Abstract
Ovarian insufficiency is described as a multifaceted issue typically encountered in the field of assisted reproduction. The three main identified diagnoses of ovarian insufficiency include premature ovarian failure (POF), poor ovarian response (POR), and advanced maternal age (AMA). Patient heterogeneity in the era of individualized medicine drives research forward leading to the emergence of novel approaches. This plethora of innovative treatments in the service of adequately managing ovarian insufficiency is called to undertake the challenge of addressing infertile patients exploring their reproductive options. This review provides an all-inclusive presentation and critical analysis on novel treatments that have not achieved routine clinical practice status yet, but have recently emerged as promising. In light of the lack of randomized controlled trials conveying safety and efficiency, clinicians are left puzzled in addressing the “how” and “for whom” these approaches may be beneficial. From ovarian injection employing platelet-rich plasma (PRP) or stem cells to artificial gametes and ovaries, ovarian transplantation, and mitochondrial replacement therapy, this descriptive review provides insight toward assisting the practitioner in decision making regarding these cutting-edge treatments. Biological mechanisms, invasiveness levels, efficiency, as well as possible complications, the current status along with bioethical concerns are discussed in the context of identifying future optimal treatment.
Keywords
From the time of birth of the first in vitro fertilization (IVF) child in 1978 till today, the common denominator in research is the focus of clinicians’ efforts to override pathophysiological barriers leading to infertility and provide solutions. From controlled ovarian hyperstimulation (COH) protocols to the emergence of intracytoplasmic sperm injection (ICSI), embryo and gamete cryopreservation, and preimplantation genetic screening (PGS), these—once introduced as—novel approaches have been successfully established as clinical routine practice 1 .
Despite the fact that several infertile couples can be successfully treated, one category of infertile patients still remains highly challenging for clinicians. This category refers to patients diagnosed with ovarian insufficiency. Ovarian insufficiency is a term describing a wide range of pathophysiological conditions where ovarian function is compromised. Several iatrogenic, pathophysiological, or physiological factors and processes can compromise ovarian function namely maternal age, autoimmune diseases, mutations in genes implicated in the regulation of ovarian function and development, chromosomal and developmental abnormalities, infectious diseases, and gonadotoxic treatments (e.g., chemotherapy or radiotherapy) 2 –5 . In the context of assisted reproductive technology (ART), women presenting with ovarian insufficiency tend to produce oocytes of compromised quality, may be unable to ovulate normally, may not be responding to COH protocols or they may present with primary failure of ovarian function 2 –5 . Considering that ART success requires good quality oocytes that will in turn provide good quality embryos of high implantation potential, patients with ovarian insufficiency present with a significantly compromised reproductive potential.
The three main patient categories with ovarian insufficiency are defined as premature ovarian failure (POF), poor ovarian response (POR), and advanced maternal age (AMA). Due to the wide spectrum of pathophysiological conditions leading to ovarian failure, managing patients with ovarian insufficiency is highly perplexing. These challenges have served as a driver for the emergence of a new era in the field of reproductive medicine which has risen dynamically in the last decade, aiming to effectively address ovarian insufficiency in the context of treating infertility. Recent studies investigating the regenerative dynamic potential of the ovaries demonstrate that follicular growth could be stimulated when an adequate ovarian environment is restored 6 . This restoration can be achieved employing factors of known regenerative potential such as stem cells, isolated growth factors, or platelet rich plasma (PRP) 7 . Additionally, recent advances in the field of stem cell biology are enabling the reprogramming of differentiated cells to stem cells and subsequently to gamete-like cells also known as artificial gametes 8 . Last but not least, recent studies suggest that mitochondrial function is strongly related to oocyte aging and fertilization failure 9,10 . These data, in combination with the recent technological development allowing nearly complete replacement of the cytoplasm of an oocyte, introduce the novel approach of mitochondrial replacement therapy for oocyte rejuvenation claiming to lead to an advanced cellular dynamic.
Our team of experts provides herein a timely and essential critical review analysis aiming to report on the novel approaches in addressing the challenging case of ovarian insufficiency, expressing considerations and concerns toward clinical application in humans. This review presents the prospective use of ovarian rejuvenation employing ovarian injection of stem cells, growth factors, and PRP, along with mitochondrial replacement therapy, and continuing to employment of artificial gametes, artificial ovaries, and ovarian transplantation. This review provides a comprehensive and critical analysis with regards to the novel treatments addressing ovarian insufficiency. The manuscript discusses the biological mechanisms entailed, the invasiveness and efficiency, along with the possible complications, the current status, and the bioethical implications of these novel approaches.
Stem Cell Transplantation
Stem cell therapy has noted considerable success in the field of regenerative medicine during the last two decades. The term “stem cells” is used in order to describe a wide spectrum of undifferentiated cells of the human body having the ability to self-renewal, to proliferate and differentiate to several organ and tissue-specific cell types. Due to their developmental ability, stem cells may represent the “pin of the arrow” in the field of regenerative medicine today.
As anticipated, following successful implementation of stem cell therapy on various systems (as mentioned above), investigating the potential use of stem cell therapy in the context of treating the female reproductive system became the focal point of cutting edge research. This trend has further focused on the context of infertility related to ovarian insufficiency. The explosion of interest witnessed by the scientific community regarding the potential use of stem cell therapy in treating infertility is depicted in a recent systematic review published by Fazelli et al. in 2018. The results provided, indicate that more than of 11,400 published studies have investigated the potential use of stem cell as an effective therapy for several conditions leading to both male and female infertility 11 . From endometrial rejuvenation in patients presenting with Asherman syndrome to restoration of ovarian function in poor responder patients, stem cells presented as an appealing therapeutic tool in the field of reproductive regenerative medicine 12,13 .
Studies have hitherto focused on the use of the multipotent stem cells and particularly of the mesenchymal stem cells (MSCs) as a potential therapeutic tool for ovarian insufficiency. Their exceptional properties including undifferentiation, proliferation, and renewal capacity are of high significance rendering stem cells a remarkable “tool” in the service of transplantation and drug targeted therapy. Additionally, the unique profile of the multipotent stem cells reduces the rejection and tumorigenic risk and increases stem cell survival rate making therapy more efficient and safer 14,15 .
Stem cell therapy could perhaps represent the missing piece of the puzzle in overcoming the challenges encountered in the management and treatment of these infertile couples. Multipotent stem cells can effectively induce tissue regeneration, mediated by the two exceptional and unique abilities characterizing them. On the one hand, these cells are able to migrate to damaged tissues and organs and differentiate to tissue-specific cells, promoting healing processes. On the other hand, several studies demonstrate that various multipotent stem cells have the ability to act as paracrine modulators. These cells are able to secrete a wide spectrum of growth factors, chemokines, and mitogenic proteins and via these are able to promote proliferation and vascularization. Among these factors are the transforming growth factor a (TGF-a), the TGF-b, the epidermal growth factor, and the insulin-like growth factor 1 (IGF-1) 16 –18 . Furthermore, multipotent stem cells are able to modulate immune response and present with anti-inflammatory properties. These cells are capable of secreting several growth factors and anti-inflammatory cytokines such as members of interleukin superfamily, tumor necrosis factor, and interferon γ (INF-γ). Moreover, several studies demonstrate that multipotent stem cells exert anti-apoptotic properties limiting tissue degeneration 19,20 . In regard to the ovarian function, several studies demonstrate that the ovarian niche is able to attract several types of undifferentiated cells from other organs and tissues and especially from bone marrow. These cells are able to migrate into the ovaries supporting folliculogenesis in a procedure known as “homing” 21 . Following the migration to ovaries, these cells secrete several growth factors and hormones orchestrating ovarian function. Considering the pathophysiological base of ovarian insufficiency involving degenerative phenomena leading to the collapse of the ovarian niche and the disruption of the molecular network controlling the ovarian vascularization, it is easy to understand the rationale behind the use of the multipotent stem cells as a potential therapeutic tool. Stem cells could restore ovarian function via the secretion of several hormones and growth factors inducing angiogenesis and tissue regeneration.
The potential therapeutic value of stem cells in ovarian failure was reported for the first time approximately 25 years ago. Sanders et al. published a retrospective observational study indicating that POF patients were able to achieve natural conceptions following bone marrow transplantation 22 . Fast forward to today and this approach albeit promising still retains its experimental procedure status. Several other published reports indicated that ovarian function restoration could be achieved following bone marrow transplantation in some POF patients previously treated with high-dose of chemotherapy or/and total body irradiation 23,24 . It has been voiced that the mechanism of action for bone marrow stem cells (BMSCs) is to migrate to ovaries through the circulation, exhibiting regenerative properties via the secretion of several hormones and growth factors. Following “homing”, BMSCs could restore the function of pre-existing undamaged follicles, leading finally to restoration of ovarian function 25 –28 .
Following the publication of aforementioned observations, numerous studies investigating the potential use of stem cells for ovarian insufficiency were conducted in animal models prior to initiating human studies. As reported, the compromised function of ovaries in POF or POR models was reversed, estrogen secretion from the compromised tissue was restored, and follicular growth was reactivated, while granulosa cell apoptosis was mitigated accompanied with the occurrence of angiogenesis. What is more, fertility restoration and live births were documented suggesting that BMSCs transplantation could be an effective treatment regarding ovarian function restoration in POF, POR, and AMA patients 29,30 . Following the encouraging published data indicating the prospective use of BMSCs in addressing ovarian insufficiency, the therapeutic potential of other sources of stem cells indicating their ability to induce similar regenerative phenomena. The results from the aforementioned studies demonstrate that there are several easily accessible novel sources of stem cells that may be used toward ovarian rejuvenation.
Hitherto, clinical studies investigating the potential therapeutic value of these cells in humans are limited. This may be attributed to the fact that there are several limitations entailed in stem cell transplantation, albeit promising outcomes have been documented. In the study of Eddesy et al., 10 women presenting with idiopathic POF were subjected to MSC autologous ovarian transplantation. Recovery of menstruation was achieved 3 months following autologous MSC transplantation for two patients and a subsequent live birth of a healthy offspring has been documented 31 . In another study published by Gabr et al., 30 women with idiopathic POF were also subjected to MSC autologous ovarian transplantation. Ovarian function restoration, including gonadotropin level reduction, and estrogen and anti-Müllerian hormone (AMH) level increase, was observed 1 month post-treatment. The therapeutic effectiveness was impressively sustained during a 48 week follow up. Three patients achieved pregnancy via IVF treatment and one patient reported a pregnancy following natural conception 32 . In the prospective observational pilot study published by Herraiz et al., the effectiveness of an autologous stem cell ovarian transplantation protocol (ASCOT protocol) was assessed in POR patients. This was the first clinical study investigating stem cell therapy for ovarian rejuvenation in POR patients. Following bone marrow mobilization with granulocyte-colony stimulating factor (G-CSF) administration and stem cell collection by apheresis from peripheral blood, the 17 patients participating in the study were submitted to CD133+ BMSC ovarian infusion via intra-arterial catheterization under ultrasonography guidance. The time frame between stem cell collection and stem cell infusion was less than 24 h for all patients. Two weeks following ASCOT treatment, a significant increase in the antral follicle count (AFC) was observed. As anticipated, an increase in AMH levels was also observed. Following COH treatment, an increase in the number of AFC and oocytes obtained was recorded in comparison to the patients’ previous medical history. However, the embryo euploidy rate was reported to be low. Five pregnancies were achieved, two following an IVF/embryo transfer (ET) cycle and three via natural conception 13 . The potential effectiveness of stem cell therapy was also recorded in a 45 years old perimenopausal woman. Eight weeks following stem cell therapy, a significant improvement regarding AFC number and AMH levels was observed. The patient achieved pregnancy via an IVF-frozen cycle. The woman was subjected to noninvasive prenatal testing indicating a normal karyotype. A live birth was finally achieved and the patient delivered via cesarean section at 38 weeks. No complications were observed in the pediatric follow up 33 .
Stem cell therapy seems to be a promising approach. Nonetheless, in order to safely recruit it in clinical application toward treating ovarian insufficiency in the context of infertility, it should be highlighted that it is equally imperative to consider both strengths and weaknesses.
Despite the fact that mesenchymal stem cells are characterized by a low immunologic profile, allogeneic stem cell transplantation could lead to severe graft rejection, posing a risk for the patients’ health. Studies in the field demonstrate that autologous stem cell transplantation is related with enhanced therapeutic efficiency compared to allogeneic transplantation in one out of the four women treated with allogeneic transplantation could face severe chronic gynecological graft disease 30,34 . One of the antipodes, autologous stem cell transplantation could serve as a potential solution in an effort to eliminate the graft rejection risk. However, hitherto, the practice of autologous stem cell transplantation entails two levels of invasiveness. The first one refers to the harvesting procedures and methods of stem cell retrieval via bone marrow aspiration. This fact coupled with the second level of invasiveness encountered at the stage of treatment application entailing stem cell injection into the ovaries, performed via laparoscopy or intra-arterial catheterization, significantly increases concerns. Thus, it is of high significance to investigate the potential use of other sources of stem cells for autologous transplantation, except from BMSCs, namely endometrial stem cells (EnSCs) and adipose derived stem cells (ADSCs). These categories of stem cells have successfully been used as a potential therapeutic tool for addressing ovarian insufficiency in animal models. Nonetheless, studies in humans have not been conducted till date. An overview of the presented studies on the novel approach of stem cells administration indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 1.
An Overview of Published Studies on the Novel Approach of Stem Cells Administration Indicating Study Design, Methods Employed, Outcome, and Adverse Effects Reported.
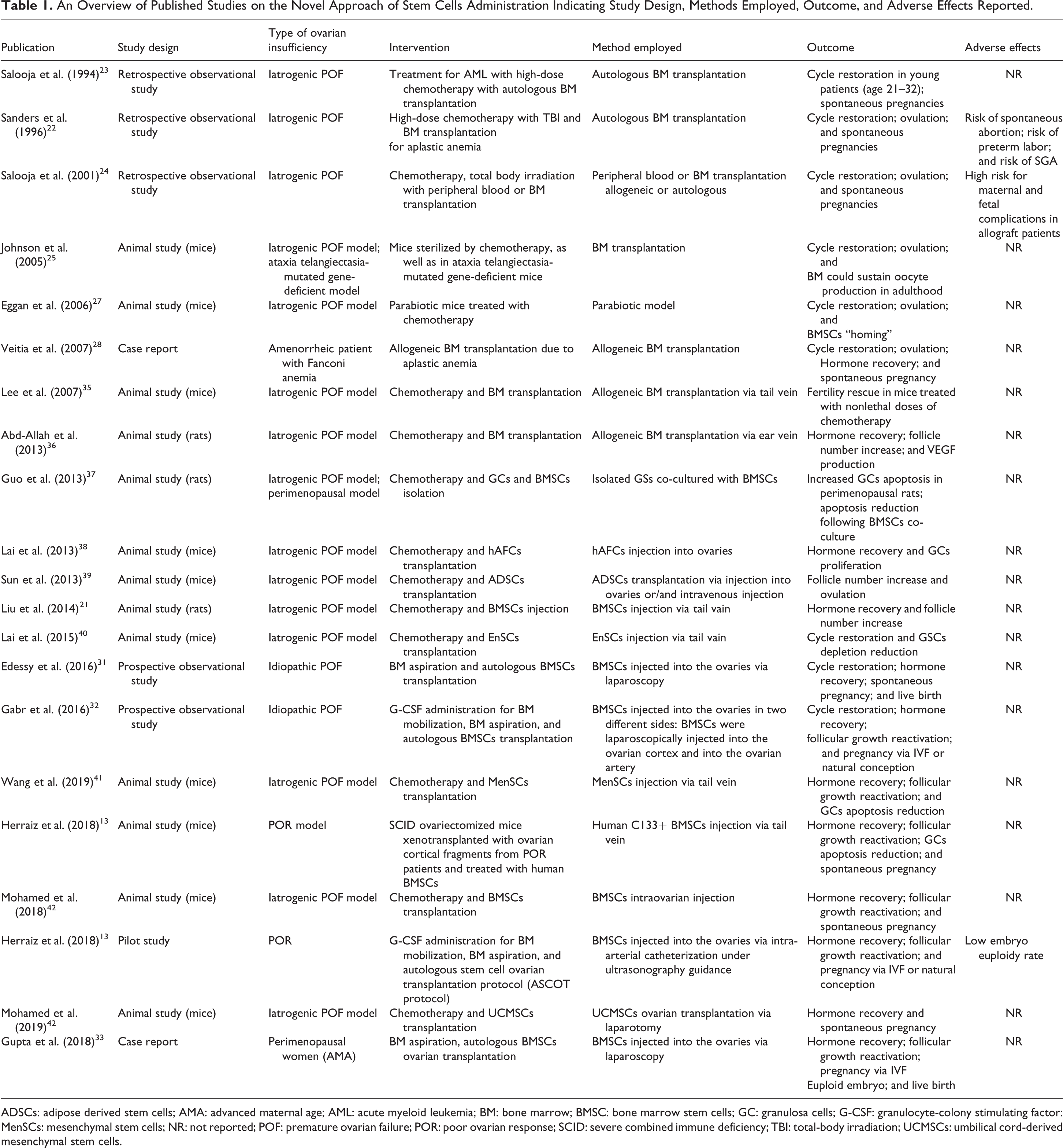
ADSCs: adipose derived stem cells; AMA: advanced maternal age; AML: acute myeloid leukemia; BM: bone marrow; BMSC: bone marrow stem cells; GC: granulosa cells; G-CSF: granulocyte-colony stimulating factor: MenSCs: mesenchymal stem cells; NR: not reported; POF: premature ovarian failure; POR: poor ovarian response; SCID: severe combined immune deficiency; TBI: total-body irradiation; UCMSCs: umbilical cord-derived mesenchymal stem cells.
Reporting on the safety of stem cell therapy in ovarian insufficiency, serious considerations should be highlighted, especially in light of the lack of published evidence addressing any side effects related to this novel approach. The most severe potential adverse effect is that unfortunately, intense cell proliferation events that occur following stem cell transplantation may induce malignant formation. It is well documented that long-term cultured mesenchymal stem cells could induce tumorigenesis and metastasis. Following stem cells, isolation from their source of origin, in vitro expansion of cell population is typically required to achieve a clinically suitable grade of stem cells. However, stem cells at higher passages could lead to malignant cell transformation 29 . Considering the aforementioned, it is of added value to determine both the appropriate dose and the specific administration time following stem cell harvesting, in order to achieve a balance between safety and efficiency. It seems that the appropriate management of patient—defined by severity— requires an individualized standardization by the practitioner in the era of precision medicine. Nonetheless, it is imperative to thoroughly understand the underlying mechanisms regulating stem cell treatment.
Regarding the appropriate administration method for ovarian stem cell transplantation, data that have been obtained till date are inconclusive and lacking clarity. Two potential administration methods have been proposed namely stem cells injection into the ovarian cortex via laparoscopy and intra-arterial ovarian catheterization under ultrasonography guidance. Both of these methods appear to be equally efficient. Considering the level of invasiveness, intra-arterial catheterization may be viewed as more patient-friendly. However, in certain cohorts of patients such as AMA women, the practice of intra-arterial catheterization targeting atrophic ovaries of compromised volume due to aging renders the procedure challenging 43 . Future randomized controlled trials (RCTs) comparing the two methods with respect to the patients’ pathophysiology will provide a deeper impact in categorically delineating this issue and addressing the benefits entailed.
In conclusion, robust data provided from future RCTs and well-designed clinical trials are vital in order to ascertain safe conclusions with respect to an array of crucial questions remaining unanswered and perhaps even undefined. This very fact justifies why beyond the promising nature of its application it still retains its experimental status. The appropriate dose and specific administration time following stem cell harvesting, the appropriate administration method, the duration of the recovery period and the potential treatment replication are all issues of value that merit further investigation. Furthermore, cost-effectiveness studies along with studies focusing on assessing patients’ behavioral patterns and response comparing stem cell therapy to the conventional treatment options should be submitted. Regarding the cost of stem cell therapy, it may be safe to conclude that it typically corresponds to a particularly high charge per treatment cycle one that could range from over two to fivefold higher than the cost for a conventional IVF cycle depending on the country where treatment is provided 44,45 . This fact alone may raise considerable bioethical issues. Considering the multifaceted nature of ovarian insufficiency implicating several different pathophysiological conditions, meticulous observations and documentations are needed in order to determine the specific cohorts of patients for whom stem cell therapy may be efficient and safe. Till then, stem cell transplantation for ovarian rejuvenation should maintain the status and classification of an experimental treatment and practitioners should abstain from embarking on clinical application outside the scope of an approved research protocol.
Platelet-Rich Plasma Intra-Ovarian Infusion
PRP is comprised of crucial ingredients identified in repair mechanisms instigating fundamental physiological procedures in a healthy individual. Numerous physiological mechanisms are involved in the multifaceted phenomenon of tissue restoration and repair process, including proliferation, angiogenesis, programmed cell death 46 , and cell migration, all of which are attributed to the valuable synergy of factors evoked by platelets namely vascular endothelial growth factor (VEGF), platelet-derived growth factor AB (PDGF-AB), and TGF-b1 47 –49 . Additionally, growth factors are coupled by the presence of chemokines and cytokines engaging in order for tissue healing to be orchestrated 50 . The rationale behind implementing PRP in the context of managing pathologies affecting tissue integrity was based on the aforementioned key components originally verified in participating in the tissue repair process. Its application may extent to restore these physiological procedures in a system identified with pathologies compromising function.
PRP has noted considerable success in various systems 51 –55 . Following successful implementation of PRP in treating pathologies related to various systems, investigating the potential use of PRP in the context of the female reproductive system was anticipated. In light of the fact that monthly ovulation in the female reproductive system has been described as micro-trauma 56 , the rational of recruiting PRP along with its vital components being involved in tissue repair, may be viewed as one way of justifying its application regarding ovarian function restoration.
Intraovarian injection of PRP in animal models and subsequent resurgence in oocyte production was initially introduced in literature describing the injection of growth factors in the ovary. These studies resulted in increased follicular development and a diminution in apoptotic events in animal models 57,58 . This radical approach claims to trigger and revive an otherwise described as a “comatose” ovary through autologous administration of enriched plasma providing a concentrated cohort of platelets capable of producing essential growth factors. Application of PRP regarding restoration of ovarian function was introduced by Sfakianoudis et al. and Sills et al. 7,43,59,60 . Regarding the method and technique describing intraovarian PRP treatment, various protocols have been described for the preparation of PRP including either commercial kits or a more affordable in-house preparation 43,60 .
Albeit reports have surfaced regarding PRP employment at the moment PRP is considered to be an experimental procedure in the context of ovarian insufficiency until robust data are provided from RCTs. The first report on the clinical application of PRP in human ovaries included perimenopausal and prematurely menopausal IVF patients with poor ovarian reserve exploring options to address infertility and pursue a pregnancy. These patients were treated employing intraovarian PRP injection 61 . Restoration of ovarian function along with restoration of the menstrual cycle was noted 1–3 months post-treatment. Further to that, the patients included were subjected to natural IVF cycles yielding a range of 1–5 oocytes per retrieval. Poor responder patients as well as patients of diminished ovarian reserve have also been investigated with reports revealing impressive hormonal restoration, improved oocyte accumulation, clinical pregnancies, and subsequent live births in a short time interval post-treatment 59,60,62 . Based on the observations regarding hormonal restoration following the PRP application, these results indicate that ovaries of compromised function following PRP application seem to mimic the physiology and adopt respective pathways along with the endocrinological behavior of a functional ovary even for a defined shorter time frame. However, the lack of conclusive data allows no further conclusions drawn on the mapping of the activated pathways regarding the interaction between PRP and the reproductive organs. An overview of the presented studies on ovarian PRP injection indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 2.
An Overview of Published Studies on the Novel Approach of PRP Administration Indicating Study Design, Methods Employed, Outcome, and Adverse Effect Reported.
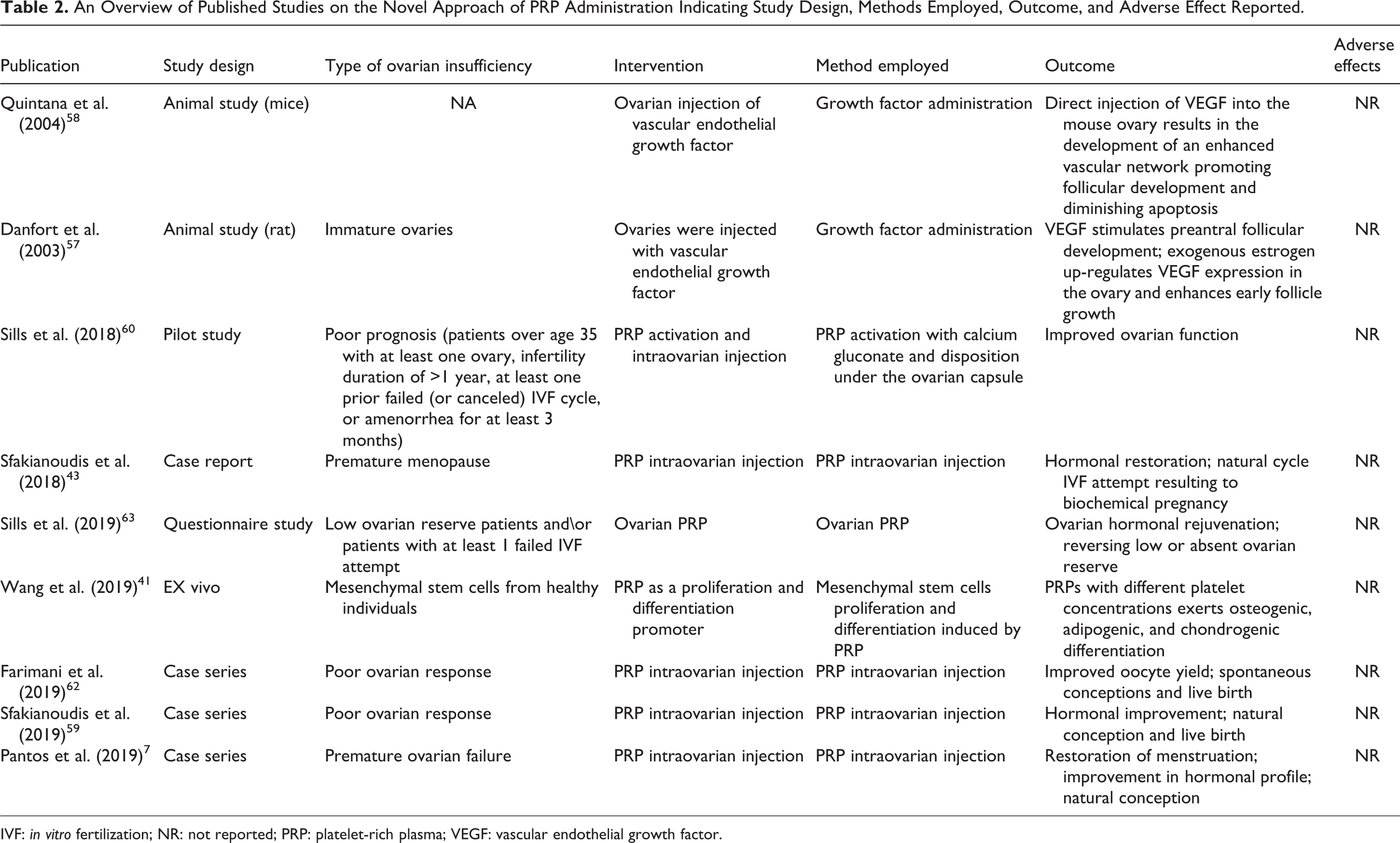
IVF: in vitro fertilization; NR: not reported; PRP: platelet-rich plasma; VEGF: vascular endothelial growth factor.
In an effort to elucidate all aspects regarding PRP application as a therapeutic option for ovarian insufficiency, it should be highlighted that both strengths and weaknesses are equally imperative to consider. PRP constitutes an autologous product minimizing the potential of side effects that may be related with the administration of a heterologous sample. Despite PRP application being described in the literature as a straightforward procedure, nonetheless, the procedure of PRP application entails predicted challenges with respect to its invasive nature and the fact that the clinical application of intraovarian injection requires meticulous handling and respective patient individualized standardization by the practitioner. For certain cohorts of patients such as menopausal women, the practice of intraovarian infusion targeting atrophic ovaries of compromised volume due to aging adds another level of complexity rendering the procedure rather demanding 43 . What is more, multiple injection sites seem to be required, thus the level of invasiveness may be viewed as considerable. Regarding the cost of this novel approach, an affordable technique has been indicatively proposed for the preparation of blood samples with an estimated cost of $10 64 . However, in clinical practice, the cost of this service is typically reaching over 100-fold of this corresponding to approximately 20% of a conventional IVF cycle cost depending on the country where treatment is provided 65 . PRP injection remains an invasive procedure performed under ultrasound guidance with the anticipated risks not adequately elucidated. Lack of published evidence addressing any side effects is far from reassuring leading to the acknowledgment of the need for well-designed clinical trials. Aside from the promising results already published for some cohorts of patients, there has been some concern voiced on the aftereffect of PRP application. The intense cell proliferation events that occur may induce malignant formations possibly due to differentiation of stem cells present in ovaries 66 . Nevertheless, the presence of stem cells in ovaries represents a heated debate 67 with opposing schools of thought emerging, supporting the norm of the restricted reserve of oocytes versus the revolutionary concept of ovarian germline stem cells capable to rejuvenate the ovarian tissue.
While investigating the possibilities of PRP application, precise primary and secondary outcomes should be decided upon, in order for a conclusive report on PRP’s effectiveness to be delivered. Undoubtedly, the molecular interaction needs to be elucidated prior to designing large RCTs that aim to convey safety in practice. It has been voiced that PRP’s concentration may play a key role in cell proliferation and differentiation of mesenchymal cells. Managing various pathologies may require different optimal concentrations. This may explain the heterogeneity in the results and outcomes documented and observed following PRP application amongst patients 41 . A personalized approach taking into account the underlying pathology should be pursued. Several factors potentially affecting the end result namely time intervals between PRP applications, volume administered, as well as the threshold of the maximum number of allowed interventions should be addressed. Pondering on whether PRP has a place in clinical practice in addressing ovarian insufficiency, a study submitted the thesis that aside from promising outcomes in ovarian physiology, an essential improvement on the treated patients’ quality of life has been noted. This could be plausibly attributed to the hormonal rebalance recorded following intervention 63 , encouraging the employment of PRP and extending its consequences beyond a strict physiological and molecular context.
The ovarian reserve issue may be a multifaceted matter, thus managing these patients may require a plethora of available tools and techniques. Hitherto, the complex dialog between PRP components and the ovary remains to be deciphered. In anticipation of solid molecular and clinical evidence to support the validity of the “ovarian rejuvenation” concept, PRP may face the risk of remaining a costly service. Nonetheless, preliminary data showcase its potential of being perceived as a ground-breaking medical innovation. Could that be enough to give it a try? Maybe. Nonetheless, we need to question whether evidence is accumulating to a satisfactory degree leaving the scientific community content in deciding to introduce routine clinical application of PRP for ovarian insufficiency.
Ovarian Tissue Transplantation
Ovarian tissue cryopreservation may not be considered as a unique novel approach. Since the first implementation, over 130 babies have been born 68 . However, it is still considered to be an experimental approach 69 . Various reports attempt to establish a commonly accepted technique for ovarian transplantation. Orthotopic transplantation may present as an optimal practice since the microenvironment around the transplanted tissue is the most suitable for follicular development 70 . On the other hand, there is limited number of tissue fragments allowed to be transferred due to space constriction, while the level of invasiveness presents as a major limitation resulting in severe pelvic adhesions 71 . It should be highlighted that natural conception may be achieved in cases of orthotopic transplantation. The method of transplanting ovarian tissue may be considered technically appealing since the utilized ovarian tissue is characterized by a solid extracellular matrix structure coupled with complex cellularity. Furthermore, biomaterials are introduced in order to enhance the structure and integrity of ovarian tissue by improving adhesion, proliferation, and differentiation of the cells involved. Natural biomaterials present with significant heterogeneity in their composition spectrum, while synthetic biomaterials’ ability to promote cellular and tissue remodeling is restricted 72 . Thus, both sources present equally with weaknesses regarding the configuration of an optimal model for ovarian tissue transplantation.
Since undergoing autologous ovarian tissue transplantation requires collecting tissue prior to acknowledging and identifying signs of ovarian deficiency, this strategy is typically addressing strictly a particular cohort of patients. These patients are women that are destined to experience an anticipated ovarian shortage following cancer therapy, without excluding the patient group that will experience anticipated gamete exhaustion due to a prognosis or even a diagnosis of POF. In cases of a diagnosis of malignancy, ovarian tissue cryopreservation for subsequent transplantation is suggested, since it could be performed in a short time-frame without delaying the initiation of therapy. From the first attempts on ovarian tissue transplantation almost 20 years ago 73 to numerous successful live births following this procedure published in literature since then 74 –78 , a new line of approaching ovarian deficiency has been introduced and established.
Valuable observations have been documented in literature reporting on restoration of fertility, ovarian activity reinstatement, endocrine function reactivation, and follicular recruitment 70,79 . What is more, transplanting ovarian tissue has been proposed strictly on the grounds of ovarian rejuvenation leading to what may be described as a hormonal ovarian reboot irrespectively of pursuing fertility treatment. Ovarian tissue transplantation enabled a hormonal restoration lasting an impressive 7-year period 79 , which was documented to be extended to a 12-year restoration period when the procedure was repeated 70 . Nonetheless, one should not fail to highlight that this invasive procedure minimizes and potentially jeopardizes the current ovarian reserve for the patient, thus bioethical concerns are raised.
A major limitation to consider is that autologous transplantation entails the risk of reintroducing cancerous cells to the treated ovary 80 . Moreover, immune rejections constitute a major complication for this line of approach, albeit the autologous source of tissue may contribute to overcoming this side effect. Ischemia has also been described as a detrimental side effect following ovarian tissue transplantation 81 . The main cause of this event is the lack of vessel anastomosis during the tissue transplantation procedure. As a result, the transplanted ovarian tissue is exposed for a certain time period of 5 days 82 in a hypoxic and ischemic environment which demands an urgent intervention to induce neovascularization. In this context, various approaches have been attempted to optimize the outcome including employment of growth factors as a supplementary tool to assist toward completion of neovascularization in a timely fashion 83 . However, their application in human models is nowhere near consideration even though current experimentation on animals has contributed favorable results. Concerns over subsequent cancer diagnosis have emerged, nonetheless, they were considered as a low probability event when tissue is sourced from young women aged 20–25 84 . The technique of cryopreservation and its respective efficiency should be further taken in to account as a challenging part of the ovarian tissue transplantation approach. It is the actual cryopreservation technique that may perhaps result in compromised outcomes misleadingly attributed to the transplantation technique. Contemplating on the logistics of performing ovarian tissue transplantation for restoring endocrine function in menopausal patients, the procedure is less invasive since the graft site may not involve the pelvic cavity. Furthermore, it may be performed under local anesthesia, while in case of an ovarian neoplasm emergence, the ovarian tissue implant could be immediately extracted 84 . An overview of the presented studies on the novel approach of ovarian tissue transplantation indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 3.
An Overview of Published Studies on the Novel Approach of Ovarian Transplantation Indicating Study Design, Methods Employed, Outcome, and Adverse Effects Reported.

NR: not reported; POF: poor ovarian response.
All of the above constitute vital indications in favor of this treatment option. Nonetheless, similarly to all hormone replacement approaches aiming to postpone the permanent hibernation of the ovaries for women reaching the end of their reproductive life-span, ovarian tissue transplantation entails risks. Apart from the concerns raised when rejuvenation is the primary patients’ interest, practitioners appear hesitant to embark on this suggested approach even in cases where patients are facing a reproductive dead-end. This may be attributed to the realization that ovarian transplantation has been around for several decades, failing nonetheless to be established as a foolproof approach in clinical practice one that could provide specialists with confidence regarding application.
Artificial Ovary
An additional step in ovarian tissue preparation prior to implantation has been proposed in an effort to address the risk of reestablishing the treated disease. It includes the formation of an ovarian tissue scaffold where oocytes appear, grow, and subsequently “harvested.” Hitherto, this approach has been applied in animal models in which granulosa cell-oocyte complexes were collected and subjected to a 3D in vitro culture. Following standard proliferation, oocyte yield was higher than a conventional IVF approach could achieve 85 . Likewise, constructing a 3D artificial ovary has been attempted in order to successfully incorporate, accommodate, and enable maturation of human oocytes 86 . Most previous attempts have failed due to the common denominator of implementing a solid structure albeit lacking adequate mimicking of vital endocrine and paracrine pathways. Three ovarian cell types were included namely theca cells, granulosa, and oocytes 86 . By including theca cells as a fundamental component of the 3D ovary, hormonal production was achieved. Interestingly, implementing collagen or alginate scaffolds failed to ascertain a similar endocrinology response. What is more, enhanced interaction between cell types was observed conspiring toward the development of a physiological model. Under these circumstances, maturation and development of the primordial oocytes may be enabled. The novelty behind this approach is indisputable; however, the fact that it constitutes what may be described as an “isolated” system lacking the complexity of numerous interactions with other physiological systems should be highlighted.
Bioengineering strategies to restore fertility and address ovarian insufficiency include transplantation of fresh or cryopreserved ovarian tissue and tissue engineering involving implementation of growth factors, stem cells, pluripotent stem cells, mesenchymal stem cells, and biomaterials 72 . The percentage of cancer patient survivors is increasing, translating to patients who despite succeeding in overcoming a disease, they will be reproductively challenged experiencing infertility strain and concerns attributed to gonadal injury during therapy 87 . Numerous successful attempts at performing ovarian tissue cryopreservation and subsequent transplantation reveal valuable data on the efficiency of the approach. However, little is known in regard to whether this approach should be considered as a method to delay the onset of menopause—a trend that appears to be rapidly evolving. In regard to the concept of the artificial ovary, further considerations should be taken into account. An overview of the presented studies regarding the novel approach of artificial ovary indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 4. It should be highlighted that exposing sensitive tissue in a challenging environment may entail certain epigenetic risks which in turn may raise bioethical issues. How far is too far when the—once considered as science fiction—concept of harvesting gametes becomes a reality? Until strong evidence accumulates, the concept of artificial ovary clearly remains at the experimental stage. With RCTs being an extrapolation at best, clinical practice will have to wait.
An Overview of Published Studies on the Novel Approach of Artificial Ovary Indicating Study Design, Methods Employed, Outcome, and Adverse Effects Reported.

BMSC: bone marrow stem cells; NR: not reported.
Artificial Gametes
There are four basic cell types employed in the formation of artificial gametes, including germ line stem cells, induced pluripotent stem cells (iPSCs), somatic cell nuclear transfer to embryonic stem cells (SCNT), and SCNT to donor oocytes. Successful implementation resulting to live birth following artificially generating gametes has only been reported in animal models 88 , thus hitherto artificial gametes constitute an experimental procedure. Based on observations in animal models where artificial gametes may be transferred to ovaries enabling natural conception, the scientific community cannot help but ponder whether this strategy may present as a useful tool in the hands of practitioners managing ovarian insufficiency in the distant or not too distant future.
The option of employing artificial gametes has been introduced as an alternative solution for patients lacking the potential to produce functional gametes such as women with premature ovarian insufficiency 89 . In human models, the development of artificial oocytes has been described, while a successful fertilization has been confirmed in one case study 88 . As reported by White et al., human oogonial stem cells (OSCs) were transplanted to human ovary tissue xenografted to mice following in vitro proliferation, resulting in the development of artificial oocytes 90 . A challenging process of gamete formation originating from human embryonic stem cells has been described; however, this route was outlined as insufficient since differentiation of embryonic cells in culture systems entails certain difficulties to perform 91 . Human amniotic stem cells have also been successfully recruited in an effort to form artificial gametes 92 . Moreover, engaging human hepatic cell lines in order for germline cells to originate resulted in the formation of follicle-like structures, subsequent blastocyst-like structures, and finally germ cell/embryonic tumors following prolonged culture 93 . An overview of the presented studies on the novel approach of stem cells administration indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 5.
An Overview of Published Studies on the Novel Approach of Artificial Gametes Indicating Study Design, Methods Employed, Outcome, and Adverse Effects Reported.

NR: not reported.
It is vital for concerns on safety to be thoroughly addressed prior to even considering the route to clinical application. With RCTs being a mere hypothesis, it appears that the experimental stage regarding artificial gametes may take longer than anticipated. Despite the biological risks entailed, there are significant ethical concerns raised in a societal level that should be addressed prior to pursuing this option as a solid novel approach for management of ovarian insufficiency 94 A clear presentation of the patients for whom this approach may be beneficial and for whom access should be granted is imperative 95 .
Mitochondrial Replacement Therapy
Mitochondria are identified as “the powerhouse of the cell.” The role of mitochondria in the development of the preimplantation embryo and its implantation potential has been studied for at least two decades 96 . The processes of fertilization and proper embryonic development require great amounts of adenosine triphosphate (ATP) which is produced by mitochondria 9 . Oocyte mitochondrial DNA (mtDNA) content has been associated with poorer oocyte quality and ovarian insufficiency 97 . The prognostic value of the copy number has been a topic of controversy in literature since a number of studies observed a statistically significant higher number of mtDNA copies in embryos that resulted in implantation failure 98 , whereas other studies observed no statistically significant difference 99 .
Nonetheless, it is evident that maternal aging is the cause for mitochondrial dysfunction through swelling and cristae disruption in oocytes 100 . The employment of mitochondria from a young healthy donor was hypothesized to provide the preimplantation embryo with a better and more potent developmental environment during the early stages 101 . This may have served as the driver leading to early attempts of mitochondrial replacement therapy, which was initially approached by performing ooplasmic transfer, as first described by Cohen 101 . Although ooplasmic transfer presented with success in enabling an improvement in embryo quality resulting to reports of clinical pregnancy and live birth 102 , the Food and Drug Administration (FDA) decided to ban its application in 2001 due to ethical and medical concerns mainly related to heteroplasmy and interrelated adverse effects 102 .
More than a decade following the FDA ban, novel approaches regarding mitochondrial transfer in order to achieve improved ovarian function have been implemented. To avoid concerns introduced by including a heterologous donor, the initial approach employed the autologous germline mitochondrial energy transfer (AUGMENT). The protocol of AUGMENT includes isolation of mitochondria from the patient’s OSCs, processed and injected into the patient’s own oocytes during ICSI 103 .
The first application of AUGMENT was performed by Fakih et al. 103 targeting women presenting with low quality oocytes and/or embryos, diagnosed with diminished ovarian reserve, ovulatory dysfunction, polycystic ovarian syndrome (PCOS), tubal factor, and endometriosis. A total of 93 women from two IVF centers were recruited. The results of the study supported a beneficial role for the AUGMENT as the clinical pregnancy rate achieved was 35% and 22% for each center respectively, in contrast to the 11% and 4% corresponding to the clinical pregnancy rate of the same patients prior to AUGMENT. In the same study, sibling oocytes from 25 patients were randomly allocated to two groups, one designed to undergo the AUGMENT therapy and a control group. Out of the 25 patients, two underwent embryo-transfer with embryos originating from the control group, for nine patients, embryo-transfer was not performed, and 14 patients underwent embryo-transfer with embryos originating from the AUGMENT group. Eight pregnancies were observed all originating from the experimental group. A second, albeit smaller study, conducted by Oktay et al. 104 included 15 women undergoing an autologous germline mitochondrial energy transfer, described as autologous mitochondrial injection (AMI) by the authors. Of the 15 women recruited, only 10 underwent embryo-transfer, and 4 of them achieved a clinical pregnancy status.
These promising results were contradicted by the interim analysis of a discontinued large-scale triple blind RCT 10 . Following the enrollment of about 31% of the study’s total population (n = 59/190), an interim analysis was performed. According to the results of the study, AUGMENT did not increase neither the fertilization rate nor the euploid blastocyst formation rate. On the contrary, blastocyst formation rate was decreased in the experimental group. Moreover, embryo grading according to Spanish Association for the study of Reproductive Biology (ASEBIR) criteria was poorer, and live birth rates were similarly lower in the experimental group. These observations in the interim analysis rendered the recruitment of more patients unnecessary and the study was terminated, although the clinical pregnancy and live birth rates presented in the study were better when compared to the other studies regarding the autologous mitochondrial transfer. It should be highlighted that patients included in the RCT were not necessarily of poor ovarian function as they were younger than 42 years old, with median AMH of 1.69 ng/ml, with an interquartile range from 0.97 to 3.24 ng/ml. This is a reason for caution in all studies employing autologous mitochondrial transfer. The population enrolled is mainly a mixed population with only a few participants being diagnosed with diminished or poor ovarian response. It may be possible that the autologous mitochondrial transfer is suitable for a specific population which still remains to be identified.
Apart from the autologous mitochondria transfer, heterologous mitochondrial transfer, known as mitochondrial replacement therapy (MRT), has been employed mainly on the grounds of avoiding transmission of mitochondrial diseases 105 –107 . There are four protocols available for MRT, the pronuclear transfer (PNT), the polar body transfer (PBT), the maternal spindle transfer (MST), and the germinal vesicle transfer (GVT) 105,108 . The three latter techniques require the employment of donor enucleated MII oocytes, whereas PNT requires the employment of a donor zygote following removal of the pronuclei and polar bodies. In PNT, both parental pronuclei are transferred to the donor zygote. GVT requires transfer of the maternal germinal vesicle enclosing the nuclear DNA to the donor enucleated oocyte, and PB requires the transfer of the polar body of the maternal MII oocyte in the enucleated donor oocyte. MST requires the transfer of the spindle transfer enclosing the nuclear DNA to an enucleated donor oocyte. PNT and PBT have been evaluated as possible methods for MRT with embryos reaching the blastocyst stage without progressing to implantation or pregnancy 109,110 . Zhang et al. 111 employed PNT to achieve a pregnancy for a 30-year-old woman with mitochondrial disease. The same group achieved a pregnancy employing MST, for a woman with Leigh syndrome 112,113 . It should be mentioned that although initially a permission for long-term follow-up was granted, the parents of the offspring decided to forego the permission, thus pediatric follow-up data are not available 114 . In the context of employing MRT, MST is the sole technique applied in an effort to address poor ovarian response. The clinical trial 115 has so far resulted in one live birth, from a 32-year-old poor responder with four previous failed IVF attempts. This technique has raised considerable ethical concerns. The European Society of Human Reproduction and Embryology (ESHRE) issued a formal thesis on strong discouragement and disapproval regarding the technique’s application in the context of addressing poor ovarian response. Practitioners should refrain from adopting the clinical practice of MST for anything else other than to avoid transmission of a mitochondrial inherited disorder for which case no other alternative is viable. It is strictly this approach that has claimed regulation status by the Human Fertilisation and Embryology Authority (HFEA) in the United Kingdom 116 . It is clear that in the context of ART treatment, there is a lack of robust or even adequate data indicating safety and effectiveness of the method, which currently maintains its experimental status. An overview of the presented studies on the novel approach of stem cells administration indicating study design, methods employed, outcome, and any adverse effects reported is provided in Table 6.
An Overview of Published Studies on the Novel Approach of MRT Indicating Study Design, Methods Employed, Outcome, and Adverse Effects Reported.

AUGMENT: autologous germline mitochondrial energy transfer; MRT: mitochondrial replacement therapy; NR: not reported; RCT: randomized controlled trial.
Long term follow-up of MRT is required in order to report on the safety and efficacy of the procedure or even to buttress the original hypothesis trail of thought driving its development. Conducting more RCTs is a requirement in the topic. The RCTs should present with strict inclusion and exclusion criteria, and should not employ a mixed population. It is of imperative importance to evaluate the safety and the efficiency of a technique that may present as one of the turning points of ART practice. Moreover, although MST appears to be the sole option in providing favorable results in humans, all protocols should be evaluated, prior to deciding on superiority in the application. Although heteroplasmy levels seem to be low by employing the novel MRT techniques, mito-nuclear interactions are a topic of research. Two meta-analyses performed in animal models have reported contradicting results 117,118 . The first chronologically published meta-analysis has reported that mito-nuclear interactions do not pose a side-effect of MRT 117 . The second meta-analysis has reported that MRT may routinely affect offspring biological and biochemical characteristics with large scale effect due to mito-nuclear interaction, which seem to be even greater in humans 118 . It is of interest that the second meta-analysis has been questioned regarding its design by the author of the first 119 . Until large scale human studies are performed, the scientific community ought to practice cautiousness and maintain a vigilant stance in order to limit possible and perhaps probable long-term side effects that currently may not be efficiently identified, understood, treated, or reversed. At the moment, the research developing and fueling this novel approach is clearly not validated to justify clinical application. Perhaps this may be a good time for the scientific community to re-examine the “certainties” that fueled the idea that replacing mitochondria may be the answer to ovarian insufficiency prior to proceeding with clinical application.
Discussion
The concept of ovarian reboot has recently attracted heated interest. This may be attributed to the increased prevalence of the diagnosis of ovarian insufficiency for cases that demand effective options on managing their subsequent infertility status. This increase may be justified on the grounds of the remarkable results and elevated survival rates of treating cancer patients in the era of precision medicine. These patients can now enjoy a prolonged life span which nonetheless may harbor infertility and reproductive dilemmas. On another note, the concept of delaying aging has been intensively promoted in media. This reality reinforces practitioners toward engaging and endeavoring novel approaches fueled by the patients’ request. On the antipode, understandably clinicians appear hesitant and practice skepticism in embarking on novel proposed options, since data are often conflicting and lack robustness. Nonetheless, reports in literature describing case series and pilot studies communicate to the scientific community that clinical application of novel—albeit not yet having reached routine clinical application status—approaches may present a real option. It may not be uncommon for certain practices to invest in optimizing a new line of treatment on premature ovarian insufficiency. The variety of approaches including stem cells, platelet-rich plasma, ovarian tissue transplantation, formation of artificial gametes, even the latest emerging idea of mitochondrial replacement technique calls for specialization and expertise by clinics and practitioners. A critical assessment providing a comparative perspective between all novel approaches, regarding issues of importance to the practitioner from cost and practical aspects of application, to novelty, ethical concerns raised, and potential for acquiring future clinical routine practice status is provided in Table 7. Despite the variation considering the respective legal framework regarding application of novel approaches in ART, patients may have access to such treatments enabled by cross-border reproductive care 120 .
A Critical Assessment Providing a Comparative Perspective between All Novel Approaches, Regarding Issues of Importance to the Practitioner from Cost and Practical Aspects of Application to Novelty, Ethical Concerns Raised, and Potential for Acquiring Future Clinical Routine Practice Status.

+: very low; ++: low; +++: moderate; ++++: high; +++++: very high.
Staying true to their hippocratic oath “first do no harm” practitioners are invited to see beyond the hype and hope entailed in cutting-edge treatments. In lack of robust RCT data that could provide a safety net, and with publications on novel approaches overwhelming the field of ART, it is our obligation to question the effectiveness and thoroughly research these options. It is forming the apposite questions that will ascertain design of studies providing a consensus on these options. “Could the hormonal and physiological changes observed following application of novel approaches be truly attributed to the success of follicular re-recruitment?” To extrapolate on that concern, since ovarian function is programmed for termination, “how can we definitively follow and confirm the exact trail along with the cause and effect relationship between novel therapeutic intervention and the emerging improvement?” Despite the principles of physiological or pathological events and the respective unidentified molecular pathways leading to a compromised ovary, “could a successful application in restoring function translate to rewinding time?” “Could the resulting follicles and oocytes carry a distinctive element compromising their potential and rendering them present, but nonetheless of poor quality and potential?”
In order to properly assess these points of heated debate, certain guidelines should be proposed and established. The common denominator of all novel approaches in the service of overcoming ovarian insufficiency as critically presented herein lies in inconclusive but rather promising data. This very fact renders all of them to be at an experimental stage rather than of established clinical routine practice. Reestablishing fertility potential may be the main objective in published literature. Nonetheless, restoring fertile status following the diagnosis of ovarian insufficient may be defined by numerous criteria, enabled by various approaches, and confirmed employing different outcome measures depending on study design and population identity. Interestingly, and as shown herein, the perception of a successful outcome varies considerably amongst researchers. This may only be delineated by concrete studies examining outcomes on a clinical as well as on a basic research level. It is of high significance to highlight that all the approaches included herein have been proposed as promising techniques aiming to address the infertility related to ovarian insufficiency. Societies of Reproductive Medicine have issued in the past statements and guidelines, clearly shifting the status if a certain technique from being an experimental procedure to one classified as ascertaining the status of clinical routine practice. Such was the case for oocyte vitrification and warming 121 . Nonetheless, hitherto strong evidence is not in place for any of the presented techniques in order for the label of experimental procedure to be lifted. In the future, well-designed studies presenting clearly indicated outcome measures, limitations, and complications, while ascertaining reproducibility may accelerate drawing a safe conclusion on whether introducing these methods to the clinical routine practice set up would be beneficial or detrimental. However, understanding the outcome of such approaches presents one side of the coin. Distinguishing the profiles of patients who respond to certain approaches and to what extent these approaches restore their ovarian function remains to be delineated in this era of individualized medicine. The molecular mechanism involved during the implementation of the aforementioned novel treatments may only be unraveled by basic research studies that could hold the potential to conclusively address the question “for whom these novel approaches may perform efficiently.” While estimating the cost and effectiveness of the aforementioned approaches, one should not fail to consider the financial and psychological cost entailed—on an individualized basis—in addressing ovarian insufficiency when opting to employ what may be described as the conventional and traditional approach of oocyte donation. The field of medical practice has experienced the phenomenon of embarking on novel ideas solely on the grounds of limited-yet-promising data accompanying a novel technique covering the unchartered territory. It is important to be provided with published data conveying transparency to enable a fair and impartial assessment, as not all of them may be perceived as equally promising. Taking into account the fact that there is an increase in the cost of health care set up 122 extending to infertility treatment, evidence-based medicine in the field of ART is in the spotlight of research aiming to a more collected and bullet proof approach to manage pathologies such as ovarian insufficiency.
There may not be a superior method waiting to be discovered in addressing the issue of premature ovarian insufficiency especially in light of patient heterogeneity. Albeit novel available options may be constantly emerging, extreme caution should be practiced as they may be introduced lacking the platform of solid and well-designed studies that should buttress and confirm their value. Embracing novel approaches and acknowledging what drives their emergence is of pivotal importance. Faced with the promise of what presents as the holy grail in treating ovarian insufficiency one should fine-tune the sense of balance between practicing enthusiasm, patience, and caution. The reproduction endocrinology and infertility world will benefit from definitively connecting the dots between cause and effect, and concurring on safety and effectiveness while practicing the golden ratio between the patients’ desire for alternative treatment and providing them with a last resort option—one not surrounded by uncertainty—in the world of evidence-based medicine.
Footnotes
Acknowledgments
The present study was conducted as part of the M.Sc. program “Research in Female Reproduction” of Medical School, National and Kapodistrian University of Athens.
Authors’ Contributions
K.S., G.M., and M.S. conceived and designed the project. A.R., S.G., D.R., E.M., P.T., and P.G. performed the literature review. K.S., A.R., S.G., D.R., E.M., P.T., and P.G. contributed to drafting and editing the manuscript. K.P., N.V., G.M., M.K., and M.S. supervised the study and revised the manuscript. All authors approved the final draft.
Ethical Approval
Our institution does not require ethical approval for reporting review articles.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
