Abstract
Acute brain injury resulting from ischemic/hemorrhagic or traumatic damage is one of the leading causes of mortality and disability worldwide and is a significant burden to society. Neuroprotective options to counteract brain damage are very limited in stroke and traumatic brain injury (TBI). Given the multifaceted nature of acute brain injury and damage progression, several therapeutic targets may need to be addressed simultaneously to interfere with the evolution of the injury and improve the patient’s outcome. Stem cells are ideal candidates since they act on various mechanisms of protection and repair, improving structural and functional outcomes after experimental stroke or TBI. Stem cells isolated from placenta offer advantages due to their early embryonic origin, ease of procurement, and ethical acceptance. We analyzed the evidence for the beneficial effects of placenta-derived stem cells in acute brain injury, with the focus on experimental studies of TBI and stroke, the engineering strategies pursued to foster cell potential, and characterization of the bioactive molecules secreted by placental cells, known as their secretome, as an alternative cell-free strategy. Results from the clinical application of placenta-derived stem cells for acute brain injury and ongoing clinical trials are summarily discussed.
Keywords
The Rationale for Using Placenta-Derived Cells for Acute Brain Injury
Acute brain injury, resulting from traumatic brain injury (TBI), ischemic stroke, or intracerebral hemorrhage (ICH), is associated with high rates of short-term mortality and long-term disability worldwide 1 . Independent of its etiology (vascular occlusion, bleeding, or mechanical injury), brain damage is amplified after the primary insult, with the activation of genomic, cellular, and/or biochemical processes that interact in a complex network leading to delayed cellular dysfunction and death. Although the extensive effects of these events and the abundance of targets offer the potential for therapeutic interventions, so far there is no pharmacological treatment that can reverse the pathologic cellular cascades and improve the outcome. Thus, there is a pressing need for therapeutic approaches aimed at protecting and repairing the injured brain.
Stem cells, with their ability to act simultaneously on multiple targets, driving the damaged microenvironment from a toxic to a more protective/regenerative activation state, are a promising strategy 2 –6 . Stem cells have been isolated from almost all body organs, raising the question of the best source for each specific pathology, since heterogeneous effects have been observed depending on the source.
According to their tissue origin, stem cells display specific differences at transcriptional and proteomic levels 7,8 . Moreover, aging negatively affects stem/progenitor cell properties 9,10 , reducing their proliferation and differentiation capacity 11,12 , their immunomodulatory effects 13 , and their therapeutic potential 14,15 . Thus, stem/progenitor cells from placenta are of special interest in a translational perspective, mainly on account of their reduced immunogenicity and high immunomodulatory potential and their broad plasticity 16,17 .
The placenta is a fetomaternal organ. The fetal placental tissues include the amniotic and chorionic membranes, the umbilical cord, and the chorionic villi. The maternal component is known as decidua 16,18 . All these placental tissues/fluids are easily available after birth, without invasive procedures, and their use is free from ethical issues.
This review examines the evidence for placenta-derived stem cell therapy for acute brain injury in experimental settings, with a focus on TBI, stroke, and ICH. We not only discuss efficacy but also the mechanisms of action and the engineering strategies used to amplify stem cell potency. One section will deal with the discussion of the bioactive molecules secreted by placental cells and the potential use of the secretome as a cell-free, better defined therapeutic strategy. Lastly, results from the clinical application of placenta-derived stem cells for acute brain injury and ongoing clinical trials will be discussed.
This is a narrative, nonsystematic, comprehensive review of the literature. The search string run in the PubMed database in January 2017 was as follows: ((“traumatic brain injury” OR “brain trauma” OR “head trauma”) OR (“Brain Ischemia”[Mesh] OR “Stroke”[Mesh] OR stroke OR “brain ischemia”)) AND ((“Stem Cells”[Mesh] OR “stem cells” OR “stem cell” OR “stromal cells” OR “stromal cell”) AND (“placenta” OR “amnion” OR “amniotic” OR “cord blood” OR “umbilical” OR “chorion” OR “chorionic” OR “trophoblasts” OR “decidua”)). We screened 301 papers for title, abstract, and full text, finally selecting 139 original articles and 60 reviews. Five of the original articles were clinical studies and 134 experimental, of which 21 focused on TBI, 93 on cerebral ischemia, 16 on neonatal/infant stroke, and 4 on ICH. Among papers on stroke, those using models of heatstroke or diabetes were outside the remit of this review and were therefore not used. Thirty-five papers were added for analysis of the secretome, based on our own files or reference lists of previously reviewed articles.
In the majority of studies, cells were of human (h) origin, with very few using rat (r) or mouse (m) cells. Cells were isolated from different placental tissues and fluids including umbilical cord blood (UCB), umbilical cord tissue (UCT), amnion (A) and amniotic fluid (AF). The specific subtypes of cells used in the field of acute brain injury are listed in Figure 1. We found no study using cells from chorion, trophoblasts, or decidua.
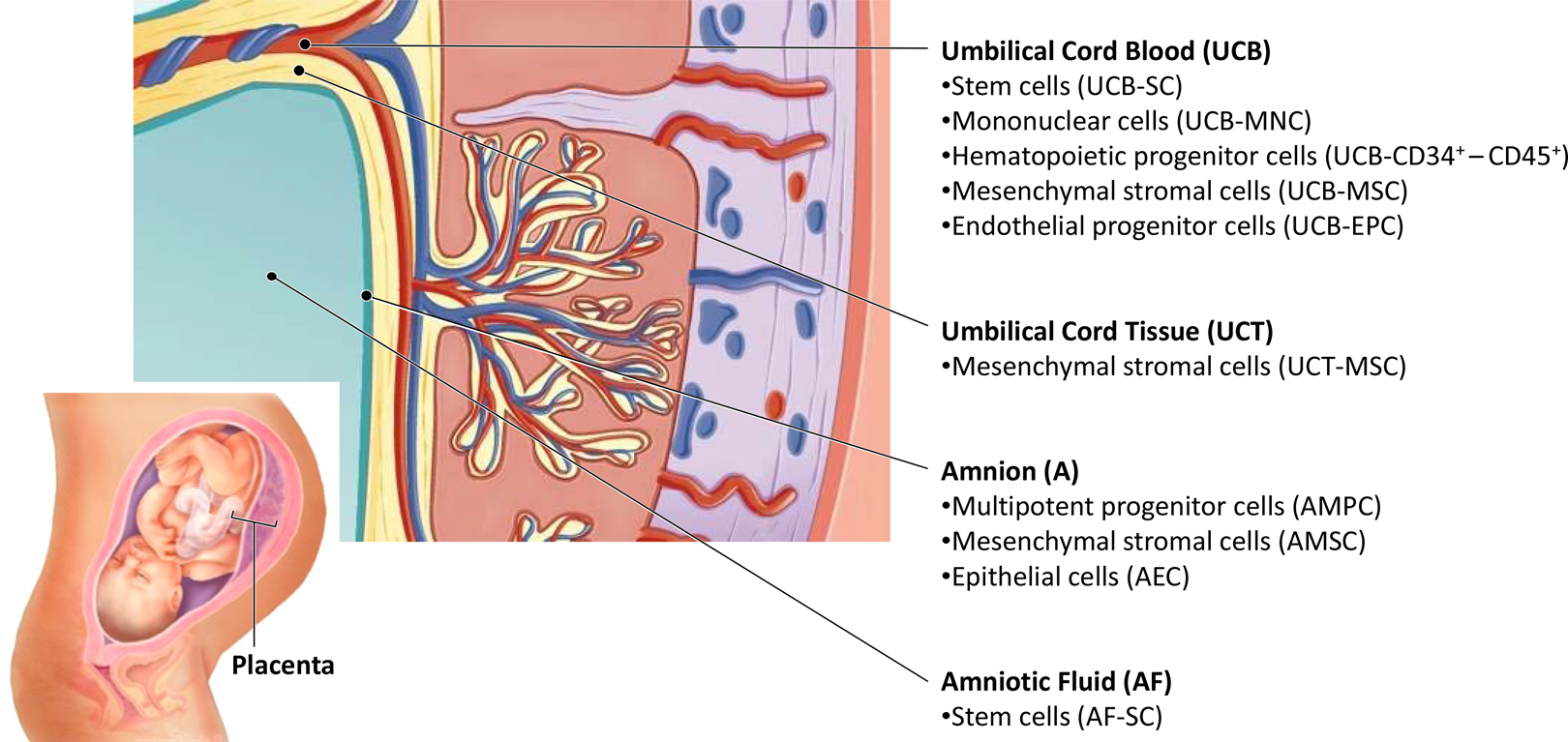
Specific subtypes of placenta-derived cells used for experimental acute brain injury.
Placenta-Derived Cell Transplantation for TBI
The literature concerning the use of placenta-derived stem cells in experimental models of TBI shows more widespread use of cells isolated from the UCB or tissue 19 –27 than cells from the amnion or AF 28,29 . No papers were found using cells from chorion, trophoblasts, or decidua.
The first paper using human umbilical cord blood stem cells (hUCB-SCs) was published by Lu and colleagues 19 who used 2 million hUCB-SCs, without selecting any specific population. Cells were intravenously infused in rats, 24 h after controlled cortical impact (CCI) brain injury and improved sensorimotor function from 2 wk post-TBI 19 . After this first experiment, the use of placenta-derived stem cells focused on investigation of the effects of specific subpopulations (endothelial, hematopoietic, or mesenchymal stromal cells [MSCs]) isolated from the hUCB.
The endothelial progenitors were selected with the aim of promoting vascular repair and stimulating neoangiogenic processes. Both systemic 22 and local (intracerebroventricular [ICV] infusion) 23 transplantation of hUCB-derived endothelial progenitor cells (hUCB-EPCs) in traumatized rodents can increase microvascularization in the injured brain and angiogenic processes 22,23 , preserving blood–brain barrier (BBB) integrity 23 and reducing brain edema 23 . ICV-transplanted hUCB-EPCs integrated into the brain microvasculature 23 , but when intravenously (IV) infused, the number of integrated hUCB-EPCs was scanty and the neovascularization was mostly due to the release of proangiogenic factors 22 .
Similar results on the vascular compartment have been obtained with hUCB-derived hematopoietic subpopulations (CD34+ or CD45+)
21,24
, with an increase in cerebral perfusion pressure
21
, stimulation of vascular endothelial growth factor (VEGF) production
24
, and increased angiogenesis
21
. Besides vascular effects, hUCB-CD34+ cells also stimulated neurogenesis, with increases in the amounts of newly generated neurons (BrdU+/NeuN+ cells), induced anti-inflammatory effects, lowering serum levels of proinflammatory cytokines (tumor necrosis factor α [TNF-α] and intercellular adhesion molecule 1 (Icam-1)) and increasing the anti-inflammatory ones (interleukin [IL] 10), and decreased toxic oxidizing radicals (
The other subpopulation widely used for TBI are MSCs isolated from hUCB 20 or hUCT 25 –27,30 . Since the data available do not allow for a distinction of enhanced efficacy of the MSCs depending on the different regions of the umbilical cord, they are collectively referred to as umbilical-MSCs. Systemic 25,27 and local 20,26 transplantation of MSCs in TBI rodents was able to induce functional improvement in sensorimotor 20,25 –27 and cognitive 20,26,27 deficits and reduce the contusion volume 20,26 . Transplanted umbilical-MSCs were found in the lesion site and their migration toward the injured tissue seemed to be driven by the stromal cell-derived factor 1/ C-X-C chemokine receptor type 4 (SDF-1/CXCR4) pathway 25 ; however, the number of cells surviving after transplantation gradually drops 20 .
The various protective mechanisms elicited by transplanted cells involve different pathways. MSCs reduce brain edema 26 , lower the number of apoptotic cells in the damaged area 25 , boost the expression of neurotrophic factors 20,26 , stimulate endogenous neurogenesis 25 –27 , and have immunoregulatory effects, reducing serum markers of proinflammatory mediators (IL-1, interferon [IFN] γ, antibrain antibody) and increasing anti-inflammatory ones (IL-10 and transforming growth factor β) 30 . Umbilical-MSCs also shift activated microglial cells in the pericontusional tissue toward a nonphagocytic phenotype with protective and healing functions 20 .
The amniotic source for stem cell therapy has been less investigated for TBI, with only 2 papers published 28,29 . Chen and colleagues used human amnion–derived multipotent progenitor cells (hAMPCs) and transplanted 2 million cells ICV immediately after penetrating ballistic-like brain injury in rats 28 . Transplanted cells reduced axonal degeneration in the corpus callosum and in the thalamus. Labeled hAMPCs migrated from the subventricular zone to the corpus callosum but not to the thalamus, and there was no evidence of differentiation toward neuronal lineage, suggesting that the effects of these cells may result from a continuous supply of secreted bioactive factors 28 .
Our group has provided evidence of the protective effects of human amnion membrane-derived MSCs (hAMSCs) in a mouse model of TBI modeled by controlled cortical impact (CCI) 29 . We compared the effects of ICV and IV transplanted cells 24 h after TBI in mice. Only ICV transplanted cells were found in the brain at acute stages after injury, but ICV and IV transplanted hAMSCs induced functional improvement in sensorimotor deficits to similar extents. Functional improvement was associated with histological protective modifications, such as neuronal and vessel rescue in the pericontusional cortex and the stimulation of endogenous neurogenesis 29 .
To summarize, the literature regarding the use of placenta-derived cell therapy for TBI varies widely, with different tissues of origin, isolation protocols, and subpopulations. Although it is hard to compare the therapeutic effects obtained in different laboratories, it is clear that infused cells have pleiotropic action on multiple targets, inducing protective/regenerative environmental changes and stimulating endogenous neuroprotective mechanisms by autocrine/paracrine factors. The presence of cells in the lesioned brain seems to be unnecessary to confer the protection, thus posing the basis for the development of a cell-free strategy using the stem cell secretome. This topic will be discussed in the section “Placenta-Derived Cell Secretome: Toward Cell-Free Therapy.”
Placenta-Derived Cell Transplantation for Stroke
The first preclinical use of placenta-derived cells for stroke dates back to 2001 31,32 . Chen and colleagues reported that IV infused hUCB-SCs after middle cerebral artery occlusion (MCAO) in rats improved functional recovery 31 , and Okawa and colleagues described the permanence of hippocampal transplanted rat-derived AECs at the site of injection, 5 wk after stroke in gerbils 32 . Since then several studies have explored the efficacy and mechanisms of action of placenta-derived cells after brain ischemia. As with TBI, we also noted for stroke that cells derived from an umbilical source have been investigated far more than amniotic cells, and we did not find any studies that used cells isolated from the chorion, trophoblast, or decidua.
The mechanisms of hUCB cell-induced recovery after stroke have been investigated with the aim of establishing whether transplanted cells must actually enter the brain to be effective. When hUCB-SCs were IV infused 24 to 48 h after MCAO, viable cells were found in the brain and improved functional recovery was reported 31,33 . Efficiency was dose dependent and higher when compared to intracranial administration at the same time point 33 . This dose-related effect was also found for hUCT-derived cells 34 . The ability of hUCT-MSCs to differentiate into neural phenotypes has been shown in vitro 35 ; however, transdifferentiation of hUCT-MSCs into fully functional neurons after in vivo acute brain injury has never been demonstrated and mechanisms other than engraftment appear to mediate the restorative effects after hUCT-MSC infusion 36,37 .
Despite the above evidence, along with the observation that hUCB-SCs have potent migratory capacity in response to ischemic brain injury 38 , probably through chemokine SDF-1/CXCL12 39 , there is evidence that systemically administered cells do not need to enter the brain to induce functional recovery after MCAO 40 . Several studies indicate that hUCB cells induce restorative effects after stroke through the secretion of trophic factors 41 . In addition, hUCT-MSCs secrete human brain–derived neurotrophic factor (BDNF), neutrophil-activating protein-2 (NAP-2), angiopoietin-2 (Ang-2), CXCL16, platelet-derived growth factor-AA (PDGF-AA), basic fibroblast growth factor (hVEGFR-3), and human vascular endothelial growth factor receptor-3 37 (see also “Placenta-Derived Cell Secretome: Toward Cell-Free Therapy” section). Although most studies show these trophic factors induce angiogenesis, neurogenesis, and synaptogenesis at the site of injury after adult and perinatal ischemia 34,42,43 , the role of hUCB-SCs in restoring spleen weight and splenic CD8+ T-cell counts after MCAO has been highlighted 44 , indicating a role in dampening the systemic inflammatory response after stroke. The immunomodulatory effects of hUCB cells are established in other inflammatory conditions and are potential mechanisms for mediating restoration after stroke. hUCB-SCs were able to reduce ischemia-induced infiltration of granulocytes, monocytes, and CD3+ T cells and activation of astrocytes and microglia in adults 45 –47 and pups 48 , in addition to reducing proinflammatory cytokines and nuclear factor κB activity 46 after stroke, all of which may contribute to the resolution of the inflammatory response after stroke and facilitate postischemic plasticity.
In terms of the subpopulations, hUCB-CD34+ cells have been prominently investigated in stroke models and have been shown to reduce brain damage in newborns 49 and to induce behavioral recovery 50,51 which is enhanced in CD34+ cells overexpressing glial cell line–derived neurotrophic factor (GDNF) 52 . hUCB-CD34+ cells have also been shown to be responsible for the angiogenic and neurogenic effects observed after hUCB-SC infusion 42 , although hUCB-CD34− cells were just as effective 50 . Boltze and colleagues not only found that hUCB-CD34+ and CD34− cells had similar behavioral effects, but also that human umbilical cord blood-derived mononuclear cells (hUCB-MNCs) were more effective 53 . Among the different cells in hUCB-MNCs (immature T and B cells, monocytes, and stem cells), monocytes were found to have the most marked effects on improving functional recovery and reducing infarct 54 . Several studies have also indicated the therapeutic benefits of hUCB- or hUCT-derived MSCs after stroke. hUCB-MSCs reduced infarction lesion size after cerebral ischemia in adult rats 55 and dogs 56 as well as rat neonates 57,58 . hUCT-MSCs also improved functional recovery in rats after MCAO 37 , possibly by reducing peripheral and cerebral proinflammatory cytokines, an observation reported for both hUCB-MSCs 57,59 and hUCT-MSCs 60 .
Despite this wealth of evidence on the comprehensive efficacy of hUCB cells in stroke, a number of studies show a lack of effects 61 –63 , so further research into different storage and processing conditions is still needed to confirm the therapeutic potential of hUCB- and hUCT-derived cells in stroke.
As regards amniotic cells, the epithelial population amniotic epithelial cells (AECs), isolated from rat 32 or human 64 tissues, can migrate to the site of ischemic injury 32 and improve functional recovery after MCAO 64 . hAMSCs can also improve functional recovery after MCAO 65,66 , accelerated by overexpression of BDNF 67 .
AF stem cells were also shown to promote functional recovery after MCAO 68 , reducing ischemic damage and promoting neurogenesis 69 , and have been used for reprogramming into induced pluripotent stem cells (iPSCs) 70 .
To summarize, the current evidence for the potential of placenta-derived cell therapy for stroke is promising. Despite the different protocols and subpopulations tested, and the consensus needed on optimum storage and processing conditions, the literature indicates that placenta-derived stem cells improve functional recovery after MCAO, most likely by promoting angiogenesis and neurogenesis and through potent immunomodulatory effects that contribute to the resolution of the inflammatory response after stroke and facilitate postischemic plasticity.
Placenta-Derived Cell Transplantation for ICH
In the experimental models of ICH, hUCB and hUCT are the only placental sources that have been investigated 71 –73 . hUCB-MNCs were tested in a dose-dependent study 71 in order to study the effects of 4, 8, or 16 million IV infused cells 1 d after intrastriatal ICH. All transplanted groups showed similar improvement in sensorimotor function after 1 wk. Cells migrated selectively to the hemorrhagic area in the right striatum, with more cells found in the highest dose group. Similarly, the lesion volume decreased in all transplanted groups compared to control, with a dose-dependent effect.
Two studies investigated the effects of hUCB-MSCs 72 or hUCT-MSCs 73 . ICV infusion of 500,000 hUCB-MSCs, 2 d after intrastriatal ICH, improved sensorimotor function, with a reduction in the lesion volume 4 wk after transplant 72 . Cells were found in proximity to the hemorrhagic area 3 d after injection, but they disappeared at later time points (4 wk), with a nonsignificant tendency toward reduction in proinflammatory markers (TNF-α, cyclooxygenase-2 (COX-2) microglial activation, and neutrophil infiltration). Xie and colleagues compared the effect of intracerebrally (200,000 cells) or IV (2 million cells) infused hUCT-MSCs after intrastriatal ICH, observing similar protection on sensorimotor deficits and lesion volume 73 . Four weeks later, intracerebrally transplanted hUCT-MSCs were still clustered in the injection site, near the hemorrhagic region, while only a small part of the IV transplanted cells could be found in the brain. Only rats that intracerebrally received hUCT-MSCs had enhanced vascular density in the peri-ICH regions of the ipsilateral hemisphere, which was attributed to the limited number of IV transplanted cells that migrated to the damaged tissue. Given the similarity of the functional benefit, it may well be that locally and systemically transplanted cells act through different mechanisms and that combining the two may exert synergistic effects.
Manipulation of Placenta-Derived Stem Cells and/or the Use of Survival-Promoting Scaffolds
To optimize the therapeutic efficacy of transplanted cells, different combinatory strategies have been employed, including the use of scaffolds (spatially guiding tissue regeneration), the genetic modification (to overexpress specific factors or promote survival), and delivery of different biomolecules (trophic factors, modulators of inflammatory molecules).
With the aim of enhancing cell engraftment and differentiation, in vitro neuronal commitment prior to in vivo transplantation has also been tried 74,75 . hUCT-MSCs untransdifferentiated or transdifferentiated into neural-like cells (hUCT-MSC-NSCs) were resuspended in 10 µL Matrigel and transplanted into the ipsilateral hippocampus of rats, 7 d after TBI induced by weight drop impact model 74 . Only 6% to 7% of the transplanted cells were found in the brain 28 d after transplantation, with no differences in survival rates between hUCT-MSCs and hUCT-MSC-NSCs. Both treated groups showed a reduction in contusion volume and an improvement in cognitive deficits compared to control groups. However, untransdifferentiated hUCT-MSCs induced a better outcome than hUCT-MSC-NSCs. In vitro experiments showed that hUCT-MSCs secrete more neurotrophic factors (BDNF and neurotrophin-3 [NT-3]) than hUCT-MSC-NSCs, suggesting that transdifferentiation before use may not provide any advantage.
Two years later, the same authors did a similar analysis using hAMSCs undifferentiated or in vitro transdifferentiated into neural stem-like cells (hAMSC-NSCs) 75 . Cells were transplanted in TBI rats using a similar protocol, with the only exception that cells were infused earlier (4 d). Both transplanted groups showed improvement in sensorimotor and cognitive functions compared to controls, but hAMSC-NSC transplantation resulted in significant enhancement of sensorimotor function compared with hAMSC-treated rats. Unlike in the previous study, hAMSC-NSCs produced higher levels of neurotrophic factors (nerve growth factor [NGF], BDNF, NT-3, GDNF, ciliary neurotrophic factor [CNTF]) than hAMSCs, again suggesting that cell replacement is unlikely. Instead, a neuroprotective effect, associated with neurotrophic factors released by the grafted cells, may contribute to functional recovery.
In a similar study where 20,000 neural stem cells differentiated from hUCB (hUCB-NSCs) were intracerebrally infused 48 h after photothrombotic stroke, extensive migration of the cells into brain-damaged cortex in the first week after transplant was observed. However, differentiation was observed in very few cells and cell survival in the brain decreased gradually 76 . Again, this suggests that the persistence of cells in the host brain is not required and differentiation does not seem the main mechanism of action.
Therapeutic hypothermia can reduce functional deficits and limit secondary damage in models of TBI 77 . In order to potentially enhance therapeutic efficacy, Tu and colleagues combined hypothermia and hUCT-MSC treatments in a rat model of severe TBI 78 . Temperature-sensitive hUCT-MSCs were generated in order to promote the survival and the activity of grafted cells in the hypothermic rats by infecting cells with a retrovirus carrying the temperature-sensitive tsA58 SV40 LT antigen gene. Hypothermia contributed to the survival, migration, and proliferation of ts-hUCT-MSCs up to 28 d after injection in the lesion cavity, promoting the improvement in sensorimotor and cognitive functions more than hUCT-MSC therapy alone 78 .
Another approach used to establish an optimal environment to support lasting viability of stem cells in situ is the use of engineered scaffolds, which make it possible to deliver cells appropriately to the injured site, optimizing their therapeutic effects. Huang and colleagues combined hUCT-MSCs overexpressing CXCR4 fused to green fluorescent protein (hUCT-MSCCXCR4/GFP), responsible for cell migration through SDF-1 signaling and usually present at low levels on MSC membrane, with a chitosan scaffold linked to recombinant BDNF 79 . The scaffold containing hUCT-MSCCXCR4/GFP was transplanted in the lesion cavity of TBI rats. CXCR4 overexpression stimulated hUCT-MSC migration toward the lesion site, and the BDNF released from scaffolds favored the differentiation of transplanted cells into neurons, contributing to the regeneration of brain tissue at the lesion boundary.
The same researchers developed another type of scaffold, RADA16-BDNF, a self-assembling peptide hydrogel that favored the differentiation of hUCT-MSCs into MAP-2-positive neuronal cells 80 . When hUCT-MSCs were cocultured with lipopolysaccharide (LPS)-activated astrocytes, the percentage of MAP-2-positive cells was higher and typical neuron-like cells with neurites extending in 3-D directions differentiated from hUCT-MSCs. RADA16-BDNF scaffolds embedded with hUCT-MSCCXCR4/GFP and LPS-activated astrocytes were then transplanted into the lesion cavity of rats subjected to moderate (2-mm-diameter) and large (5-mm-diameter) lesions. hUCT-MSCCXCR4/GFP migrated from the scaffold to the injured tissue, with a lesion size–dependent efficacy in reducing the lesion size 80 .
Transgenically modified hUCT-MSCs engineered to overexpress hepatocyte growth factor (hUCT-MSCsHGF) have been used to enhance the therapeutic potential for ICH damage 81 . hUCT-MSCs or hUCT-MSCsHGF (600,000) were transplanted ICV 7 d after ICH, modeled by collagenase VII injected into the left internal capsule. Both transplanted groups showed better motor performance than untreated animals, and hUCT-MSCHGF-transplanted rats demonstrated a greater functional recovery than hUCT-MSC-transplanted rats. Neurological recovery was coupled with nerve fiber remyelination (upregulation of myelin basic protein(MBP)) and axonal regeneration (upregulation of growth-associated protein 43 (GAP-43)), demonstrating the pivotal role of growth factors in placenta-derived stem cell–induced protection.
To conclude, different strategies have been developed to enhance the therapeutic effects of transplanted cells. Understanding the mechanisms of protection induced by placenta-derived stem cells will help identify the best strategy for maximizing their effect.
Placenta-Derived Cell Secretome: Toward Cell-Free Therapy
The beneficial effects of cell therapy for acute brain injury are mediated by the release of soluble factors with multilevel effects providing support for surviving host cells, offering protection from toxic stimuli from the damaged brain tissue, and stimulating endogenous protective/reparative mechanisms. The view of secreted factors as mediators of brain protection raises the possibility of a cell-free therapeutic strategy, overcoming important issues related to intrinsic cell heterogeneity and safety concerns. In vitro 29,82 –86 and in vivo 86 –88 evidence shows that the conditioned medium (CM) from placenta-derived cell cultures can induce effects similar to the cell counterparts, demonstrating the feasibility of this approach. The CM composition is influenced by different factors: besides the organ of origin and the donor’s genetic background, several variables (i.e., cell density, cell passage, culture medium, days of collection, presence of preconditioning stimuli) may affect the release of soluble molecules. We recently provided evidence of comparable protective effects of hAMSC and CM treatments, indicating that cross talk between hAMSCs and the damaged tissue is not vital for the release of bioactive factors 29 . However, whether the in vivo challenge to the transplanted cells enhances or otherwise changes the release of their bioactive factors calls for further investigations.
A number of cell-released soluble factors have been proposed as mediators of the protection, and different classes of molecules have been investigated (Figure 2). Among the different classes of molecules that could be released in the secretome, the proteic component is the most widely investigated. A recent review by Bai and colleagues provided an overview of the secretion properties of umbilical-MSCs 89 , identifying more than 90 cytokines belonging to the families of ILs, TNFs, IFNs, colony-stimulating factors, growth factors, and chemokines. These proteins have important potential interest for brain pathologies, in view of their numerous physiological functions, executing anti-inflammatory and immunomodulatory effects; arousing angiogenesis, wound healing, and neurogenesis; and stimulating antiapoptotic, antiscarring, and matrix remodeling effects 89 . Pires and colleagues also identified other classes of proteins released by UCT-MSCs involved in protection against oxidative stress, anti-excitotoxicity effects, proteasomal degradation, and regulators of toxic protein deposition 90 .

Categories of molecules secreted by placenta-derived stem cells.
Nucleic acids are another class of molecules that have recently aroused great interest. MicroRNAs (miRs) are small noncoding sequences of RNA that can regulate the gene expression of target cells and are being studied for therapy as they play a key role in pathology. Packaged inside extracellular vesicles, miR can be released into the extracellular space and mediate nonhormonal intracellular communication 91 . The miR profile of UCT-MSCs was recently characterized, with distinct expression from fibroblasts 92 . Forty-two miRs have been found to be expressed differently in UCT-MSCs and fibroblasts, and among the 15 that were upregulated, miR-21, miR-146a, and miR-181 displayed the most significant expression. Analysis of the downstream targets unraveled the involvement of these miRs in the inflammatory pathways, regulating macrophage phenotype and inflammation ablation for tissue repair 93 –95 . In the brain, miR-21 mediates neuronal and microglial survival after ischemic injury, reducing the levels of the apoptosis-inducing factor Fas ligand 96,97 ; miR-181 regulates the expression of IL-10 anti-inflammatory cytokines in astrocytes 98 and reduces the neuronal apoptosis induced by microglia activated after an ischemic insult, suppressing TNF-α expression 99 .
Another hUCB-MSC-released miR involved in neurorestorative effects is miR-126 100 , which is vital for regulating the function of endothelial cells, angiogenesis, and vascular integrity 101 . Chen and colleagues showed that hUCB-MSC treatment of stroke in type 2 diabetes mellitus mice restored the serum and cerebral levels of miR-126, which was downregulated after injury 100 . Cell therapy promotes vascular remodeling and angiogenesis and reduced brain hemorrhage and BBB leakage. These effects were coupled with white matter remodeling, anti-inflammatory effects, and a generally better functional outcome. All of these effects were significantly attenuated in mice treated with hUCB-MSCs in which miR-126 expression was inhibited, indicating an important role for this miR in hUCB-MSC-induced protection 100 .
Metabolites are small molecules (molecular weight <2,000 Da) that include endogenous compounds (peptides, lipids, amino acids, nucleic acids, carbohydrates, organic acids, vitamins) or exogenous compounds (food additives, drugs, toxins, pollutants, and other chemicals that humans come into contact with) 102 . It is not easy to define the metabolomics profile of human cells because thousands of compounds belong to this class. In an in vitro model of acute brain injury obtained by exposing cortical organotypic brain slices to oxygen-glucose deprivation, we have recently demonstrated that the CM from hAMSCs was protective, and we identified a CM subfraction containing small metabolites (molecular weight <700 Da) with marked protective properties 29 . We further analyzed the CM active subfraction, by profiling 6 metabolic classes, and identified a pool of selectively enriched metabolites. These included molecules such as lysine, taurine, α-aminoadipic-acid, and spermidine, for all of which neuroprotective effects have been reported 103 –107 . However, our metabolomics analysis was targeted a priori; thus, we cannot exclude that additional metabolites with protective potential are present in the CM subfraction. Another metabolite that is attracting interest for the protection induced by stem cells is prostaglandin E2 (PGE2). PGE2 is highly secreted by placenta-derived stem cells 7,108 –110 , and there is increasing evidence of its immunosuppressive effects on lymphocytes 109,111 –113 and monocytes/macrophages 114,115 and a role for brain protection 116 –119 .
To conclude, the secretome of placenta-derived stem cells holds great promise for the treatment of acute brain injury. Different molecules have been identified as possible drivers of beneficial effects. The exact mechanisms of action are still not clear and numerous factors, rather than any single one, probably contribute synergistically to the neuroprotective effects.
Clinical Use of Placenta-Derived Cells in Acute Brain Injury
Placenta-derived stem cells are still only at their dawn in the clinical setting compared to the more widely used bone marrow–derived stem cells, so only a few studies investigating the feasibility and safety of this approach for acute brain injury have been conducted so far (Table 1).

Abbreviations: hUCB, human umbilical cord blood; SPIO, supermagnetic iron oxide; hUCT-MSCs, human umbilical cord tissue-derived mesenchymal stromal cells; BM-MSCs, bone marrow-derived mesenchymal stromal cells; UC-MSCs, umbilical cord-derived mesenchymal stromal cells.
hUCB-derived stem cells for acute brain injury were first used as an experimental neurorestorative treatment in a 16-mo-old child in a permanent vegetative state resulting from a cardiac arrest–induced global hypoxic/ischemic brain injury when he was 9 mo old 120 . The child had his own hUCB deposited in a blood bank; autologous hUCB cells were isolated and committed to neuronal lineage (hUCB-NSCs), then labeled with supermagnetic iron oxide (SPIO) and ICV transplanted. The child underwent 4 transplantations of 12 million hUCB-NSCs each, the first was when he was 16 mo old, followed by 3 consecutive monthly injections. Magnetic resonance imaging (MRI) monitoring 24 h posttransplant showed hUCB-NSCs extending along the ependyma of the lateral ventricle. The SPIO signal progressively decreased but was still visible 7 d, 1 mo, and 2 mo after transplant, and disappeared by 4 mo. There were no signs of brain edema, hemorrhage, or tumor in the 6 mo of observation 120 or in the follow-up at 33 mo 121 . The only side effect was transient moderate fever the day after transplant. Thus, the authors affirm the feasibility and the safety of intracerebral transplant of hUCB-NSCs in the brain.
Jiang and colleagues ran a pilot study to assess the safety of hUCT-MSCs delivered by a catheter through perilesional vessels to treat stroke of the MCA territory, in order to release more stem cells to the infarcted area compared to the more commonly used intravenous transplantation 122 . Four males (between 40 and 59 y) with stroke (3 ischemic and 1 hemorrhagic) were treated with 20 million hUCT-MSCs between 11 and 50 d after the stroke onset. Patients were monitored for the presence of additional ischemic strokes: angiography 20 min after cell delivery did not indicate any new blood flow impairment and MRI prior to discharge did not reveal any new infarct. After transplant, there was no sign of immune response (fever or rash), demonstrating the feasibility of intra-arterial delivery of hUCT-MSCs. The trial was not powered to assess hUCT-MSC efficacy and no conclusions could be drawn about the therapeutic potential.
Chen and colleagues employed a multiple cell transplantation approach for stroke patients in the chronic phase 123 . Ten stroke patients (6 ischemic and 4 hemorrhagic), aged 42 to 87 y, were transplanted with different cell populations: olfactory ensheathing cells from aborted human fetal olfactory bulbs, Schwann cells from aborted human fetal sciatic nerve, neural progenitor cells from aborted human subependymal zone of fetal brain, and/or hUCT-MSC collected after birth. Patients received one or more doses of cells, ranging from 1 to 23 million cells, using different delivery routes (intracranial, intrathecal, or intravascular) and different intervals from stroke onset (from 6 mo to 20 y). These heterogeneous treatments/protocols led to no adverse events during or after the procedure or during the long-term follow-up. The authors state that all patients achieved some degree of improvement in neurological function, speech, muscle tension, balance, pain relief, and respiratory stability. Again, the small sample size limits the value of the findings on combinatory cell therapy, so the results must be interpreted with caution.
A retrospective analysis compared the effect of human bone marrow-derived mesenchymal stromal cells (hBM-MSCs) and hUCT-MSCs or a conventional surgical approach for ICH 124 . A total of 24 patients were treated with conventional hematoma removal surgery within 6 h of the hemorrhage. Patients scheduled for MSC treatment received 2 transplants of hBM-MSCs (n = 7) or hUCT-MSCs (n = 9) into the hematoma cavity 2 and 3 wk after injury. All the transplanted patients had a shorter hematoma reabsorption time and a better outcome at 5 y than untreated patients. Importantly, patients receiving hUCT-MSCs had a better outcome than hBM-MSC-treated patients starting from 3 mo after injury, suggesting that placenta-derived stem cells have higher therapeutic potential than adult stem cells 124 .
Only one clinical study using placenta-derived stem cells in TBI has been published 125 . Forty patients with TBI at chronic stages (range: 1-11 y post-TBI) were randomly allocated to treatment with hUCT-MSCs or vehicle, and follow-up was obtained at 6 mo posttreatment. Twenty patients in the stem cell group received 4 hUCT-MSC transplants, each containing 10 million stem cells (over an interval of 5-7 d) by lumbar puncture. All patients were analyzed by Fugl-Meyer assessment (FMA) 126 , a multi-item scale assessing motor function, sensory function, balance, joint range of motion, and joint pain, and by Functional Independence Measure (FIM) 127 , a multi-item rating scale assessing self-care, bowel and bladder management, mobility, communication, cognition, and psychosocial adjustment. During stem cell transplantation, patients were monitored for body temperature, heart rates, blood pressures, oxygen saturations, and respiratory rates, and no obvious abnormalities were found. Four (20%) patients experienced headache and mild dizziness within 48 h post lumbar puncture. At 6 mo, patients received head and spinal cord MRI examinations and no abnormalities related to the stem cell transplantation were found. Rating scales at 6 mo indicated that while the control group had FMA and FIM scores not significantly different from the baseline time point, the hUCT-MSC-treated patients had slightly better FMA and FIM scores 125 . Thus, the preliminary findings of the therapeutic potential of hUCT-MSCs demonstrate the feasibility and safety of this approach for acute brain injury. Further research is now needed to validate and strengthen these results in order to offer cell therapy for patients with acute brain injury.
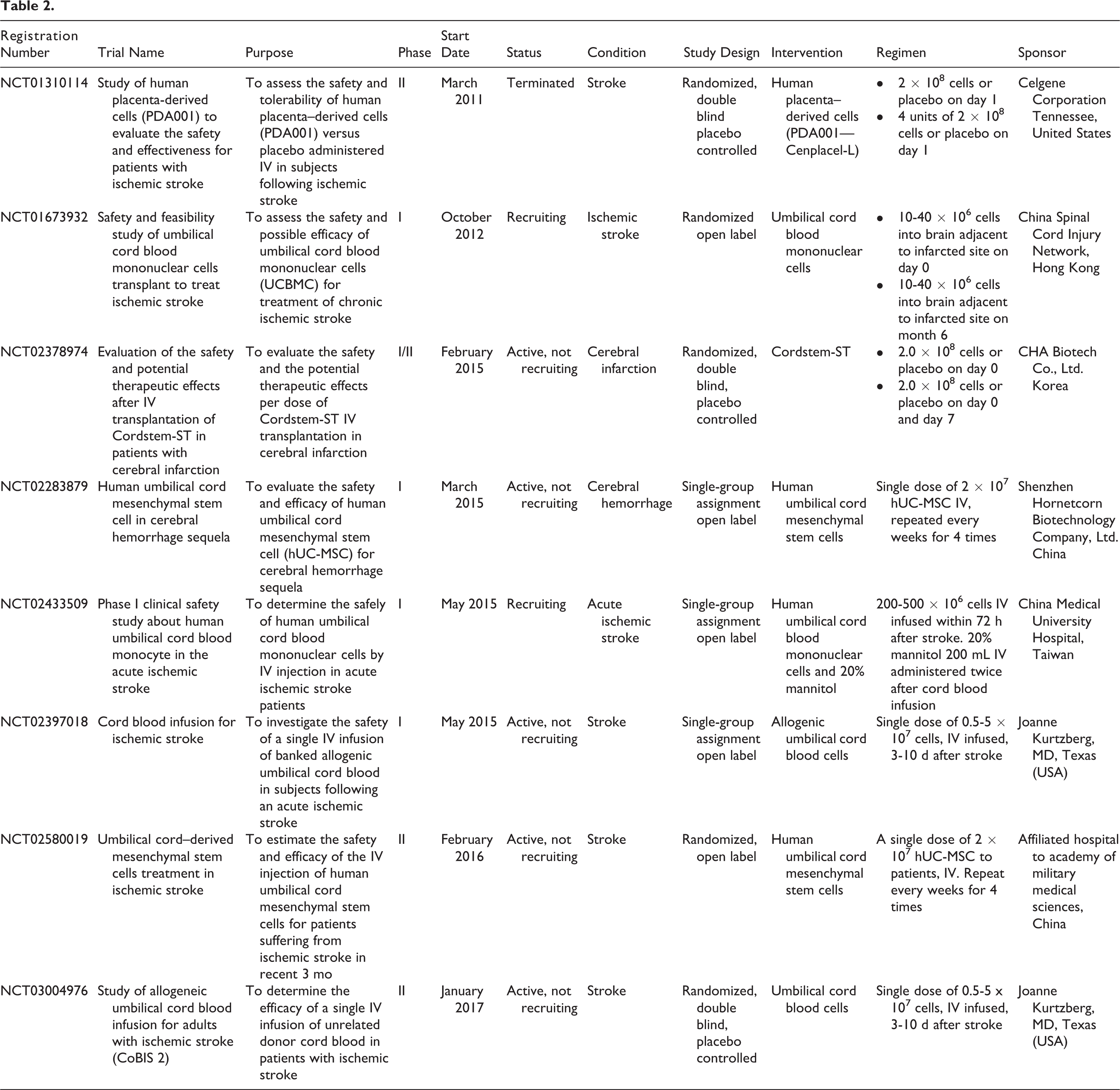
Currently, 8 ongoing phase I or II clinical trials are present in the world’s largest registry clinicaltrials.gov using placenta-derived stem cells for acute brain injuries (Table 2). Seven trials target stroke, 1 cerebral hemorrhage, and none have been designed for TBI. All trials are single center and use UC-derived stem cells. Three trials are designed as single-group assignment open label, 2 as randomized open label, and 3 as randomized double-blind placebo-controlled trials. Thus, several conclusions will be drawn at the end of these trials, posing the bases for the construction of a larger phase 3 trial.
Conclusions
There is growing experimental evidence for the efficacy of placenta-derived stem cells for acute brain injury. The multitarget potential of cell therapy fulfills the need of the damaged brain in which numerous injury mechanisms are triggered. Infused cells can reprogram the local inflammatory microenvironment from detrimental to beneficial, favoring protective/pro-regenerative changes in the lesioned tissue and contributing to permanent improvement in neurological function. Even in case of predifferentiation into neuronal lineage, cell replacement is unlikely, whereas the main mechanisms of action through which infused cells confer protection is through the secretion of bioactive factors, suggesting the potential for a cell-free strategy based on the cell secretome. Different classes of molecules have been identified as mediators of protective effects. However, the pool of factors sufficient and necessary to promote brain protection/repair has not yet been pinpointed and further studies are needed to identify a controlled, standardized “secretome-based” cell-free strategy. Clinical studies are in their infancy, with few reports demonstrating the safety and preliminary efficacy of cell transplantation. The ongoing trials are crucial to assess the efficacy of placenta-derived stem cells for acute brain injury.
Footnotes
Authors’ Note
Francesca Pischiutta received a fellowship from “Fondazione Umberto Veronesi” (FUV, 2016).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the association “Amici del Mario Negri.”
