Abstract
Purpose:
Death domain–associated protein (DAXX) is a multifunctional nuclear protein involved in apoptosis, transcription, deoxyribonucleic acid damage response, and tumorigenesis. However, the role of DAXX in breast cancer development and progression remains elusive. In this study, we examined the expression patterns and function of DAXX in human breast cancer samples and cell lines.
Methods:
Immunohistochemistry was used to analyze the expression and localization patterns of DAXX. Additionally, we investigated whether DAXX played an intrinsic role in the cellular response to damage induced by ultraviolet (UV) irradiation in MDA-MB-231 breast cancer cells (isolated at M D Anderson from a pleural effusion of a patient with invasive ductal carcinoma).
Results:
Our results showed that nucleus size, chromatin organization, and DAXX localization were altered in breast cancer tissues compared with those in control tissues. Compared with cytoplasmic and nuclear expression in benign breast tissues, DAXX was colocalized with promyelocytic leukemia in nuclei with a granular distribution. Endogenous DAXX messenger ribonucleic acid levels were upregulated upon UV radiation in MDA-MB-231 cells. DAXX-deficient cells tended to be more sensitive to irradiation than control cells. Conversely, DAXX-overexpressing cells exhibited reduced phosphorylated histone H2AX (γ-H2AX) accumulation, increased cell survival, and resistance to UV-induced damage. The protective effects of DAXX may be related to the activation of the ataxia telangiectasia mutated (ATM)-checkpoint kinase 2 (ATM-CHK2)-cell division cycle 25c (CDC25c) signaling pathways in Gap2/Mitosis (G2/M) checkpoint and ultimately cell cycle arrest at G2/M phase.
Conclusions:
Taken together, these results suggested that DAXX may be an essential component in breast cancer initiation, malignant progression, and radioresistance.
Introduction
Breast cancer is the most frequently diagnosed malignancy and the second leading cause of cancer-related death among women 1 . Moreover, the incidence of breast cancer has been increasing in recent years, with higher rates of breast cancer occurring in younger patients 2 . Breast cancer is a heterogeneous disease characterized by gene mutations and dysregulation of numerous cellular pathways, such as pathways involved in cell cycle progression, angiogenesis, and apoptosis 3 . Treatments aimed at targeting such aberrant genes and pathways may play an important role in breast cancer therapy and could help improve clinical outcomes in patients with breast cancer. Death domain–associated protein (DAXX) was originally discovered as a proapoptotic protein that specifically bound to the death domain of the transmembrane receptor as tumor necrosis factor receptor superfamily member 6 (fas) in the cytoplasm and was later confirmed to have potential antiapoptotic activity 4 . DAXX has also been shown to be localized primarily in the nucleus at promyelocytic leukemia nuclear bodies (PML-NBs), also known as PML oncogene domains 5 , where it functions as a regulator of transcription. In recent years, accumulating evidence has suggested that DAXX expression is closely associated with tumorigenesis. For example, approximately 43% of pancreatic neuroendocrine tumors (PanNETs) have inactivating mutations in alpha thalassemia/mental retardation syndrome X-linked (ATRX) or DAXX genes 6 –8 .
The role of DAXX signaling in cancer diseases remains controversial, with data describing both repressing and promoting effects on cancer development. DAXX is reported to be overexpressed in human ovarian surface epithelial tumors, promote the proliferation of ovarian cancer cells, and protect ovarian cancer cells from x-ray irradiation and chemotherapy-induced deoxyribonucleic acid (DNA) damage 9 . Similarly, DAXX functions as a novel anaphase promoting complex (APC) inhibitor, is frequently overexpressed in prostate cancer, and may play roles in the development and progression of prostate cancer 10 . Recent studies have shown that DAXX acts to negatively regulate cancer growth and invasion. Shi et al. report works in triple negative breast cancer (TNBC) show DAXX as a modulator of DNA damage repair and suppressor of TNBC progression to sensitize tumors to the poly (ADP-ribose) polymerases(PARP) inhibitor by repressing DNA repair protein RAD51 homolog 1 (RAD51) functions 11 . DAXX is also verified to inhibit tumor recurrence in estrogen receptor-positive (ER+) breast cancer by recruiting DNA methyltransferase 1 (DNMT1) to methylate some promoters of stem genes and suppress tumor-initiating cells 12 . Moreover, investigation reveals that absence or depletion of DAXX protein increases cellular taxol resistance in breast cancer patients. DAXX functions as a trigger for taxol sensitization in combination with ras association domain family member 1 (RASSF1) and defines a mitotic stress checkpoint that enables cells to exit mitosis, together with the recently reported function of DAXX in the transcription repression of the prometastatic tyrosine kinase receptor c terminus of MNNG HOS transforming gene (c-MET) 13 . The heterogenetic roles of DAXX suggest the requirement to fully understand the role and the potential therapeutic implications.
Here, we investigated the expression, localization, and function of DAXX in human breast cancer tissues and cell lines upon ultraviolet (UV)-dependent induction of the DNA damage response.
Materials and Methods
Clinical Tissue Samples
Ninety-three paraffin-embedded tumor sections from patients who underwent mastectomy were provided by the Department of Breast Surgery at the Third Affiliated Hospital of Harbin Medical University (Harbin, China). The samples included 51 samples from patients with breast carcinoma and 42 benign breast tissues as controls (benign hyperplasia, n = 16; fibroadenoma, n = 26). The following clinical data were collected: patient age; estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) expressions; and lymph node status (Table 1).
Association of DAXX Expression with Clinicopathological Features in Human Breast Cancer.

DAXX: death domain–associated protein; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2.
Immunohistochemistry (IHC)
Paraffin-embedded tissue sections were heated at 60°C for 2 h, deparaffinized in xylene, and hydrated through a graded alcohol series. The sections were washed three times with phosphate-buffered saline (PBS), and endogenous peroxidase activity was quenched using 3% H2O2 for 20 min at room temperature. Antigen retrieval was performed by microwave treatment using 0.01 M citric acid buffer (pH 6.0), and samples were then cooled for 40 min at room temperature. The slides were washed three times with PBS, blocked with 5% bovine serum albumin (BSA) for 30 min at room temperature, and incubated overnight at 4°C with rabbit anti-DAXX monoclonal antibodies (1:150 dilution; Sigma-Aldrich, St. Louis, MO, USA) or anti-γ-H2AX (1:200 dilution; Cell Signaling Technology, Danvers, MA, USA). The sections were washed three times with PBS and incubated for 40 min with secondary antibodies. Slides were then stained with diaminobenzidine substrate, counterstained with hematoxylin, dehydrated, and mounted. Staining intensity and intracellular localization of DAXX were scored by pathologists.
Cell Culture
Michigan Cancer Foundation-7 (MCF-7), MDA-MB-231,and Sloan Kettering-Breast Cancer Research (SK-BR-3) human breast cancer cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, Grand Island, NY, USA). MDA-MB-231 cells were cultured in L-15 (Gibco) and supplemented with 10% fetal bovine serum (FBS; Hyclone, Biological Industries, Beit Haemek LTD, Israel) and 1% penicillin/streptomycin (P/S; Gibco). All cells were maintained in a humidified chamber at 37°C with 5% CO2.
Transfection
The pSRG (pSUPERretro-IRES-hrGFP) plasmid, DAXX overexpression (pLEGFP-DAXX, GFP-DAXX) plasmid, and DAXX-shRNA plasmid (pSuper-shDAXX, sh-DAXX) were kindly provided by Prof. Heng-Yu Fan (Life Science Institute, Zhejiang University). Nontargeting oligonucleotides (shCON) were designed and synthesized as vehicle of sh-DAXX. At 24 h before transfection, cells were seeded in 6-well plates at 0.5 × 105 cells/well for MCF-7 and SK-BR-3 cells or 0.8 × 105 cells/well for MDA-MB-231 cells in complete culture medium. The medium was changed to P/S-free DMEM or L-15 at 2–3 h before transfection, and cells were transfected with 500 µl Opti-MEM (Gibco), 3 µg plasmid, and 3 µl Lipofectamine LTX (Invitrogen, Carlsbad, CA, USA) per well, according to the manufacturer’s protocol. Stable transfected cells were selected with G418 (Sigma) for more than 2 weeks.
Ultraviolet Irradiation
Cells were washed with PBS and then removed from dishes prior to exposure to ultraviolet C (UVC) irradiation (254 nm) at intensities of 25 or 50 J/m2. Fresh medium was added to the culture immediately after irradiation. The radiated cells were reincubated in a humidified chamber at 37°C with 5% CO2.
Colony Formation Assays
At 48 h after transfection, cells were irradiated at 25 or 50 J/m2 for clonogenicity assays. After irradiation, cells were reincubated in fresh medium for 2 weeks. For colony formation assays, stably transfected cells selected using G418 (800 mg/ml) were trypsinized, counted, seeded into new 6-well culture plates (1 × 104 cells/well), and incubated for 2 weeks. Surviving colonies (≥ 50 cells per colony) were counted and photographed after staining with crystal violet. Each experiment was performed three times.
Wound Healing Assays
Stably transfected MDA-MB-231 cells were seeded in six-well plates in complete L-15 medium and cultured overnight. The medium was changed to FBS-free L-15, and cell monolayers were scraped in a straight line using a 200-µl pipette tip. The “wound” was then photographed at 0, 12, and 24 h using a phase-contrast inverted microscope (Nikon TE2000-U, Tokyo, Japan). To better understand the role of DAXX in MDA-MB-231 cells, we analyzed the tumor migration rate in GFP-DAXX and sh-DAXX cells. Each experiment was performed three times.
RNA Extraction and Quantitative Reverse Transcription Polymerase Chain Reaction (qPCR)
Total ribonucleic acid (RNA) was extracted from cultured cells using TRIzol reagent (Ambion, Carlsbad, CA, USA) and reverse transcribed using a High Capacity cDNA Reverse Transcription Kit (ABI, Foster City, CA, USA). qPCR was carried out with 1 µl cDNA, 10 µl TransStart Top Green qPCR SuperMix (TransGen Co. Ltd, Beijing, China), and primers in 20-µl reaction volume on a CFX96 Real-Time Thermal Cycler (Bio-Rad, Hercules, CA, USA). The following primer pairs were used: 5′-GATACCTTCCCTGACTATGGGG-3′ and 5′-GTAACCTGATGCCCACATCTC-3′ for DAXX; 5′-CCATCGTCCACCGCAAAT-3′ and 5′-GCTGTCACCTTCACCGTTCC-3′ for ACTIN. Relative expression was calculated after normalization to h-actin. Samples were run in triplicate.
Western Blotting
Cells were harvested using lysis buffer with 1% phenylmethylsulfonyl fluoride (PMSF), and the protein was collected by centrifugation at 17,418 × g for 30 min. Protein extracts (20–30 μg) were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes (Millipore, Eppendorf, MA, USA). Blots were blocked with 5% nonfat milk in PBS-Tween20 (PBS-T) for 2 h and incubated overnight at 4°C with primary antibodies targeting DAXX (1:1,000 dilution; Sigma-Aldrich), γ-H2AX/phosphorylated CDC25c (p-CDC25C)//CDC25c)/phosphorylated CDC2 (p-CDC2)/β-TUBULIN (1:1,000 dilution; Cell Signaling Technology), or
Immunofluorescence
Cells were seeded in 24-well plates, treated, washed with PBS containing 0.25% TritonX-100, fixed with 4% paraformaldehyde at 4°C for 20 min followed by ice-cold 75% ethanol for 5 min, and then maintained in blocking buffer (PBS containing 0.25% TritonX-100 and 2% BSA). Next, cells were incubated overnight at 4°C with primary antibodies targeting h-DAXX (1:150 dilution), PML (1:50 dilution; Santa Cruz), or γ-H2AX/phosphorylated CHK2 (p-CHK2, 1:200 dilution). After washing with PBS-T, cells were incubated with secondary antibodies (antigoat Alexa 488-conjugated antibodies or antirabbit Alexa 546-conjugated antibodies; 1:200 dilution; Santa Cruz) for 1 h at room temperature. Nuclei were counterstained with Hoechst (1:100 dilution) for 2 min at room temperature. Slides were analyzed immediately with a fluorescence microscope (Nikon).
Statistical Analysis
Statistical comparisons in Tables 1 and 2 were analyzed by χ2 tests, and a P-value less than 0.05 represents a significant difference. Other statistical comparisons of mean values were performed using Student’s t-tests. Differences with P-values of less than 0.05 were considered statistically significant.
Summary of DAXX Expression and Localization in Human Breast Tumor Tissues.

N: nucleus; C: cytoplasm.
Results
DAXX Expression Varied in Human Breast Tumor Tissues
First, we examined the expression and localization patterns of DAXX in human breast tumors using IHC. We found that DAXX was ubiquitously expressed in benign and malignant tissue lesions, and heterogeneous immunolabeling for DAXX was observed within each individual sample (Fig. 1A). However, there were no significant differences in DAXX levels between benign and malignant breast tissue specimens (P > 0.05; Table 2). Notably, in benign breast tissues, DAXX was localized in both the nucleus and the cytoplasm, showing a diffuse or patchy distribution with small, condensed nuclei (Fig. 1B, a and b), whereas that in breast cancer tissues showed nuclear staining only, with a granular distribution and large, open nuclei with prominent, visible nucleoli (Fig. 1B, c and d). Thus, the distribution and expression patterns of DAXX in breast cancer tissues differed from those in benign breast tissues. Furthermore, analysis of DAXX levels and clinicopathological features in 51 malignant breast tissues (Table 1) showed that there were no significant correlations between DAXX expression levels and patient age or tumor pathological characteristics (size, ER/PR/HER2 expression, and lymph node status). Taken together, these results suggested that DAXX localization, rather than DAXX expression, may play an important role in the progression of breast cancer.
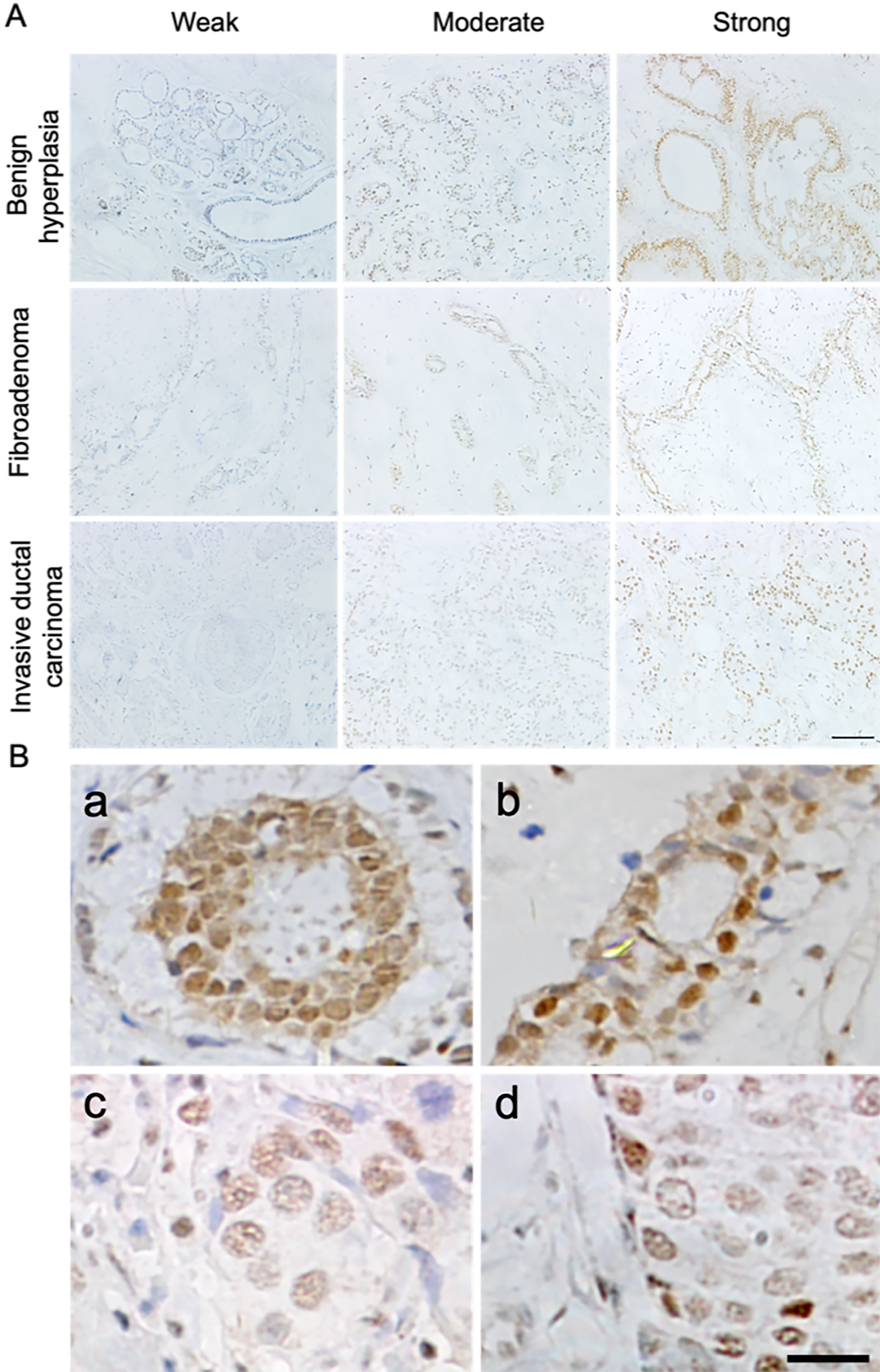
Immunohistochemical expression and localization of DAXX in human breast tumor samples. (A) Heterogeneous immunostaining for DAXX was observed in human benign and malignant breast tissue lesions. Scale bar = 100 μm. (B) Immunochemistry results for the localization of DAXX in human breast tumor sections (a: benign breast hyperplasia; b: breast fibroadenoma; c: breast medullary carcinoma; d: breast simplex carcinoma). Scale bar = 20 μm. DAXX: death domain–associated protein.
Patterns of DAXX Expression in Human Breast Cancer Cell Lines
The expression and subcellular localization of DAXX were also examined in breast cancer cell lines. As expected, the localization patterns in breast cancer cells were consistent with those in breast cancer specimens (supplemental Figure S1A). DAXX was mainly localized to PML-NBs with a discrete speckled nuclear pattern and no cytoplasmic staining. Importantly, no significant differences in DAXX expression levels were observed by qPCR and western blotting among several breast cancer cell lines (supplemental Figure S1B–D).
For further analysis of the effects of DAXX in breast cancer cells, we used lipofectamine (LTX) transfection reagents to overexpress or silence DAXX in breast cancer cells. We found that the green fluorescent protein (GFP)-DAXX and GFP-shRNA DAXX (sh-DAXX) ectopic fusion proteins were predominantly localized in the nucleus (Fig. 2A) and that endogenous DAXX was effectively overexpressed and silenced, respectively, following transfection (Fig. 2B–E).

DAXX expression in human breast cancer cell lines. (A) Immunofluorescence analysis of the localization of DAXX in cultured human breast cancer cells transfected with GFP-DAXX or sh-DAXX (green). Nuclei were stained with Hoechst (blue). Scale bar = 20 μm. (B) Western blot analysis of the effects of DAXX overexpression. (C) The data shown are representative of relative protein as compared with β-tubulin. *P < 0.05. (D and E) qPCR and western blot analysis of the effects of DAXX depletion. Total RNA and protein were isolated at 48 and 72 h, respectively, after transfection. *P < 0.05; **P < 0.01. DAXX: death domain–associated protein; GFP: green fluorescent protein; qPCR: quantitative reverse transcription polymerase chain reaction; shCON: cells transfected with control shRNA; shRNA: cells transfected with DAXX shRNA; RNA: ribonucleic acid.
DAXX Promoted Cell Proliferation and Metastasis in Breast Cancer Cell Lines
To further investigate the potential effects of DAXX in breast cancer cells, we performed colony formation assays in MDA-MB-231 cells stably transfected with vector, GFP-DAXX, or shRNAs targeting DAXX. As shown in Fig. 3A, B, compared with vector-transfected cells, overexpression of DAXX markedly increased colony formation abilities, whereas DAXX depletion decreased colony formation in tumor cells. Next, the effects of DAXX expression on cell motility at 0 and 24 h were assessed using wound healing assays. We found that cell migration was significantly inhibited by DAXX depletion, and the cells were unable to migrate to fill the wounded area, even at 24 h after scraping (Fig. 3C).

DAXX promoted colony formation and migration in breast cancer cells. (A) Colony formation assays following transfection with vector, GFP-DAXX, or sh-DAXX in MDA-MB-231 cells. Colony numbers were counted after 2 weeks (> 50 cells per clone). Scale bar = 500 μm. (B) Quantification of average colony numbers of MDA-MB-231 cells and their derivatives (GFP-DAXX and sh-DAXX cell lines). *P < 0.05, **P < 0.01. (C) Wound healing assays in MDA-MB-231 cells transfected with GFP-DAXX or sh-DAXX cultured in FBS-free medium. Scale bar = 100 μm. DAXX: death domain–associated protein; sh: shRNA; shRNA: cells transfected with DAXX shRNA; GFP: green fluorescent protein.
DAXX Protected Breast Cancer Cells from UV-Induced Damage
Next, we examined whether DAXX was involved in the cellular response to UV-induced damage by measuring endogenous DAXX expression in MDA-MB-231 cells after UVC radiation (50 J/m2). We found that DAXX messenger-RNA (mRNA) was upregulated after irradiation, and this elevated expression persisted for at least 12 h following irradiation with 50 J/m2. However, DAXX protein expression was not upregulated as expected (Fig. 4A, B), suggesting that UVC irradiation induced DAXX expression at the transcriptional level but not at the translational level. The localization of DAXX upon UVC was then analyzed by immunofluorescence staining (Fig. 4C). Interestingly, following UVC irradiation, DAXX remained localized in the nucleus.

DAXX reduced the degree of UV-induced DNA damage. (A) Changes in endogenous DAXX mRNA levels following exposure to UV radiation. Cells were irradiated at 50 J/m2 and harvested at the indicated times after irradiation. Control cells were not irradiated. Total RNA from the cells was analyzed by qPCR. (B) Western blot analysis showing endogenous DAXX protein levels after UV irradiation. β-Tubulin was used as a loading control and for normalization. (C) Immunofluorescence results showing the localization of DAXX after UV irradiation. DAXX (red), PML (green), nuclei (blue). Scale bar = 20 μm. (D) Immunofluorescence staining of γ-H2AX or p-CHK2 in MDA-MB-231 cells with or without UV irradiation. Scale bar = 10 μm. (E) Immunofluorescence staining results showing the level of γ-H2AX in GFP-DAXX- and sh-DAXX-positive cells with or without UV irradiation. Green: GFP-DAXX- or sh-DAXX-positive cells, red: γ-H2AX staining, blue: Hoechst staining. Scale bar = 10 μm. (F) Western blot results showing the levels of γ-H2AX in control (CON), GFP-DAXX, and sh-DAXX cells after UV irradiation. The irradiated cells were harvested at 30 and 60 min after UV treatment. (G) Western blot analysis for the levels of γ-H2AX in the GFP-DAXX and control groups following irradiation with different doses (0, 25, or 50 J/m2). All cells were harvested at 60 min after UV treatment. (H) Immunochemistry analysis of DAXX and γ-H2AX in human breast invasive ductal carcinoma sections. a, b, and c show high, moderate, and low expressions of DAXX, respectively; d, e, and f show low, moderate, and high expressions of γ-H2AX, respectively. DAXX: death domain–associated protein; UV: ultraviolet; DNA: deoxyribonucleic acid; RNA: ribonucleic acid; mRNA: messenger RNA; GFP: green fluorescent protein; qPCR: quantitative reverse transcription polymerase chain reaction; sh-DAXX: cells transfected with DAXX shRNA; PML: promyelocytic leukemia.
We then examined the association of DAXX expression with the cellular response to UV damage. Previous studies have shown that H2AX expression and CHK2 phosphorylation are key events in the DNA damage response and markers of double-stranded breaks 10 . As shown in Fig. 4D, based on immunofluorescence staining for γ-H2AX and phospho-Chk2, severe cellular damage was observed in MDA-MB-231 cells within 30 min after irradiation. In addition, γ-H2AX expression was obviously reduced at 30 min after irradiation in DAXX-overexpressing cells compared with that in control cells. Conversely, γ-H2AX expression was increased in cells depleted of DAXX expression as compared with that in adjacent untransfected control cells (Fig. 4E). As expected, western blot analysis also showed that DAXX overexpression suppressed UVC-induced γ-H2AX accumulation at all time points tested (Fig. 4F, G).
Further exploration of DAXX and γ-H2AX levels in breast invasive ductal carcinoma tissues by IHC showed that tissue specimens with higher levels of DAXX expression exhibited reduced γ-H2AX staining, whereas tissues with lower DAXX expression exhibited higher γ-H2AX accumulation (Fig. 4H). Taken together, our results suggested that DAXX protected breast cancer cells from DNA damage upon UVC irradiation.
DAXX Overexpression Enhanced Cellular Resistance to UV Irradiation in Breast Cancer Cells
To determine the effects of DAXX expression on cell survival upon UV stress, the irradiated cells were subjected to colony formation assays after UV exposure. As depicted in Fig. 5A, B, overexpression of DAXX increased colony numbers, whereas DAXX knockdown dramatically sensitized MDA-MB-231 cells to both doses of UVC irradiation compared with vector control cells. These results demonstrated that MDA-MB-231 cells expressing GFP-DAXX exhibited greater resistance to UVC irradiation.

DAXX increased cellular resistance to UV irradiation. (A) Cell survival ability of MDA-MB-231 cells and their derivatives (GFP-DAXX and sh-DAXX cells) after UV irradiation, as determined by clonogenic assays. (B) Quantification of average colony numbers of MDA-MB-231 cells and their derivatives (GFP-DAXX and sh-DAXX cells) after irradiation with different doses. *P < 0.05. (C) Western blot analysis showing the levels of G2/M checkpoint proteins after UV irradiation in MDA-MB-231 cells. (D) Western blot analysis of the expression of cell apoptosis–related proteins in MDA-MB-231 cells after UV irradiation. β-Tubulin was used as a loading control. DAXX: death domain–associated protein; UV: ultraviolet; sh-DAXX: cells transfected with DAXX shRNA; p-CDC25c: phosphorylated CDC25c; p-CDC2: phosphorylated CDC2; BCL2: B-cell lymphoma 2; BAX: BCL2 associated X; GFP: green fluorescent protein.
DAXX Altered the G2/M Transition and Decreased Apoptosis Rates
The increases in cell survival and clonogenicity following DAXX overexpression could be associated with the induction of cell cycle arrest or apoptosis. Cells are most sensitive to irradiation during the G2/M phase, and G2/M-phase arrest is a major cause of cell death induced by irradiation or anticancer drugs 14 . Thus, we examined the expression levels of proteins associated with G2/M cell cycle checkpoints and apoptosis to identify the possible mechanisms underlying the observed prosurvival effects of DAXX. First, we analyzed the expression and activity of CDC25c and CDC2 kinase, which are involved in the transition between the G2 phase and mitosis. As shown in Fig. 5C (supplemental Figure S2A–D), compared with the vector control cells, MDA-MB-231 cells overexpressing DAXX showed decreases in CDC25c-Ser216 and CDC2-Tyr15 phosphorylation on inhibitory sites. Conversely, in DAXX-knockdown cells, the protein phosphorylation levels were increased compared with those in control cells (shCON), suggesting increased arrest at the G2/M boundary. Thus, we concluded that DAXX was necessary and sufficient for cell cycle progression through the G2/M boundary following UV irradiation.
We next investigated the expression of Bcl-2 and Bax, which play pivotal roles in apoptosis. As shown in Fig. 5D (supplemental Figure S2 E–G), DAXX overexpression resulted in upregulation of Bcl-2 and downregulation of Bax relative to the expression in control cells. Conversely, in DAXX-knockdown cells, the opposite results were observed following UV damage. These results, together with the results shown in Fig. 5C, implied that the protective effects of DAXX against UV-induced damage may be mediated through promotion of the G2/M transition and decreasing apoptosis.
Discussion
In this study, we examined the expression, localization, and function of DAXX in breast cancer cells and tissues. Our results demonstrated that DAXX was differentially localized in breast cancer cells and tissues relative to that in control samples. Moreover, DAXX played a role in cancer-related cell processes, including cell proliferation and migration. These results provide important insights into the role of DAXX in breast cancer.
Previous studies have suggested that DAXX functions mainly in apoptosis and transcription. Moreover, accumulating evidence has revealed that DAXX has an additional role in the pathogenesis of multiple types of cancer, including prostate cancer 10 , ovarian cancer 9 , PanNETs 6 –8 , lymphoma 15 , and neuroblastoma 16 . In many of these studies, DAXX protein levels have been shown to be upregulated or downregulated depending on the cancer type. However, in our study, there were no significant differences in DAXX protein levels in breast cancer tissues compared with that in benign breast tissues. Interestingly, we found that nucleus size, chromatin organization, and DAXX localization were different in breast cancer tissues relative to those in control tissues. Previous reports have demonstrated that cancer cells generally display aberrant epigenetic events, with genomic DNA hypomethylation and promoter hypermethylation of certain genes 17,18 . Nucleus size primarily depends on the DNA content and the status of chromatin organization, the latter of which is associated with epigenetic events, such as DNA methylation and histone modifications, which are precisely regulated by the chromatin remodeler DAXX. Indeed, DAXX is a predominantly nuclear protein that is often located within PML-NBs and regulates chromatin organization status 19,20 . Moreover, DAXX has been shown to possess transcriptional repression activity through direct protein–protein interactions and recruitment of DNMTs, histone deacetylases, or chromatin-modifying factors to promoters of target genes 21,22 . Furthermore, recent studies in multiple types of cancer tissues have linked the expression of DAXX to tumor aggressiveness in patients with cancer 8,23 . Importantly, our in vitro results showed that DAXX promoted the proliferation and migration of breast cancer cells. These results, together with our findings of altered nucleus size, chromatin organization, and DAXX localization in breast cancer tissues, demonstrated the potential role of DAXX in the carcinogenesis and malignant progression of breast cancer.
Accumulating evidence has shown that DAXX is localized in both the nucleus and the cytoplasm and has dual functions. Nuclear DAXX is believed to participate in transcriptional regulation, whereas cytoplasmic DAXX is reported to act as a proapoptotic protein involved in FAS-induced apoptosis. In addition, DAXX has been shown to exhibit dynamic intracellular translocation 24 . However, it is still unclear whether DAXX is exported from the nucleus to the cytoplasm or remains exclusively in the nucleus upon exposure to stress stimuli, and cell line– or stimuli-dependent phenomena may exist. In this study, we found that DAXX localization was not altered by UVC exposure, suggesting that DAXX may have functions in transcriptional regulation, rather than in the cytoplasmic apoptotic response, in breast cancer cells after UV irradiation.
In addition, we also found that endogenous DAXX mRNA expression was upregulated soon after exposure to UV irradiation; however, total protein levels were not elevated as expected, consistent with a microarray experiment showing that DAXX mRNA is upregulated in breast cancer cells upon exposure to curcumin relative to that in mammary epithelial cell lines 25 . Moreover, a previous study showed that DAXX is phosphorylated rapidly to induce p53 activation in response to DNA damage through the ataxia telangiectasia mutated(ATM)/Ataxia telangiectasia and Rad3 related (ATR) signaling pathway 26 . Therefore, we postulated that DAXX may play an intrinsic role in the DNA damage-induced response depending on the level of phosphorylation. Our results also suggested that DAXX depletion resulted in cell death and elevated UV-induced DNA lesions in breast cancer cells. These results were further verified by the increase in γH2AX accumulation in human breast ductal carcinoma tissues exhibiting lower DAXX levels. Taken together, these results strongly indicated that DAXX was important for maintaining genome stability and suppressing DNA damage-induced cell death.
In general, cell cycle regulation is regarded as the most important determinant of radiosensitivity. The cell cycle is greatly affected upon irradiation by activating cell cycle checkpoints, and relative radiation sensitivity mainly depends on the cell cycle phase, with irradiated cells being most sensitive during G2/M phase, less sensitive in G1 phase, and least sensitive in S phase 14 . Therefore, cell cycle arrest at G2/M is the major reason for cell death induced by irradiation or anticancer agents. As already noted, CDC2 and CDC25c kinases, which are required for cell cycle progression into mitosis, function as key checkpoint proteins in the DNA damage-induced G2/M signaling cascade pathway. When DNA damage occurs in cells, the activity of CDC25c kinase is inhibited through phosphorylation of Ser216, which is moderated by CHK1/2 kinases 27 . Because of the inactivity of CDC25c, CDC2 kinase fails to be dephosphorylated at Thr15 by functional CDC25c and cannot enter into mitosis owing to the block in CDC2/cyclin B kinase activation; this finally leads to G2/M arrest 28 . In this study, we showed that the levels of CDC25c and CDC2 phosphorylation were decreased in DAXX-overexpressing cells and increased in DAXX-knockdown cells compared with those in control cells, suggesting that DAXX-overexpressing cells have a shorter G2/M delay, that is, DAXX depletion can exacerbate G2/M arrest. Additionally, a previous study showed that a longer G2/M delay was induced by radiation in radiosensitive cell lines compared with that in matched normal or resistant cells 29,30 . Therefore, we propose that DAXX depletion may arrest cells in the radiosensitive G2/M phase of the cell cycle, causing breast cancer cells to be more sensitive to UV irradiation and subsequently promoting apoptosis.
In conclusion, our IHC analysis showed altered expression patterns of DAXX in breast cancer tissues compared with benign breast tissues. Additionally, DAXX was shown to play an important role in the cellular response to UV-induced DNA damage and to exert protective effects through moderation and restoration of cell cycle arrest at G2/M. However, its exact role in the regulation of G2/M arrest and apoptosis has not been well studied and remains unclear. Our future challenges will focus on the generation of suitable animal models to be used to explore DAXX functions in vivo.
Supplemental Material
Supplemental_Fig.S2 - Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint
Supplemental_Fig.S2 for Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint by Zhiyan Shan, Li Liu, Jingling Shen, Haiyue Hao, Honghong Zhang, Lei Lei, Feng Liu and Zhipeng Wang in Cell Transplantation
Supplemental Material
Supplemental_Fig.S3 - Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint
Supplemental_Fig.S3 for Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint by Zhiyan Shan, Li Liu, Jingling Shen, Haiyue Hao, Honghong Zhang, Lei Lei, Feng Liu and Zhipeng Wang in Cell Transplantation
Supplemental Material
Supplementary_Fig.S1 - Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint
Supplementary_Fig.S1 for Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint by Zhiyan Shan, Li Liu, Jingling Shen, Haiyue Hao, Honghong Zhang, Lei Lei, Feng Liu and Zhipeng Wang in Cell Transplantation
Supplemental Material
Supplemental Material, Title_and_description_for_all_supplemental_material - Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint
Supplemental Material, Title_and_description_for_all_supplemental_material for Enhanced UV Resistance Role of Death Domain–Associated Protein in Human MDA-MB-231 Breast Cancer Cells by Regulation of G2 DNA Damage Checkpoint by Zhiyan Shan, Li Liu, Jingling Shen, Haiyue Hao, Honghong Zhang, Lei Lei, Feng Liu and Zhipeng Wang in Cell Transplantation
Footnotes
Acknowledgments
We appreciate Dr. Hengyu Fan for providing the vector described in this paper.
Ethical Approval
This study was fully concerned with relevant ethical principles and codes in the research design. The isolation of breast tissue samples from patients and the usage of specimens for studying were approved by the Institutional Research Board of Harbin Medical University.
Statement of Informed Consent
The breast tissue samples were obtained with donors’ written informed consent.
Statement of Human and Animal Rights
The procedures and the experiments we’ve performed on humans respected the ethical standards in the Declaration of Helsinki and other international regulations. This article does not contain any studies with animal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant nos. 81772970, 31671252) and the Scientific Research Startup Fund of Heilongjiang Postdoctoral Program (grant no. LBH-Q19149).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
