTTGGTGAGCTTC-3′; IL-1β (F), 5′-TGTGAAATGCCACCTTTTGA-3′, and (R) 5′-TGAGTGATACTGCCTGCCTG-3′; MMP-3 (F) 5′-TGAACAATGGACAAAGGATACAACAGG-3′, (R) 5′-ATCATCTTGAGACAGGCGGAACC-3′; MMP-13 (F) 5′-AGTGGTGGTGATGAAGATGATTTGTC-3′, (R) 5′- TCTAAGGTGTTATCGTCAAGTTTGCC-3′; TNF-α (F) 5′-ACGGCATGGATC TCAAAGAC-3′, (R) 5′-GTGGGTGAGGAGCACGTAGT-3′; IL-6 (F) 5′-CTG CAAGAGACTTCCATCCAGTT-3′, (R) 5′-GAAGTA GGG AAG GCC GTG G-3′; β-actin: forward, 5′-GCAAGCAGGAGTATGACGAG-3′, reverse, 5′-CAAATAAAGCCATGCCAATC-3′. The relative mRNA levels of indicated target genes were normalized to that of β-actin.
Western Blot
After three treatments, chondrocytes were lysed by a radio immunoprecipitation assay (RIPA buffer; Cell Signaling Technology, Boston, MA, USA). Subsequently, 30 µg of intracellular proteins was electrophoretically resolved on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis to separate the proteins with different molecular weight. Then western blotting analysis was performed as previously described 20 . The separated proteins were transferred to a polyvinylidene difluoride membrane and incubated with specific primary antibodies (TRIM8, inducible nitric oxide synthase [iNOS], cyclooxygenase-2 (COX-2), aggrecan, collagen-II, p-p65, p65, and p-IκBα) and secondary antibody (Abcam). Finally, the immunoreactive bands were detected by enhanced chemiluminescence system (Millipore, Billerica, MA, USA).
Enzyme-linked immunosorbent assay
The chondrocytes medium was collected and centrifuged at 15,000 rpm for 10 min to collect the supernatant. To test the role of TRIM8 in inflammation and extracellular matrix (ECM) degradation, the levels of prostaglandin E2 (PGE2), TNF-α, IL-6, MMP-3, MMP-13, ADAMTS-4, and ADAMTS-5 were measured using commercial ELISA kits (R&D Systems, Minneapolis, MN, USA).
Statistical Analysis
All data were expressed as mean ± SD. Statistical comparisons among different groups (more than two groups) were performed by one-way analysis of variance using SPSS version 13.0 software (SPSS Inc., Chicago, IL, USA). Statistical comparisons between two groups were carried out using Student’s t-test. Differences were considered statistically significant when P <0.05.
Results
The expression of TRIM8 was significantly increased in chondrocytes from OA patients and IL-1β-stimulated OA chondrocytes.
Initially, we examined the expression levels of TRIM8 in chondrocytes from OA patients. We found that the mRNA expression levels of TRIM8 were significantly increased in chondrocytes from OA patients as compared to normal ones (Fig. 1A). Next, we assessed the expression pattern of TRIM8 in IL-1β-stimulated OA chondrocytes. The quantitative real-time polymerase chain reaction results revealed that at the mRNA level, the TRIM8 expression was significantly increased by IL-1β in chondrocytes as compared to the control group (Fig. 1B). Similarly, IL-1β treatment also greatly upregulated the protein expression level of TRIM8 in OA chondrocytes (Fig. 1C).

Graphical presentation of the TRIM8 expression levels in chondrocytes from osteoarthritis (OA) patients and IL-1β-stimulated OA chondrocytes. (A) The mRNA levels of TRIM8 in human primary chondrocytes isolated from OA patients and normal controls were measured by qRT-PCR analysis. n =5. *P < 0.05. (B) The qRT-PCR analysis showing the expression pattern of TRIM8 in IL-1β-stimulated OA chondrocytes. *P < 0.05. (C) The western blot analysis showing the increased protein level of TRIM8 in IL-1β-stimulated OA chondrocytes. n = 4. *P < 0.05. IL-1β: interleukin 1 beta; OA: osteoarthritis; qRT-PCR: quantitative real-time polymerase chain reaction; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Suppressed the Production of NO and PGE2 in IL-1β-Induced OA Chondrocytes
The role of TRIM8 in OA was explored by investigating the effect of TRIM8 on IL-1β-induced inflammation in OA chondrocytes. Chondrocytes were transfected with si-TRIM8 orsi-scramble, followed by the confirmation of transfection efficiency. The results showed that transfection with si-TRIM8 significantly suppressed the protein expression of TRIM8 expression in OA chondrocytes under IL-1β stimulation (Fig. 2A). As shown in Fig. 2B, C, IL-1β significantly induced the production of NO and PGE2, compared to that in the control chondrocytes. Moreover, knockdown of TRIM8 suppressed the IL-1β-induced production of NO and PGE2 in OA chondrocytes.

Knockdown of TRIM8 suppressed the production of NO and PGE2 in IL-1β-induced OA chondrocytes. (A) Confirmation of transfection efficiency by western blot after transfection with si-TRIM8 or si-scramble. *P < 0.05. (B, C) Effect of TRIM8 knockdown on the production of NO and PGE2 in IL-1β-induced OA chondrocytes. n = 3. *P < 0.05. IL-1β: interleukin 1 beta; NO: nitric oxide; OA: osteoarthritis; PGE2: prostaglandin E2; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Suppressed iNOS and COX-2 Expression in IL-1β-Induced OA Chondrocytes
Further, we examined the effect of TRIM8 on the protein expression of iNOS and COX-2. Western blot analysis showed that chondrocytes transfected with si-TRIM8 exhibited significant decreases in the protein expression levels of iNOS and COX-2 induced by IL-1β (Fig. 3).

Knockdown of TRIM8 suppressed iNOS and COX-2 expression in IL-1β-induced OA chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 24 h. The protein expression levels of iNOS and COX-2 were measured by western blot analysis. n = 4. *P < 0.05. COX-2: cyclooxygenase-2; IL-1β: interleukin 1 beta; iNOS: inducible nitric oxide synthase; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Suppressed the Levels of TNF-α and IL-6 in IL-1β-Stimulated OA Chondrocytes
Furthermore, we also investigated the effect of TRIM8 on the production of TNF-α and IL-6. ELISA demonstrated that IL-1β-stimulated production of TNF-α and IL-6 in OA chondrocytes was blocked by knockdown of TRIM8 (Fig. 4A, B). In addition, we found that knockdown of TRIM8 significantly suppressed the mRNA expression levels of TNF-α and IL-6 induced by IL-1β in OA chondrocytes (Fig. 4C, D).

Knockdown of TRIM8 suppressed the levels of TNF-α and IL-6 in IL-1β-stimulated OA chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 24 h. (A and B) The levels of TNF-α and IL-6 in culture supernatant were determined by ELISA. (C and D) The mRNA expression levels of TNF-α and IL-6 were determined by quantitative real-time polymerase chain reaction. n = 6. *P < 0.05. IL-1β: interleukin 1 beta; TNF-α: tumor necrosis factor alpha; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Decreased the Levels of MMP-3 and MMP-13 in IL-1β-Stimulated Chondrocytes
Next, we examined the effect of TRIM8 on the production of MMPs including MMP-3 and MMP-13, which are important for cartilage degradation. ELISA results illustrated that IL-1β stimulated the production of MMP-3 and MMP-13 in chondrocytes, while knockdown of TRIM8 prevented the IL-1β-stimulated MMP-3 and MMP-13 production (Fig. 5A, B). Similarly, we observed that TRIM8 knockdown efficiently suppressed the IL-1β-induced MMP-3 and MMP-13 mRNA expressions in OA chondrocytes (Fig. 5C, D).
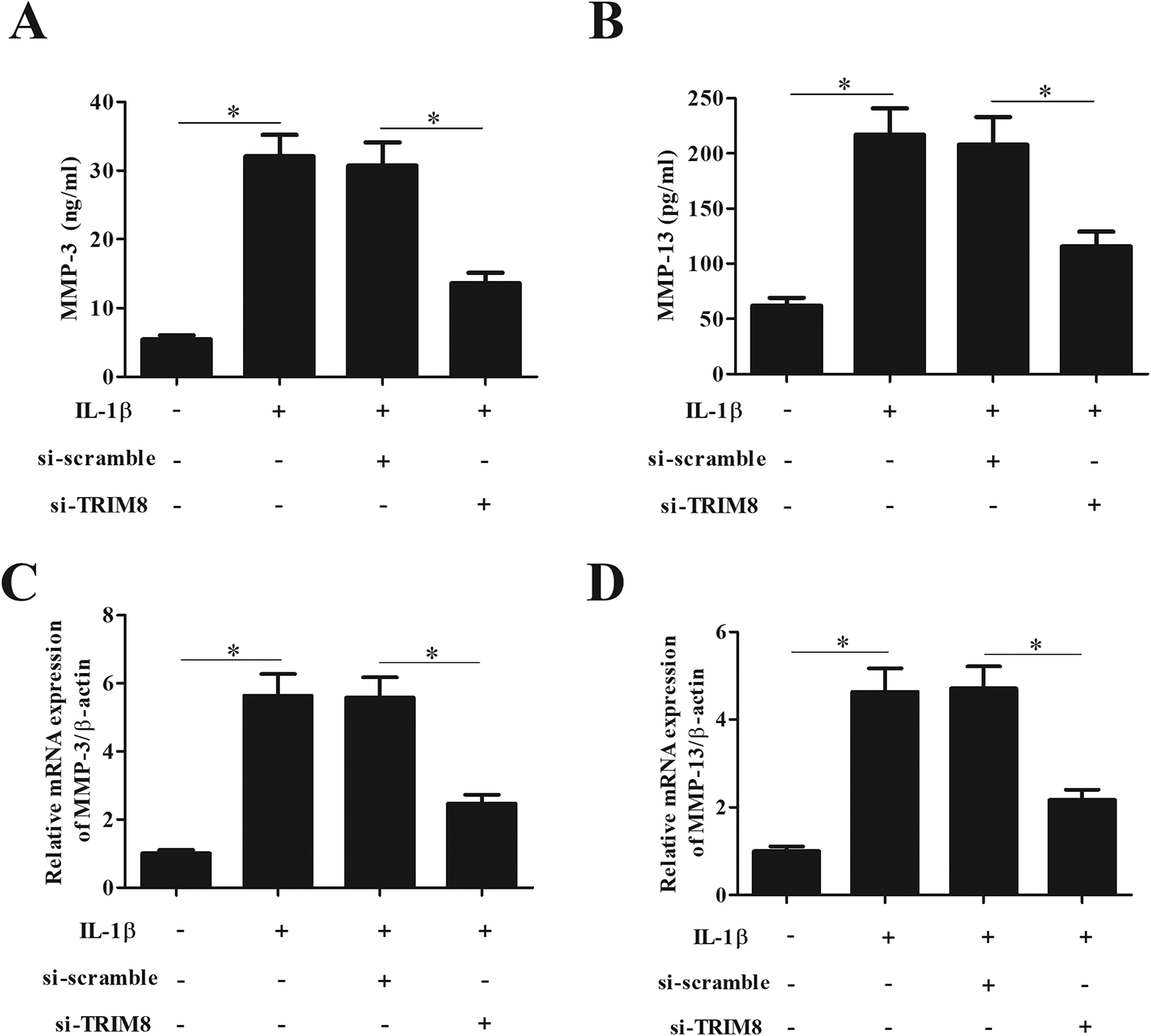
Knockdown of TRIM8 decreased the levels of MMP-3 and MMP-13 in IL-1β-stimulated chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 24 h. (A, B) The effects of TRIM8 knockdown on the production of matrix-degrading enzymes MMPs including MMP-3 and MMP-13 were examined by ELISA. (C, D) The mRNA expression levels of MMP-3 and MMP-13 were determined by quantitative real-time polymerase chain reaction. n = 5. *P < 0.05. IL-1β: interleukin 1 beta; MMP: matrix metalloproteinase; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Attenuated the Production of ADAMTS-4 and ADAMTS-5 in IL-1β-Induced Chondrocytes
The effect of TRIM8 on the other ECM degradation enzymes ADAMTS-4 and ADAMTS-5 was explored using ELISA. IL-1β stimulation caused significant increases in the expression of ADAMTS-4 and ADAMTS-5. However, knockdown of TRIM8 counteracted the IL-1β-mediated production of ADAMTS-4 and ADAMTS-5 in chondrocytes (Fig. 6A, B).

Knockdown of TRIM8 attenuated the production of ADAMTS-4 and ADAMTS-5 in IL-1β-induced chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 24 h. (A and B) The effects of TRIM8 knockdown on the production of matrix-degrading enzymes ADAMTSs including ADAMTS-4 and ADAMTS-5 were examined by ELISA. n = 5. *P < 0.05. IL-1β: interleukin 1 beta; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Increased the Expression of Aggrecan and Collagen-II in IL-1β-Induced Chondrocytes
In addition, the protein expression levels of ECM proteins including aggrecan and collagen-II were also detected using western blot assay. As indicated in Fig. 7, knockdown of TRIM8 improved ECM deposition in IL-1β-induced chondrocytes with increased protein expression levels of aggrecan and collagen-II.

Knockdown of TRIM8 increased the expression of aggrecan and collagen-II in IL-1β-induced chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 24 h. (A, B) The protein expression levels of extracellular matrix proteins including aggrecan and collagen-II were detected by western blot. n = 4. *P < 0.05. IL-1β: interleukin 1 beta; TRIM8: tripartite motif 8.
Knockdown of TRIM8 Prevented IL-1β-Induced NF-κB Activation in Chondrocytes
NF-κB signaling pathway was found to be crucial in the development of OA. Thus, we explored the effect of TRIM8 on NF-κB signaling pathway in chondrocytes. The results demonstrated that IL-1β treatment greatly induced NF-κB activation in chondrocytes, as proved by increased p-p65 and p-IκBα levels. However, the IL-1β-mediated alternations of p-p65 and p-IκBα levels were restored by knockdown of TRIM8 (Fig. 8).
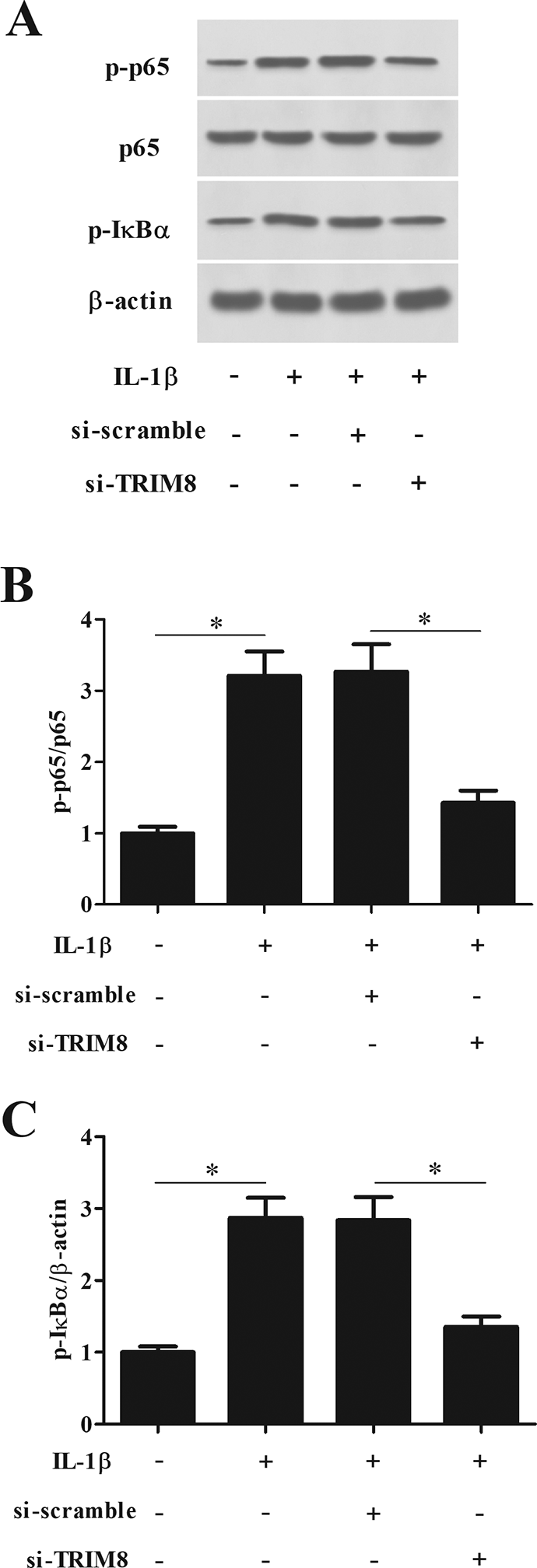
Knockdown of TRIM8 prevented IL-1β-induced NF-κB activation in chondrocytes. Chondrocytes were transfected with si-TRIM8 or si-scramble, and then treated with IL-1β (10 ng/ml) stimulation for 1 h. (A) To determine the effect of TRIM8 knockdown on the NF-κB activation, the levels of p65, p-p65, and p-IκBα were detected by western blot analysis. (B and C) Quantification analysis of p-p65 and p-IκBα. n = 4. *P < 0.05. IκB: inhibitory κB; IL-1β: interleukin 1 beta; NF-κB: nuclear factor kappa B; TRIM8: tripartite motif 8.
Discussion
OA is one of the most prevalent joint disorders associated with inflammation. Advances in the understanding of OA pathogenesis have provided insights into the role of chondrocytes in OA 6 . Chondrocytes are important for the maintenance of proper cartilage matrix; however, loss of function and apoptosis would lead to the failure of the articular cartilage 6 . IL-1β, a major cytokine mediating cartilage degradation in OA, has been reported to decrease chondrocytes’ viability 21 . A mass of previous studies has applied IL-1β to facilitate the synthesis of proinflammatory cytokines that eventually enhance cartilage degradation. In the current study, we also isolated the human OA chondrocytes and stimulated with IL-1β to induce inflammation.
Elevated levels of TNF-α, IL-1, and IL-6 have been found in the cartilage samples of OA patients, confirming their important roles in OA pathogenesis 22 . Our results showed that IL-1β induced the production of proinflammatory cytokines including TNF-α and IL-6. IL-1β also induced the production of a large amount of NO and PGE2, which are crucial inflammatory mediators and regulators of cartilage matrix synthesis and degeneration. Besides, some other intermediate molecules, such as iNOS and COX-2, were also induced by IL-1β, which might contribute to the increased production of NO and PGE2, respectively 23 . IL-1β suppressed the synthesis of ECM proteins including aggrecan and collagen-II in chondrocytes. In addition, IL-1β induced production of matrix-degrading enzymes, such as MMPs and A disintegrin and metalloproteinase with thrombospondin motif (ADAMTSs), which are closely involved in the breakdown of the articular cartilage 24 .
Previous findings indicate that TRIM8 is involved in various inflammatory responses. TRIM8 positively regulates proinflammatory responses in Pseudomonas aeruginosa-induced keratitis through promoting K63-linked polyubiquitination of transforming growth factor β-activated kinase 1 (TAK1) 25 . TRIM8 negatively regulates TLR3/4-mediated inflammatory and innate immune responses by blocking TRIF–TBK1 interaction 26 . Blocking of TRIM8 inhibits lipopolysaccharide-induced inflammatory response with decreased levels of TNF-α, IL-1β, and IL-6 in lung epithelial cells 18 . Inhibition of TRIM8 restrains I/R-mediated cerebral injury, which is associated with inflammation and apoptosis 18 . Our results showed that TRIM8 expression was significantly increased in OA tissues. In vitro studies showed that IL-1β induced the TRIM8 expression in chondrocytes. Knockdown of TRIM8 attenuated IL-1β-induced production of proinflammatory cytokines and inflammatory mediators. Knockdown of TRIM8 might prevent the cartilage matrix degradation through inhibiting the expression of matrix-degrading enzymes and inducing the synthesis of ECM proteins.
The NF-κB signaling pathway has multifunctional roles in a wide range of biological processes, such as cell survival, apoptosis, aging, inflammation, and immune responses 27 . According to the previous studies, NF-κB signaling is activated in OA chondrocytes and widely involved in OA pathophysiology through induction of various inflammation-related factors and ECM-degrading enzymes 28,29 . Overall, NF-κB signaling affects chondrocytes survival, synovial inflammation, and cartilage matrix remodeling, implying a distinctive role in OA pathogenesis 30 . The NF-κB signaling is recognized as a disease-contributing factor and has become a therapeutic target for OA. TRIM8 was found to regulate inflammatory responses through NF-κB signaling 31 . Thus, we explored the effect of TRIM8 on NF-κB signaling in IL-1β-induced chondrocytes. We found that knockdown of TRIM8 decreased the levels of p-p65 and p-IκBα, indicating that TRIM8 knockdown prevented IL-1β-induced NF-κB activation in chondrocytes.
There existed several limitations in this study. A major limitation is that our findings are based on the in vitro experiments. Future in vivo experiments are needed to verify the role of TRIM8 in OA. Secondly, the exact molecular mechanisms by which TRIM8 regulates NF-κB pathway in chondrocytes need to be further explored in the future study.
The current study has provided experimental evidence that TRIM8 was involved in the progression of OA. This study proved that knockdown of TRIM8 lessened IL-1β-induced inflammation and ECM degradation in chondrocytes. The effects of TRIM8 on IL-1β-induced chondrocytes were attributed to the inhibition of NF-κB activation. These findings provided important insights into the role of TRIM8 in OA.
Supplemental Material
Supplemental Material, Supplementary_Figure_1 - Knockdown of TRIM8 Attenuates IL-1β-induced Inflammatory Response in Osteoarthritis Chondrocytes Through the Inactivation of NF-κB Pathway
Supplemental Material, Supplementary_Figure_1 for Knockdown of TRIM8 Attenuates IL-1β-induced Inflammatory Response in Osteoarthritis Chondrocytes Through the Inactivation of NF-κB Pathway by Ruoxi Liu, Hao Wu and Huanjin Song in Cell Transplantation
Footnotes
Ethical Approval
This study was approved by the Ethics Committee at the Second Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Second Affiliated Hospital of Xi’an Jiaotong University Ethics Committee (Approval Number: 2020283)-approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
