Abstract
Severe spinal cord injury (SCI) is caused by external mechanical injury, resulting in unrecoverable neurological injury. Recent studies have shown that exosomes derived from bone marrow mesenchymal stem cells (BMSCs-Exos) might be valuable paracrine molecules in the treatment of SCI. In this study, we designed SCI models in vivo and in vitro and then investigated the possible mechanism of successful repair by BMSCs-Exos. In vivo, we established one Sham group and two SCI model groups. The Basso, Beattie, Bresnahan (BBB) scores showed that BMSCs-Exos could effectively promote the recovery of spinal cord function. The results of the Nissl staining, immunohistochemistry, and TUNEL/NeuN/DAPI double staining showed that BMSCs-Exos inhibited neuronal apoptosis. Western blot analysis showed that the protein expression level of Bcl-2 was significantly increased in the BMSCs-Exos group compared with the PBS group, while the protein expression levels of Bax, cleaved caspase-3, and cleaved caspase-9 were significantly decreased. The results of western bolt and qRT-PCR demonstrated that BMSCs-Exos could activate the Wnt/β-catenin signaling pathway effectively. In vitro, we found that inhibition of the Wnt/β-catenin signaling pathway could promote neuronal apoptosis following lipopolysaccharide (LPS) induction. These results demonstrated that BMSCs-Exos may be a promising therapeutic for SCI by activating the Wnt/β-catenin signaling pathway.
Introduction
Spinal cord injury (SCI) is a devastating disease with serious complications and, as yet, there is no effective cure. According to World Health Organization survey data, SCI affects more than 180,000 patients every year worldwide 1,2 . SCI causes a series of complex pathological changes, including apoptosis or death of neurons and oligodendrocytes, inflammation, ischemia, excitotoxicity, axonal degeneration, and demyelination, all of which ultimately lead to spinal cord dysfunction 3,4 . Most SCI research focuses on the reconstruction of functional synaptic neurons, the inhibition of neural inflammation, the promotion of intrinsic recovery, the elimination of extrinsic disorders, the reduction of reducing lesion, and other fields 5 –7 .
Transplantation of mesenchymal stem cells (MSCs), including bone marrow mesenchymal stem cells (BMSCs), neural stem cells, human umbilical cord blood cells, embryonic stem cells, and Schwann cells, has been investigated as a potential treatment for SCI 8,9 . However, the effectiveness of transplantation of MSCs is limited by disadvantages such as immunological rejection, genetic variation, the lower survival rate of transplanted cells, and complicated operation steps 10 –13 . Therapeutic effects of transplantation of stem cells in SCI have been demonstrated in several studies, and a paracrine mechanism plays a key role in this role of stem cells 14 . Exosomes have an important role in the paracrine process 15 . Exosomes (40–120 nm in diameter) are small particles which are secreted by living cells and released into the extracellular fluid 16,17 . The formation and secretion of exosomes are related to protein, signal protein, cytoskeleton protein, and growth factors 18 . Exosomes contain precise targeted molecules, chaperones, enzymes, and membrane trafficking proteins. One study demonstrated that cognitive impairments caused by traumatic brain injury could be remarkably reduced by extracellular vesicles derived from MSCs 19 . Exosomes have been widely reported to be involved in the recovery of SCI 13,20 . Therefore, we hypothesized that exosomes derived from BMSCs can also effectively restore functional defects caused by SCI.
The Wnt/β-catenin signaling pathway is a well-characterized family of glycoproteins and is correlated with diverse physiological and pathological processes including cell proliferation and patterning, neural development, neuronal survival and axonal guidance, and cell polarity and motility 21,22 . There is growing evidence showing that Wnt signaling may be involved in SCI and repair 23 –25 . It has been investigated as a potential therapeutic approach in the central nervous system (CNS) 26,27 . Thus, we reason that functional behavioral recovery after SCI may be promoted by exosomes derived from BMSCs (BMSCs-Exos) via the Wnt/β-catenin signaling pathway.
However, the ability of BMSCs-Exos to promote spinal cord recovery after SCI is unclear, and the relationship between BMSCs-Exos and Wnt/β-catenin signaling pathways in SCI has not been reported. The purpose of this study was to investigate the neuroprotective mechanism of BMSCs-Exos in the treatment of SCI, and to further explore whether they might exert effects by activating the Wnt/β-catenin signaling pathway.
Materials and Methods
Acquisition and Identification of BMSCs
BMSCs were obtained from E28 male Wistar rats and cultured in the T75 cell culture flask (Corning Incorporated, Corning, NY, USA) with a cell concentration of 1 × 105/ml 28 . The BMSCs were cultured in an incubator with a humidified atmosphere containing 5% CO2 at 37°C for 48 h and then expanded with Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F-12, Thermo Fisher Scientific, Waltham, MA, USA) with 10% fetal bovine serum (FBS, Gibco Life Technologies, Carlsbad, CA, USA) and 1% penicillin-streptomycin (Solarbio, Beijing, China). The medium was changed every 2–3 days. BMSCs were passaged when the cells reached 80–90% confluency in the cell culture dishes. Flow cytometry was used to identify the surface makers of BMSCs. BMSCs single-cell suspension was prepared by EDTA (Gibco Life Technologies), and cells were collected by centrifugation and washed twice with PBS solution. After that, 1% PBS was used to block nonspecific binding for 30 min at 4°C. Subsequently, BMSCs were incubated with primary antibodies to CD29 (Biolegend, San Diego, CA, USA), CD90 (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), CD34 (Santa Cruz Biotechnology, Inc. Santa Cruz, CA, USA), and CD45 (Biolegend) for 1 h at 4°C, respectively. BD FACSCalibur flow cytometer (Becton, Dickinson and Company) acquired data. All the data analysis was performed using Flowjo VX10 (Stanford University, Palo Alto, CA, USA).
Isolation and Purification of Exosomes
After P2 BMSCs reached 80–90% confluency in the cell culture dishes (Corning Incorporated), the culture medium was replaced with an exosome-depleted FBS-containing medium (System Biosciences, Mountain View, CA, USA), and the culture was continued for 48 h. The cell culture supernatant was dispensed into a 15 ml centrifuge tube (Corning Incorporated), and the mixture was centrifuged at room temperature, 300 g for 10 min, and then the supernatant was collected. The collected supernatant was transferred to a new 15 ml centrifuge tube (Corning Incorporated), centrifuged at 2000 g for 10 min, and the supernatant was then collected and transferred to a matching centrifuge tube (10 ml, Ultra-Clear tube, Beckman Coulter, Brea, CA, USA), centrifuged at 10,000 g for 30 min at 4°C, and the supernatant was collected. The collected supernatant was again transferred to a new ultracentrifuge (Beckman Coulter), centrifuged at 100,000 g for 6 h at 4°C, and the supernatant was discarded. All the steps were performed in a cell ultra clean platform. The precipitate in the centrifuge tube was washed with 100 µl of PBS. The desired solution was stored in a –80°C freezer. The acquired exosomes were observed by transmission electron microscopy (TEM, Tecnai, FEI, Hillsboro, OR, USA). Western blot was used to examine the exosome surface markers of CD9 (1:1000, Abcam, Cambridge, UK), CD63 (1:1000, Abcam), and CD81 (1:1000, Abcam).
Neuron Cell Culture
Neuron cells were extracted from the spinal cords of postnatal day 1 Wistar rats and cultured as previously described 29,30 . EDTA (0.25%, Gibco Life Technologies) was used to digest the fragmented tissue at 37°C for 20 min. DMEM/F12 (Thermo Fisher Scientific) containing 10% FBS (Gibco Life Technologies) was used for terminating digestion. The cells were then coated with poly-D-lysine (Sigma-Aldrich, St. Louis, MO, USA) in prepared medium for 8 h. Then the medium was changed to the neurobasal media including B27 (1%, Gibco Life Technologies), GlutaMAX (0.25%, Gibco Life Technologies), and penicillin/streptomycin (0.5%, Gibco Life Technologies). All cells were cultured at 37°C and in 5% CO2.
Treatment of Cells
Lipopolysaccharide (LPS, 100 ng/ml, Sigma-Aldrich) was used to culture neuronal cells to mimic neuronal cell damage. The antagonist XAV939 (1 μM; Selleckchem, Houston, TX, USA) was used to suppress the Wnt/β-catenin signaling pathway 31 . Neuron cells were randomly divided into five groups: (-) control group; (a) LPS group; (b) LPS + XAV939 group; (c) LPS + BMSCs-Exos group; (d) LPS + XAV939 + BMSCs-Exos group. BMSCs-Exos was used to grow neuron cells at a concentration of 100 µg/µl, as described previously 32 .
Treatment of Animals and Exosomes
Some 150 adult male Wistar rats (150–200 g) were purchased from the Laboratory Animal Center of Shandong University (Jinan, Shandong province, China). All animals were randomly assigned into three groups: Sham group, PBS-treated group and BMSCs-Exos-treated group (n=50/group). Animals were kept in a specific pathogen-free laboratory at 24 ± 0.5°C with a 12 h light–dark cycle.
Allen’s method was used to make the SCI model. The T9–T11 laminas were removed in all rats. A 10 g metal weight was dropped from a height of 5 cm to hit the T9–T11 spinal cord of rats in the BMSCs-Exos group and PBS group, and the metal left the spinal cord immediately after the strike. The wounds of each group of rats were finally sutured. After SCI, rats in the BMSCs-Exos treatment group were injected with BMSCs-Exos (200 μg exosomes mixed in 200 μl PBS) immediately, and rats in the PBS treatment group were injected with the same amount of PBS. We then injected the same amount of BMSCs-Exos and PBS into different groups every 3 days until the 27th day after SCI 32 . The Sham group received no injections.
Behavioral Assessment
Motor function after SCI at 1, 3, 7, 14, 21, and 28 days was determined by the Basso, Beattie, Bresnahan (BBB) locomotor rating scale. Three observations of freely moving animals were performed double-blind to assess the ability of all the rats. The average locomotor scores were calculated and recorded.
Western Blot Analysis
Three rats were selected in each group randomly and anesthetized (10% chloral hydrate, 0.33 ml/kg i.p.) at 3, 7, 14, 21, and 28 days after SCI. Spinal cord tissues (1 cm cephalad and caudally around the epicenter) were obtained from the rats. The proteins from the spinal cord tissue and neuron cells were extracted. Western blot analysis was performed as described previously 33 . Primary antibodies anti-β-catenin (1:1000, Cell Signaling Technology, Inc., Danvers, MA, USA), anti-TCF-4 (1:1000, Cell Signaling Technology, Inc.), anti-cleaved caspase-3 (1:1000, Cell Signaling Technology, Inc.), anti-cleaved caspase-9 (1:1000, Cell Signaling Technology, Inc.), anti-Bcl-2 (1:1000, Abcam), anti-Bax (1:1000, Abcam), anti-NeuN (1:1000, Abcam), and anti-β-actin (1:1000, Abcam) were used to compare expression in each group. The results were visualized through using enhanced chemiluminescence reagent (Thermo Fisher Scientific). The density of the protein bands was analyzed using software ImageJ (NIH, Bethesda, MD, USA).
Quantitative RT-PCR Analysis
Three rats were selected in each group randomly and anesthetized (10% chloral hydrate, 0.33 ml/kg i.p.) at 3, 7, 14, 21, and 28 days after SCI. The total RNA was extracted from injured spinal cord by Trizol Reagent (Thermo Fisher Scientific) and used for detection of the relative expression of mRNA. In the study, the mRNA expression of lymphoid enhancer factor-1 (LEF-1: forward primer 5′-TCAGGCAAGCCTACCCATCTTC-3′ and reverse primer 5′-GGGTGCTCCTGTTTGACCTGA-3′; Takara Biomedical Technology, Beijing, China) and T-cell factor-1 (TCF-1: forward primer 5′-AAGATGACACGGATGACGATGG-3′ and reverse primer 5′-GTGTTGCTGCAGGTACGACTTGA-3′, Takara) was used to detect the activation of the Wnt/β-catenin signaling pathway, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH: forward primer 5′-GGCACAGTCAAGGCTGAGAATG-3′ and reverse primer 5′-ATGGTGGTGAAGACGCCAGTA-3′, Takara) was used as internal control. Cycling conditions were as follows: 30 s at 95°C, followed by 40 cycles of 5 s at 95°C, 10 s at 55°C, 15 s at 72°C, and finally melting curve analysis was recorded.
Tissue Preparation
Three rats were selected in each group randomly at 14 days after SCI and anesthetized by 10% chloral hydrate (0.33 ml/kg), perfused transcardially by 150 or 200 ml normal saline and 500 ml 4% paraformaldehyde in 0.01 M PBS. The T9–T11 segments (including the injured epicenter) of the spinal cord were carefully removed from the rats and immersed in 4% paraformaldehyde overnight at 4°C and then embedded in paraffin, and blocked sections were taken with a microtome and mounted onto slides.
Nissl Staining
To observe the overall morphology of the spinal cord, the sections (20 µm thickness) were incubated in cresyl violet (Beyotime, Beijing, China) for Nissl staining. Five randomly selected areas from lesions of gray matter were used to count the Nissl staining-positive cells in the three groups.
Immunohistochemistry
Microwave retrieval of antigens on the sections was done, and then the tissue sections were immersed by 3% H2O2 to block endogenous oxidase. Antigen impurities were blocked by sera at room temperature for 20 or 30 min, and then samples were washed three times with 0.01 M PBS, and the sections were incubated with primary antibody anti-NeuN (1:100, Abcam) overnight at 4°C. The sections were washed with 0.01 M PBS and then incubated with secondary antibody for 20 min. Anti-biotin-labeled peroxidase solution was used to terminate the reaction. Finally, diaminobenzidine (DAB) colorized the sections, which were counterstained with hematoxylin, dehydrated with absolute ethanol and fixed with neutral glue; the nuclei were then observed under 200× microscope. Five view fields were randomly chosen and the number of neuron cells counted from each section. The results were determined by the number of neurons.
TUNEL Staining
TUNEL staining was used to identify the apoptotic rate of neurons in the spinal cord lesion. The sections were incubated overnight with the primary antibody anti-NeuN (1:100, Abcam) and goat anti-rabbit IgG combined with Alexa Fluor-488 (1:400, Invitrogen Corporation, Carlsbad, CA, USA), and following the instructions for the TUNEL reaction mixture (In Situ Cell Death Detection Kit, TMR red; Roche, Mannheim, Germany) the samples were incubated for 1 h in a humidified atmosphere in the dark. The sections were analyzed by fluorescence microscopy (Olympus Corporation, Tokyo, Japan) and the TUNEL-positive cells were counted to calculate the apoptotic rate in each section.
Statistical Analysis
Statistical analyses were performed using Graph Prism Program, Version 7.0 (GraphPad Software, Inc., La Jolla, CA, USA). All data are presented as mean ± SD. The comparison of two groups and among multiple groups was performed using unpaired Student’s t-test and ANOVA, followed by Least Significant Difference post-hoc test, respectively. Statistical significance was considered at p<0.05.
Results
Identification of BMSCs and BMSCs-Exos
The morphology of BMSCs (P3) was observed by optical microscope. BMSCs with swirling and radial arrangement were observed under light microscope (Fig. 1A). The markers CD29 and CD90 were detected in almost all P3 BMSCs. However, the BMSCs scarcely expressed CD34 and CD 45 (Fig. 1B). Ultracentrifugation isolated exosomes from third-generation BMSCs culture supernatant. Transmission electron microscopy (TEM) was used to observe the exosome samples, which presented numerous saucer-shaped vesicles (Fig. 1C). The specific exosome markers CD9, CD63, and CD81 were determined by Western blot (Fig. 1D).

Identification of BMSCs-Exos. (A) Morphology of P3 BMSCs. (B) Phenotypic characterizations (CD29, CD90, CD34, CD45) of P3 BMSCs were identified by flow cytometry. (C) Morphology of exosomes observed by TEM. (D) Western blot analyzed the specific exosome surface markers.
Exosomes Treatment Improves Locomotor Functional Recovery after SCI
At 1, 3, 7, 14, 21, and 28 days after successful SCI modeling of the rats, BBB scores were measured to assess the effect of BMSCs-Exos on locomotor functional recovery (Fig. 2A). Following SCI, the hind limbs of the rats were paralyzed in the BMSCs-Exos and the PBS groups. The locomotor functional recovery of the two groups with different treatment methods improved gradually at 3 days after SCI. The scores of the BMSCs-Exos group were significantly higher than the PBS group between 2 and 4 weeks after SCI (p<0.05). Nissl staining revealed that the overall morphology of the spinal cord and the number of surviving neuronal cells in the BMSCs-Exos group was significantly better than the PBS group at 14 days after SCI (Fig. 2B).

BMSCs-Exos promotes recovery of motor function. (A) Locomotor functional recovery of rats was evaluated by the Basso, Beattie, and Bresnahan (BBB) scores in three groups. Compared with the PBS group, the activity of BMSCs-Exos group was significantly improved within 7–14 days after SCI. **p<0.01 versus the Sham group; ##p<0.01 versus the PBS-treated group. n = 10 per group. (B) The Nissl staining showed the overall morphology of spinal cord and assessed the survival of neurons at 14 days after SCI. Scale bars = 100 μm. (C) Number of gray matter neurons; columns represent the mean ± SD, **p<0.01 versus the sham group; ##p<0.01 versus the vehicle group, n = 3 per group.
Exosomes Inhibit Neuronal Apoptosis in SCI Rats
Western blot results revealed that the protein expression levels of Bax, cleaved caspase-3, and cleaved caspase-9 were obviously up-regulated, and the protein expression level of Bcl-2 was down-regulated in the SCI models (Fig. 3A). However, treatment with exosomes reduced the protein expression levels of Bax, cleaved caspase-3, and cleaved caspase-9 significantly and increased Bcl-2 expression after SCI. The results showed that the Sham group had the largest number of neuronal cells, and the number of neuronal cells in the BMSCs-Exos group was significantly larger than that in PBS group (Fig. 3B–F). Neuronal cells from the three groups were used to detect the neuronal apoptosis ratio by TUNEL/NeuN/DAPI double staining. The results showed that the proportion of apoptotic neurons in the BMSCs-Exos group was significantly lower than that in PBS group (Fig. 3G–H). Moreover, the immunofluorescence results indicated that the BMSCs-Exos group had more neurons than the PBS group (Fig. 3I–J).

Exosomes inhibit neuronal apoptosis. (A) Western blot analysis of the protein expression levels of Bcl-2, Bax, cleaved caspase-3, and cleaved caspase-9 in the spinal cord lesion at 3, 7, 14, 21, 28 days after SCI. (B–F) BMSCs-Exos treatment significantly increased Bcl-2 expression, decreased Bax, cleaved caspase-3, cleaved caspase-9 expression in the spinal cord neurons. Compared with the PBS group, NeuN expression was significantly increased in the BMSCs-Exos group. β-actin was used as the loading control. (G, H) TUNEL/NeuN/DAPI double staining and immunohistochemistry analysis of neuronal cells apoptosis rate in the spinal cord lesion at 14 days. (I, J) BMSCs-Exos reduced the rate of apoptosis-positive cells after spinal cord injury, scale bar = 20 μm. The number and morphology of neurons in BMSCs-Exos group were better than that in PBS group, scale bar = 20 μm *p<0.05, **p<0.01 compared with the Sham group; #p<0.05, ##p<0.01 compared with the PBS-treated group; n = 3 per group.
Exosomes Derived from BMSCs Activate the Wnt/β-catenin Signaling Pathway after SCI
The Western blot results showed that the Wnt/β-catenin signaling pathway was activated after SCI. The BMSCs-Exos group had significantly higher protein expression levels of TCF-4 and β-catenin than the PBS-treated group at 3, 7, 14, 21, and 28 days after SCI (Fig. 4A–D). In addition, after treatment with exosomes, the mRNA expression levels of LEF-1 and TCF-1 were significantly enhanced at 3, 7, 14, 21, and 28 days after SCI. In conclusion, our results revealed that BMSCs-Exos treatment could further enhance the Wnt/β-catenin signaling pathway (Fig. 4E–F).
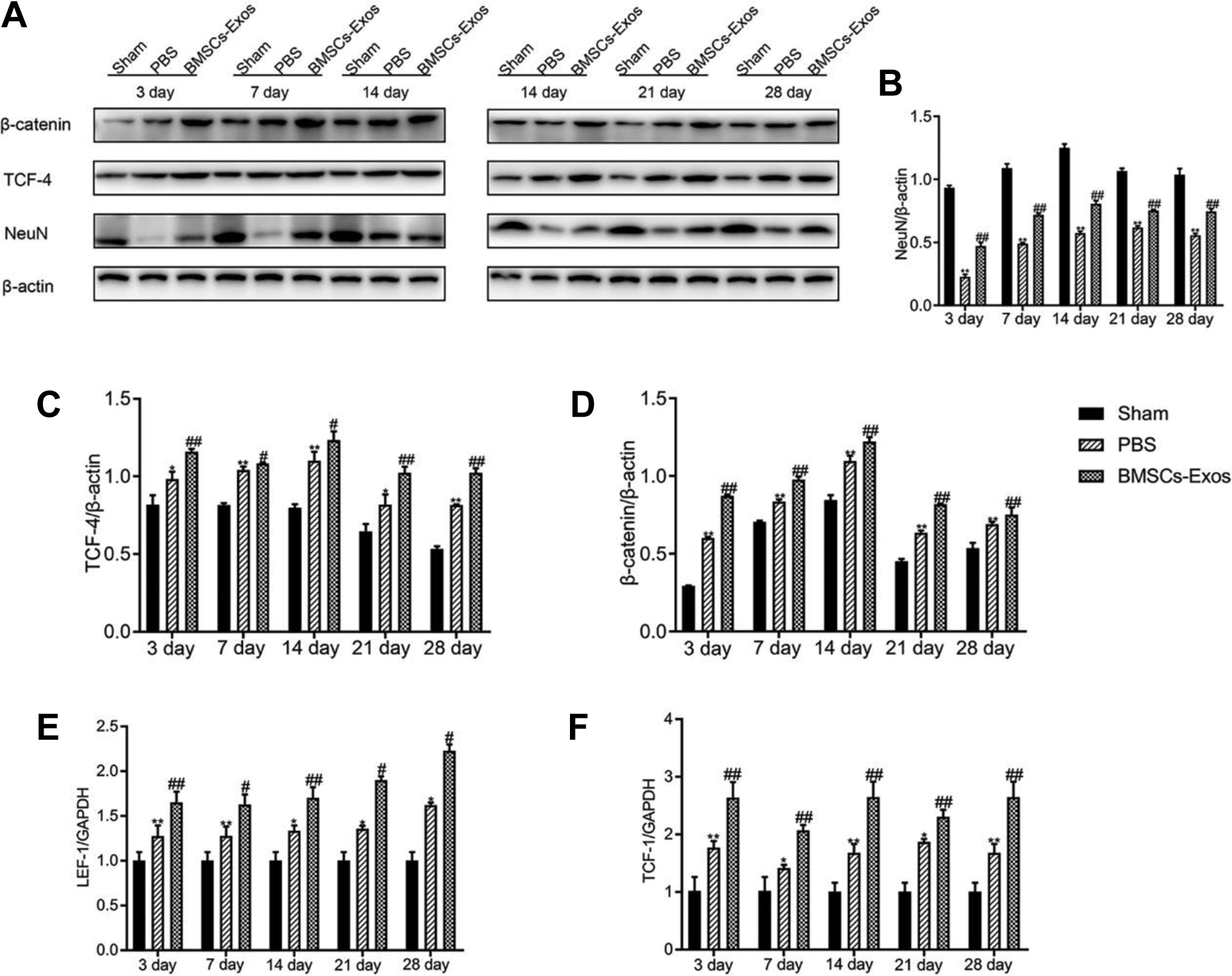
Exosomes derived from BMSCs activate the Wnt/β-catenin signaling pathway. (A) The protein expression levels of β-catenin and TCF-4 in the spinal cord neurons at 3, 7, 14, 21, and 28 days after SCI in three groups, respectively, were detected using Western blot analysis. (B) The protein expression level of NeuN was significantly increased compared with the PBS-treated group. (C, D) BMSCs-Exos treatment significantly increased β-catenin and TCF-4 expression in the spinal cord neurons compared with the PBS-treated group. (E, F) BMSCs-Exos treatment increased the mRNA expression levels of lymphoid enhancer factor-1 (LEF-1) and T-cell factor-1 (TCF-1) after SCI. *p<0.05, **p<0.01 compared with the Sham group, #p<0.05, ##p<0.01 compared with the PBS-treated group; n = 3 per group.
Inhibition of Wnt/β-catenin Signaling Reduced the Anti-apoptotic Effect of BMSCs-Exos in Primary Neurons
Primary spinal cord neurons were treated with LPS to establish an injury and apoptosis model. BMSCs-Exos and XAV939 were used to elucidate that possible mechanism of anti-apoptosis of BMSCs-Exos in vitro. The western blot analysis demonstrated that LPS-induced apoptosis up-regulated the levels of the pro-apoptotic proteins Bax, cleaved caspase-3, and cleaved caspase-9, and down-regulated the level of the anti-apoptotic protein Bcl-2. BMSCs-Exos could significantly increase the number of neuron cells in the LPS-induced neuronal apoptosis model, and this effect could be reversed by the Wnt/β-catenin signaling pathway inhibitor (Fig. 5A–F).

Inhibition of Wnt/β-catenin signaling reduced the anti-apoptotic effect of BMSCs-Exos. (A) Western blot was used to demonstrate the protein levels of Bcl-2, Bax, cleaved caspase 3, cleaved caspase 9, and Neun in the five experimental groups. (B–F) The expression of Bax, cleaved caspase 3, and cleaved caspase 9 decreased in the BMSCs-Exos group. XAV939 could significantly increase the expression patterns of pro-apoptotic proteins while reducing Bcl-2 expression. The anti-apoptotic effects of BMSCs-Exos were reversed after suppressing the Wnt/β-catenin signaling pathway. β-actin was used as the loading control. *p<0.05, **p<0.01, compared with LPS and XAV939 groups; n = 3 per group.
Discussion
SCI can cause devastating disabilities, for instance severe motor dysfunction or paralysis—and even death—leading to a reduction in the quality of life of patients with SCI 34 . Various drugs, surgery, hyperbaric oxygen, physical therapy, and other treatments have been used to treat SCI and its sequelae for more than 50 years in the clinic, but the therapeutic effects of these methods are not very satisfactory 35 . Inhibition of apoptosis of neurons is an important factor in the recovery of spinal cord function 36,37 .
Recently, a series of studies have shown that exosomes secreted by various cells have a positive effect on the repair of SCI, and there are no significant differences, and even an improvement in therapeutic effects and functional properties, between transplantation of exosomes and direct stem cell transplantation 34,38,39 . A growing number of researchers regard transplantation of exosomes as a potential alternative to stem cell transplantation 40,41 . In our study, from the BBB score results, we found that with the prolongation of recovery time, the activity ability of SCI rats improved by better degrees in BMSCs-Exos group. In addition, the results of Nissl staining and immunohistochemistry showed that the overall morphology of the spinal cord and the morphology of neuronal cells were more complete, and the number of neuronal cells was significantly increased in the BMSCs-Exos group compared with the PBS group. The behavioral improvement of the rats and the morphological examination of the tissues prove that exosomes can effectively improve the symptoms of SCI. Our experimental results are similar to previous studies 42,43 .
Apoptosis is a response of cells to physiological or pathological stimulation, which is caused by environmental changes or adaptation to injury 44 . Some diseases, such as cancer, autoimmune diseases, AIDS, and degenerative diseases of the CNS, may be caused by the defect of apoptosis regulation 45,46 . Secondary death of neurons and oligodendrocytes is an important cause of functional defects after SCI 47 . The degree of apoptosis affects functional recovery of SCI, and its occurrence plays an important role in neuronal survival and axonal regeneration 48 . Bcl-2, Bax, and caspase family members are the principal molecular components that may participate in regulating cell apoptosis. SCI promotes the secretion of the pro-apoptotic proteins Bax, cleaved caspase-3, and cleaved caspase-9, but suppresses the secretion of the anti-apoptotic protein Bcl-2 49,50 . In our study, the protein expression levels of apoptotic proteins (Bax, cleaved caspase-3, cleaved caspase-9) was significantly lower in the BMSCs-Exos group than in the PBS group, while the anti-apoptotic protein (Bcl-2) level was observed to be significantly elevated. Moreover, the results of the TUNEL/NeuN/DAPI double staining showed that the apoptosis rate of neurons decreased significantly after BMSCs-Exos therapy. These results, combined with the BBB score and tissue staining, indicate that the exosomes promote neurological recovery by inhibiting neuronal apoptosis.
It has been reported that activation of the Wnt/β-catenin signaling pathway has important effects (stimulation of axon regeneration and inhibition of nerve cell apoptosis) on improving motor function recovery and inhibiting apoptosis of neuron cells 51 –53 . Recent studies have demonstrated that the activation of the Wnt/β-catenin signaling pathway plays a key role in suppressing neuronal apoptosis after acute SCI 54 –57 . Large protein complexes are formed by the accumulation and translocation in the nucleus of Wnt ligands binding with frizzled receptors, low-density lipoprotein receptor-related proteins 5 or 6 (lrp5/6), and β-catenin proteins. In the nucleus, β-catenin binds to TCF/LEF transcriptional activator and activates transcription of downstream Wnt target genes, which participate in pathophysiological changes in a variety of diseases 49,58 . In our study, western bolt results showed that expression of β-catenin and TCF-4 was increased in injured spinal cord neurons. The results of qRT-PCR also showed that expression levels of LEF-1 and TCF-1 in the BMSCs-Exos group were higher than in the PBS group at 3, 7, 14, 21, and 28 days after SCI at the mRNA level. Our in vitro experiments found that BMSCs-Exos significantly reduced the expression levels of cleaved caspase-3, cleaved caspase-9, and Bax proteins, and markedly increased Bcl-2 levels in the primary neurons. Furthermore, the protective effect of BMSCs-Exos was inhibited when the Wnt/β-catenin signaling pathway was suppressed with XAV939. All the results indicated that the Wnt/β-catenin signaling pathway was activated after SCI and that the pathway could promote the recovery of spinal cord function, which could be enhanced by exosomes derived from BMSCs, at least partially.
Our study is the first to investigate the potential for exosomes from BMSCs to reduce tissue damage, improve functional recovery, and inhibit neural cell apoptosis after SCI by activating the Wnt/β-catenin signaling pathway. The results of our study may provide a new molecular mechanism to promote neuroprotection by exosomes. In addition, activation of the Wnt/β-catenin signaling pathway by substances in the exosome is worth investigating.
Footnotes
Ethical Approval
Ethical approval to this original research was obtained from the Ethical Committee of Qilu Hospital of Shandong University, Shandong Province, China.
Statement of Human and Animal Rights
The animal experiments in this study were approved by the Animal Care and Research Committee of Qilu Hospital of Shandong University. All experiments were performed with relevant laws and guidelines, and following institutional guidelines of Qilu Hospital of Shandong University.
This article does not contain any studies with human participants performed by any of the authors.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC) (No. 81602361) and Jinan Science and Technology Project (No. 201805042).
