Abstract
Bilirubin is a natural cytoprotective agent and physiologic doses have proven to be beneficial in various models of organ and cellular transplantation. Recently, we showed that bilirubin has protective effects in models of pancreatic islet transplantation, preventing cell death associated with islet stress and suppressing the release of damage-associated molecular patterns. Despite these promising therapeutic attributes, the natural bilirubin used in these research studies is animal-derived (porcine), making it unsuitable for clinical application. In the current study, we synthesized two bilirubin analogs that can be produced without the use of animal-derived products. Antioxidant activity for the analogs was measured using the ferric-reducing-ability-of-plasma (FRAP) and 2,2V-azinobis(3-ethylbenzothiazoline-6-sulfonate) (ABTS) assays. Dose-dependent cytotoxicity and cytoprotective effects were then demonstrated in isolated rat islets. Compound
Introduction
Diabetes mellitus (DM) is a debilitating and progressive disease that has profound effects not only on the affected individual, but on the entire healthcare system 1 . Treatment with insulin injections is tedious and fails to replicate normal physiology and affected patients are at risk of developing progressive hypertension, vascular complications, kidney disease, and blindness 1 – 3 . Pancreatic islet cell transplantation offers a potential noninvasive cure for patients with type 1 diabetes mellitus (T1DM), although application of this therapy has been limited because of a significant loss of islet cell mass and function occurring after implantation4,5. A prolonged period of ischemia occurs from the time of donor harvest of islet cells until the point of revascularization within the recipient. Hypoxemia suffered by the islet cells during the transplantation process results in the formation of numerous reactive oxygen species, ultimately causing cellular necrosis and apoptosis via intracellular signaling pathways. Beta cells are particularly vulnerable to free radical injury due to relatively weak activity of catalase, superoxide dismutase, and glutathione peroxidase in these cells 5,6 , making cell death following islet isolation a critical hurdle to successful islet transplantation in patients with T1DM.
Induction of the heme oxygenase-1 (HO-1) system during various forms of cellular insult and injury is an important endogenous cytoprotective mechanism against oxidative stress, and upregulation of this system in transplanted islet cells has shown improved antiapoptotic effects and in vivo function 7 –9 . Biliverdin, one of the three byproducts of the breakdown of heme by heme oxygenase, is rapidly reduced to bilirubin via biliverdin reductase in what is thought to be an evolutionary adaptation to produce this cytoprotectant during cell stress 10 . Bilirubin has been shown to be an important mediator of cytoprotection via powerful endogenous antioxidant activity and anti-inflammatory effects 7,11 –13 . Bilirubin has not only been shown to provide potent cytoprotection of transplanted islets via induction of HO-1 and significant scavenging of free oxygen and nitrogen radicals, but it has also been shown to suppress the innate immune system resulting in improved donor tolerance to islet grafts 14 –16 .
Although bilirubin has been established as a promising adjuvant therapy to survival of islets during the transplantation process, continued research into the value of bilirubin as a pharmaceutical agent has been hindered by the fact that bilirubin is currently only available as a naturally derived compound from porcine sources 17 . Strict federal regulations placed on medical devices that are composed of, or are exposed to, animal-derived materials has deterred scientists from pursuit of compounds such as bilirubin for medicinal use 18 . Synthesis of novel compounds, structurally similar to bilirubin, could serve as candidates for safely sourced drugs with similar cytoprotective effects to natural bilirubin, without the concerns and limitations associated with procurement from a live animal source.
The objective of this study was to design and synthesize simplified structural fragments of bilirubin and to screen the candidate compounds for relative antioxidant activity using standard laboratory methods. Selected compounds were then evaluated for dose-dependent cytotoxic effects in isolated murine islets. Finally, in vitro antioxidant and cytoprotective effects were assessed in a relevant model of hypoxic stress. We hypothesized that successful development and synthesis of such a compound could be achieved and that cytotoxicity in islet cells would be dose-dependent and similar to cytotoxicity seen with natural bilirubin. We further hypothesized that the antioxidant and cytoprotective effects of these analogs would be similar to natural bilirubin.
Materials and Methods
Development and Synthesis
As an initial evaluation as to whether the effects of bilirubin could be mimicked by simplified bis-pyrroles, we targeted Compounds
Briefly, synthesis of Compound
Compound
Structure and purity of both bis-pyrrole compounds were confirmed via 1H nuclear magnetic resonance spectroscopy using established methods 19 . Complete compound characterization data for each compound is provided in the supplemental information for this manuscript.
Relative Antioxidant Screening
Following synthesis, candidate molecules were screened for relative antioxidant activity and compared to natural bilirubin as well as to standard antioxidant assay controls (quercetin and Trolox) using the ferric reducing ability of plasma (FRAP) assay and the 2,2V-azinobis(3-ethylbenzothiazoline-6-sulfonate) (ABTS) radical cation assay. Stock solutions of each compound were prepared for both assays and concentrations of 800, 600, 400, 300, 200, and 100 μmol were produced by dilution of the stock solution with 100% ethanol. Briefly, stock solutions were prepared for each compound as follows: Bilirubin (Sigma-Aldrich, St. Louis, MO, USA)—5.8 mg was suspended in 1 ml 50% ethanol in water + 10 µl 2 M NaOH, the mixture was sonicated for complete dissolution, then 100% ethanol was added to a final volume of 10 ml; Compound
FRAP Assay
The FRAP assay was completed evaluating all bis-pyrrole compounds following guidelines as previously reported 20 . The FRAP assay is based on electron-donating antioxidants that reduce ferric-tripyridyltriazine (Fe3+ TPTZ) complex, a colorless compound, to ferrous complex (Fe2+-TPTZ), resulting in the development of an intense blue color with an absorption maximum at 593 nm. FRAP reagent was prepared by mixing acetate buffer, TPTZ solution, and FeCl3·6H2O and then prewarmed to 37°C prior to use in the assay 20 . Sample stock solutions were prepared as above. Reagent–sample reaction mixtures were created in a 96-well microplate and were incubated at 37°C. Absorbance was measured at 593 nm and subtracted from the absorbance of the buffer-only blank well after 2 h.
ABTS Radical Cation Assay
The ABTS assay was performed on all bis-pyrrole compounds using previously reported methods 21 . ABTS solution was combined with 2.45 mmol/l potassium persulfate to produce colored 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) radical cation (ABTS·+). Dilution of the deep green ABTS·+ solution with ethanol to an absorbance of 0.7 at 734 nm was performed. During the assay, the ABTS·+ is decolorized by antioxidants according to their concentrations and antioxidant capacities. Stock solutions of antioxidant test bis-pyrrole compounds (as above) were mixed with oxidized ABTS·+ and decolorization of the samples (absorptance) was measured at 734 nm, 10 min following reagent–sample mixing.
Biological Testing
Based on the strong performance of Compound
Islet Isolation
Experiments were approved by the Institutional Animal Care and Use Committee at North Carolina State University and were performed according to the NIH Guidelines for Animal Care and Use. Wistar rats were purchased from Charles River Laboratories, Wilmington, NC, USA. Rats were euthanized using CO2 inhalation and harvest of islet cells was performed immediately after confirmation of death. Pancreatic islets were isolated using the standard enzymatic digestion methods described by Zmuda et al. 22 Digestion of pancreatic tissue was performed by injection of 5 ml of 2 mg/ml Collagenase IV (Sigma-Aldrich) into the common bile duct. Pancreata were maintained on ice until digestion was initiated by immersion in a 37°C water bath for 17 min. Islet purification was performed using a Ficoll density gradient separation technique, and islet yield was calculated before plating in standard RPMI 1640 media (Invitrogen, Carlsbad, CA, USA) with 10% fetal bovine serum and 1% Pen-Strep (Invitrogen, Carlsbad, CA, USA). Islets were then incubated at 37°C and 5% CO2 for 24 h to allow stabilization after isolation stress. Experiments were performed in triplicate, and for each experiment, islets from a single rat were hand-picked and divided into separate plates to allow comparison of different compounds and at different concentrations.
Cytotoxicity
Following post-isolation incubation, islets were washed and replated with 300 islets per 35 mm2 well. Islets were then suspended in 3 ml of media containing either Compound
Hypoxic Challenge
To assess biologic effects following hypoxic injury, further rat islets were isolated and incubated overnight under standard conditions. Islets were then hand-selected, and aliquots of 300 islets were distributed into individual wells of 12-well cell culture plates. Individual wells (n = 3) of islets were assigned to one of five treatment groups; Compound
Islet Cell Viability
Islet cell death following both cytotoxicity and oxidative challenge experiments was determined based on propidium iodide (PI) exclusion. Islets were incubated for 15 min with Hoechst 33258 and PI stains followed by examination via epifluorescent photomicroscopy. Images were analyzed using NIH Image J software, and the percentage of PI-positive cells present in each islet, indicating % cell death in each individual islet, was calculated using a custom islet macro as previously described 24 .
Statistical Analysis
Islet viability data were compared between groups using a one-way analysis of variance and post hoc pairwise comparisons were made using Tukey’s honestly significant difference test. StatView software was utilized to calculate all statistics (SPSS Inc., Chicago, IL, USA). P-values of ≤0.05 were considered statistically significant.
Results
Synthesis
The synthesis and purification of bis-pyrrole compounds

Chemical equation for the synthesis of Compound 1, a simple bis-pyrrole with a system of conjugated double bonds and potentially reactive hydrogen atoms, similar to tetra-pyrrole, bilirubin. Calculated MW for C16H23ON2 [M + H]+ is 259.1805, measured MW 259.1796.

Chemical equation for the synthesis of Compound
FRAP
Results of antioxidant activities based on the FRAP assay are illustrated in Fig. 3. Order of antioxidant activity of tested compounds from highest to lowest activity was quercetin > bilirubin > Compound
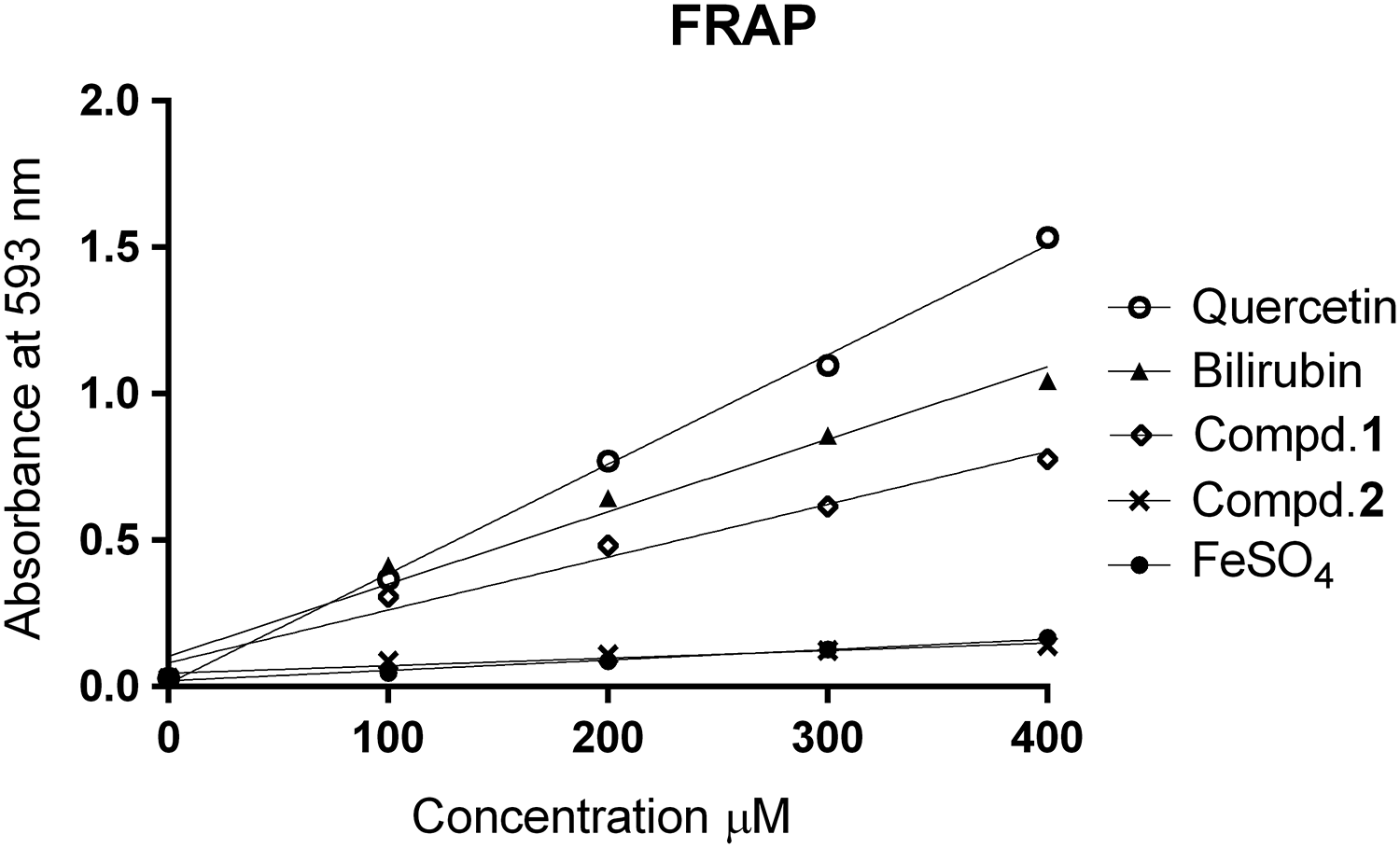
Antioxidant capacity of Compound
ABTS
The antioxidant activity results based on the ABTS assay are illustrated in Fig. 4. Antioxidant effect was similar to that demonstrated in the FRAP assay results and antioxidant activity from highest to lowest was as follows: quercetin > bilirubin > Compound

Antioxidant activity of Compound
Islet Viability Following Cytotoxicity
Dose-dependent cytotoxicity was evident in murine islets in both treatment groups with significant increases in percent cell death seen with natural bilirubin and Compound

Islet cell death (%) following treatment with increasing concentrations (μM) of Compound
Islet Viability Following Hypoxic Injury
Overall, rat pancreatic islets subjected to hypoxic injury showed significantly higher survival (P < 0.0001) when treated with Compound

Islet cell death (%) following hypoxic injury and treatment with Compound
Discussion
Bilirubin has been established as a powerful endogenous cytoprotectant, mediating improved outcomes in several models of health and disease including ischemia reperfusion injury, endothelial damage, and myocardial infarction, as well as solid organ transplantation via significant antioxidant activity and immunomodulatory effects
7,12
–16,25
–27
. However, our study is the first to evaluate the feasibility of using a synthetic, significantly simplified, bilirubin analog, which could provide a safe, sterile, easily mass-produced drug for use in islet cell transplantation while avoiding the shortcomings of bilirubin procured from an animal source. We were able to successfully synthesize a novel bis-pyrrole compound and demonstrate that Compound
The FRAP and ABTS*+ assays are established methods of evaluating antioxidant activity of complex biologic substances, such as those found in plasma, and the ABTS*+ assay has been used previously to confirm the powerful antioxidant capacity of bilirubin
20,21,28
. Consistent with previous reports, natural bilirubin was found to have robust antioxidant activity based on these methods, and, encouragingly, the antioxidant activity of Compound
Quercetin, a flavonoid with known vigorous antioxidant activity, was used as a positive control in our study when evaluating the relative antioxidant capacity of our novel analogs
32
. It is worth noting that, although both natural bilirubin and Compound
Dose-dependent cytotoxicity was seen in islet cells treated with increasing concentrations of both natural bilirubin as well as Compound
A protective effect on murine islets following hypoxic stress was seen when cells were treated with Compound
In contrast to some previous reports, we were not able to show a significant protective effect on isolated murine islet cells following hypoxic injury treated with natural bilirubin at 10 to 20 μM when compared to vehicle control islets. However, similar reports out of our laboratory have suggested that, although significant improvements in cell survival are seen in murine islets treated with natural bilirubin following subjection to 3 h of hypoxia, prolonged exposure to hypoxic conditions exceeding 24 h may overwhelm bilirubin’s protective antioxidant mechanisms
24,34
. Hypoxia and nutrient deprivation due to impaired diffusion, resulting in a gradual transition from apoptosis to necrosis within the central cells of hypoxic pancreatic islets, is thought to be the primary mechanism of death when isolated islets are subjected to prolonged hypoxic conditions
35
. Impaired diffusion of bilirubin, especially to the central cells of the hypoxic islet, likely contributes to islet cell loss under prolonged hypoxic conditions, and perhaps improved diffusion of a smaller and less complex bilirubin analog, such as Compound
These findings also highlight another dilemma with bilirubin as a therapeutic for islet allograft transplantation, namely bilirubin’s poor bioavailability, being an insoluble compound in water at physiologic pH and a highly protein-bound substance in plasma
7
. Although Compound
Some limitations should be taken into consideration when interpreting the results of this study. In vitro studies are inherently unable to adequately predict the viability, functionality, and therefore ultimate success of islet cells transplanted in vivo, regardless of therapy employed. Additional practical aspects of Compound
Based on the synthetic nature of the bilirubin analog Compound
Conclusions
In conclusion, Compound
Supplemental Material
Supplemental_Information - In Vitro Evaluation of a Novel Synthetic Bilirubin Analog as an Antioxidant and Cytoprotective Agent for Pancreatic Islet Transplantation
Supplemental_Information for In Vitro Evaluation of a Novel Synthetic Bilirubin Analog as an Antioxidant and Cytoprotective Agent for Pancreatic Islet Transplantation by Emilee J. Luckring, Patrick D. Parker, Homayoun Hani, Mary H. Grace, Mary A. Lila, Joshua G. Pierce and Christopher A. Adin in Cell Transplantation
Footnotes
Data Availability
The nuclear magnetic resonance data used to support the findings of this article are included within the article and within the supplementary information files. The FRAP, ABTS*+, cytotoxicity, and viability following hypoxic injury data are included within the article.
Ethical Approval
This study was approved by the Institutional Animal Care and Use Committee of North Carolina State University, Raleigh, NC, USA.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the NIH Guidelines for Animal Care and Use, Washington, DC, USA.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the North Carolina State University Research and Innovation Seed Funding Program (grant number 77266).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
