Abstract
Although the clinical application of new drugs has been shown to be effective in slowing disease progression and improving the quality of life in patients with pulmonary fibrosis, the damaged lung tissue does not recover with these drugs. Thus, there is an urgent need to establish regenerative therapy, such as stem cell therapy or tissue engineering. Moreover, the clinical application of mesenchymal stem cell (MSC) therapy has been shown to be safe in humans with idiopathic pulmonary fibrosis (IPF). It seems that a combination of MSC transplantation and pharmaceutical therapy might have additional benefits; however, the experimental design for its efficacy is still lacking. In this review, we provide an overview of the mechanisms that were identified when IPF was treated with MSC transplantation or new drugs. To maximize the therapeutic effect, we suggest that MSC transplantation is combined with drug application for synergistic effects. This review provides clinicians and scientists with the most efficient medical options, in the hope that this will spur on future research and lead to an eventual cure for this disease.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a specific form of chronic and progressive fibrosing interstitial pneumonia of unknown cause 1 . It occurs mainly in older adults (median age at diagnosis 66 years, range 55–75 years) 2 , is limited to the lungs, and is characterized by fibroblast proliferation and extracellular matrix remodeling 3 , which results in progressively worsening dyspnea and pulmonary function, usually with poor prognosis. The diagnosis of IPF requires the histopathologic and/or radiologic pattern of usual interstitial pneumonitis on high-resolution computed tomography. Other known causes of interstitial lung disease (ILD) (e.g. connective tissue diseases, drug toxicity, and other environmental exposures) must be excluded first. IPF is a fatal lung disease, with median survival ranging from 3 to 5 years 3 . The majority of patients demonstrate a slow and gradual progression over many years, and some patients remain stable while others have an accelerated decline 4 . A small minority of IPF patients (approximately 5–10%) may suffer from acute exacerbation annually 1 . The incidence of IPF is reported to be an estimated 4.6 to 16.3 cases per 100,000 worldwide. Based on healthcare claims data from the United States, an estimated prevalence of between 14.0 and 42.7 per 100,000 persons was reported 5 .
The antifibrotic drugs nintedanib and pirfenidone have been shown to significantly reduce the decline in forced vital capacity (FVC), decrease acute exacerbation, and improve mortality rates in patients with IPF in several studies 6 –8 . Recently, they have been recommended conditionally in several guidelines for the treatment of IPF 9 ; however, current medication can only slow the progression of IPF but does not reverse the fibrosis process. There is still a lack of effective options for treating IPF, although abundant possible targets from animal studies have shown their potential. Even though some off label drugs, such as inhaled NAC, could shorten the time of investigation through meta-analysis, the response is relatively limited compared with specific target therapy 10 . Previous studies have provided results for both stem cell and pharmacological treatments, but data on a combination of these are still lacking. In this review, we have summarized the experimental and clinical evidence and possible mechanisms of action to present the potential options for this intractable disease.
Treatment Strategies in IPF
Compared with other diseases of the respiratory system, IPF is hard to predict and prognosticate. The main cause of IPF is still unclear, but it may involve genetic, environmental, or toxic components 11 . Bleomycin (BLM) is commonly used to induce symptoms of IPF in animal models, as it produces an oxidative stress specific to alveolar epithelial cells 12 . In these models, the immune cells and fibroblasts gradually infiltrate the lungs of BLM-treated mice. On the other hand, differentiation and proliferation of myofibroblasts from fibroblasts produce excessive extracellular matrix. Consequently, anti-inflammatories have been commonly used as a treatment option in previous studies; however, the treatment was stopped in one trial due to an increasing risk of death in IPF patients with no clinical benefit 13 . Other inducers, such as transforming growth factor β (TGF-β) and fibroblast growth factor (FGF), have been used as targets for blockage by inhibitors or antibodies 14 . In recent years, there is evidence of an increasing trend on the use of stem cell therapy in IPF; its use almost comparable to studies on new drug development and compound studies (Fig 1). This trend suggests the potential benefit of stem cell treatment in IPF. The first successful stem cell transplantation was carried out 50 years ago 15 , but even now there are potential exciting applications that deserve our attention.
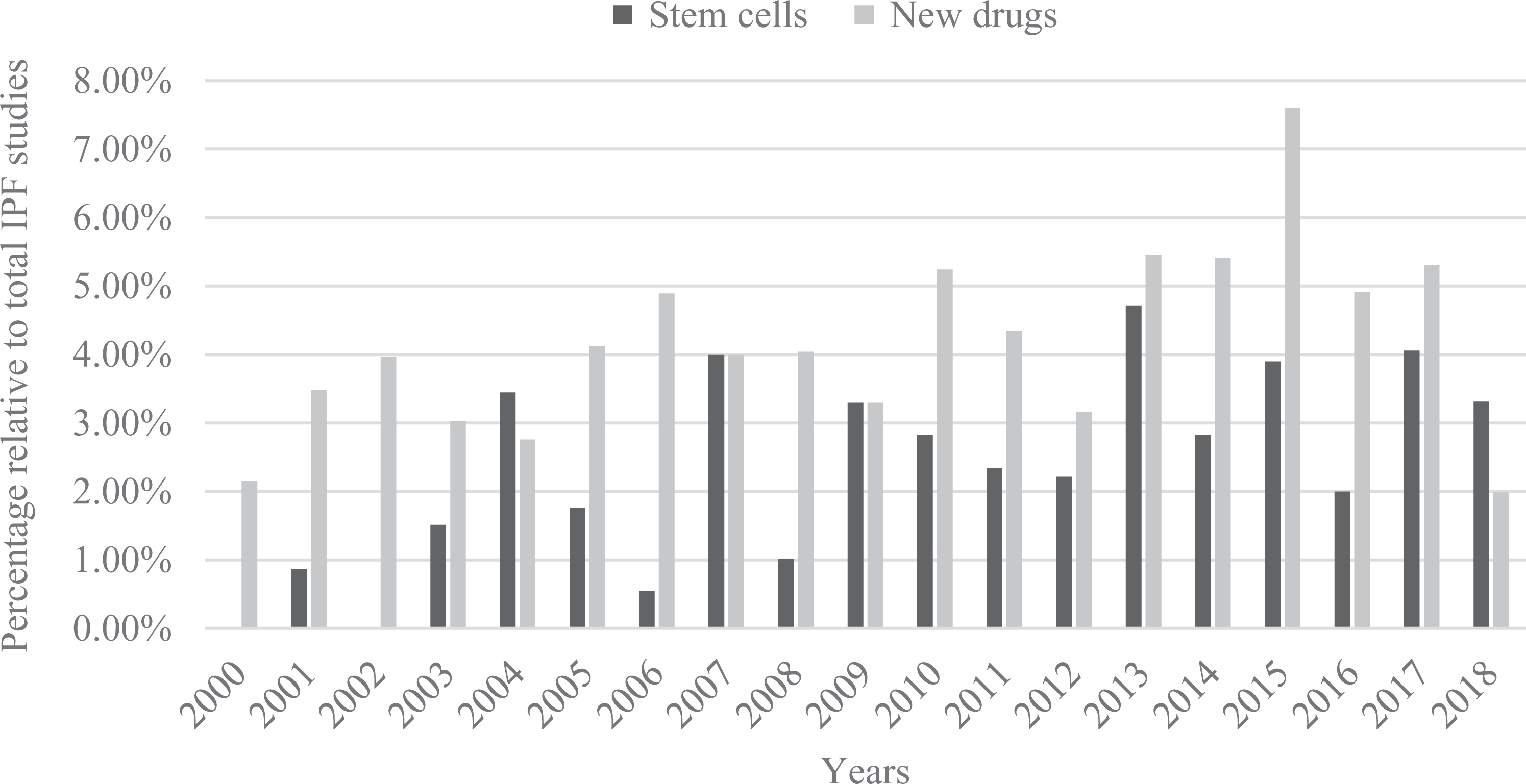
Publications of new drugs or stem cells development and each percentage of total IPF studies.
Developmental Drugs for Treatment of IPF
The two Food and Drug Administration (FDA)-approved pharmacological targets for treatment of IPF are TGF-β (pirfenidone) and PDGF/VEGF/FGF (nintedanib). The efficacy of pirfenidone is impressive, as it was found to significantly lower the relative risk of death compared to the placebo group at week 52 16 . The data collected from four phase III trials showed that the mean life expectancy was 8.72 years for patients given pirfenidone versus 6.24 years for those receiving best supportive care 17 . On the other hand, in a 52-week phase III trial involving 1,061 subjects, nintedanib was found to slow disease progression, but there was no statistically significant difference for the primary endpoint 18 . Although the preclinical studies are quite epromising, the human trials did not show consistent results, not including the cases waiting for FDA approval. Potential developmental drugs provide additional possible targets for improving current therapy. Pamrevlumab, an anti-connective tissue growth factor antibody (FG3019), is under phase III trials (NCT01890265). Another drug that targets alpha v beta 6 (αvβ6) integrin has also completed a phase II trial involving IPF participants (NCT01371304). In short, new drug development for IPF is a highly risky and ineffective business.
Current Stem Cell Therapies for IPF
Despite the fact that MSC therapy has been used in the clinic for approximately 10 years, more than 75% of studies are still in phase II or earlier 19 . Lung diseases account for 23 of the 493 transplantation studies, according to https://www.clinicaltrials.gov/. More recently, an allogeneic bone marrow MSC (BMSC) transplantation study in IPF patients assigned three cohorts to a single IV infusion of 20, 100, or 200 × 106 human BMSC 20 . There is also a case report of a 56-year-old man with IPF who received human umbilical cord-derived mesenchymal stem cell (HUC-MSC) transplantation with intravenous infusion 21 . Both studies, observed over 1 year, did not register any adverse events. The authors of a phase I trial of adipose-derived stem cell (ADSC) treatment for IPF reported on their experiences of acquiring FDA approval (NCT01385644). The protocol used three rounds of endobronchial infusion with Stromal vascular fraction (SVF), totaling 1.5×106 cells, and the cell fate was traced by 99mTc-HMPAO 22 . These reports suggest that stem cell transplantation in IPF is initially safe and with some benefits, and the standardized protocol might encourage scientists to explore the potential for MSC therapy in IPF patients. Data from previous clinical studies suggest that the transplanted cell type is important for IPF treatment. Of the problems that may arise between the MSC source and the receiver during allograft, placenta-derived MSCs such as umbilical cord blood or amnion stem cell provides less complications. 23 . It has been well-established that management of host rejection through HLA typing in placenta-derived MSCs can be performed easily in clinical applications. 24 . The results with amniotic stem cells are the most abundant results in clinical studies. Although their primary endpoints were usually for safety and not efficacy, stem cells still hold promise for better outcomes than most known drugs 25 .
Mechanisms of Stem Cell Therapy in IPF
The progression of IPF is a complex event consisting, at minimum, of epithelial-dependent fibroblast-activated processes and poor response to anti-inflammatories. These effects involve downstream pathways of epithelial mesenchymal transition (EMT) and an immune response 3 , as shown in Fig 2. Some EMT regulators like WISP-1 and BMP4 have been found to have a role in fibroblast differentiation or collagen production through Wnt and BMP pathways, respectively 26,27 . In regard to an immune response, the PGE2 pathways can prevent Fas ligand-induced apoptosis in myofibroblasts and fibroblasts; however, due to the lower expression of PGE2, alveolar type II cells are still sensitive to apoptosis 28 . The differences in expression might explain the apoptosis in alveolar epithelial cells but not in myofibroblasts or fibroblasts. Targeting of cell cycle-related proteins has also been reported as a potential treatment for IPF. S-phase kinase-associated protein 2 (Skp2) is an effector protein that enhances degradation of p27 29 and is essential for the progression of IPF. Increased expression of p27 with an antagonist for Skp2, SZL-P1-41, could inhibit BLM-induced pulmonary fibrosis 30 . Additionally, treatment with BLM in fibroblasts also induces ER stress-related proteins, such as GRP78, CHOP, and ATF4, and enhances proliferation of fibroblasts through a PI3K/Akt pathway 31 . The activation of the PI3K/Akt pathway is another upstream regulator of EMT 32 and makes the response more unpredictable and irreversible. Although there are few connections between each signaling pathway, the evidence has shown that the use of each inhibitor, such as LY294002 and Tauroursodeoxycholic acid (TUDCA), is efficient at ameliorating pulmonary fibrosis.

Mechanism assuming that the fibrogenesis consists of alveolar cell apoptosis and immune cell infiltration (left). The transplanted stem cells circulate through the pulmonary artery (bottom) and act through immunomodulation, secretion of growth factor, and differentiation into alveolar-like cells (right).
Regulation of Immune Response or Secretome
Bone marrow (BM)-derived MSCs are used more often in mechanism studies than in clinical studies 33 , perhaps because the source is easier to obtain. It is thought that the injected cells could repair lung tissue with its immunomodulatory effects. From the immunological perspective, the BM-derived MSCs had lower levels of expression of the immunosuppressive molecules PDL-1 and CD1a than placenta-derived MSCs, and IFN-γ induction also showed lower potential to stimulate T cell proliferation in BM-derived MSCs 34 . However, placenta-derived MSCs had a suppressive effect in cytokine-stimulated natural killer lymphocytes 35 , though the cell type is still hard to ascertain. Furthermore, the process of IPF is usually accompanied by inflammation and profibrotic cytokinesis. To produce an anti-inflammatory effect, MSCs reduce IL-1β, IL-6, TNF-α, TGF-β, and vascular endothelial growth factor (VEGF) in BLM-induced lung injury 36 . Although these studies did not find leftover stem cells in injured lungs, the secreted circulating mediators were taken into account to benefit the therapeutic effects for IPF 37 .
Stem cell therapy also benefits from secretion of isolated exosomes that contain proteins and RNA from their own cell origin 38 . These extracellular vesicles of between 10 and 100 nm in size, called exosomes, allow communication between distal cells and their cargos 39 . The effects induced by exosomes may be associated with the modulation of the immune response and suppression of macrophage phenotype 40 . To confirm this theory of stem cell secretion, Tan et al. purified exosomes from amniotic stem cells and administered them intranasally. They found that pulmonary capacity was recovered in the exosome-treated groups in BLM-induced pulmonary fibrosis 41 . Moreover, the specific delivery potential of exosomes has been reported to be due to the presence of membrane proteins 42 , making the use of exosomes instead of MSCs alone a better proposition. However, a contradictory result suggested that MSC-isolated microvesicles (MV) reduced pulmonary fibrosis, and that MSCs have a better treatment effect than MV 43 . Unfortunately, these authors did not reveal the amount of MSCs used for isolating the MV, and the result is also lacking in dosage effectiveness. Nevertheless, these studies suggest that the treatment potential of MSC therapy is positive regardless of its presence in the lung tissue.
Evidence for Differentiation into Functional Alveolar Cells
The injected MSCs were found in the lung epithelium and expressed an epithelium-like phenotype 44 ; but researchers also found that their presence is not necessary for an effect, since the condition medium could also inhibit the effect of inflammatory cytokines and macrophage-produced TNF-α 45 . The anti-inflammatory molecule TSG-6 can be secreted and homed to the injury site of myocardiocytes by the embolized-MSCs in the lung 46 . Evidence of MSC differentiation into lung epithelial cells was found in nude mice engrafted with amnion-derived stem cells 47 , and the CXCR4/SDF1 axis is thought to be involved in stem cell plasticity. However, in immunocompetent mice, injection of allogenic amnion-derived stem cells did not find labeled cells in lung, brain, heart, spleen, liver, and kidney 37 . It seems that the ability to engraft is determined by the innate immune responses. Although the cost is higher and time-scale longer, autogenic MSC transplantation is still considered better than allogenic MSC transplantation in IPF.
Influential Factors for Clinical Outcome
Data from a large-scale meta-analysis indicate that the most important factors for stem cell therapies are dosage and time points 48 . A double dosage of autologous stem cell transplantation produced a two-fold better outcome (10% vs 20%) in a study on humans 49 . Ghadiri et al. summarized five clinical studies in IPF patients involving autogenic or allogenic MSC therapy; however, four of them are still under patient recruitment 50 . The route of transplantation is also conflicting, since animal studies did not compare the efficiency between intravenous and intranasal transplantation. Interestingly, MSC transplantation via the intraperitoneal route is also effective for treating IPF, which suggests that direct contact of stem cells with lung tissue is not necessary 51 .
Comparison of Drugs and Stem Cells in Clinical Potential
Intravenous injection of ADSC on day 3, 6, and 9 after BLM-induction in mice has comparable therapeutic effects with the approved drug pirfenidone 52 . However, there are no beneficial effects when injected on day 14, 17, and 20, due to its diminished response of anti-inflammation and inhibited profibrotic genes 53 . In current pharmacological studies, the targeting of drugs to specific organs or tissues is still under investigation. In the case of stem cell transplantation, tissue repair from migrating stem cells is a natural characteristic of MSCs 19 , which is also found in bleomycin-induced lung injury or infarcted myocardium 44,54,55 . On the other hand, pharmacological applications have more detailed studies for absorption, distribution, metabolism, and excretion, and the mechanisms are clear in most clinical drugs. The manufacturing process of MSCs is determined by the donor and source (i.e. autogenic transplantation of MSCs is more expensive than allogenic). But the effect of engraftment in autogenic transplantation is more impressive than other known drugs and might apply to regenerative medicine. Advancements in MSC purification and modification might be a future goal when the limits of current medication are reached.
Problems and Future Breakthroughs
The ethical issues in stem cell transplantation include the potential of these cells to develop into a human being 56 . However, MSCs lack such potential, so this issue is not relevant here. Other concerns that arise are the promotion of tumor growth and metastasis, and overestimating therapeutic potential 57 . One of the problems we encountered in this review was characterizaton of the culturing method or media, since little is known about which characteristics would be changed from their original niche 58 . Autologous transplantation is performed once a culture and expansion method, which is as similar to the natural niche as possible, is formalized 59 . Considering the economic concerns, HLA-matched banking for allogenic transplantation and mass production is rational 60 . Although it is not necessary to evaluate the effects of graft-versus-host disease, cases with immunosuppressive agents should be evaluated with the total adding effect.
Conclusion
Recently, a convincing new drug has been discovered that slows the decline of Forced Vital Capacity (FVC) as well as prolonging survival, but it does not rebuild the normal structure of lung tissue. The final goal of therapy should be the recovery of pulmonary histological structure or retainment of the cellular functions of each cell type. While there is no evidence suggesting MSC differentiates into alveolar epithelial cells, the theory that supplies remaining lung progenitor cell proliferation is still expectable. MSC transplantation is as effective as pharmaceutical therapies, but has relatively unclear mechanisms of action.
Here we have listed mechanisms sufficient for treating IPF, such as immunomodulatory effect, secretion of cytokines, and suppression of myofibroblast differentiation; however, MSCs neither act as an inhibitor of signaling pathways, nor as an agonist of receptors. To counter this disadvantage, a combination of drugs and MSCs might be of benefit. MSC treatment lacks the ability to improve myofibroblast apoptosis 61 , re-epithelization of mesenchymal type cells (or mesenchymal to epithelial transition, MET) 62 , and TGF-β inhibition 63 . MSC-derived MV or exosomes might provide another option. Neutralizing antibodies or drugs approved for marketing can support such limitations. Rather than applying for new drug development trial approval at the Institutional Review Board (IRB), which can be both time-consuming and cost inefficient, use of approved drugs proves to be a better strategy. These suggestions provide more options for physicians and patients.
In summary, we concluded that current MSC therapies have observed positive results in clinical and animal studies. Moreover, molecular targets involved in the process of fibrogenesis, such as cytokines or growth factors, can work alongside MSCs with synergistic effect. Here we discussed some of the known mechanisms involved and studies based on these mechanisms. Exploring possible combinations for making the best therapeutic strategies should be the next step in the pursuit of better outcomes in the treatment of IPF, bearing in mind that both a molecular approach and cell therapy have their own disadvantages. However, two heads are better than one as they say, so selecting the positive features from each approach might be the best way forward.
Footnotes
Acknowledgments
This study was funded by Buddhist Tzu Chi Bioinnovation Center, Tzu Chi Foundation, Hualien, Taiwan; and Ministry of Science and Technology, Taiwan (MOST 106-2320-B-303-001-MY3 and MOST 106-2320-B-303-002-MY3).
Author Contribution
Horng-Jyh Harn and Li-Ing Ho contributed equally to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
