Abstract
Adoptive transfer of alloantigen-specific immunomodulatory cells generated ex vivo with anti-CD80/CD86 mAbs (2D10.4/IT2.2) holds promise for operational tolerance after transplantation. However, good manufacturing practice is required to allow widespread clinical application. Belatacept, a clinically approved cytotoxic T-lymphocyte antigen 4-immunoglobulin that also binds CD80/CD86, could be an alternative agent for 2D10.4/IT2.2. With the goal of generating an optimal cell treatment with clinically approved reagents, we evaluated the donor-specific immunomodulatory effects of belatacept- and 2D10.4/IT2.2-generated immunomodulatory cells. Immunomodulatory cells were generated by coculturing responder human peripheral blood mononuclear cells (PBMCs) (50 × 106 cells) with irradiated donor PBMCs (20 × 106 cells) from eight human leukocyte antigen-mismatched responder–donor pairs in the presence of either 2D10.4/IT2.2 (3 μg/106 cells) or belatacept (40 μg/106 cells). After 14 days of coculture, the frequencies of CD4+ T cells, CD8+ T cells, and natural killer cells as well as interferon gamma (IFN-γ) production in the 2D10.4/IT2.2- and belatacept-treated groups were lower than those in the control group. The percentage of CD19+ B cells was higher in the 2D10.4/IT2.2- and belatacept-treated groups than in the control group. The frequency of CD4+CD25+CD127lowFOXP3+ T cells increased from 4.1±1.0% (preculture) to 7.1±2.6% and 7.3±2.6% (day 14) in the 2D10.4/IT2.2- and belatacept-treated groups, respectively (
Keywords
Introduction
Clinical transplantation is a well-established treatment option for patients with end-stage organ disease, and the outcome of transplantation has improved substantially over the last half-century. However, most of the current immunosuppressive regimens are still inadequate to permit long-term graft acceptance and function. Only 55% of transplant recipients retain a functional graft at 10 years following transplantation 1 . In addition, because of the non-specificity of immunosuppressants such as calcineurin inhibitors, anti-metabolites and steroids, these recipients are at significantly increased risk for concomitant adverse effects, including opportunistic infections, metabolic disorders, nephrotoxicity, and malignancies 2,3 . Although strategies to minimize the chronic use of immunosuppression, such as T cell-depleting therapy, have been attempted, complete withdrawal of immunosuppressive drugs following liver transplantation has mainly proven unsuccessful 4,5 . Strategies to induce specific immune-tolerogenic effects towards transplanted organs and/or tissues, thus avoiding the chronic use of conventional immunosuppressive agents, are essential for further improving long-term outcomes following transplantation.
One promising strategy is the use of suppressor/anergic cells to minimize the dosage of immunosuppressants and/or to induce operational tolerance 6 –8 . After the identification of the role of regulatory T cells (Tregs) in self-tolerance 9 , adoptive transfer of Tregs has been attempted in organ transplantation 10,11 . The use of expanded natural Tregs or donor antigen-specific Tregs has shown promise in preclinical models and early clinical trials 12 –14 . However, Treg purification and expansion methods, for example using anti-CD3/CD28 coated beads together with rapamycin 15 or interleukin (IL)-2 16 , require highly specialized facilities and complicated techniques, which become quite costly and, therefore, hamper the possibility for widespread application.
Donor antigen-specific immunomodulatory cells can be generated by culturing recipient lymphocytes stimulated by donor antigens while blocking the CD28/B7 costimulatory pathway 17 –20 . Bashuda et al. established a method to generate donor-specific immunomodulatory cells ex vivo using anti-CD80/86 monoclonal antibodies (2D10.4/IT2.2) and showed that, in rhesus monkeys, long-term renal graft function was achieved even after complete withdrawal of immunosuppressants after infusing the animals with ex vivo generated immunomodulatory cells 21 . This strategy was also applied in a clinical trial comprising 10 consecutive adult patients undergoing living donor liver transplantation and was proven safe with promising efficacy 22 . In this pilot study, donor-specific immunomodulatory cells were generated ex vivo by coculturing recipient lymphocytes with irradiated donor cells in the presence of 2D10.4/IT2.2 for two weeks, which were thereafter transferred to the recipients without any additional Treg purification. Of the 10 patients, seven have completed successful weaning and cessation of immunosuppressive agents for more than four years. 23 This approach holds promise for the induction of operational tolerance and appears to be simpler and more cost effective than other Treg purification and expansion strategies. However, to allow its widespread use in a deceased donor transplant setting, good manufacturing practice of alloantigen-specific immunomodulatory cells is required to generate cells that comply with EMA and FDA regulations; 2D10.4/IT2.2, which was used in these studies, is not clinically approved in Europe and the USA.
In terms of the CD80/CD86-CD28 interaction, Davies et al. demonstrated that belatacept, a clinically approved cytotoxic T-lymphocyte antigen 4-immunoglobulin (CTLA4-Ig), can be used ex vivo to shift human peripheral blood mononuclear cells (PBMCs) to HLA-mismatched alloantigens with high efficiency and specificity 20 . Considering the promising results of the clinical trial demonstrated by Todo 22 and the notion that belatacept possesses high affinity for CD80/CD86 24 , we hypothesize that belatacept can be used for the clinically approved ex vivo alloanergization of recipient human PBMCs with similar efficacy to 2D10.4/IT2.2. Herein, we compared the efficacy of ex vivo-generated alloantigen-specific immunomodulatory cells in the presence of either 2D10.4/IT2.2 or belatacept using HLA-mismatched human PBMCs. The results of this study will supply crucial information for the widespread clinical application of this tolerance induction strategy.
Materials and Methods
Isolation of human PBMCs
PBMCs were isolated from healthy volunteer blood donors using Lymphoprep™ density-gradient centrifugation (Axis-Shield, Oslo, Norway) following a protocol approved by the institutional review board. Informed written consent was obtained from all healthy volunteers, and the Regional Ethics Committee at Stockholm, Sweden, approved this study, including the consent procedure (Dnr: 2014/1565-31/4).
In brief, freshly drawn blood from healthy volunteers was collected into tubes containing an acid–citrate–dextrose solution and centrifuged to separate the plasma and cell fraction, the latter of which was mixed with a double volume of phosphate-buffered saline (PBS) and laid on top of Lymphoprep™. After centrifugation for 35 min at 400
Reagents
Anti-CD80/86 mAbs (2D10.4 and IT2.2, respectively) and CTLA4-Ig (belatacept) were purchased from e-Bioscience (Stockholm, Sweden) and Bristol-Myers Squibb AB (Stockholm, Sweden), respectively.
Ex vivo Generation of Donor-Specific Immunomodulatory Cells
Donor antigen-specific immunomodulatory cells were generated ex vivo in the presence of anti-CD80/CD86 mAbs (2D10.4/IT2.2) or belatacept using human leukocyte antigen (HLA)-mismatched PBMCs based on previous protocols 21,22 . Responder PBMCs (50 × 106 cells) were cocultured with irradiated (30 Gy) donor PBMCs (20 × 106 cells) in 25 cm2 culture flasks (Corning, NY, USA) in RPMI 1640 culture medium containing heat-inactivated responder serum (0.15 mL; 1%, v/v). The final volume was 15 mL, and the cultures were treated with 10 µg/mL (3 µg/106 cells) 2D10.4 and 10 µg/mL IT2.2, or 133 µg/mL (40 µg/106 cells) belatacept 20 , or no antibodies (sham treatment; control group). On day 7, the generated cells were collected from each flask, and two million cells were separated for cell composition analysis using flow cytometry. The remaining cells were centrifuged to separate the culture supernatant and cells. Cells were suspended in fresh medium containing 1% recipient serum. Irradiated donor PBMCs (20 × 106 cells), culture media, and either 2D10.4 and IT2.2 (10 μg/mL, individually) or belatacept (133 μg/mL) were replenished according to the original culture conditions. On day 14, generated cells were collected, washed three times with PBS and used for further analysis. Cell number and viability were assessed by standard Trypan blue (Sigma-Aldrich, Stockholm, Sweden) exclusion staining.
Characteristics of Responder and Donor Pairs
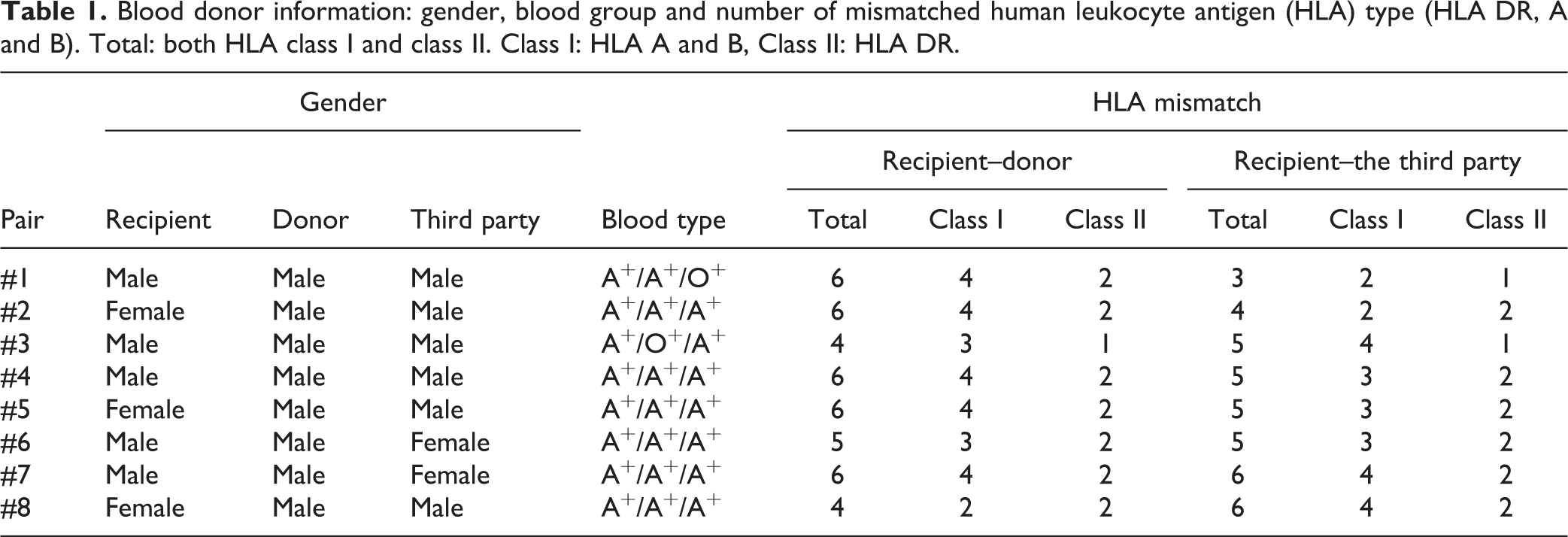
PBMCs isolated from the blood of healthy volunteer donors were cocultured in ABO blood type-compatible responder and donor pairs (eight pairs). A third-party stimulator was chosen from non-related individuals, among which five sets of the donor and third-party pairs shared one or two HLA antigens. The gender, blood type, and number of mismatched HLA types in each pair are shown in Table 1.
Blood donor information: gender, blood group and number of mismatched human leukocyte antigen (HLA) type (HLA DR, A and B). Total: both HLA class I and class II. Class I: HLA A and B, Class II: HLA DR.
Cell Composition Analysis of the Generated Cells (Flow Cytometry)
Freshly isolated responders or the generated cells were analyzed with flow cytometry on days 0, 7, and 14. For the analysis of lymphocyte and monocyte lineage markers, the following conjugated antibodies (Abs) were used: anti-CD4-PerCP Cy5.5, anti-CD8-APC H7, anti-CD3-HV450, and anti-CD19-HV500 (all from BD Biosciences, San Jose, USA); and anti-CD16-PE, anti-CD56-PE, anti-CD14-PC7, and anti-CD45-APC AF700 (all from Beckman Coulter, Marseille, France). CD subsets were determined using the Lineage Cocktail 1 (lin 1; FITC-conjugated Abs against CD3, CD14, CD16, CD20, and CD56; all from BD Biosciences), anti-CD123-PECF594, anti-CD11c-PC7 (both from BD Biosciences), and anti-HLA DR-APC (Beckman Coulter). Tregs were identified using anti-CD127-PE, anti-CD4-PerCP Cy5.5 (BD Biosciences), anti-CD25 PC7 (Biolegend, San Diego, USA), and anti-FOXP3-AF647, clone 236A/E7 (BD Biosciences). The staining procedure, including fixation and permeabilization, was performed according to the manufacturer’s instructions (eBiosciences, San Diego, CA, USA). To prevent non-specific binding, mouse immunoglobulin G1 was added prior to incubation with the FOXP3 antibody. The data were acquired with a Navios flow cytometry instrument and analyzed using Kaluza software (Beckman Coulter, Brea, USA).
Comparison of FOXP3 Isoform Gene Expression in the Generated Cells
mRNA was extracted from frozen pelleted cells with a RNeasy Kit (Qiagen, Hilden, Germany, product id: 74104) according to the manufacturer’s instructions. cDNA was produced with a SuperScript® VILO cDNA Synthesis Kit (Life Technologies, Carlsbad, CA, USA, product id: 11754050) according to the standard protocol. The cDNA was added to a SYBR Green Master Mix (Life Technologies, product id: 4367659) and subjected to quantitative polymerase chain reaction. Primers were used for amplification of full-length FOXP3 mRNA and FOXP3 mRNA lacking exon 2. GAPDH was used as a reference gene. The data were analyzed with StepOne software v2.1, and Ct values were obtained and used for the calculation of relative mRNA expression.
Examining the Donor-specific Activities of the Generated Cells with the Cytokine Enzyme-linked Immunospot Assay
IL-10 and IFN-γ production from the generated cells collected after 14 days of culture was examined by the enzyme-linked immunospot (ELISPOT) assay (Mabtec AB, Nacka, Sweden).
In the IL-10 ELISPOT assay, different concentrations of the generated cells (5 × 104, 2.5 × 104, 1.25 × 104, and 6.25 × 103 cells/well) were stimulated with the irradiated donor or third-party PBMCs (1 × 105 cells/well) for 24 h in an anti-human IL-10 mAb-precoated ELISPOT plate.
In the IFN-γ ELISPOT assay, freshly harvested responder PBMCs (105 cells/well) were cocultured with the irradiated donor or third-party PBMCs (105 cells) in the presence or absence of varying numbers of generated cells (5 × 104, 2.5 × 104, 1.25 × 104, or 6.25 × 103/well) for 72 h in an anti-human IFN-γ mAb-precoated ELISPOT plate.
The IL-10 or IFN-γ spots were detected by biotinylated anti-human IL-10 or IFN-γ antibodies, and the spots were enumerated by ELISPOT software (AID, Strasburg, Germany) and expressed as spot-forming cells/105 cells.
Examining the Immunomodulatory Effect of Generated Cells: Proliferation Assay
The suppressive function of the generated cells was measured by the mixed lymphocyte reaction (MLR). Responder cells (PBMCs) were stimulated with irradiated responder, donor or third-party cells (PBMCs, 105/well in a 96-well round-bottom plate), in the presence or absence of varying numbers of generated cells (5 × 104, 2.5 × 104, 1.25 × 104, or 6.25 × 103/well). All measurements were performed in triplicate. After one week of culture in RPMI 1640 medium containing 1% heat-inactivated responder serum, 3H-thymidine (1 μCi/well; PerkinElmer Inc., Waltham, MA, USA) was added. The cells were pulsed with 3H-thymidine (1 µCi/well) 18 h before estimating thymidine incorporation with a β-counter (Wallac Sverige AB, Upplands Väsby, Sweden).
The percentage of the response was calculated as the cpm of the MLR with addition of generated cells divided by the cpm of the MLR without the addition of generated cells.
Examining the Immunomodulatory Effect of Generated Cells: Measurement of Cytokines in Culture Supernatant
IFN-γ and IL-10 protein levels in the coculture supernatants (at days 7 and 14) and in the MLR supernatants were quantified using enzyme-linked immunosorbent assay (ELISA; human IFN-γ and IL-10 ELISA kits were from Mabtech AB) following the manufacturer’s protocol. All measurements were performed in triplicate.
Statistical Analysis
Quantitative results are presented as the mean values ± standard deviation (SD). The Wilcoxon matched pairs signed rank test or two-way ANOVA with Tukey’s multiple comparison test was carried out for statistical analysis. A
Results
Comparison of Cell Number and Viability of the Generated Cells after Coculture in the Presence or Absence of 2D10.4/IT2.2 or Belatacept
After 14 days of coculture, the cell number decreased from 50 × 106 cells to 25.2 × 106 cells in the 2D10.4/IT2.2 group and 26.3 × 106 cells in the belatacept group; these numbers were significantly lower than those of the untreated control group (34.7 × 106 cells) (

Cell number during the coculture. Viable cell numbers were counted before coculturing (50 × 106 per flask, open bars) and after seven days (grey bars) and 14 days of coculture (black bars). During the 14-day culture period, the cell numbers decreased to 25.2 × 106 per flask in the 2D10.4/IT2.2 group and 26.3 × 106 in the belatacept group; they were significantly lower than those of the untreated control group (34.7 × 106 cells). The results are from eight responder–donor pairs and are expressed as the mean ± SD (
Cytokine Profiles During and After the Generation of Immunomodulatory Cells
IFN-γ and IL-10 protein levels in the coculture supernatants at day 7 and day 14 were quantified by ELISA. Compared with the untreated control group, groups that had either 2D10.4/IT2.2 or belatacept added showed significantly inhibited IFN-γ production during coculture. There was no significant difference between the 2D10.4/IT2.2 and belatacept groups (Fig. 2). The concentration of IL-10 was low, and no significant difference was observed between either the 2D10.4/IT2.2 or belatacept groups and the untreated control group (data not shown).
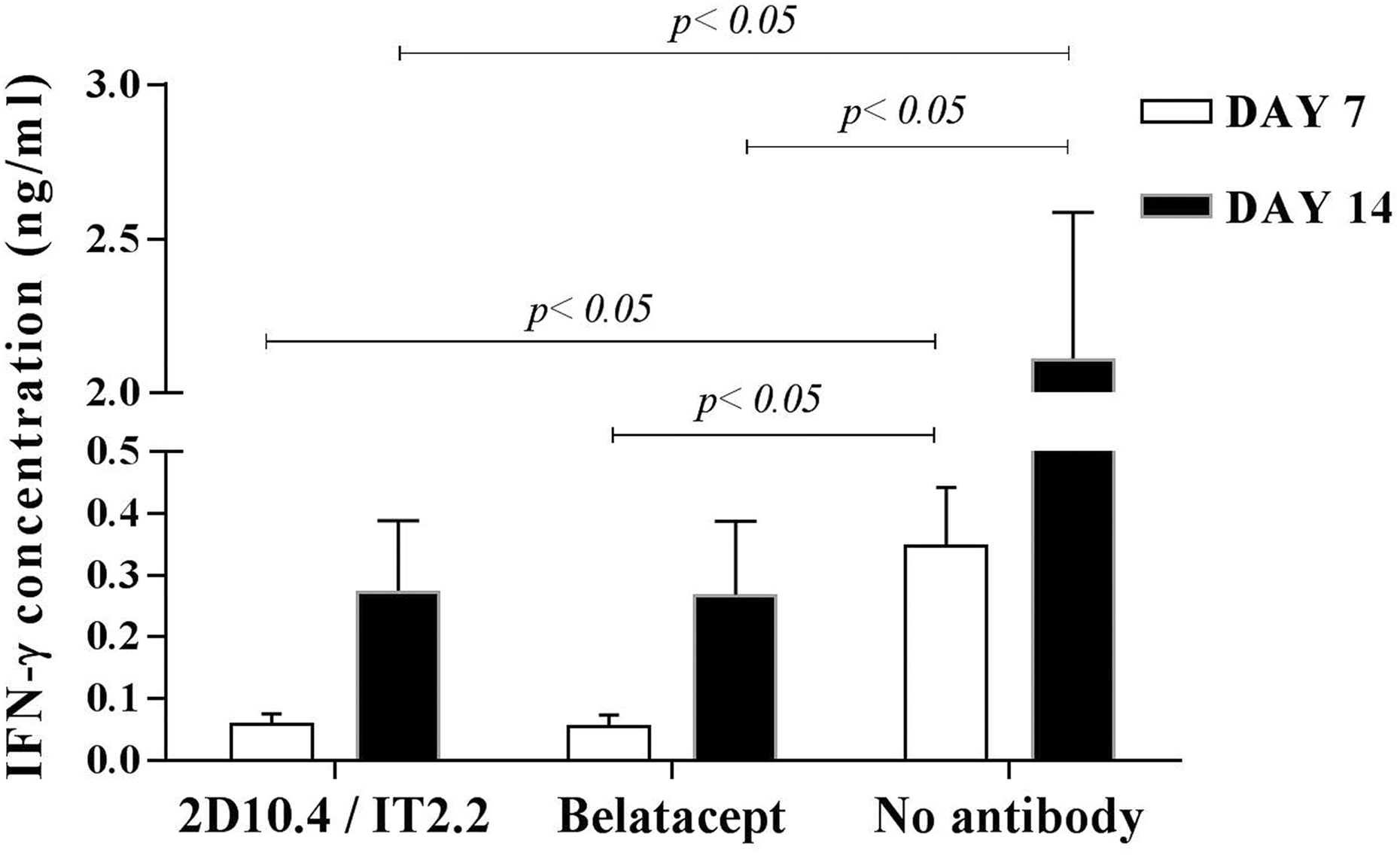
Interferon-γ (IFN-γ) levels in the coculture supernatants. The supernatants were collected from the coculture flask at days 7 (open bars) and 14 (filled bars), and the IFN-γ protein levels were quantified by enzyme-linked immunosorbent assay. The addition of either 2D10.4/IT2.2 or belatacept significantly inhibited IFN-γ production during coculturing compared with that in the untreated control group. There was no significant difference between the 2D10.4/IT2.2- and belatacept-treated groups. The results are expressed as the mean ± SD (
Changes in the Cell Composition of Generated Cells in Relation to Costimulation Blockade Treatment
The composition of generated cells was analyzed by flow cytometry before culture on days 7 and 14 (Fig. 3). During the 14 days of coculture, the CD3+ T cell population was increased in all the groups. The most marked increase in CD4+ T cells was noted in the 2D10.4/IT2.2-treated group (Fig. 3(A) and (B)). CD8+ T cells, and the number of CD3-CD16+/CD56+ natural killer (NK) cells gradually decreased in the 2D10.4/IT2.2- and belatacept-treated groups (Fig. 3(A) and (B)). The CD4–CD8– double negative CD3+ T cell population, which contains the TCRγδ T cell population, was not changed in any of the groups (Fig. 3(B)). The frequency of B cells was similar in the 2D10.4/IT2.2- and belatacept-treated groups, while it was significantly lower in the untreated control group (Fig. 3(A)). CD14+CD45+ monocytes and Lin1–CD11c+HLA DR+ myeloid dendritic cells decreased to almost undetectable levels, which could be, at least partly, due to firm adhesion to the plastic culture flasks (Fig. 3(A) and Supplemental Material Fig. 1 online).

Changes in cell composition before and after culture. Cells were collected before and at seven and 14 days after culture with irradiated donor stimulators. Assessment of the composition of the cell population was examined by flow cytometry. The values are depicted as the lower quartile, median, and upper quartile (boxes) with minimum and maximum ranges. Boxes with thin lines show precultured cells, those with light grey-filled boxes represent cells cultured for seven days, and dark grey-filled boxes represent cells cultured for 14 days. (A) Cells were gated for CD45+ cells (all hematopoietic cells except red blood cells and plasma cells). The percent composition of CD3+ T cells, CD3-CD16/56+ cells (natural killer (NK) cells), CD19+ B cells, and CD14+ monocytes are shown. The percentage of Lineage negative cells was calculated as lin-1 negative population in lymphocyte gated cells (see Supplemental Fig. 1). (B) Percentage of CD4+, CD8+ or CD4-CD8- cell populations within the total CD3+ T cell population are shown. (
As shown in Fig. 1, after 14 days of culture, the total cell number in the untreated control group was significantly higher than that in the 2D10.4/IT2.2- and belatacept-treated groups. Based on the cell composition analysis and total cell numbers within each flask, the generated cell numbers of the different cell subsets were calculated and compared (Fig. 4). The cell number of each subset was similar between the 2D10.4/IT2.2- and belatacept-treated groups throughout the entire culture period. On day 14, the numbers of CD4+ (non-Tregs) or CD8+ T cells and NK cells in the untreated control group were significantly higher than those in the 2D10.4/IT2.2- and belatacept-treated groups. The numbers of Tregs and B cells were not significantly different in any of the conditions. There was no significant difference between the 2D10.4/IT2.2- and belatacept-treated groups in any phenotypes, including Tregs.

Comparison of different subset cell numbers after 14 days of culture. Based on the cell numbers in the flask and the percentage of each subset of cells, different subsets of the generated cell numbers in each flask were calculated. The estimated numbers were compared among the three groups. Black bars represent the 2D10.4/IT2.2-treated group, grey bars represent the belatacept-treated group, and white bars represent the untreated control group. The non-regulatory T cell (Treg) population was calculated as total number of CD4+ T cells minus CD4+CD25+CD127lowFOXP3+ T cells. There was no significant difference between the 2D10.4/IT2.2- and belatacept-treated groups. The mean ± SD is shown (
Comparison of the Treg Population in CD4+ T Cells during 14 Days of Culturing
Several studies have indicated that the immunosuppressive effect is correlated with the percent expression of CD25+CD127lowFOXP3+ (Treg) in CD4+ T cells. Here, we compared the percentage of Tregs in the CD4+ T cell population during culture (Fig. 5(A)). Within the CD4+ T cell subset, CD4+CD25+CD127lowFOXP3+ Tregs were significantly expanded at day 14 from 4.1±1.0% to 7.1±2.6% and 7.3±2.6% in the 2D10.4/IT2.2- and belatacept-treated groups, respectively, and to 6.7±1.9 in the control group (mean±SD,

Detection of CD4+CD25+CD127lowFOXP3+ regulatory T cells (Tregs) among the generated cells. (A) One of the typical examples of Treg gating in different treatment groups. The left panel (Day 0) represents cells before culturing, and the three panels to the right represent the generated cells with three different treatments, as indicated in the figure. (B) The percentage of Tregs within the CD4+ T cell population was compared during culturing. The values are depicted as the lower, median, and upper quartile (boxes) with minimum and maximum ranges. Boxes with thin lines show precultured cells, thick line cells cultured for seven days and grey-filled boxes cells cultured for 14 days. (
Comparison of Relative Expression of FOXP3 Isotype mRNA in Generated Cells
As shown in Fig. 5, the Treg populations in CD4+ T cell subset in the three groups, that is, the 2D10.4/IT2.2-, belatacept-, and vehicle-treated groups, after 14 days of culture were not significantly different. We further analyzed mRNA expression of FOXP3 in the generated cells to investigate phenotype differences. Full-length FOXP3 mRNA expression did not increase in the 2D10.4/IT2.2- or belatacept-treated groups compared with the control group (Fig. 6(A)). In contrast, delta-2 FOXP3 mRNA expression was significantly increased in both the 2D10.4/IT2.2- and belatacept-treatment groups compared with the control group (

FOXP3 isoforms. The mRNA expression levels of FOXP3 in the generated cells at day 14 were evaluated. (A) Full-length FOXP3 mRNA expression did not increase in the 2D10.4/IT2.2 or belatacept-treated groups compared with the untreated control group. (B) On the other hand, delta-2 FOXP3 mRNA expression significantly increased in both the 2D10.4/IT2.2- and belatacept-treated groups compared with the no-antibody treated group. There was no significant difference between the 2D10.4/IT2.2- and the belatacept-treated groups. The values are depicted as lower quartile, median, and upper quartile (boxes) with minimum and maximum ranges;
Generated Cells’ Donor-specific Activities: IL-10 Production (ELISPOT)
To quantitate the number of IL-10-producing cells in the generated cells via exposure to donor antigen, we performed an ELISPOT assay. Naïve responder cells did not produce IL-10 after 24 h of exposure to allogeneic antigens (data not shown). When generated cells from the 2D10.4/IT2.2- or belatacept-treated groups were cultured with specific donor cells, the number of IL-10-producing cells increased significantly in a dose-dependent manner based on generated cell numbers (Fig. 7). This tendency was also noted against the third-party antigens, although the increase in the number of IL-10-producing cells was not significant compared with those in stimulator alone (Fig. 7). There was no significant difference between the 2D10.4/IT2.2- and belatacept-treated groups regarding IL-10 production. With the generated cells from the untreated control group, the IL-10-producing cell numbers tended to increase in a dose-dependent fashion; however, this did not reach statistical significance.

Interleukin (IL)-10 enzyme-linked immunospot (ELISPOT) assay. The numbers of IL-10-producing cells in generated cells after 24 h of exposure to donor or third-party antigens were examined by ELISPOT. The numbers on the x-axis indicate the ratio of the generated cells to the irradiated stimulator (105/well): w/o = no generated cells, 1:16 = 1/16 (6.25 × 103/well), 1:8 = 1.25 × 103/well, 1:4 = 2.5 × 104/well, and 1:2 = 5 × 104/well. The results are shown as the spot number/well of generated cells stimulated with donor (black bars) or third-party (open bars) antigen subtracted with the spot number with autologous stimulators (background). The results are expressed as the mean ± SD;
Generated Cells’ Donor-specific Immunomodulatory Function: Number of IFN-γ-producing Cells in MLR
IFN-γ production from naïve responder cells was detected 72 h after alloantigen stimulation. Therefore, the effect of the addition of generated cells on IFN-γ production in the MLR was evaluated after 72 h by the ELISPOT assay. The addition of generated cells from the 2D10.4/IT2.2- and belatacept-treated groups did not enhance the frequencies of IFN-γ-producing cells in naïve responder cells after stimulation with donor or third-party antigens. In contrast, the addition of generated cells from the untreated control group, which generated high amounts of donor-specific memory effector cells, significantly increased the number of IFN-γ-producing cells in a dose-dependent fashion under stimulation with donor antigens. Nevertheless, no significant differences between the 2D10.4/IT2.2 and belatacept groups were seen (Fig. 8).

Interferon (IFN)-γ enzyme-linked immunospot (ELISPOT) assay. Freshly isolated responder cells (105/well) were incubated with the irradiated stimulator (105/well) in the presence or absence of different concentrations of generated cells. At 72 h after incubation, the numbers of IFN-γ-producing cells were examined by ELISPOT. The results shown are the spot number/well with donor (black bars) or third-party (open bars) stimulators subtracted with the spot number with autologous stimulators. The numbers on the x-axis indicate the ratio of the generated cells to the naïve responder (105/well): w/o = no generated cells, 1:16 = 1/16 (6.25 × 103/well), 1:8 = 1.25 × 103/well, 1:4 = 2.5 × 104/well, and 1:2 = 5 × 104/well. The numbers of IFN-γ-producing cells in the mixed lymphocyte reaction (MLR) did not increase when the generated cells from the 2D10.4/IT2.2- and belatacept-treated groups were added. In contrast, the spot number in MLR-stimulated donor-specific antigens increased significantly after the addition of generated cells from the untreated control group. The results are expressed as the mean ± SD,
Generated Cells’ Donor-specific Immunomodulatory Function: MLR Proliferation Assay and Cytokine Profiles in Culture Supernatant
The generated cells were collected after 14 days of coculture, and the effect of the generated cells on freshly isolated naïve responder cells was examined in MLR. Peak proliferation of the naïve responders in MLR was observed on day 7; therefore, the effect of the addition of the generated cells in MLR was analyzed on day 7. The generated cells from both the 2D10.4/IT2.2- and belatacept-treatment groups effectively impeded the proliferative responses of freshly isolated responder PBMCs against donor antigens in MLRs in a dose-dependent manner. The suppressive effect of the generated cells was milder against the third-party antigen than against the specific donor stimulator. No significant differences between the 2D10.4/IT2.2- and belatacept-treatment groups were seen (Fig. 9). In contrast, the generated cells from the untreated control group did not inhibit responder proliferation against donor cells or against the third-party stimulators.

Inhibition of the proliferative response in mixed lymphocyte reaction (MLR). Freshly isolated responder cells (105/well) were incubated with the irradiated stimulator (105/well) in the presence or absence of different concentrations of generated cells. The numbers on the of x-axis indicate the ratio of the generated cells to the naïve responder: w/o = no generated cells, 1:16 = 1/16 (6.25 × 103/well), 1:8 = 1.25 × 103/well, 1:4 = 2.5 × 104/well, and 1:2 = 5 × 104/well. The proliferation of the MLR on day 7 was assessed by the [3 H]thymidine incorporation assay. The proliferation against donor antigens and the third-party stimulator is shown in black and grey bars, respectively. The results are expressed as % response = cpm with generated cell × 100 cpm without generated cells Each bar indicates the mean ± SD,
In the MLR culture supernatant from day 6, we also detected that IFN-γ production was down-regulated and IL-10 production increased when generated cells from either the 2D10.4/IT2.2- or belatacept-treated groups were added, compared with the production in the supernatant of MLR culture lacking generated cells (
Discussion
In the present study, we evaluated and compared the efficacy of ex vivo-generated alloantigen-specific immunomodulatory cells in the presence of anti-CD80/CD86 mAbs (2D10.4/IT2.2) or CTLA4-Ig (belatacept) by using human PBMCs from HLA-mismatched responder and donor pairs. During ex vivo generation, the addition of 2D10.4/IT2.2 or belatacept suppressed the production of IFN-γ and the expansion of non-Treg CD4+ T cells, CD8+ T cells and NK cells while expanding the CD4+CD25+CD127lowFOXP3+ T cell subset. Total FOXP3 mRNA expression was similar in all three groups; however, higher expression of delta-2 FOXP3 mRNA was observed in the 2D10.4/IT2.2- and belatacept-treated groups.
We also demonstrated that the cells generated with belatacept in vitro exhibited comparable immunomodulatory effects to those generated with 2D10.4/IT2.2 in vitro, while the generated cells from the control groups exhibited strong donor-specific memory cell responses. Upon re-exposure to donor antigens, the donor-specific memory cells proliferate quickly and produce high quantities of IFN-γ, while the regulatory cells do not proliferate but produce IL-10. Therefore, as a functional comparison, we also evaluated IFN-γ and IL-10 production from these generated cells as well as the effect of the generated cells towards naïve recipient PBMCs upon stimulation with donor-antigen in MLR. In fact, cells generated with 2D10.4/IT2.2 or belatacept, upon exposure to donor antigens, showed lower IFN-γ production and higher IL-10 production than cells in the control groups. The proliferation of naïve cells against donor stimulators was significantly suppressed by the addition of 2D10.4/IT2.2- or belatacept-generated cells, confirming that 2D10.4/IT2.2- and belatacept-generated cells possess donor-specific immunomodulatory activities. Our results are in line with previous findings reported by Davies et al., who demonstrated that the generation of HLA-mismatched human PBMCs with belatacept resulted in a great reduction in subsequent allo-responses 20 . However, our strategy contains several methodological differences compared with Davies’ report 20 , specifically, the recipient–donor cell ratio and the culture period.
In the current study, the donor specificity of the generated cells was examined by comparing the responses of freshly isolated responder PBMCs against those of donor antigens and third-party stimulators. The generated cell inhibitory responses against donor antigens were stronger than those against third-party antigens; however, a similar tendency was often observed upon third-party stimulation, especially with the highest concentration of generated cells. It is possible that some shared epitopes between donor and the third-party antigens may enhance the generated cell response against third-party stimulators. Considering these facts, immunomodulatory cells generated by ex vivo activation could possess activity against donor-specific antigens and, to a lesser extent, against third-party antigens.
Ex vivo programming of generated cells with donor-specific antigens would be important because the injected cells need to settle inside the grafts or the draining lymph nodes to control donor-specific immune responses. Recently, it has been demonstrated that adoptive transfer of alloantigen-specific immunomodulatory cells generated with 2D10.4/IT2.2 allowed the early withdrawal of immunosuppressants and promoted graft acceptance after non-human primate kidney transplantation 21 and after clinical living donor liver transplantation 22 . Given these promising immunomodulatory effects of the ex vivo generated cells together with our findings that 2D10.4/IT2.2 and belatacept exhibit similar efficacies in the generation of immunomodulatory cells, belatacept appears to be a promising agent that can replace 2D10.4/IT2.2 for clinical grade ex vivo generation of alloantigen-specific immunomodulatory cells.
To investigate the mechanism of the efficacy exerted by ex vivo-generated cells in our protocol, it is important to address which cells are important and responsible for the donor-specific immunomodulatory effects. Tregs have been reported to suppress the ability of both CD4+ and CD8+ T cells by preventing their differentiation, activation, and proliferation through the secretion of inhibitory factors, such as IL-10, IL-35, and transforming growth factor (TGF)-β 25 –27 . Tregs can also diminish effector T cells directly in a perforin-dependent and granzyme-dependent manner or suppress their activation 28,29 by blocking galectin-1 binding to activated T cells 30 . Given these properties, Tregs are supposed to be a key player in the immunomodulatory effects of cells generated with either 2D10.4/IT2.2 or belatacept in our protocol. In our study, the actual numbers of CD4+CD25+CD127lowFOXP3+ T cells (Tregs) were maintained or slightly increased during the 14 days of culture in all three groups, while the number of other cell types was diminished. Individual inspection according to each responder and donor pair revealed that the percentage (and the number) of Tregs (CD4+CD25+CD127lowFOXP3+) was not correlated to the degree of immunomodulatory effects seen on MLRs, the inhibition of IFN-γ, or promotion of IL-10 production. Likewise, as reported in the clinical study by Todo, the dose of CD4+CD25+FOXP3+ Tregs infused into the liver transplant recipients was not correlated with the outcome of successful induction of operational tolerance, and the dose of Tregs (0.43 to 6.37 × 106/kg) was much lower than that in other Treg-based cell therapies 22 . However, in our comparison, the delta-2 FOXP3 mRNA expression, which is an abundant FOXP3 isoform that bestows Tregs with significant suppressive activity, was significantly higher in both the belatacept- and 2D10.4/IT2.2-treated groups than in the untreated control group. Forced expression of delta-2 FOXP3 is believed to convert CD4+CD25– cells into CD4+CD25hi cells with immunosuppressive activities. 31,32,33 One explanation could be that Treg expansion and/or conversion could occur later during allo-restimulation 34 . It has also been suggested that the present biological markers of Treg may not fully identify its function 35 and that different subtypes of Tregs (polyclonal or antigen-specific) differ in their immunomodulatory capacities 36 .
Considering that the entire population of generated cells was evaluated and used in our protocol, cells other than Tregs could be contributing to the immunomodulatory function together with Tregs 37,38 . Moreover, the impact of regulatory B cells (Bregs) on immunomodulation and tolerance following transplantation 39 –41 has been increasingly recognized. Bregs can be induced by inflammation, toll-like receptor stimulation, CD40 activation and some types of auto-antigens to suppress both innate and adaptive immune responses and to enhance Treg function. Bregs have been reported to produce high levels of IL-10, IL-35, and TGF-β for Tregs. Therefore, the IL-10 production observed in our study could be associated with Breg activity. The role of other regulatory cell types, such as regulatory dendritic cells and regulatory macrophages, is increasingly recognized in the quest towards achieving operational tolerance after transplantation 38,42 . Although we could not clearly delineate which cells were important for the immunomodulatory effects, continued investigation of the dynamics within the generated cell population could provide the opportunity to expand and deepen our current knowledge on the ex vivo generation of immunomodulatory cells.
Belatacept binds to CD80/CD86 with high specificity and affinity. Regarding clinical transplantation, the BENEFIT study recently reported that belatacept improves long-term allograft and patient survival after kidney transplantation 43 . At a mechanistic level, belatacept and 2D10.4/IT2.2 block both CD28- and CTLA 4-mediated signals through their binding to CD80/CD86 on antigen-presenting cells 44 , and selective CD28 blockade was reported to effectively promote FOXP3+ Treg function in non-human primates 45 . Therefore, blocking the CTLA 4-mediated signals may detract from the attenuation of alloreactive T cell responses as well as Treg numbers. CTLA 4 signals activate Tregs by enhancing the expression of indoleamine 2,3-dioxygenase (IDO), a tryptophan catabolizing pathway 46 , and CD80 binds to the programmed death-ligand 1 47 . Indeed, selective CTLA-4 signaling was reported to play an important role in antigen-specific adaptive Treg induction 48 . Considering the balance of CD28-dependent costimulatory and CTLA 4-mediated coinhibitory signals, it is relevant to identify the optimal dose of belatacept for the effective ex vivo generation of immunomodulatory cells. It should be noted that some reports have indicated that high levels of belatacept could inhibit Treg expansion 49 –51 . In the current study, we chose the belatacept dose of 40 μg/106 cells; based on a previous report 20 , the optimal inhibition of secondary alloresponses was observed at a belatacept dose of 40 μg/106 cells compared with that at a dose of 10 μg/106 cells.
In a more recent ongoing study, we observed that the cells generated with belatacept at a dose of 1.0 or 10 μg/106 cells possessed comparable immunomodulatory effects compared with those generated with 40 μg/106 cells belatacept (data not shown). Furthermore, we noticed that the cells generated with a lower dose of belatacept (1 μg/106 cells) showed a tendency towards a higher Treg population and produced more IL-10 by donor antigen stimulation than did cells generated with a higher dose of belatacept (10–40 μg/106 cells). However, considering the earlier mentioned discrepancy of the percentage and total number of Tregs within the generated cell product and their subsequent immunomodulatory effects shown in both our results and the clinical pilot data reported by Todo 22 , the absolute number of Tregs may be an insensitive parameter to predict the efficacy of this strategy. Further studies to find the proper range of agents and in-depth characterization of the immunomodulatory effects of different cell subpopulations within the generated cells are currently in progress, the results of which may reveal important mechanisms and contribute to reducing the costs for clinical application.
In summary, alloantigen-specific immunomodulatory cells generated with belatacept and 2D10.4/IT2.2 show comparable effective donor-antigen specific inhibitory effects on the immune system. Given the promising outcome in the clinical trial with the adoptive transfer of cells generated with 2D10.4/IT2.2 ex vivo, we suggest belatacept as an approved agent for the ex vivo generation of clinical grade alloantigen-specific immunomodulatory cells for clinical use.
Supplemental Material
SUPPLEMENTAL_FIGURE_1 - Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade
SUPPLEMENTAL_FIGURE_1 for Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade by M. Watanabe, Makiko Kumagai-Braesch, M. Yao, S. Thunberg, D. Berglund, F. Sellberg, C. Jorns, S. Lind Enoksson, J. Henriksson, T. Lundgren, M. Uhlin, E. Berglund, and B.-G. Ericzon in Cell Transplantation
Supplemental Material
Supplemental_figure_1 - Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade
Supplemental_figure_1 for Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade by M. Watanabe, Makiko Kumagai-Braesch, M. Yao, S. Thunberg, D. Berglund, F. Sellberg, C. Jorns, S. Lind Enoksson, J. Henriksson, T. Lundgren, M. Uhlin, E. Berglund, and B.-G. Ericzon in Cell Transplantation
Supplemental Material
Supplemental Material, 20180621Cell_TX_Figures_suppl2 - Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade
Supplemental Material, 20180621Cell_TX_Figures_suppl2 for Ex Vivo Generation of Donor Antigen-Specific Immunomodulatory Cells: A Comparison Study of Anti-CD80/86 mAbs and CTLA4-lg Costimulatory Blockade by M. Watanabe, Makiko Kumagai-Braesch, M. Yao, S. Thunberg, D. Berglund, F. Sellberg, C. Jorns, S. Lind Enoksson, J. Henriksson, T. Lundgren, M. Uhlin, E. Berglund, and B.-G. Ericzon in Cell Transplantation
Footnotes
Acknowledgments
We thank Ms. Helene Johanson and Dr. Antonio Romano (Department of Transplantation Surgery, CLINTEC, Karolinska Institutet) for their contributions to the experiments.
Ethical Approval
This study, including the consent procedure, was approved by the Regional Ethics Committee, Stockholm, Sweden (Dnr: 2014/1565-31/4).
Statement of Human and Animal Rights
Our study design and methods respects the human rights and safety of the study participants according to the declaration of Helsinki. This study does not contain any animal subjects.
Statement of Informed Consent
All study participants were given written and oral information of the study and all participants signed a written consent before participation of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the regional agreement on medical training and clinical research (ALF) between the Stockholm County Council and the Karolinska Institutet, the Stockholm County Council (clinical postdoctoral appointment), the Foundation for International Surgical Cooperation, Professor Lars-Erik Gelin’s Foundation, and Emil and Vera Cornell’s Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
