Abstract
The selection of optimal pancreas donors is one of the key factors in determining the ultimate outcome of clinical islet isolation. North American Islet Donor Score (NAIDS) allows for estimating the chance of the success of islet isolation. It was developed based on the data from over 1000 donors from 11 islet isolation centers and validated in the University of Alberta, Edmonton, on the cohort from the most active islet transplant center. Now we aimed to also validate it in our much less active program. Areas under the receiver operating characteristic curves (AUROCs) and logistic regression analyses were obtained to test if NAIDS would better predict successful islet isolation (defined as post-purification islet yield >400,000 islet equivalents (IEQ)) than previously described Edmonton islet donor score (IDS) and our modified version of IDS. We analyzed the donor scores with reference to 82 of our islet isolation outcomes. The success rate increased proportionally as NAIDS increased, from 0% success in NAIDS < 50 points to 40% success in NAIDS ≥ 80 points. AUROCs were 0.67 (95% confidence interval (CI) 0.55–0.79) for NAIDS, 0.58 (95% CI 0.44–0.71) for modified IDS, and 0.51 (95% CI 0.37–0.65) for IDS and did not differ significantly. However, based on logistic regression analyses, NAIDS was the only statistically significant predictor of successful isolation (p = 0.01). The main advantage of NAIDS is an enhanced ability to discriminate poor-quality donors than previously used scoring systems at University of Chicago, with 0% chance for success when NAIDS was <50 as compared with 40% success rate for IDS <50. NAIDS was found to be the most useful available tool for donor pancreas selection in clinical and research practice in our center, allowing for identification and rejection of poor-quality donors, saving time and resources.
Keywords
Introduction
Pancreatic islet transplantation (ITx) is a therapeutic option for patients with “brittle” type 1 diabetes (T1DM) with the potential to improve metabolic control and prevent hypoglycemia. Currently, most patients require subsequent islet infusions to maintain long-term islet graft function and the beneficial effect of procedure-improved glucose control. The majority of patients (>90%) maintain at least partial islet function for over 5 years, some for over 10 years 1 –3 . Pancreatic islet mass, represented by islet equivalent (IEQ), is the most important parameter that correlates with clinical outcome after transplantation 4 . For this reason, it is the most important parameter in decision making for clinical islet transplantation. The selection of an optimal pancreas donor is one key factor in determining the ultimate outcome of human islet isolation. To facilitate the selection of a suitable organ, methods for the systematic, quantitative assessment of donor quality have been developed for both pancreas transplantation 5,6 and ITx 7,8 . In 2005, based on their own experience, the Edmonton group developed the Pancreas Donor Scoring for the purpose of islet transplantation (IDS) 7 . This model identified factors predictive of poor isolation outcomes, including donor factors as well as the physical properties of the pancreas. IDS ranges between 0 and 100 points; the higher the score, the higher the chance for islet isolation success. This success is defined as islet yield >300,000 IEQ. Since then, the scoring system has been adopted in many islet centers. At University of Chicago, we first internally modified IDS by excluding factors related to pancreas quality of preservation in order to enable early decision making based only on donor-derived data, and to avoid unnecessary organ procurement and shipment (Table 1; data unpublished). Later, we developed and validated a new pancreas scoring system (NAIDS) based on data from multiple islet centers 8 . We designed and conducted a retrospective, multicenter analysis of data from 11 islet isolation centers in North America. The data set from 1056 deceased donors was used for the development of the scoring system to predict islet isolation success (defined as post-purification islet yield >400,000 islet equivalents). Next, NAIDS was validated on a cohort in the University of Alberta, Edmonton. It is crucial that limitations and applicability of assessment tools are recognized prior to their use in clinical practice; therefore, this study aimed to validate NAIDS externally, for use at our ITx program, which is much less active than Edmonton. To assess the utility of NAIDS we reviewed the outcomes of islet isolation procedures performed at our institution with reference to donor scores calculated according to IDS, our modification of IDS, and NAIDS.
The University of Chicago modification of Islet Donor Score (IDS).

* Refer to local reference values
** Specific biomarkers are defined as blood glucose, amylase, lipase. Abnormal value in Illinois is Lipase >190U/L, Amylase >200 U/L and Glucose >200 mg/dL(or >11.1 mmol/L)
*** Other biomarkers include AST, ALT, Creatinine, BUN. Abnormal value in Illinois is ALT >50IU/L, AST >40IU/L, Creatinine >1.38 mg/dL and BUN>24 mg/L.
Materials and Methods
Study Design
The charts of all deceased donors of pancreatic islets were retrospectively reviewed to assess their donor points according to the previously described Edmonton IDS, our modification of IDS, and NAIDS. The values of all islet donor scores were analyzed with reference to post-purification islet yield as expressed in IEQ. Successful islet isolation was initially defined as post-purification islet yield greater than 400,000 IEQ. We also analyzed scenarios in which successful isolation was considered either 350,000 IEQ or 300,000 IEQ. Cases other than successful islet isolation were labeled as failed islet isolation. All donor data were entered in REDCap Project in the University of Chicago website before analysis.
Donor Selection
We pre-selected only brain dead; HIV, HBV (HBsAg, HBcAb) and HCV seronegative; NAT negative; and non CDC high-risk donors according to Centers for Disease Control 9 with HbA1c below 6. Next, the primary investigator, a transplant surgeon, made a decision of acceptance or rejection based on modified IDS score, urgency of the transplant, surgical comfort of procuring surgeon, and expected cold ischemia time (CIT) below 12 h. Pancreata were then shipped to the islet isolation facility for processing, in cold (4°C) preservation solution SPS1 (Organ Recovery System, USA) or histidine-tryptophan-ketoglutarate (Köhler Chemie GmbH, Germany).
Islet Isolation
Islets were isolated in a good manufacturing practice (GMP) facility at the University of Chicago using the Ricordi standard semiautomated procedure 10 . Briefly, collagenase (Liberase; Roche, Indianapolis, IN) solution was infused through the main pancreatic duct, and the organ was digested in the Ricordi Chamber (Biorep Technologies, Miami, FL). After digestion, all tissue was collected, and islets were purified with a continuous density gradient in the COBE 2991 Cell Processor (Caridian BCT, Lakewood, CO). Islet yield was determined by manual count of dithizone-stained samples, converting the different islet sizes into IEQ 11 .
Islet Donor Scores
IDS
This continuous model consists of nine donor factors (age, body mass index (BMI), cause of death, length of hospital stay, amylase/lipase activity, use of vasopressors, blood glucose, social and medical history), two transplant/procurement factors (CIT and procurement team), and seven pancreas physical properties (size, quality of flush, consistency, quality of procurement, damage, quality of packaging, fat content). The combination of the donor quality, transplant/procurement factors, and pancreas quality factors was given a numerical score from 0 to 100 to determine the quality of a pancreas for islet isolation. The higher the score, the higher the chance for islet isolation success, defined as islet yield >300,000 IEQ 7 .
Modified IDS
Our modification of the IDS model included only eight donor factors and a single procurement factor (Table 1). The pancreas quality factors were excluded to allow actual organ selection before the procurement. The range of donor points extends from 0 to 100 and as in IDS, the higher the score, the higher the chance for islet isolation success.
NAIDS
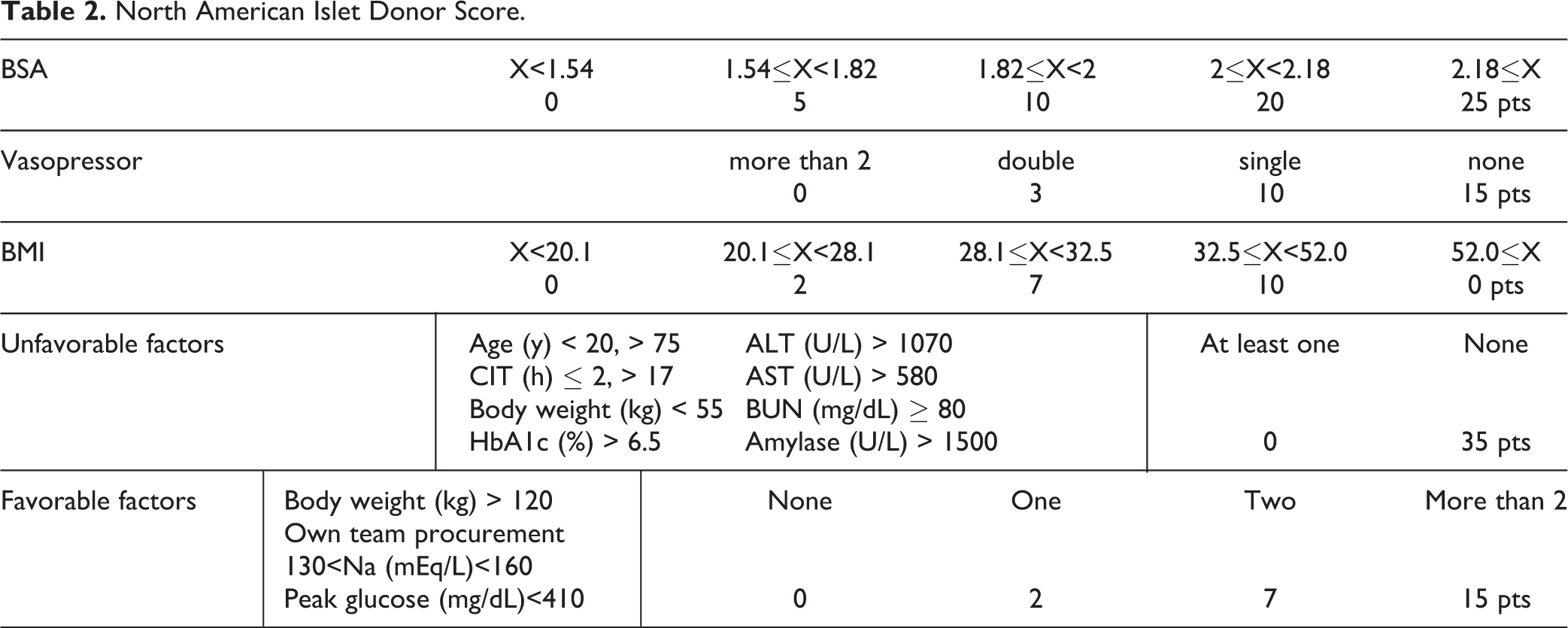
The NAIDS is a comprehensive scoring system. It consists of donor variables that predict islet isolation success with post-purification IEQ yielding over 400,000 out of a total of 100 points. A higher NAIDS corresponds to a higher success rate. Donor body surface area (BSA), BMI, and use of vasopressors were recognized as main donor variables included in the NAIDS. There were also two supplemental composite factors—favorable (based on serum Na and glucose, along with body weight (BW) and own organ-procuring team), and unfavorable (based on age, CIT, BW, serum HbA1c, liver enzymes, blood urea nitrogen (BUN) and amylase). BSA is given a maximum of 25 points, followed by BMI with 10 points. The absence of vasopressor usage is awarded a maximum of 15 points. If there are no unfavorable factors, 35 points are given. Favorable factors are given a maximum of 15 points (Table 2) 8 .
North American Islet Donor Score.
Statistical Analysis
Descriptive statistics are expressed as mean ± standard deviation or median with interquartile range (IQR) as appropriate. Data were tested for normality. Receiver operating characteristic curves were made for IDS, modified IDS, and NAIDS values. The area under the receiver operating characteristic (AUROC) curves were compared between the donor scores to determine which of the indices detected the outcome (i.e., post-purification islet yield >400,000 IEQ or >350,000 IEQ or 300,000 IEQ) with greater discrimination. An AUROC ≤ 0.5 was considered no discrimination, an AUROC between 0.7 and 0.8 was considered acceptable, an AUROC between 0.8 and 0.9 was considered excellent, and an AUROC > 0.9 was considered outstanding. Univariate logistic regression analysis was performed to identify the predictor of successful islet isolation among the donor scores. A p value of less than 0.05 was considered statistically significant. The statistical analyses were performed using Statistica 12 (StatSoft, Poland) software.
Results
Donor Characteristics
The data set consisted of 82 consecutive human islet isolation procedures performed at our institution between 2007 and 2011, 18 research-grade (to train new personnel and optimize the conditions) and 64 clinical-grade isolations (with the goal to transplant) (Table 6). Two research-grade isolations were excluded from the analysis due to technical failure and early termination of the cell processing.
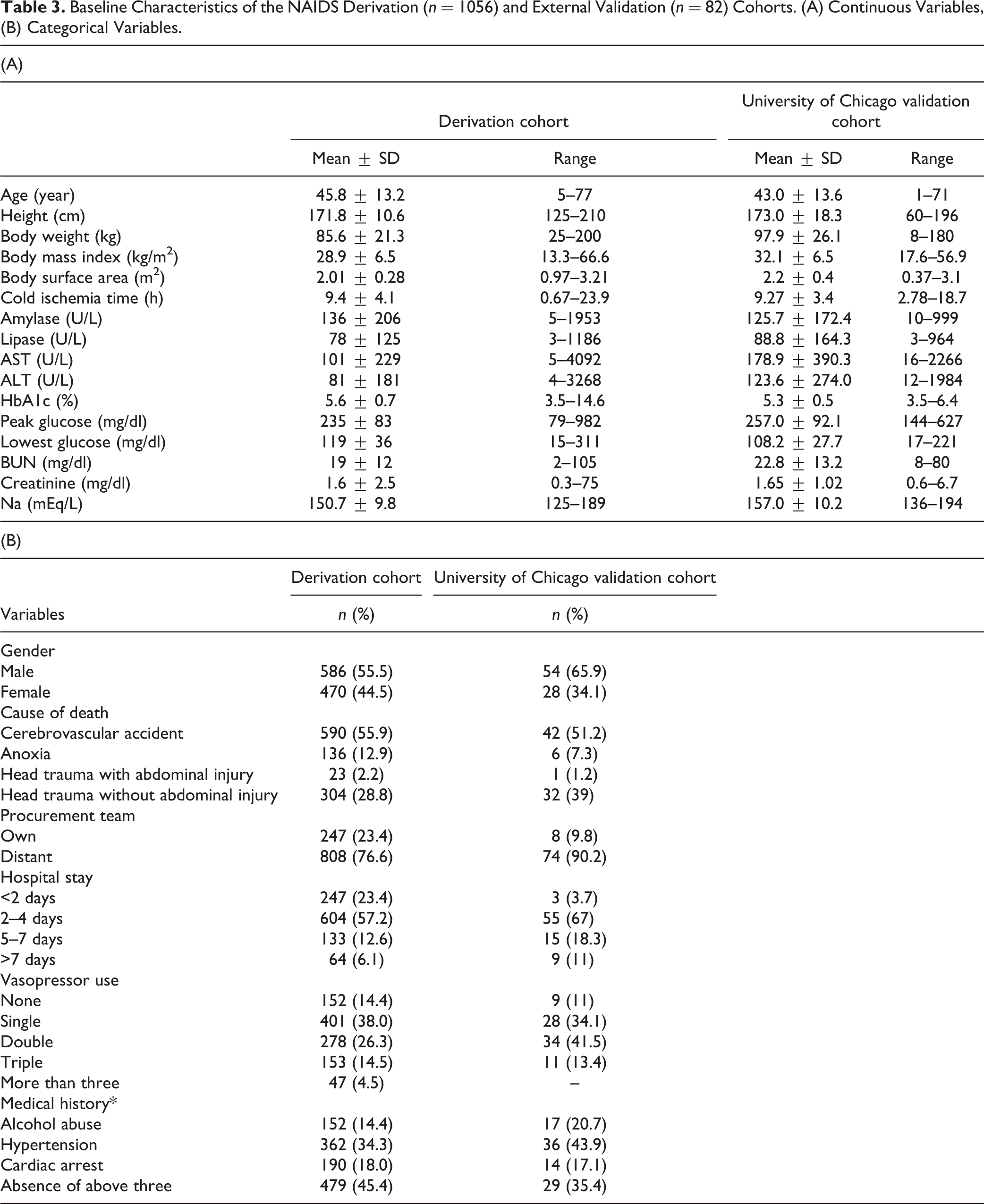
The external validation cohort included a significantly lower number of patients than both the derivation and Edmonton validation cohorts. The average donor for this cohort was similar to the reference donor described in the NAIDS derivation model in terms of age, BMI, and gender, but there were significantly more deaths due to head trauma without abdominal injury. We also used pancreata procured by a distant team significantly more often. The donor demographics for this cohort and the cohort used to derive the NAIDS are shown in Table 3.
Baseline Characteristics of the NAIDS Derivation (n = 1056) and External Validation (n = 82) Cohorts. (A) Continuous Variables, (B) Categorical Variables.
Donors were grouped into five strata depending on the total number of points achieved in each of the scoring systems. The distribution of results differed according to the scoring model used; that is, less than 4% of University of Chicago donors had ≥ 80 points in IDS, only 4.9% according to modified IDS in comparison to 30.5% of donors with ≥ 80 points when NAIDS was calculated. Examination of NAID scores showed that the organs utilized were similar between derivation and original validation cohort, with 51.6% and 56.4% of pancreata from donors with NAIDS above 60 points in the derivation and original validation cohort, respectively. However, analysis of NAIDS distribution in our center’s cohort showed skewed scores, with few low-score donors and a preponderance toward a higher proportion of high NAID scores with 75.6% of scores above 60 points (Table 4).
The Characteristics of Utilized Pancreata.

Univariate Logistic Regression Analysis
Of the 82 analyzed cases, 25 (30.5%) were successful islet isolations. This rate was similar to the NAIDS derivation cohort, but lower than 50.5% (90/179) in the Edmonton validation cohort. Logistic regression confirmed that NAIDS was a statistical significant predictor of the isolation success defined as 400,000 IEQ with odds ratio (OR) of 1.05 (95% confidence interval (CI) 1.01–1.09) with p = 0.01, whereas IDS and modified IDS were not (p = 0.83 and 0.26, respectively). When the cut-off for isolation success was set as 350,000 and 300,000 IEQ the results were similar with OR of 1.04 (95% CI 1.01–1.07) with p = 0.01 and OR of 1.03 (95% CI 1.00–1.05) and p = 0.03 for NAIDS, respectively.
Success Rate Based on Various Donor Scores
The success rate increased proportionally as the NAIDS increased, from 0% success in NAIDS < 50 points (0/13) to 40.0% success (10/25) in NAID score ≥ 80 points. When the NAIDS was between 50 and 80 points, the chance for success was approximately 34% (15/44). Distribution of the success rate depending on the NAIDS in the University of Chicago cohort was similar to the distribution of the success rate in the Edmonton and multicenter derivation cohorts (Fig. 1). Distribution of the success rate for scores between 50 and 80 was similar for NAIDS and IDS. Similarly, when the total number of points exceeded 70, the chance for islet yield greater than 400 kIEQ was around 40% irrespective of the scoring system used. However, when NAIDS was < 50 there was 0% chance for success, while when IDS was < 50 there was still 40% chance of success with the threshold set at 400 kIEQ (p = 0.02). With the total number of points below 60, the difference in the number of successes between NAIDS and IDS was of borderline significance (p = 0.09).
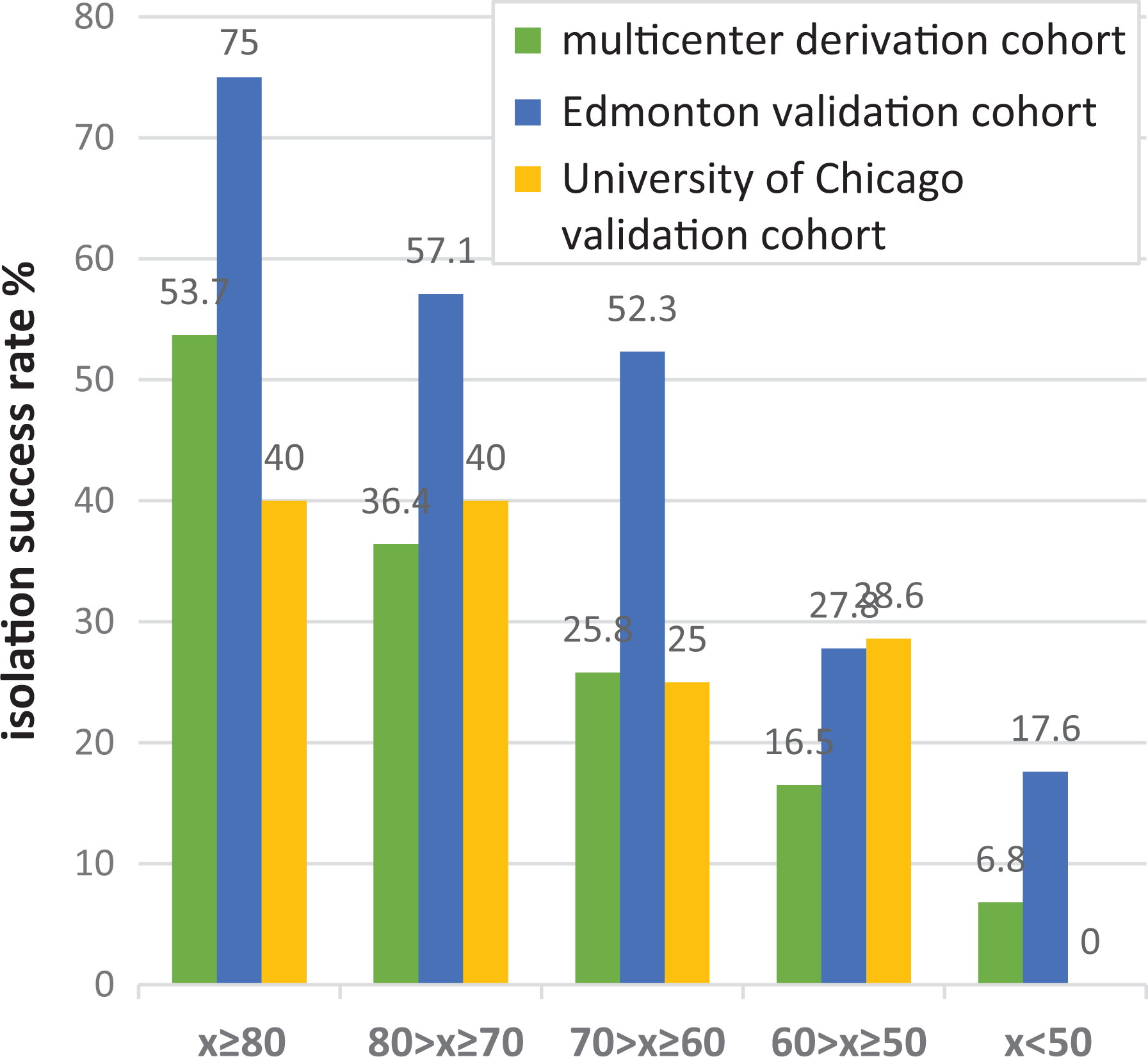
Distribution of the isolation success rate (post-purification islet yield greater than 400,000 IEQ) depending on NAID score in the multicenter derivation cohort (n = 1056), Edmonton validation cohort (n = 179), and University of Chicago (n = 82).
We identified 32 isolations when the difference in scores, assuming pancreas from the same donor, between NAIDS and IDS was equal or greater than 19.5 points (e.g., NAIDS 84 points and IDS 64 points; NAIDS 47 points with IDS 81 points). These included 24 cases when NAIDS was greater and eight cases when IDS was higher. When NAIDS was greater than IDS then isolation success, defined as post-purification islet yield greater than 400,000 IEQ, was achieved in 11 out of 24 (46%) cases. In eight cases when IDS was at least 19.5 points greater than NAIDS, there were no successful isolations (p = 0.029).
Validation of the NAIDS
For validation of the NAIDS, we plotted the receiver operating characteristic curves using the multicenter derivation (n = 1056) and the University of Chicago validation cohort data (n = 82) (Figure 4). The AUROC for validation cohort data for isolation success defined as post-purification islet yield greater than 400,000 IEQ was 0.67 (95% CI 0.551–0.79) (Table 5, Fig. 2), which was not significantly different (p = 0.39) from that obtained from the development cohort. Furthermore, a similar proportional increase in a success rate was observed as the NAIDS increased in the validation cohort (Fig. 3). AUROCs for modified IDS and IDS were 0.58 (95% CI 0.44–0.71) and 0.51 (95% CI 0.37–0.65), respectively. However, the difference between the AUROCs for various scoring systems did not reach statistical significance. The results were comparable when the threshold for isolation success was set at either 350 kIEQ or 300 kIEQ.
The Areas Under Receiver Operating Curves (AUROC) of the NAID, Modified ID, and ID Scores for the Detection of Isolation Success (n = 82).

AUROC: area under receiver operating curve; CI: confidence interval; IDS: Islet Donor Score; NAIDS: North American Islet Donor Score
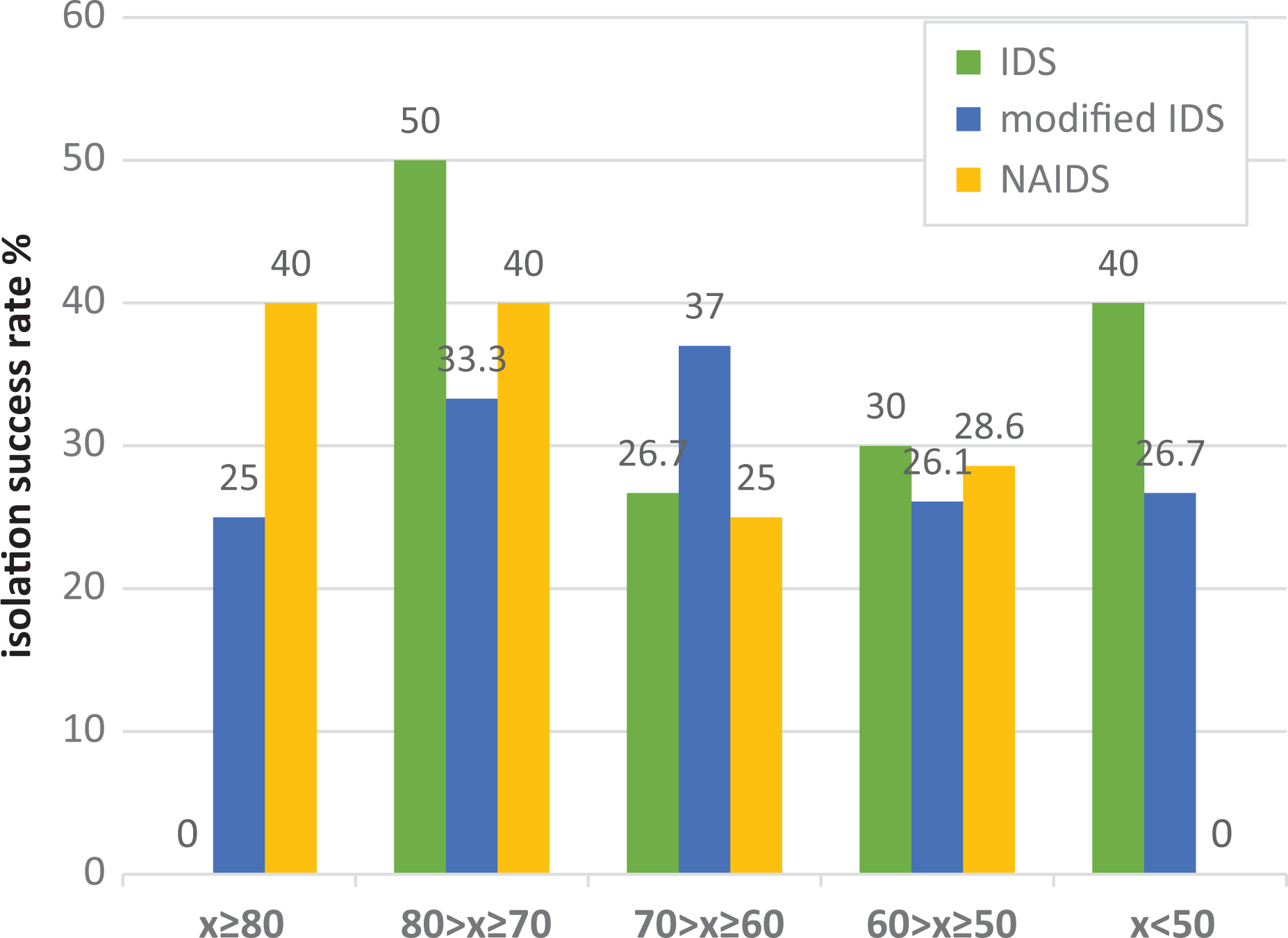
Distribution of the isolation success (postpurification islet yield greater than 400,000 IEQ) rate according to various donor scores (IDS, modified IDS and NAIDS) in University of Chicago validation cohort (N = 82).
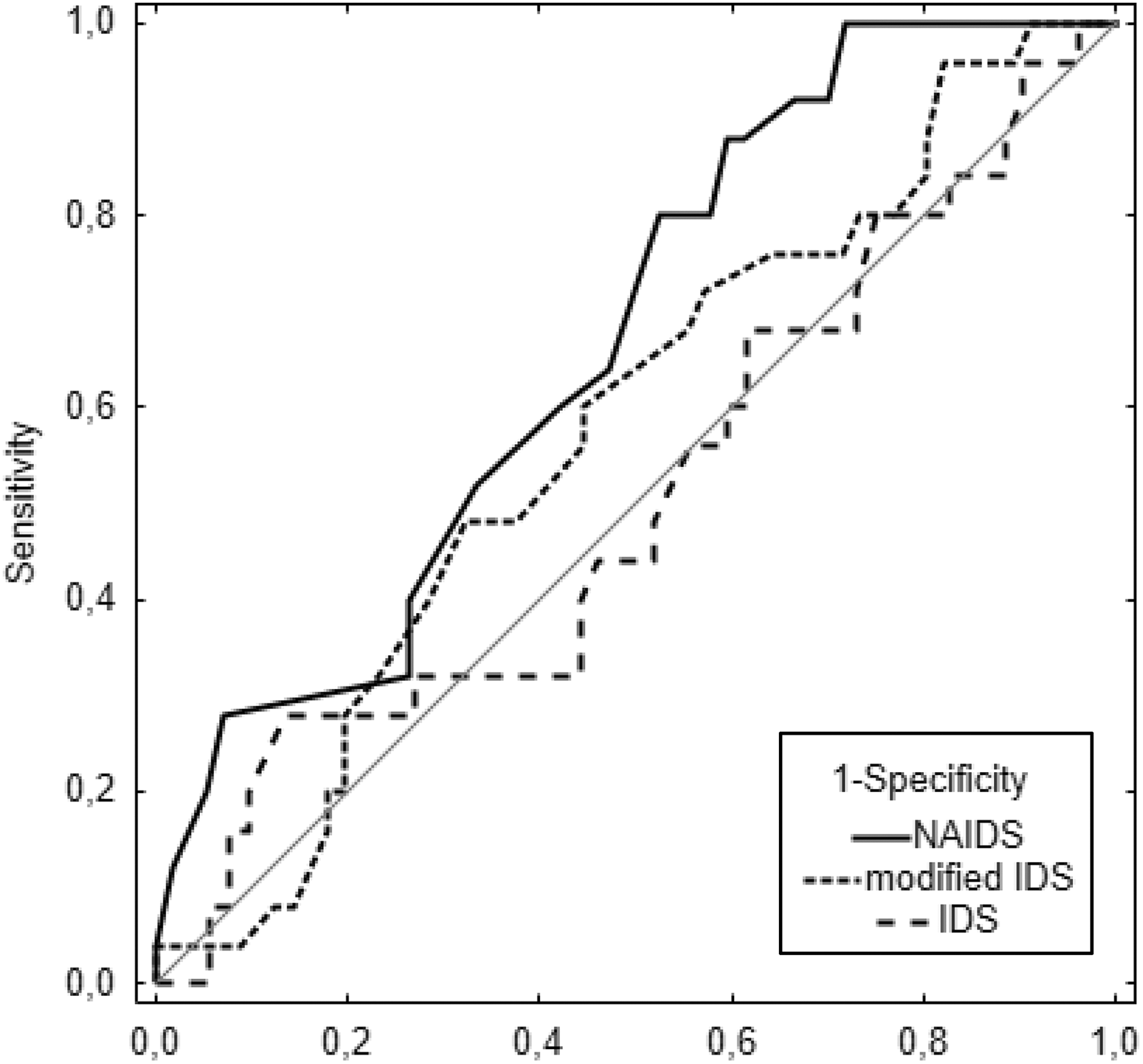
Receiver operating characteristics (ROC) curves of the NAIDS, modified IDS, and old IDS in the University of Chicago validation cohort for the identification of islet isolation success defined as total post-purification IEQ > 400,000.

Receiver operating characteristics (ROC) curves of the NAIDS in original derivation cohort (n = 1056), original derivation cohort (Edmonton) (n = 179), and the University of Chicago validation cohort (n = 82) for the identification of islet isolation success defined as total post-purification IEQ > 400,000.
University of Chicago Isolation Characteristics (n = 82).

* data for 45 infused final islet preparations
** endotoxin level was < 5 Eu/kg in all cases
Discussion
The outcome of human islet isolation depends on donor-, pancreas-, and isolation-related variables 12,13 . Despite improvements and standardization of isolation procedures, the outcome of human islet isolation remains unpredictable and highly variable. The most experienced centers are only able to obtain a sufficient islet yield from 30–50% of isolation procedures 8 . A careful selection of pancreata for processing should allow higher transplant efficiencies. Therefore, a reliable method of systematic, quantitative assessment of donor quality is highly desired. Since its introduction in 2005, IDS has been the mainstay of donor evaluation for the isolation of pancreatic islets. Once the clinical utility of a new donor score is established, internal and external validation is required prior to the implementation into clinical practice to assess the applicability in a particular context. The objective of this single-center study was to compare IDS, our modification of IDS, and recently introduced NAIDS in our own medium-volume center in their ability to facilitate recognition of a suitable pancreas donor among all reported donors.
NAIDS incorporates five criteria (including two composite ones) for the estimation of pancreas weight and quality. The rationale for the choice and arrangement of these variables has been described in detail in the original NAIDS development study 8 . Briefly, 50 out of a maximum of 100 points are assigned for BSA, BMI, and the number of vasopressors used in the donor. BMI has long been shown by many authors to positively correlate with islet isolation outcome 13 –18 . Recently, a positive association has also been found between islet size, islet insulin content (normalized for size), basal or stimulated insulin secretion, and BMI 19 . However, on the other hand, Olehnik and colleagues observed high inter-individual heterogeneity of beta-cell mass, with no clinically significant correlation with BMI. BMI is used as a clinical indicator of obesity, but it in fact does not differentiate between adipose tissue and muscle mass 20 . This could, at least in part, explain the inter-individual heterogeneity of beta-cell mass irrespective of BMI value. It is also possible that the composition of extracellular matrix structure in obese donors favors liberating islets from exocrine tissues and therefore results in a higher islet yield, when the islet number is in fact similar or lower. Kin et al. reported that BW and BSA were more strongly correlated with pancreas weight 21 . To ensure a more accurate estimation of isolation outcomes, NAIDS uses combined variables of BSA, BMI, and BW, for which positive correlation with pancreas size and islet yield were previously described 8,22 . According to NAIDS, the lack of donor vasopressor treatment predicts a favorable outcome. However, conflicting results regarding the influence of vasopressor use in donors on the isolation outcomes have been reported with no effect 4,18 , elevated yield 16 , or reduced islet yield 14 . The need for vasopressors reflects the cardiovascular status of the multiorgan donor with unsatisfactory tissue perfusion and progressive organ damage, including the pancreas. Ischemia of the pancreatic tissue leads to more extensive islet cell damage or death prior to or during the very stressful islet isolation process, exposing islets to mechanical (shaking, spinning) and enzymatic injury during the digestion phase. The effectiveness of digestion is more related to the enzyme quality, as well as donor age, which affects the extracellular matrix composition including the type of collagen present, rather than the size of the pancreas 23 . For that reason, young donors (i.e., below 20 years of age) are not preferred due to poor results of the digestion phase 23 .
NAIDS contains almost the same variables as the previous IDS; however, the number of points assigned for each factor and the formula of the score calculation is different. Therefore, there may be a substantial difference in the distribution of results according to scoring model used; that is, one donor could have been awarded a significantly lower number of points in one of the scoring system as compared with another, despite very similar donor data. AUROC was higher for NAIDS than for IDS, although for our relatively low number of cases the difference was not statistically significant. Even though in our validation cohort the AUROC for the identification of isolation success did not reach the threshold of 0.70 typically considered useful for clinical decision making, it still performed best out of the three scoring systems. Still, there was a clean-cut trend when analyzing the success rate based on various donor scores. We noticed that when the NAIDS is below 50, the chance of success was 0%, so it is not worth accepting and processing the pancreas for islet isolation; however, we can still consider isolating the islets when the score is between 50 and 80 as the chance of success is around 30%. With a score over 80 and the chance of success around 40%, we should proceed with the islet isolation in our center. The rate of successful isolations was similar irrespective of the scoring system used whenever the sum of points exceeded 70. Our results indicate that with the higher total number of points achieved, either all scoring systems are equally useful or we were unable to show the difference due to a small number of cases included and a variety of factors affecting isolation outcomes, which are not included in any of the scoring systems.
The main advantage in guiding clinical decisions using NAIDS over IDS and modified IDS is that it was more effective in identification of donors who would most likely fail. So if we rejected pancreata based on donor IDS or modified IDS, we would reject potentially good organs. On the other hand, pancreas rejection based on NAIDS improves the cost-effectiveness of isolation process, saving time and valuable resources. In cases when the use of different scoring systems generates divergent results, NAIDS better reflects the pancreas donor quality.
The critical message of our study is that NAIDS allows assessment of the pancreas donor quality prior to processing, not only in the most experienced center—Edmonton—but also in smaller centers. We externally validated it. NAIDS assists in making a difficult decision—whether to accept donor and pancreas for processing or reject due to low chance for success—easier. The cost of each islet isolation is around $20,000 in the US, in addition to the cost of the GMP facility maintenance and an on-call isolation team 24/7. From the economic perspective, one can assume that it would be optimal to wait for a perfect donor (NAIDS > 80) in order to maximize the chance for successful isolation and not to waste time and money on isolation failures. However, such strategy may result in rare pancreas processing and inefficiency due to high cost of the team and facility maintenance without productive work. Each islet center needs to develop its own donor selection strategy and islet isolation rate per month based on overall pancreas donor availability, which depends on the local, regional, or national efficiency of procurement. It also depends on activity of the pancreas transplant programs, or competitive islet transplant centers. Overall population BMI might be higher in the USA compared with European countries, which can relatively increase availability of higher NAIDS donors in the USA compared with other countries. Between January 2013 and April 2015, we participated in a multicenter trial with competitive enrollment, so as a center we were continuously fully active, ready to accept pancreata for processing. Over a 2-year period, we performed 40 islet isolations with an overall success rate of 50% (20/40) with IEQ > 400,000, which resulted in clinical transplants in our 12 patients. Among those, there were 13 (32.5%) donors with NAIDS ≥ 80, 19 (47.5%) with NAIDS ≥ 60, and 8 (20%) below 60. The success rate was 62%, 50%, and 25%, respectively for the NAIDS groups. Taking into consideration all the above factors, our previous experience, and data with different scoring systems, we decided not to process pancreata with NAIDS below 60.
Of note, there was a substantial difference in the percentage of successful isolations between the Edmonton isolation cohort and the University of Chicago isolation cohort when donors were stratified according to NAIDS. The success of the isolation depends on three main components: donor quality, pancreas procurement quality, and experience and efficiency of the islet isolation team. NAIDS takes into consideration only two of them: donor characteristic and procurement team. Islet isolation is still a very complex process and, despite standardization, decisions made based on the experience of the leader of the isolation team play very important role in the ultimate outcome. Poor decisions may lead to failure despite processing a perfect pancreas. The Edmonton group has the most experienced team and islet isolation system based on thousands of isolations during the last 20 years. For that reason, the results of processing the same quality pancreas are overall better there than in our and other centers. In addition, most pancreata are procured for us by distant teams with different levels of experience in pancreas procurement. Very poor procurement with pancreas injury, ischemia, poor perfusion, and inefficient organ cooling or preservation may remain undetectable by the receiving islet center but severely compromise pancreas quality, and may completely preclude success of islet isolation. This most likely also affected our outcomes. Again, there is much more experience with pancreas procurement in Edmonton and Canada. Altogether, despite the substantial difference in success rates in outcomes between our center and Edmonton in respective ranges of scoring systems, NAIDS allowed us to properly stratify the quality of the pancreas donor, especially to identify poor-quality donors and to avoid many isolation failures.
Several shortcomings of this study should be mentioned. This was a small study restricted to a single center with particular strategies for donor selection and islet isolation. We believe that we could have demonstrated better efficacy of NAIDS in an adequately powered study with a significantly larger number of cases included. Data were collected prospectively and analyzed retrospectively with no actual donor selection decisions based on only estimation of NAIDS. Despite these limitations, we believe that our study is of importance. This study is the first external validation that assesses the performance of NAIDS in a population external to and independent from the original derivation and validation cohorts.
In conclusion, NAIDS allows for acceptable correlation of the score with the outcome of the islet isolation. It allows us to tailor donor selection strategy, excluding donors with minimal chance for success with a score below 50 and accept remaining pancreata with a chance of success of 30–40%. Validation of NAIDS against data from our center shows that it is currently the best available tool for donor pancreas selection in clinical and research practice, allowing for adjusting the donor selection strategy to the center’s logistic and financial resources.
Footnotes
Ethical Approval
This study protocol was reviewed and approved by the Institutional Review Board (IRB) of the Division of Biological Sciences at the University of Chicago and was identified as nonhuman subject research, as determined by Federal Regulations.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by US Public Health Service Grant DK-020595 to the University of Chicago Diabetes Research and Training Center as well as Illinois Department of Public Health Grant “Pancreatic Islet Transplantation.”
