Abstract
Liver transplantation has been deemed the best choice for end-stage liver disease patients but immune rejection after surgery is still a serious problem. Patients have to take immunosuppressive drugs for a long time after liver transplantation, and this often leads to many side effects. Mesenchymal stem cells (MSCs) gradually became of interest to researchers because of their powerful immunomodulatory effects. In the past, a large number of in vitro and in vivo studies have demonstrated the great potential of MSCs for participation in posttransplant immunomodulation. In addition, MSCs also have properties that may potentially benefit patients undergoing liver transplantation. This article aims to provide an overview of the current understanding of the immunomodulation achieved by the application of MSCs in liver transplantation, to discuss the problems that may be encountered when using MSCs in clinical practice, and to describe some of the underlying capabilities of MSCs in liver transplantation. Cell–cell contact, soluble molecules, and exosomes have been suggested to be critical approaches to MSCs’ immunoregulation in vitro; however, the exact mechanism, especially in vivo, is still unclear. In recent years, the clinical safety of MSCs has been proven by a series of clinical trials. The obstacles to the clinical application of MSCs are decreasing, but large sample clinical trials involving MSCs are still needed to further study their clinical effects.
Introduction
Liver transplantation has been deemed the best therapy for end-stage liver diseases, but recipients usually have to live with life-long immunosuppression 1 (Figure 1). Although the standard pharmacological immunosuppressive treatment commonly used in clinical practice can achieve the favorable results of long grafts and patients survival rates, the side effects (Figure 1) caused by the treatment are significant 2 . In addition to the drug toxicities, the risks of malignancies and opportunistic infections have been reported to be increasing significantly 3 . Immunomodulatory cell therapy, as a complementary plan to standard pharmacological immunosuppression, appears to be a solution to this problem. In transplantation cases, the ultimate goal of immunomodulatory cell therapy is to prolong solid-organ allograft survival with reduced, or even no, usage of systemic pharmacological immunosuppression 4 .

Common classes of immunosuppressive drugs in liver transplantation and the main side effects of immunosuppressive drugs.
Mesenchymal stem cells (MSCs), a subpopulation of multipotent and non-hematopoietic cells initially isolated from bone marrow and reported by Friedenstein et al. in 1970 5,6 , are also called mesenchymal stromal cells and have been the focus of transplant scientists due to their great potential capacities for tissue repair and immunomodulation. Although no reports of large-scale clinical practices involving MSCs for liver transplantation have been found, and MSCs investigations remain mostly at the preclinical stage, the powerful immunomodulatory properties shown in recent reports make MSCs a promising candidate therapy in clinical liver transplantation. In addition, the effects of MSCs can be improved through gene modification and pretreatment, and the potential properties of MSCs show great promise in liver transplantation.
Research Status of MSCs
Cell therapy for immune rejection after organ transplantation is characterized mainly by its powerful immunomodulatory function. Although the immunomodulatory property seems to be a shared feature of all stromal cells including MSCs and fibroblasts, the characteristics of MSCs (such as easy cultivation, expansion, and storage in vitro) make them more appropriate for organ transplantation 7 –9 . Based on these characteristics, MSCs have been studied for immunosuppression after transplantation of various organs, including the liver, kidney, skin, etc.
MSCs can be isolated from diverse sources, such as umbilical cord blood, peripheral blood, and adipose tissue, and all MSCs share many characteristics, including cell phenotype and immunomodulatory properties 10 –15 . MSCs can also be isolated from some other species such as mice, rats, and rabbits, and great differences exist among them in many aspects, such as the mechanisms of their immunomodulatory properties 16 . Moreover, MSCs isolated from different human tissues do not have the identical properties. For example, all MSCs secrete different cytokines, which may affect their immunosuppressive effects 17 . A unique and definitive marker has not yet been found for MSCs isolated from various sources; however, it is known that they lack the expression of major histocompatibility complex class II and costimulatory molecules (including CD80, CD86, and CD40), and this characteristic appears to explain the low immunogenicity of MSCs 18 . Due to this low immunogenicity, in in vitro mixed lymphocyte reaction (MLR) experiments, even MSCs from third-party sources barely caused a lymphocyte response 19,20 . Thus, some authors believe that MSCs from the recipient, donor, and even third-party sources could be feasible in organ transplantation, although this is not yet widely accepted due to the lack of research evidence in the human body and the effects not being entirely satisfactory 21 .
In general, as the first tissue from which MSCs were isolated, bone marrow (BM) is currently the main source of MSCs for investigation. In clinical studies of organ transplantation, the most widely accepted practice is to choose MSCs isolated from the BM of the donor or the recipient; however, researchers are keen on seeking alternative sources of MSCs such as urine, placenta, and adipose tissue, due to the limited quantities and difficulty in isolating BM-MSCs. MSCs from other sources also have their own immunoregulatory characteristics. Although it is still unclear, and there is a lack of clinical evidence about which type of MSCs is most suitable for immunoregulation after liver transplantation, recent studies have revealed differences in MSCs from diverse sources. For instance, Rho et al. demonstrated that adipose-MSCs (A-MSCs) and BM-MSCs expressed high levels of IL-10 and TGF-β, and dermal skin-MSCs expressed high levels of IFN-γ 22 . The same dose of melatonin treatment for A-MSCs and BM-MSCs had different degrees of influence on viability 23 . Moreover, the function of MSCs from the same source may also be different. A study showed that aged BM-MSCs secrete more IL-6 and exerted stronger immunomodulatory effects than young BM-MSCs 24 . Considering that the differences among these MSCs may have different outcomes in patients undergoing liver transplantation, the above studies indicate that doctors could choose personalized medicine regimens based on MSCs according to the conditions of their patients in the future, though much research is still needed to achieve this goal.
MSC In Vitro Studies
The results of in vitro experiments have indicated the powerful immunomodulatory capacity of MSCs, which could influence almost all cells involved in the immune reaction (Table 1). For B cells, MSCs can inhibit B cell differentiation and proliferation, most likely by directly contact between MSCs and T cells, and by arresting B cells in the G0/G1 phase of the cell cycle 25 –28 . For natural killer (NK) cells, MSCs can inhibit their activation and proliferation in a dose-dependent manner 29,37 . Secreted factors, including interferon-γ (IFN-γ), prostaglandin E2 (PGE2), transforming growth factor β (TGF-β), and indoleamine 2,3-dioxygenase (IDO), as well as cell-to-cell contact might be involved in this process 29,30 . Regarding dendritic cells (DCs), MSCs could influence their differentiation, maturation, and function by direct interaction and soluble mediators, such as prostaglandin E2 (PGE2) and IL-6 31 . In detail, MSCs can interfere with the differentiation of DCs, maintain DCs in the immature stage and suppress pro-inflammatory cytokine release by DCs 31,32 . And, dependent on maturation inducers, coculture of MSCs with DCs could also upregulate the expression of PD-L1 in DCs, and results indicate that MSCs could induce tolerogenic DCs by manipulating the molecular phenotype of DCs 33 . Additionally, researchers have become gradually aware of the importance of exosomes from MSCs in immunoregulation, and have performed large scale studies 38 . The role of MSC-derived exosomes in liver transplantation will be discussed in the following sections.
The Effects of MSCs on Immune Cells.
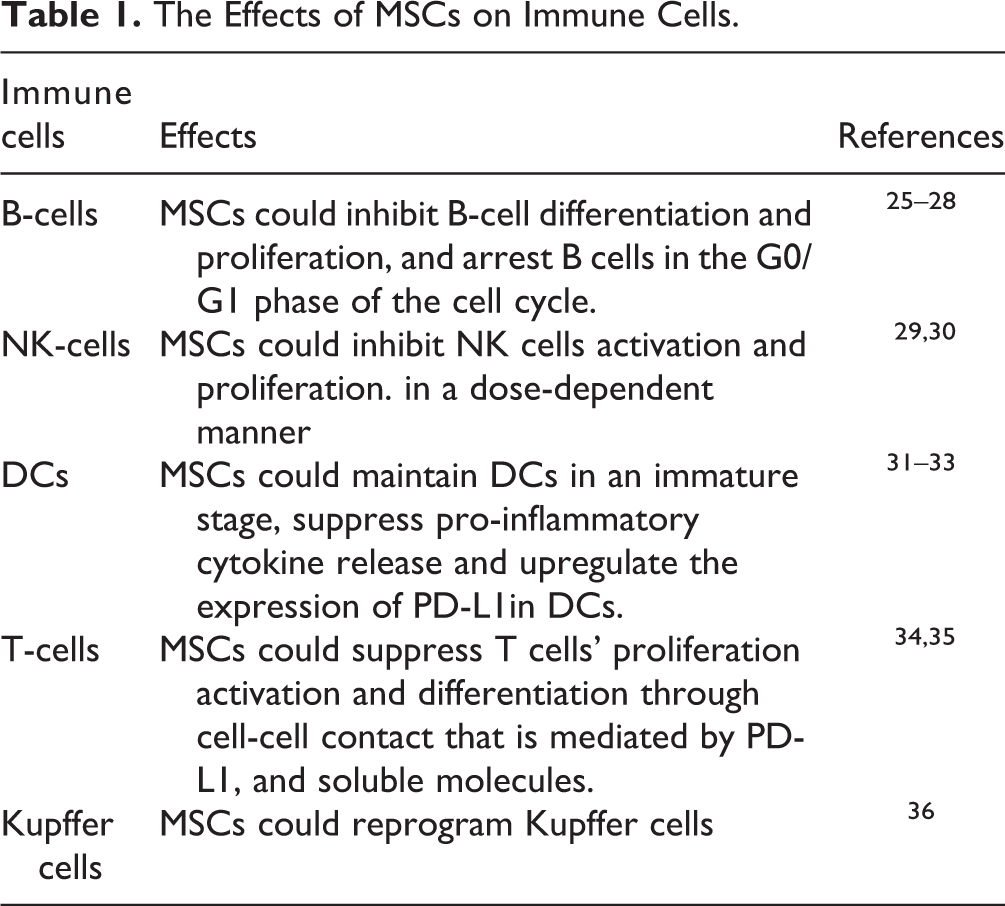
For investigations in organ transplantation, suppressing the function of T cells plays the most important role among the immunomodulatory properties of MSCs 39 . Previous studies have confirmed that MSCs can inhibit T cell activation and proliferation, promote T cell apoptosis in vitro, and regulate the T cell subset ratio by increasing Treg and Th2, as well as decreasing Th1 and Th17 cells 40 –42 . Regarding the mechanism of T cell suppression, some authors believe that the inhibition of DC maturation and phenotype is closely involved 43,44 . However, if only CD4+ T cells (with phytohemagglutinin stimulation) and MSCs are present, the immunosuppressive effect of MSCs on T-cell proliferation still works 45 . Cell–cell contact was deemed to be important for this immunosuppressive property of MSCs. Specifically, cell–cell contact was found to be crucial for regulatory T cell induction, and direct contact through the programmed death (PD) 1 inhibitory pathway was considered to be involved in the inhibitory effects on T cell activation and proliferation 34 . As well as direct contact, soluble molecules are also thought to mediate T cell suppression because MSCs could still suppress T cell proliferation even without cell–cell contact 35 . Molecules possibly involved in human MSCs, as they are known, include transforming growth factor (TGF), hepatocyte growth factor (HGF), IDO, PGE2, insulin-like growth factor binding proteins, heme oxygenase-1 (HO), human histocompatibility antigen-G5 (HLA-G5), chemokine (C-C motif) ligand 2 (CCL2), IL-10, galectin-1, and galectin-3 35,46 –55 . All these molecules were found to be involved in T cell suppression. There have been some recent studies on the mechanism of interaction between cytokines derived from MSCs and T-cells. For instance, studies have shown that cytokines derived from MSCs could participate in the inhibition of T cells by regulating expression of the FoxP3 gene 34,38 . However, the mechanism governing cytokine interactions between MSCs and FoxP3 is not completely clear, and these studies explained only part of the mechanisms linking cytokines and T-cells. Thus, the mechanism of the effects of MSC-derived cytokines on T cells is still the focus of research.
Macrophages have also been found to be involved in the immunosuppressive capacity of MSCs, and recently authors have begun to pay attention to MSC regulation of macrophages to achieve the goal of treating some specific diseases 56 . Németh et al. observed inhibition of the systemic inflammatory response and prolonged survival of rats after infusing MSCs into rats with sepsis 57 . Moreover, the immunoregulatory property of MSCs still existed after T cells, B cells, or NK cells were depleted, but the effect was eliminated when macrophages were depleted 56 . This result proved the critical role played by macrophages in MSC-mediated immunoregulation. This observation might be quite important in the application of MSCs in liver transplantation and we shall further discuss this issue below.
MSC In Vivo Studies
Concerning in vivo studies, MSCs have been found to play a role in various diseases, such as relieving illness in patients with systemic lupus erythematosus (SLE), ameliorating diabetes in a murine model, reducing inflammatory MRI parameters in multiple sclerosis (MS) patients, and preventing lethal graft-versus-host disease (GVHD) in clinical patients 58 -61 . Although much knowledge has been accumulated in term of the immunoregulatory properties of MSCs, the exact mechanism remains undefined and unclear 62 . Moreover, because of the different conditions between in vivo and in vitro studies, research data in vitro could not comprehensively explain results in vivo. For example, due to lack of direct cell–cell interaction, the cell–cell contact that was deemed critical in MSC immunosuppression in vitro is no longer believed to be critical. Therefore, the mechanism of the immunoregulatory property of MSCs in vivo needs to be investigated further.
In transplantation, their immunosuppressive property and low immunogenicity could allow MSCs to induce immunosuppression in vivo without rejection, which makes MSCs suitable for organ transplantation. In animal experiments (Table 2), many successful cases of MSC application in organ transplantation, including heart, skin, kidney, and liver 83 –85 , have been reported, supporting the use of MSCs in clinical patients. In clinical trials, since BM-MSCs were first used as immunomodulators in a patient with GVHD, several studies have supported the safety and feasibility of BM-MSC use in autologous and allogeneic organ transplantation through a large number of experiments 86 –88 . In the past, researchers were generally concerned about the long-term complications of MSCs, such as tumors and infections. To date, all clinical studies on MSCs and liver transplantation that have been published have confirmed the safety and feasibility of use of MSCs in the human body 89 –92 . Additionally, dozens of studies in other tissues have also proven the safety of MSC transplantation 93 . However, because the sample size of the previously described liver transplantation study was small and the follow-up period was short, the role of MSCs in cancer progression remains controversial, and more samples and longer-term clinical trials are still needed to support the application of MSCs in liver transplantation patients, especially those with hepatocellular carcinoma.
Liver Transplantation Trials Based on MSCs in Animal Models.

Current clinical studies focus mainly on the safety and feasibility of MSCs, but some in vivo animal trials that improved the immunomodulatory capacity and homing ability of MSCs through phenotypic modification or pretreatment have been carried out and have acquired good results. For example, researchers have enhanced the efficiency of MSCs by expressing the TNF-αR-Fc and HO-1 genes in porcine islet xenotransplantation mouse models 94 . However, whether this treatment can benefit liver transplant recipients needs further study.
Current Status of Clinical Liver Transplantation
Since the first case of human orthotopic liver transplantation was described in 1963, liver transplantation has undergone 50 years of development, with the 1-year survival rate increasing from 25% to more than 85% 34,95 . However, the problem of immune rejection after surgery still exists, with more than 30% of liver transplant recipients still suffering varying degrees of rejection (Figure 2) 96 . Nevertheless, the incidence and severity of rejection after liver transplantation remains much lower than that in other solid organs, which makes the liver an “immunologically privileged organ”. From the perspective of physiology, given its exposure to numerous foreign materials from the gut, the liver is necessarily a tolerogenic organ, and the tolerance of the liver plays a key role in preventing potentially devastating inflammatory responses against both food and normal enteric bacteria 97 . Although the phenomenon of “liver tolerance” has been described for decades, the exact mechanism is yet not thoroughly understood. All the theories involving T cell anergy induction and regulatory DC induction cannot completely explain this phenomenon 98 . However, it is sure that the liver microenvironment, especially the environment around the hepatic sinusoid, plays a critical role in maintaining liver tolerance. Xia et al. described changes in the microenvironment status that could affect the state of liver tolerance 99 . On the other hand, changing and influencing the local immune microenvironment is considered to be an important MSC immunomodulatory property, which makes MSCs excellent candidate cells for promoting and maintaining liver graft tolerance.

Mechanism of immune rejection after liver transplantation. Donor antigens are recognized by antigen-presenting cells (APCs) and transferred to T cells. After activation, T cells differentiate into various effector T cells to secrete inflammatory factors or directly participate in immune rejection. B cells are activated with or without the help of Th2 cells after recognition of the donor antigens, secreting antibodies and cytokines that act on donor cells.
Although some immunocytes including NK cells, DCs, and T cells have already been deemed to be involved in the liver tolerance mechanism, some other factors should be considered for the explanation of liver tolerance, considering that those cells exist all over the body 100 . Kupffer cells (KCs)—the macrophages located in the hepatic sinusoid—account for 75%–80% of the macrophages in the whole body, and are known to be one of the key factors that make the microenvironment of the hepatic sinusoid different from that in other organs 101 . Several decades ago, Callery et al. reported that blocking KCs could inhibit the induction of liver tolerance, thus proving the critical role of KCs in liver tolerance 102 . However, the role of KCs in immunity is ambiguous because they could both prevent liver injury and aggravate liver injury under different conditions 103 . Our previous results and those of other studies demonstrated that switching KCs from pro-inflammatory to anti-inflammatory status could promote hepatic immune tolerance 36,104 . Considering the influence of MSCs on macrophages, and the similarities between KCs and macrophages, MSCs could possibly reprogram the immune status of KCs. In fact, we already have evidence indicating that the MSC-dependent reprogramming of KCs is crucial for liver transplant tolerance 36 . Given that macrophages might be one of the major target cells through which MSCs can induce their immunomodulatory effects, and that KCs (as they are essentially macrophages) are key factors in liver transplant tolerance induction, we believe that, compared with other solid-organ transplantation, liver transplantation might be more suitable for immunoregulation involving MSCs.
MSCs in Animal Models
Since animal models of solid-organ transplantation involving MSCs were reported in 2006 in heart transplantation, many reports have described using MSCs in liver transplantation 105,106 . Such studies have indicated that different MSC sources, including recipients, donors, and third-parties, could all lead to liver allograft tolerance. Although some investigation results from in vitro experiments have been verified in vivo [for example, MSCs overexpressing interleukin-10 (IL-10) could lead to liver transplant tolerance in rats more effectively than normal MSC], due to their complexity, in vivo investigations are quite different from in vitro studies, and more research is needed 36,80 . For example, the ratio of MSCs to T-cells or peripheral blood mononuclear cells (PBMCs), which has already been determined in in vitro studies, would mean a large number of cells in in vivo experiments, and such a large number of cells would easily cause pulmonary embolism 107 . Thus, the approach and dose of MSC infusion are quite important in vivo investigation. The timing and methods of cell administration (pretransplant versus posttransplant) are also crucial because the infusion scheme can determine the final migratory location of MSCs, thus affecting MSC stimulation and interactions within the local environments and their immunomodulatory capacity 108,109 . Although several studies have reported that MSC infusion after transplantation may lead to graft dysfunction and failure to prolong graft survival, more studies still recommended a posttransplant infusion scheme, because the posttransplant infusion scheme can promote intragraft MSC migration, which can improve MSC tissue repair properties 110 . Moreover, pretransplant infusion may limit MSC homing into grafts and promote their migration to the lungs, where they might lose viability after 24 h 111 .
In most animal liver transplantation studies, liver function improvements and amelioration of inflammation can be observed in the early posttransplant phase (7 days after surgery) while anesis of liver inflammation during this phase can be attributed to the control of rejection as well as the relief of ischemia reperfusion injury (IRI) 110,112 . In fact, MSCs have been regarded as a protective factor against IRI, and MSC-mediated IRI protection might be a consequence of their inherent immunosuppressive capacity, similar to inhibition of rejection 113 . For example, Zheng and his group proved that MSCs, through the release of anti-inflammatory factors, significantly alleviate hepatic IRI 114 . Moreover, researchers also believe that MSCs could stabilize neovascularization by inhibiting inflammation, which would improve the success rate of liver transplantation 115 . To sum up, in liver transplantation, the immunomodulatory capacity of MSCs could potentially be helpful to promote liver transplantation, reduce hepatic ischemia reperfusion injury, and control liver transplantation rejection.
MSCs in Clinical Trials
More than 10 years have passed since MSCs were first used as an immunomodulator in humans (in a patient with GVHD in 2004) 116 . To date, although several cases of MSC application in the clinic have been reported (Table 3), clinical trials with large sample sizes on liver transplantation involving MSCs are still insufficient. The reasons might lie partly in the following intrinsic constraints and obstacles in the application of MSC in clinical liver transplantation: (1) Ethical problems: after decades of development, clinical liver transplantation has become quite a safe and efficient therapy for end-stage liver diseases; thus, any alteration in clinical standards would bring about much ethical doubt. In addition, ethical issues have also contributed greatly to the delay in stem cells being brought into clinical trials in many countries or regions. (For instance, wide applications of stem cells in preclinical studies were prohibited in China until July 2015.) (2) Immune environment issues: to ensure the best survival of the liver graft, recipients generally have to be severely immunosuppressed in the induction period, which means that any additional cell therapy would encounter a condition of systemic immunosuppression and would usually meet high concentrations of pharmacological immunosuppressors that would probably influence the function of transplanted MSCs. (3) Sources and pretreatment of MSCs: although the low immunogenicity of MSCs is well known, MSCs cannot be completely immunoprivileged in vivo 110 . Moreover, it is well documented that the culture conditions and pretreatment can influence the immunomodulatory potentials of MSCs; therefore the source and pretreatment of MSCs that are chosen for clinical use need further study. (4) Timing, approach, and dosage of MSC infusion: as previously mentioned, each of these factors is quite important in MSC application; however, the experience of in vitro studies and animal experiments cannot inform clinical applications. Thus, more studies with larger animals (for example, pigs) with dose escalation should be conducted. (5) Safety concerns: due to a lack of knowledge, crucial safety concerns need to be considered, such as whether MSCs increase tumor incidence or opportunistic infections risk, similar to immunosuppressive drugs, and whether MSCs have the potential for maldifferentiation given that MSCs possess multilineage differentiation potential. On the other hand, however, due to the different types of liver diseases that warrant liver transplantation, these safety problems might not be critical in all clinical transplantation cases. For example, due to the potential risk of tumor promotion, MSCs may not be suitable for patients diagnosed with early stage liver cancer but might be feasible for the patients with liver cirrhosis.
Clinical Trials of Liver Transplantation Based on MSCs Carried Out Up to August 1, 2019. All Data from https://clinicaltrials.gov/ and PubMed.
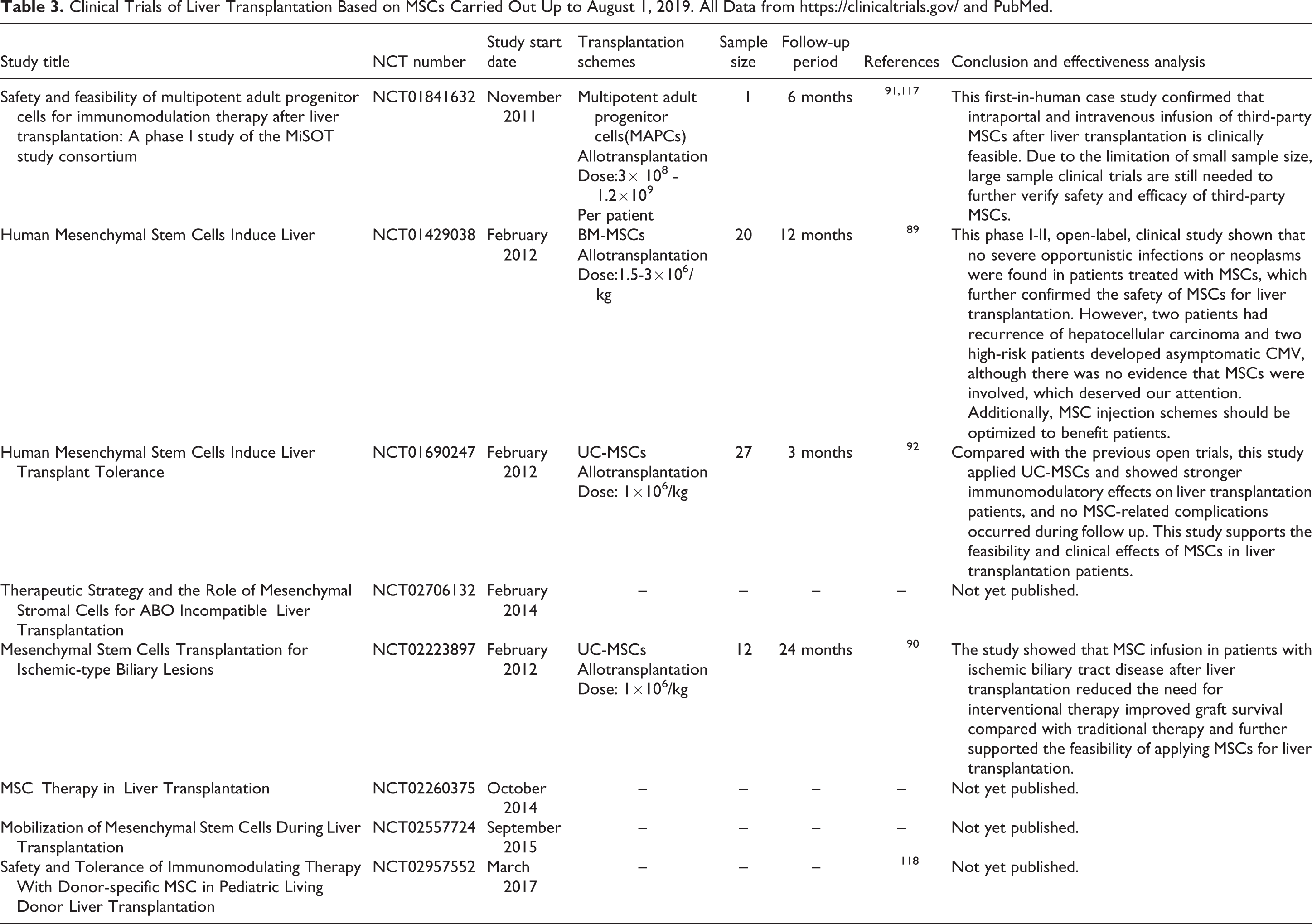
Despite the many constraints and obstacles, the application of MSCs in clinical use has not been stopped. From reports of a series of phase II clinical studies in Europe on the treatment of acute steroid-resistant GVHD, MSCs have shown substantially promising results 116 . These results not only suggest that MSCs might still be effective in GVHD even after the failure of immunosuppressive drugs, but also indicate the possibility of MSCs application in solid-organ transplantation. The MiSOT-I trial, performed by the Mesenchymal Stem Cells in Solid Organ Transplantation (MiSOT) organization, was initiated in 2013. To date, much feedback information regarding clinical research involving MSC application in liver and kidney transplantation has been summarized 110 . Such feedback, combined with other clinical trials of liver transplantation, includes mainly the following aspects: (1) The timing of MSC infusion: MSCs need to be used in the early posttransplantation phase because of their long-term pro-tolerogenic effects. Because of persistent chronic rejection, MSCs should be used posttransplantation if the function of the graft is stable or deteriorating. Moreover, the long-term effects of MSCs on chronic rejection are still expected. (2) Use of concomitant immunosuppression: Considering the existence of immunogenicity and drug targets of MSCs, the drug combinations used with MSCs may be a promising way to improve future clinical transplantation. (3) Sources of MSCs: all sources of MSC, including BM, adipose tissue and other human tissues share similarly high therapeutic potentials. Regarding immunogenicity, based on the outcomes of liver transplantation trials, MSCs were proven to be safe and effective. (4) Safety concerns: regarding the potential of MSC maldifferentiation, evidence for malignant transformation of MSCs was not found in all studies, but further attention is still needed. For the issue of opportunistic infections, four recent clinical trials of MSCs in liver transplantation showed no increased incidence of infection in patients after receiving MSCs 89 –92 . However, given the small sample size and short follow-up period of the above-mentioned trials, and a clinical trial based by Reinders et al. on renal transplantation that described that three of six patients infused with MSCs had opportunistic but mild viral infections, the possibility of infection after MSC injection should still be taken seriously 87 . (5) Immunoregulation results: Although MSCs were proven to be effective in alloimmune reaction treatments by a large number of in vivo and in vitro experiments, a clinical trial on liver transplantation above mentioned lacked the protective effects of MSCs in patients with liver transplantation, and, after withdrawal of the immunosuppressive agents, immunological rejection occurred successively in MSC-treated patients and disappeared after resumption of the immunosuppressive agents 89 . This result was most likely due to the choice of immunosuppressants and MSCs; and an inappropriate application scheme of MSCs. Another clinical trial of liver transplantation showed that, in comparison to using immunosuppressive drugs alone, MSCs could better improve liver tissue, and were able to regulate the proportion of T lymphocyte subsets to promote immune tolerance during the acute rejection phase after liver transplantation. However, the long-term status of patients could not be observed, due to the short follow-up period of only 12 weeks 92 . For induction therapy, although no clinical trials related to liver transplantation have been reported, two clinical trials on kidney transplantation confirmed the safety of MSCs as induction therapy for organ transplantation 119,120 . Moreover, one of the above studies showed that MSCs were superior to IL-2 for induction therapy 120 .
Concerning liver transplantation, specifically, information on MSC application in clinical liver surgery is still lacking. The available information on MSC application in liver transplantation is encouraging; however, large-scale trials are still needed. Nevertheless, the experience of researchers in the design of investigations is still helpful and meaningful for subsequent studies.
MSCs Combined with Immunosuppressive Drugs in Liver Transplantation
At present, there is a consensus on the use of calcineurin inhibitors (CNIs) such as tacrolimus and cyclosporine, as immune maintenance drugs after liver transplantation, supplemented with antimetabolites such as acetazolamide (AZA) and mycophenolic acid (MPA), or m-TOR inhibitors to achieve a lower dose of CNI and alleviate side effects 121 . Glucocorticoids, interleukin-2 and antithymosin are recommended for induction therapy of liver transplantation 121 . Previous clinical studies on liver transplantation are also based on the application of MSCs combined with tacrolimus and mycophenolate mofetil (MMF) in the immune maintenance therapy of liver transplantation patients. Given the low immunogenicity of MSCs, and given that there are still targets on which immunosuppressive drugs act, this issue should be considered when developing a combination regimen of MSCs and immunosuppressive drugs 122 .
It was found that short-term exposure to immunosuppressive agents in vitro had no toxicity and did not induce apoptosis in MSCs, but long-term exposure to tacrolimus at high doses for 7 days could significantly promote the death of MSCs 122 . Even exposure to MPA and rapamycin at therapeutic doses inhibited the proliferation of MSCs 122 . These results suggested that short-term and low-dose immunosuppressive agents combined with MSCs seemed to achieve better results in patients undergoing liver transplantation. In addition, the added protective agents seemed to be effective in reducing the toxicity of immunosuppressants on MSCs. For example, combination with oxytocin counteracted the cytotoxicity of tacrolimus on MSCs 123 . Researchers also found that different kinds of immunosuppressants have heterogeneous interaction effects with MSCs. For CNIs, MSCs combined with tacrolimus demonstrated considerable coordination in immune regulation after organ transplantation. Kuo et al. found that MSCs plus tacrolimus improved graft survival in a miniature swine hind-limb model 124 . Suda and his group also showed that the combination of MSCs and tacrolimus could relieve ischemic brain damage in rats 125 . A prospective, nonrandomized study on kidney transplantation proved that the use of MSCs could offset the dose of tacrolimus used in patients without increasing the risk of rejection, and indicated that combined use with MSCs could decrease tacrolimus dosage and thus alleviate adverse reactions in organ transplantation patients 126 . However, the synergistic effects of tacrolimus combined with MSCs on immune regulation have been controversial in some in vitro studies. Poncelet et al. reported that MSCs cocultured with tacrolimus effectively increased the inhibitory effect on T cells 127 , but Buron et al. showed an antagonistic effect of MSCs and tacrolimus on T cell inhibition 128 . This may be attributed to the difference in the origin of the MSCs in the above-mentioned studies. Cyclosporine was also found to interact with MSCs. Some studies have shown that cyclosporine synergistically promotes immunosuppression with MSCs 129 . However, some studies have found that cyclosporine and MSCs have antagonistic effects, which may be attributed to the differences in experimental treatment methods, such as lymphocyte stimulation, and the basic condition of the experimental subjects 128 . Exhilaratingly, MMF/MPA showed better synergistic immunosuppressive action with MSCs than other immunosuppressive drugs in both in vitro and in vivo studies. For example, a study showed that MSCs combined with MMF could significantly increase heart transplant survival compared with single-use MSCs or MSCs combined with CNIs in a rat model 130 . In an in vitro study, MSCs combined with MPA showed stronger inhibitory effects on lymphocyte proliferation than CNIs, m-TOR inhibitors, and dexamethasone 128 . These studies seemed to suggest that MMF/MPA was more suitable for combined use with MSCs than other immunosuppressants. Additionally, both steroid hormones and m-TOR inhibitors also exhibited synergistic immunosuppressive effects with MSCs in animal models 131,132 . Given the potential role of MSCs in liver transplantation, these effects should be further studied and considered when formulating a compatibility program for liver transplantation patients 132 . Taken together, the above findings imply that multiple combination regimens based on MSCs could be designed as induction or maintenance therapies for liver transplant patients with different disease conditions in the future.
Potential Therapeutic Properties of MSCs in Liver Transplantation
In addition to posttransplant immunosuppression, MSCs are also able to promote liver regeneration, angiogenesis, and anti-fibrosis, which are potentially helpful for liver transplantation in patients (Figure 3). For liver regeneration, studies found that MSCs could promote liver reparation after partial hepatectomy in mice with hepatic steatosis (HS) 133 . MSCs that increased angiogenesis in an animal model of acute liver failure and that relieve hepatic cirrhosis in animal and clinical trials were also found 134 –136 . Cytokines and factors secreted by MSCs through the paracrine pathway appear to play a major role in these processes. For example, MSCs could secrete cytokines such as IL-6, hepatocyte growth factor (HGF), and IGFBP-2 to promote hepatocyte regeneration and inhibit hepatocyte apoptosis. Metalloproteinase (MMPs) and insulin growth factor-like-I (IGF-I) released by MSCs could effectively reduce collagen accumulation and counter liver fibrosis 137,138 . MSCs were able to secrete various angiogenic factors to promote angiogenesis. Molecules such as VEGF-A, VWF, SDF-1α, Cyr61, MMP2, and MMP9 are involved in this process 139 –142 . In addition, MSCs can also participate in these processes by regulating the immune response and inhibiting the release of inflammatory factors 143 . Moreover, studies show that overexpression of related cytokines such as HGF, in MSCs can alleviate liver function damage, reduce liver fibrosis, and improve graft survival compared with normal MSCs 67 –69 . These studies suggest that MSCs have potential therapeutic properties in liver transplantation, and looking for ways to enhance these properties of MSCs and thereby enhance the effect of MSCs on liver transplantation seems to be a promising approach but it still needs be further evaluated in clinical trials.
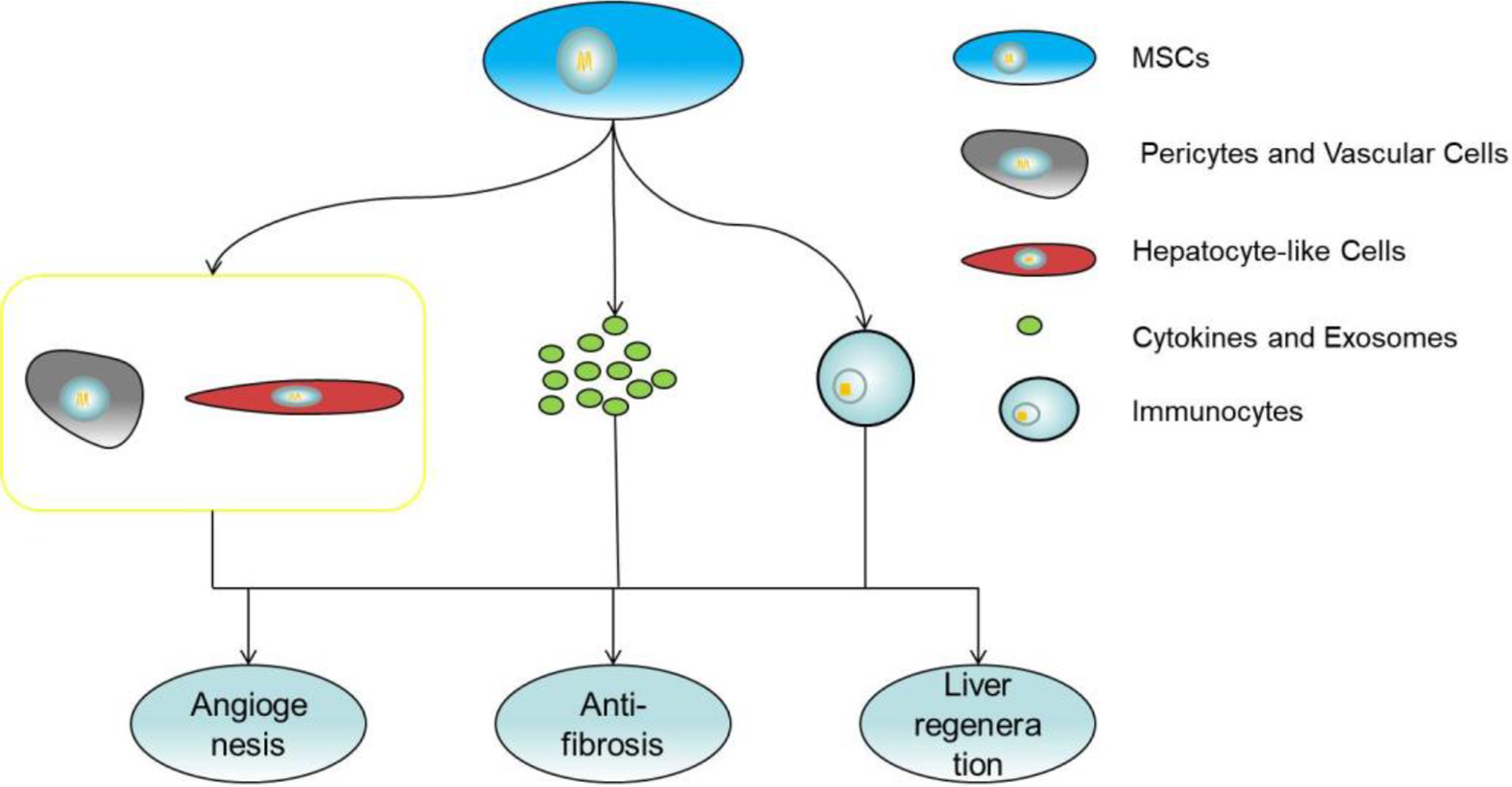
MSCs promote liver regeneration, angiogenesis and count fibrosis by secreting cytokines and exosomes, regulating inflammation and differentiation.
Despite their immunomodulatory capacity as a multipotent stem cells, MSCs have multidirectional differentiation characteristics. In previous studies, researchers proved that MSCs could differentiate into pericytes and vascular cells in vivo 139 . It should be mentioned that MSCs have the potential to differentiate into hepatocyte-like cells both in vitro and vivo 137,144 . Hepatocyte-like cells derived from MSCs have gene expression profiles similar to those of hepatocytes and perform several hepatocyte-like functions that promote the recovery of damaged liver function. These studies show a potential mechanism for MSCs to help patients undergoing liver transplantation. However, given that the immune regulation of MSCs is impaired during the process of differentiating into hepatocyte-like cells, it seems unwise to only pursue the promotion of differentiation of MSCs into hepatocytes in the application of liver transplantation.
Exosomes, as a type of extracellular vesicle (EV) are defined as lipid bilayer particles 20–150 nm in diameter released by various cells 145 . Despite being initially regarded as a waste product of cell rejection, exosomes, which are rich in proteins, coding and non-coding RNA, and even DNA, are closely associated with many diseases and physiological processes, and have attracted the attention of researchers in recent years. Given that exosomes from MSCs display powerful physiological regulatory functions, have lower ethical considerations and immunogenicity compared with stem cell therapy, and have no increased risk of cancer, they seem to be a promising approach to acellular therapy. For liver transplantation, exosomes released from MSCs have been proven to have immune regulation, tissue repair and angiogenesis functions similar to those of source cells. One study showed that injecting exosomes derived from BM-MSCs into rats with GVHD every week could inhibit Th17 cells and Tregs, and enhance pathological conditions 146 . Liver fibrosis was also alleviated after transplantation of MSC-derived exosomes by reducing oxidative stress and inhibiting hepatocyte apoptosis in a liver injury model induced by CCL4 147 . Whole-body administration of exosomes derived from MSCs promoted liver repair and angiogenesis in damaged areas 148,149 . In addition, a study showed that injecting MSC-derived exosomes into mice with hepatocellular carcinoma could effectively inhibit tumor growth and enhance chemosensitivity due to the abundance of anti-tumor microRNAs in the exosomes 150 . This study indicated that exosomes derived from MSCs may be particularly suitable for liver transplantation patients with malignant tumors, though the actual effects still need to be measured further by clinical trials. Together, exosomes derived from MSCs can be used not only as a potential mechanism for explaining the effect of MSCs on liver transplantation, but also as a new tool that may be used to benefit patients undergoing liver transplantation at some future date.
Conclusions
According to the available information from investigations including in vitro, animal, and phase I and II clinical studies, MSC infusion is generally feasible and safe for liver transplantation, but we suggest the need for a cautious approach for certain types of patients, such as patients with tumors. The application of MSCs could promote immune tolerance in patients with liver transplantation; however, whether MSCs could replace immunosuppressive drugs is questionable.
Due to the heterogeneity of MSCs, it is clear that the immunoregulatory function of MSCs can be improved by choosing appropriate sources, providing feasible pretreatment, compatible with practicable immunosuppressants and gene modification; however, whether the above schemes are actually beneficial for liver transplant patients without increasing the risk of complications, and even help to completely replace immunosuppressive drugs, still requires further clinical trial verification. It is known that soluble molecules, exosomes and direct cell-to-cell contact are considered to be the main ways whereby MSCs exert their immunosuppressive effects, but the exact mechanisms of immune regulation are not yet fully understood. Additionally, exosomes show promising therapeutic prospects, but whether exosomes can replace cell therapy in liver transplantation needs further confirmation. The MSC characteristics of promoting liver regeneration, angiogenesis, and alleviating liver fibrosis are helpful for liver transplantation patients. Improving these properties of MSCs may better benefit liver transplantation patients. The ability of MSCs to undergo multidirectional differentiation is also involved in these processes. However, it remains unclear whether regulating the differentiation of MSCs can benefit liver transplant recipients.
In summary, MSC-based therapy is a promising treatment for liver transplantation, but the exact mechanisms and effects of MSCs on liver transplantation, as well as the problems that might be encountered in the clinical applications, still require further study; such studies will greatly aid the future clinical use of MSCs in liver transplantation.
Footnotes
Author Notes
Yu You and Di-guang Wen contributed equally to this work.
Author Contributions
YY, WDG, and LZJ performed research and wrote the first draft. GJP collected and analyzed the data. All authors contributed to the design and interpretation of the study and to further drafts. LZJ is the guarantor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Science Foundation of China (No. 81170442, 81470899, 81702357), National Scholarship Foundation (No.201208505116), and Outstanding young talent fund of the second hospital of CQMU (2011).
