Abstract
Damage to bones resulting from trauma and tumors poses a significant challenge to human health. Consequently, current research in bone damage healing centers on developing three-dimensional (3D) scaffolding materials that facilitate and enhance the regeneration of fractured bone tissues. In this context, the careful selection of materials and preparation processes is essential for creating demanding scaffolds for bone tissue engineering. This is done to optimize the regeneration of fractured bones. This study comprehensively analyses the latest scientific advancements and difficulties in developing scaffolds for bone tissue creation. Initially, we clarified the composition and process by which bone tissue repairs itself. The review summarizes the primary uses of materials, both inorganic and organic, in scaffolds for bone tissue engineering. In addition, we present a comprehensive study of the most recent advancements in the mainstream techniques used to prepare scaffolds for bone tissue engineering. We also examine the distinct advantages of each method in great detail. This article thoroughly examines potential paths and obstacles in bone tissue engineering scaffolds for clinical applications.
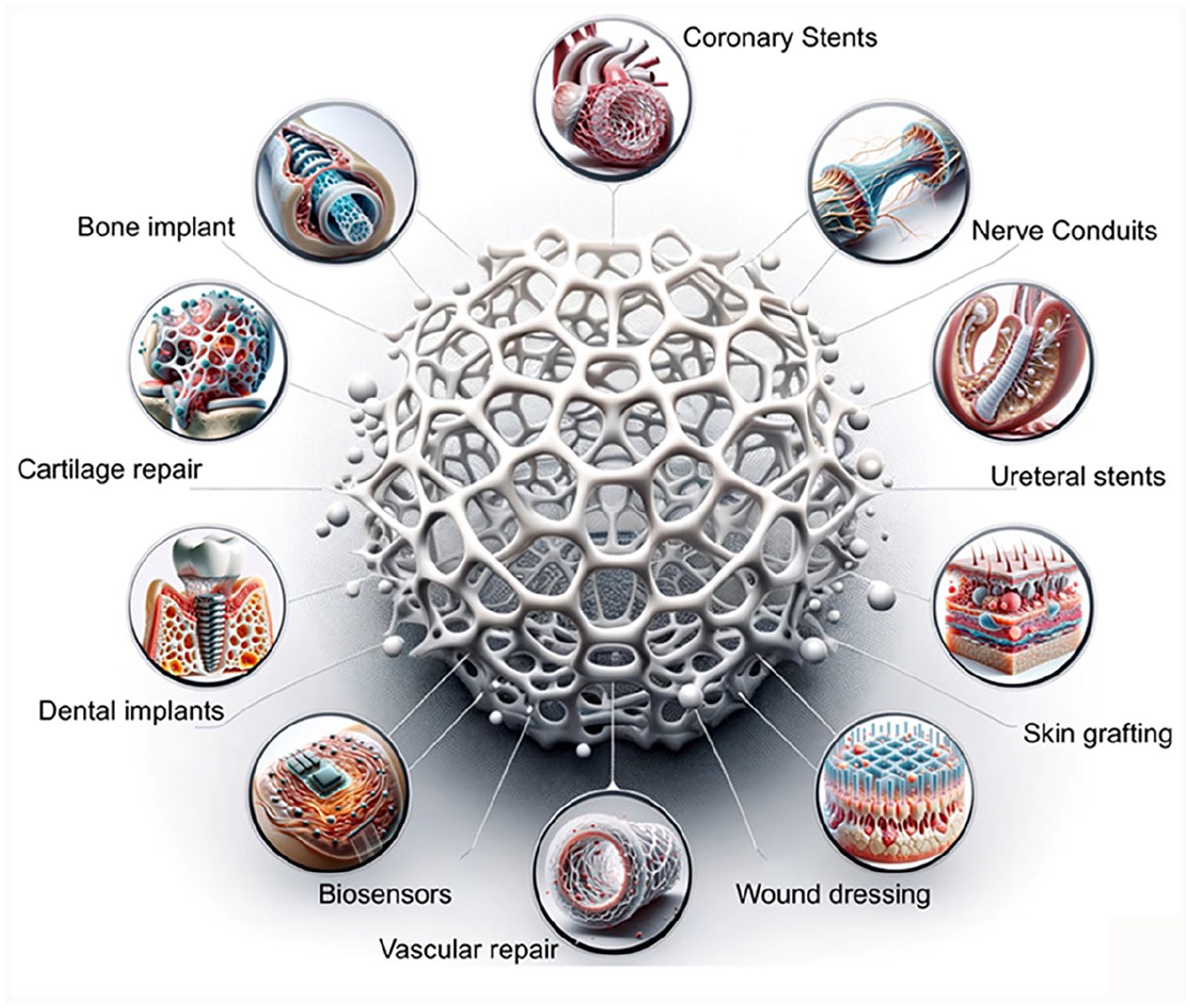
Introduction
Three-dimensional (3D) printing, or additive manufacturing, is a cutting-edge technique that enables the production of 3D items by building them layer by layer, using a digital design as a blueprint1–4. The process commences with a digital file, such as a computer-aided design (CAD) model, serves as a blueprint for the desired object5,6. The computerized model is segmented into thin, cross-sectional layers, then printed or deposited using various materials and techniques7,8. Multiple 3D printing techniques are available, each with unique features and materials that can be used9,10. Widely used methods include fused deposition modeling (FDM), stereolithography (SLA), selective laser sintering (SLS), and binder jetting. In addition, ceramics are utilized in 3D printing, which has significantly progressed in various industries, such as electronics and healthcare. Ceramic powders are employed in ceramic 3D printing11,12.
Hybrid materials (HMs) denote one of the most emergent material classes at the edge of technological advancements 13 . Material properties achieved via a synergetic combination of more than one component on the molecular scale make HMs attractive for several applications 14 . There are several approaches to the classification of HM. They can be based on the source of origin, bonding, properties, and formation route, and highly favored materials in 3D printing 15 . Commonly utilized materials include PCL, PLA/HA, PCL/GelMA, Silk Fibroin/Bioactive Glass, Chitosan/β-TCP, and PLGA/Collagen. Every hybrid variety has unique attributes, such as its durability, flexibility, resistance to temperature changes, and ease of printing16,17. 3D printing utilizes metal as a substitute material 18 .
Metals such as cobalt-chrome, titanium, stainless steel, and aluminum are usually cast-off in additive manufacturing19,20. Durable metal components are produced through the layer-by-layer fusion of metal powders utilizing selective laser melting (SLM) or electron beam melting (EBM) techniques in metal 3D printing 21 . In addition, ceramics are used in 3D printing, which has advanced uses in industries including electronics and healthcare. Ceramic powders are used in ceramic 3D printing 22 .
The human skeletal system functions as a fundamental framework, offering structural support and safeguarding the organism23,24. Nevertheless, various causes, such as the natural process of aging, physical injuries, or medical conditions, can contribute to the diminished strength or impairment of bones, ultimately resulting in substantial health complications25,26. Lately, there has been an increasing fascination with advancing novel techniques for identifying, measuring, and identifying diseases through various means 27 . To properly treat bone illnesses and injuries, it is necessary to utilize tissue engineering and regenerative medicine procedures 28 . One practical approach in biomedical applications is the utilization of bone scaffolds. These scaffolds are like a 3D framework for tissue regeneration and promote the development of new tissues and bones 29 . Because bone tissue is self-repairing and regenerating, minor flaws typically disappear independently. However, when bone abnormalities grow more significant than a critical size barrier (about >2 cm), the healing process is inadequate and frequently fails to mend30–32. Every year, 4 million people worldwide need bone replacement surgery or grafts33,34. As a result, treating bone abnormalities effectively is crucial from a clinical standpoint35,36. In clinics, bone grafting is a mainstay. Depending on the circumstances, the defect site can heal successfully using different grafts37,38.
Customized solutions for individual patients: 3D printing produces bone scaffolds tailored to each patient’s needs 39 . This individualized strategy optimizes the efficacy of the treatment, enhancing bone rejuvenation consequences and minimizing the likelihood of problems 40 . Biocompatibility: 3D-printed bone scaffolds can be fabricated using biocompatible materials, such as bioceramics or biodegradable polymers, which are highly compatible with the human body 41 . These compounds enhance cell attachment, proliferation, and differentiation, aiding the regeneration of new bone tissue 42 . This article concisely summarizes the recent advancements in the research and enhancement of 3D printing methods for producing scaffolds utilized in bone tissue engineering 43 .
Critical Requirements for 3D Printed Scaffolds
3D printing has significantly transformed the field of tissue engineering and regenerative medicine, specifically in the field of bone scaffolds44,45. Researchers can develop scaffolds that imitate the form and function of genuine bone by using biomaterials, advanced 3D printing technology, and detailed design procedures46,47. Scaffolds provide customized mechanical characteristics, such as rigidity, durability, and adaptability, together with interconnected porous structures that improve the infiltration of cells and the exchange of nutrients48,49. 3D-printed bone scaffolds are an excellent choice for patients requiring bone regeneration as long as they fulfill the criteria of biocompatibility, scaffold structure, sterilization, and regulatory compliance19,50. These scaffolds offer individualized bone repair, tissue regeneration, and enhanced quality of life. To achieve this objective, the initial and most crucial stage is thoroughly comprehending the requirements51,52. They are given in Table 1.
Biomedical Application and Properties of Hybrid Scaffolds for Clinical Application.
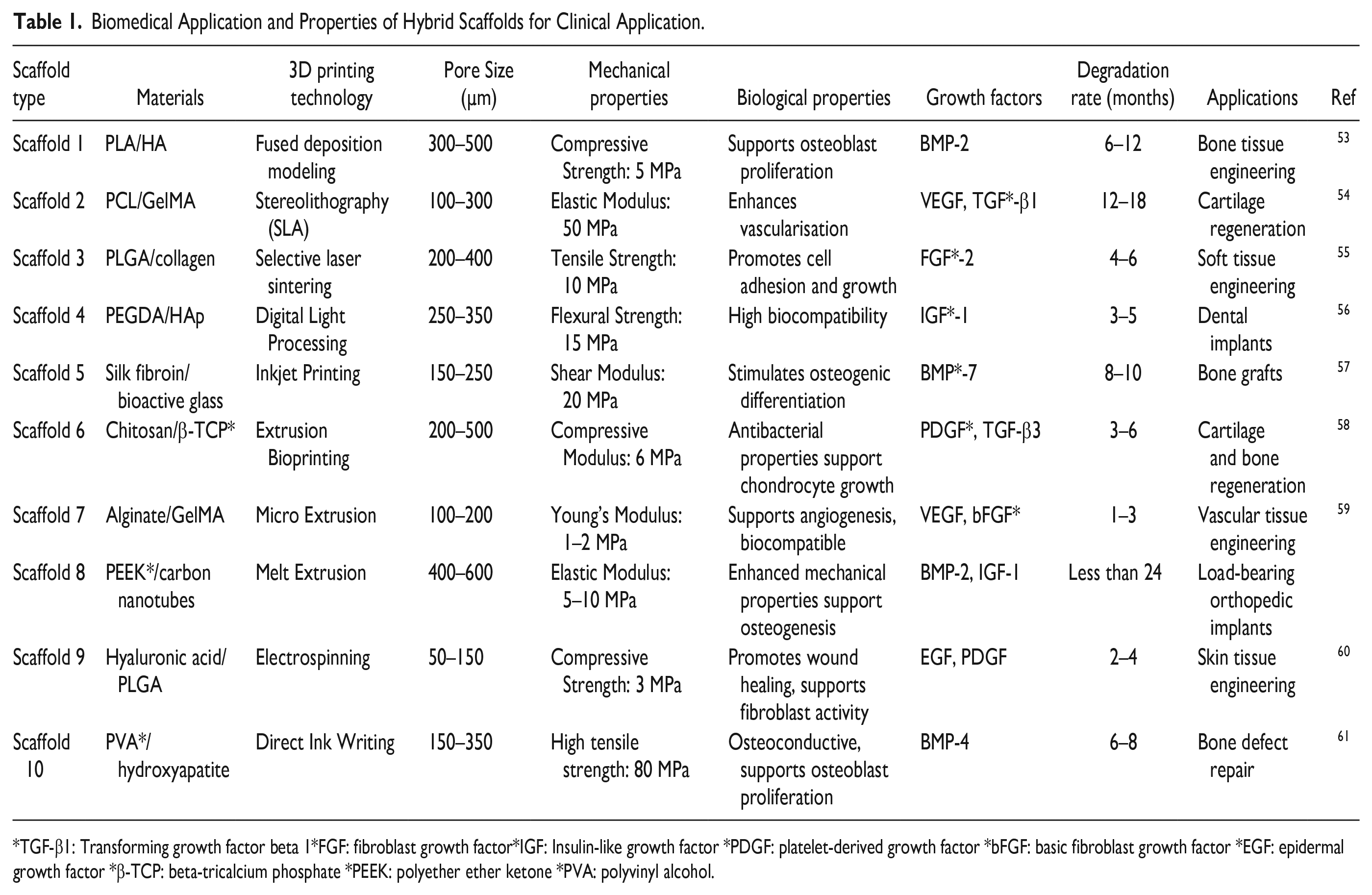
TGF-β1: Transforming growth factor beta 1*FGF: fibroblast growth factor*IGF: Insulin-like growth factor *PDGF: platelet-derived growth factor *bFGF: basic fibroblast growth factor *EGF: epidermal growth factor *β-TCP: beta-tricalcium phosphate *PEEK: polyether ether ketone *PVA: polyvinyl alcohol.
While there have been thorough examinations of scaffolds made from metal, ceramic, and polymers, more research is needed on hybrid scaffolds for advanced therapeutic purposes62,63. It is imperative to thoroughly investigate how hybrid scaffolds might effectively address crucial therapeutic requirements64,65. This encompasses a brief comprehension of their function in biomimetics, accurate bone regeneration, focused drug administration, tumor therapy, and infection treatment66,67. Exploring the incorporation of biomimetic characteristics into these scaffolds remains an unexplored domain, and conducting research is crucial to enhance compositions for more efficient bone regeneration68,69. Furthermore, there are unexplored possibilities in regulated drug administration, targeted treatments, and infection control70,71.
The Mechanism for Repairing Bone Tissue
The bone tissue repair and healing process is intricate and consists of several phases72,73. These stages primarily include inflammation and the formation of a blood clot, the recruitment and multiplication of stem cells, the development of blood vessels, the specialization of mesenchymal stem cells (MSCs), and the final phase of tissue remodeling74,75. Fractures disrupt a specific area of the blood vessels and nearby tissue, creating a hematoma and the following inflammatory phase. Within 24 hours following the fracture, the soft matrix at the hematoma site attracts immune cells to facilitate an inflammatory response76,77. After some time (week), the hematoma and inflammatory response are resolved, and the hematoma site is replaced by granulation tissue78,79. Following the inflammatory and hematoma phase, many cells, including osteoblasts and endothelial cells, become active at the location of the defect. In addition to the proliferation of stem cells, angiogenesis occurs throughout the bone tissue healing process80,81. Many blood vessels in bone are crucial for bone regeneration Fig. 1. The process of ultimate bone remodeling is a physiological phenomenon significantly influenced by the interaction between osteoblasts and osteoclasts82,83. An essential objective is to integrate the attributes and principles of each step in bone tissue restoration with scaffolds, enhancing the performance of the scaffold materials through targeted alterations to attain superior and more effective outcomes in treating bone abnormalities 84 .
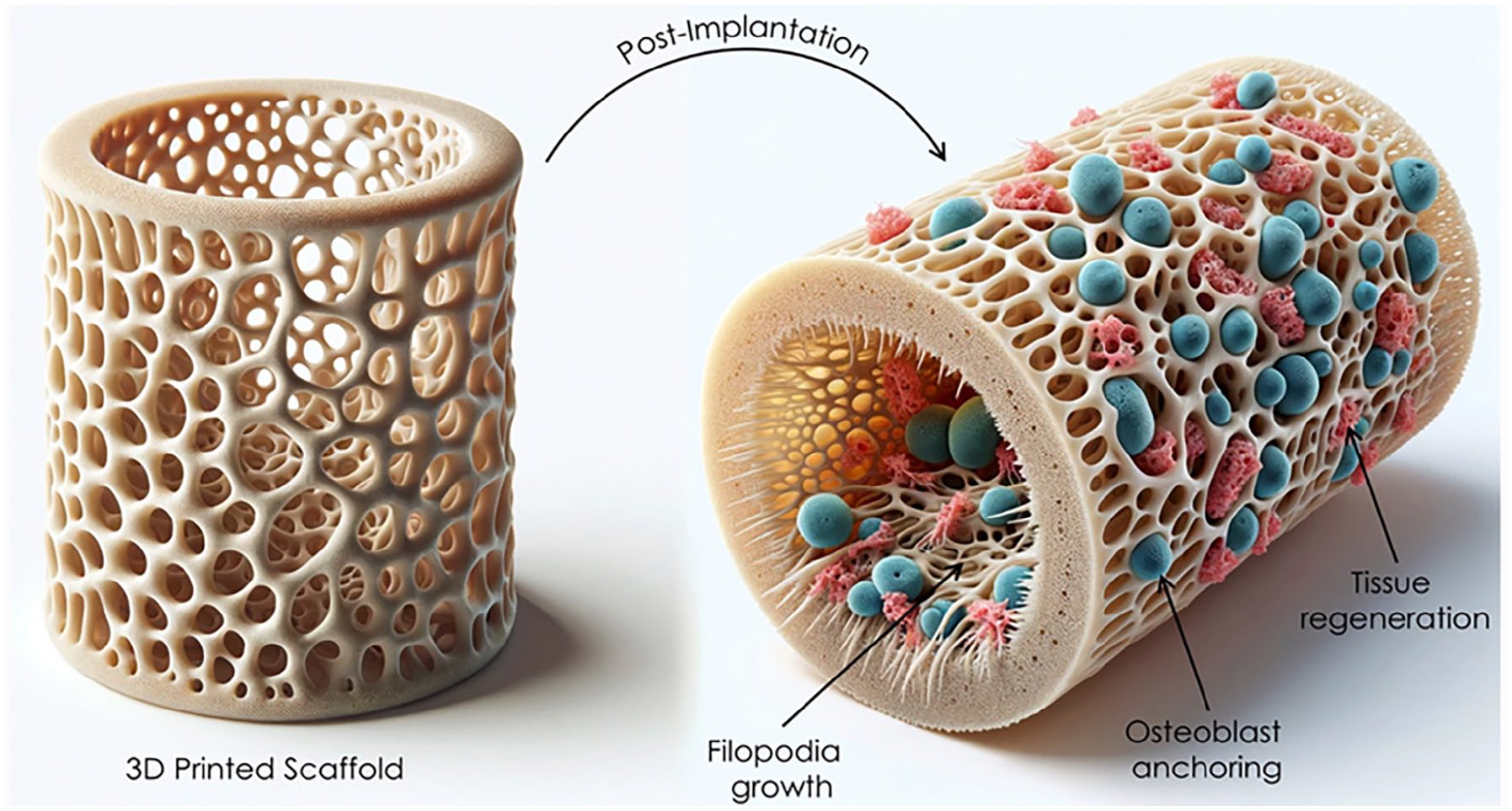
Schematic illustration of bioactive response of 3D-printed scaffolds during physiological immersion.
The Scaffold Structure Design Ensures Safety and Efficiency on Construction Sites
A 3D model of the scaffold can be created and transformed into a printable setup like Stereolithography (STL)85,86. Customized scaffold shapes could be generated via CAD software based on specific patient structural anatomical data about the lesion87,88. A 3D model is created by analyzing the anatomical data of the affected area, which is collected using computed tomography (CT) or magnetic resonance imaging (MRI) 89 . Med CAD design and interfaces, inverse engineering surfaces, and STL-triangle shape model converting procedures are various approaches to creating CAD models from therapeutic pictures 90 . Scaffolds and porous architecture are essential in tissue regeneration as they maintain tissue volume, fulfill temporary mechanical roles, and transport biofactors 91 . An effective scaffold will integrate mechanical functionality with the delivery of biofactors, facilitating a gradual transformation from scaffold to regenerated tissue as the previous scaffold breaks down 92 .
Consequently, the scaffold structure should imitate the inherent properties of genuine bone, including interconnecting pores93,94. The scaffold’s porosity is crucial for cell intrusion, nutrition exchange, and tissue amalgamation. Researchers can use 3D printing to design and fabricate pores of varying sizes and forms95–97. Fig. 2 explains the different properties required to construct the 3D scaffolds including their design, (shape, pore size, morphology) biofunctionality, (hemocompatibility, bioactivity, angiogenesis and osteointegration) manufacturing (electrospinning, SLA and 3D printing) strength (mechanical integrity, poor strength, stiffness, load bearing capacity and toughness) microstructures (degradation rate, chemical stability, processibility and crystallinity).
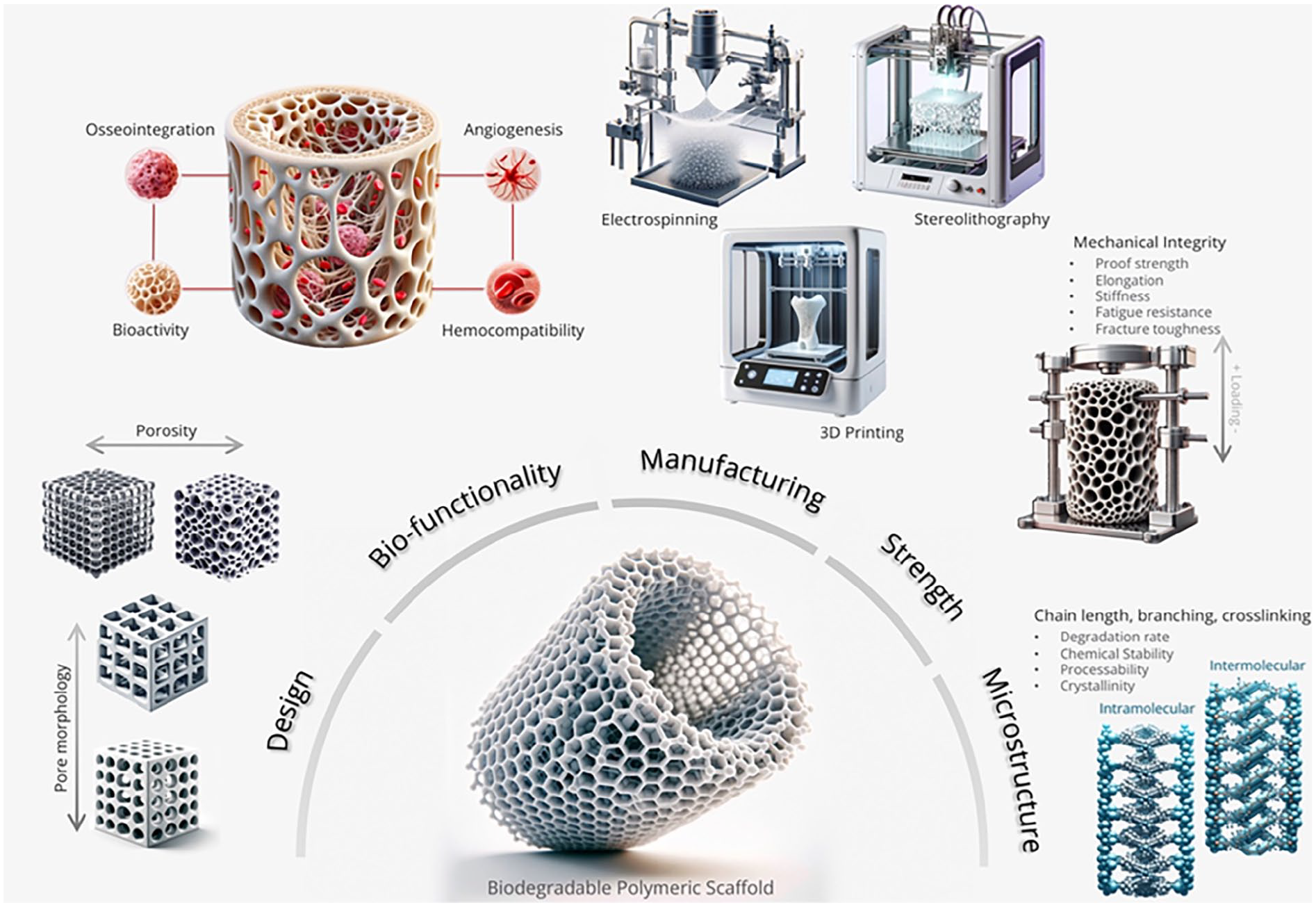
Multifaceted evaluation parameters, including design, functionality, manufacturing, mechanical integrity, and microstructure, are required for the development of an ideal scaffold.
Tissue Engineering
Tissue engineering is a promising therapy option that uses engineering concepts to alleviate tissue damage98,99. The field consists of three essential elements: scaffolds, cells, and growth factors. Bioreactors play a vital role in tissue engineering3,100. The ideal scaffolds function as structural reinforcement for damaged tissue, transforming the growth factors produced by cancer cells into stimuli that promote tissue regeneration. Conventional approaches to treating tissue injury have faced substantial challenges throughout the years 101 .
Recent progress in bone tissue engineering has integrated growth factors and other biomolecules into scaffolds to direct cell behavior during the regeneration of organs and tissues102,103. In the past 20 years, advances have been achieved in developing and applying biological scaffold materials46,104. The three primary components of bone tissue engineering are bone progenitor cells, bone growth factors, and scaffolds90,105. These elements work together to promote cell adhesion, maintain cell function, and imitate the natural process of bone tissue regeneration106,107. The scaffold must serve as a transient template for cell regeneration of new bone tissue while also being capable of degradation to allow for replacement by the newly formed bone tissue108,109. The scaffold’s primary function is to create an ideal microenvironment for cells, facilitating new tissue production and distribution of nutrients between the cells and their surroundings 110 .
Bone Scaffold Formation
Temporary constructs known as porous scaffolds facilitate the regeneration of bone tissue by providing an appropriate environment. They promote cell attachment, growth, specialization, and migration to the injury site111,112. Scaffolds are essential to tissue engineering because they offer the best extracellular matrix (ECM) for progenitor cell proliferation and differentiation 113 . These cells can enter the scaffold and start the development, differentiation, multiplication, and migration processes because they react to biochemical and physical cues in their environment114,115. When the environment is favorable, the cells secrete ECM and produce new tissues 116 . The best scaffolds have strong adhesion to bone-forming cells, biodegradability, robustness, and consistency in their mechanical properties 117 . The cells must be able to move toward the scaffold, stick to it, and multiply. Another essential feature is the scaffold’s connected porosity, which permits precise cell development and spreading inside the porous structure 118 .
This facilitates efficient angiogenesis in the surrounding tissue. The objective of constructing bone scaffolds is to produce an environment demonstrating biophysical, biomechanical, and biochemical features responsible for cell growth, specialization, and viability. Polymers, ceramics, and metals are the primary categories of biodegradable materials that have lately been examined in clinical and research settings119,120. Therefore, a scaffold is a crucial element in tissue engineering, and to fulfill its essential function, it must possess the features mentioned above 121 .
Clinical Applications of 3D-printed Scaffolds
Wound and Infection Healing
A wound is a disruption or break in the skin caused by physical or thermal damage or a pathological cause 122 . The nature and severity of wounds vary depending on their underlying etiology, clinical manifestations, healing processes, or anatomical location 123 . Regardless of their characteristics, wounds pose a significant healthcare challenge in the development of chronic diseases as they can raise healthcare expenses and complicate both internal and external health 124 . Wound healing involves a range of well-coordinated molecular processes, including hemostasis, inflammation, proliferation, and remodeling 125 .
Using 3D scaffolds with stem cell delivery has shown great potential in regenerative medicine. Wang et al. explained the examples of cell types include bone marrow mesenchymal stem cells, human umbilical cord perivascular cells (HUCPVC), and amniotic fluid-derived cells 126 . Stem, endothelial progenitor, and circulating angiogenic cells (CACs) are frequently studied. Haki et al. utilize early endothelial progenitor cells (EPCs), also known as CACs, derived from peripheral blood mononuclear cell fraction and can be used locally to address nonhealing diabetic foot ulcers127,128. Lv et al. does the augmentation in the formation of new blood vessels and a higher proportion of wound healing was noted when a scaffold made of collagen was used to transfer CACs to a diabetic rabbit ear wound (specifically, an ulcer produced by alloxan) 129 . A 3D membrane scaffold, derived from a freeze-dried conditioned medium of bone marrow mesenchymal stem cells (FBMSC-CM), effectively expedited wound healing and improved the formation of new blood vessels (neovascularization) and the growth of epithelial tissue (epithelialization) by enhancing the presence of nourishing elements in the wound area130,131. Fig. 3 explains the phenomena of wound healing for a long time and how hemostasis occurs due to 3D implant scaffolds, the formation of new cells, and epithelisation leading toward the remodeling of skin or wound repair.
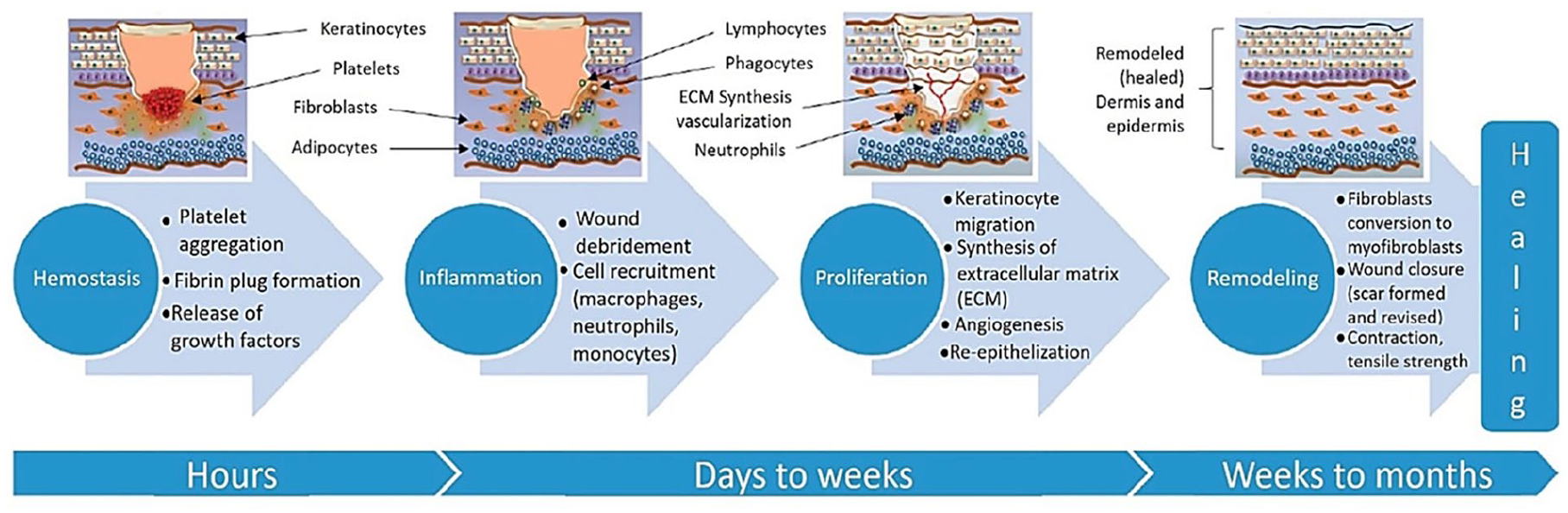
Mechanism of wound healing from hours to months adopted with permission 122 .
Tumor Therapy
3D scaffolds represent a promising solution in tumor therapy by providing a controlled and replicable microenvironment for studying cancer progression and testing treatments 111 . Many 3D scaffolds are used to treat tumors like novel bioceramic scaffolds. These scaffolds can be engineered to mimic the ECM of tumors, allowing for more accurate modeling of tumor growth, invasion, and metastasis 115 . By incorporating bioactive molecules, such as drugs, growth factors, or genetic material, 3D scaffolds can deliver targeted therapies directly to the tumor site, potentially enhancing the efficacy and reducing the side effects of conventional treatments 129 . Moreover, Fig. 4 explains the mechanism of how local sites of proteins and membranes are being damaged by the heat-generated functional scaffolds, leading to apoptosis and cell death. As a result, scaffolds exhibiting superior photothermal or magnetothermal properties are highly effective as localized treatment agents. The ability to personalize these scaffolds based on patient-specific tumor characteristics facilitates the development of tailored therapeutic strategies, paving the way for more effective and individualized cancer treatments125,129.
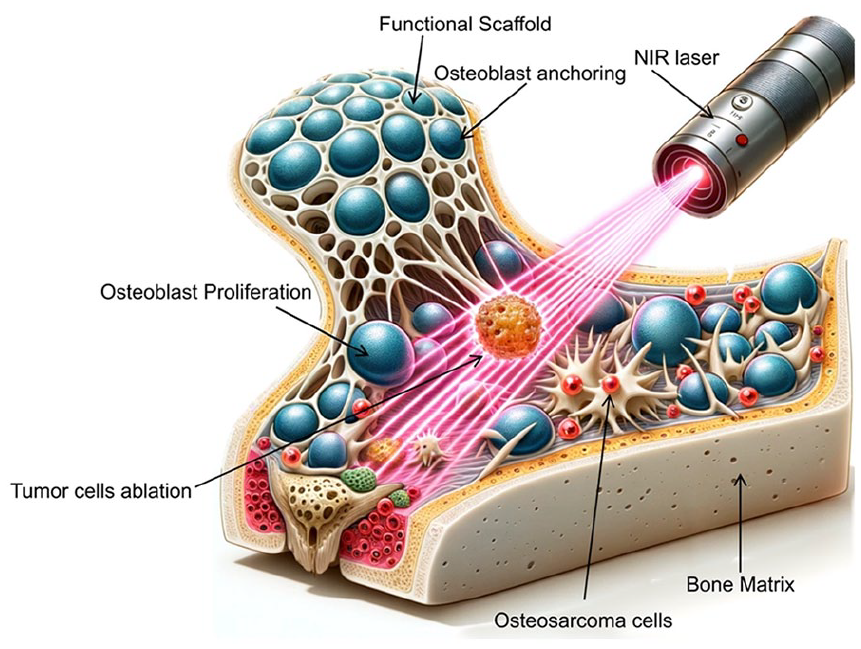
Schematic illustration of a bifunctional scaffold with significant potential for clinical use in bone tumor therapy. The scaffold demonstrates pronounced capabilities for both regenerating bone tissue and treating tumors.
Cartilage and Spine Injury
Cartilage is a soft bone with a minimal capacity for regeneration. When a lesion, such as osteoarthritis, is created, more innovative explanations are needed. Lan et al. 132 employed a scaffold using human nasal and chondrocytes using type 1 collagen. The scaffold was implanted in the mouse skin, and after 9 weeks, the printed cartilage regenerated its original shape and size. Cell viability decreased during the construction of the 3D structure. Similarly, after subcutaneous implantation, Beketov et al. 133 produced the scaffold using the bio-ink consisting of 4% collagen and chondrocytes. The cartilage tissue contains a high amount of COL2 and glycosaminoglycan (GAG).
A spine injury can be caused by accidents, swelling, dislocation, extrusion, and ischemia, leading to the damage of irreparable nerve cells. Neural stem cells (NSC) and their spatial distribution in the spinal cord is the route toward its successful repair again. Liu et al. 134 used different HMs to prepare the scaffold, such as bio-ink containing the mixed NSCs, hyaluronic acid derivatives, and chitosan. After implantation in the rats, it restored the locomotor abilities, called viability, and renewed axons. Similarly, another study explained how 3D-generated scaffolds with the help of gelatin combined with oligodendrocytes and NSCs improved motor skills and the production of new axons and neurons after implantation28,135.
Heart Disease and Liver Failure
Decrease of cardiac fibrosis, cardiomyocyte hypertrophy, and increase of vascular formation using the 3D-constructed scaffold to treat the congenital heart disease of right ventricle failure in the rat model by using the bio-ink containing progenitor cells laden cardiac ECM gelatin and neonatal human c-kit 136 . Similarly, to produce elongated cells that can contract, embedding the printed construct between 2 layers of rat omentum over a week using hydrogel with iPSCs differentiated into cardiomyocytes and patient-specific endothelial cells 137 .
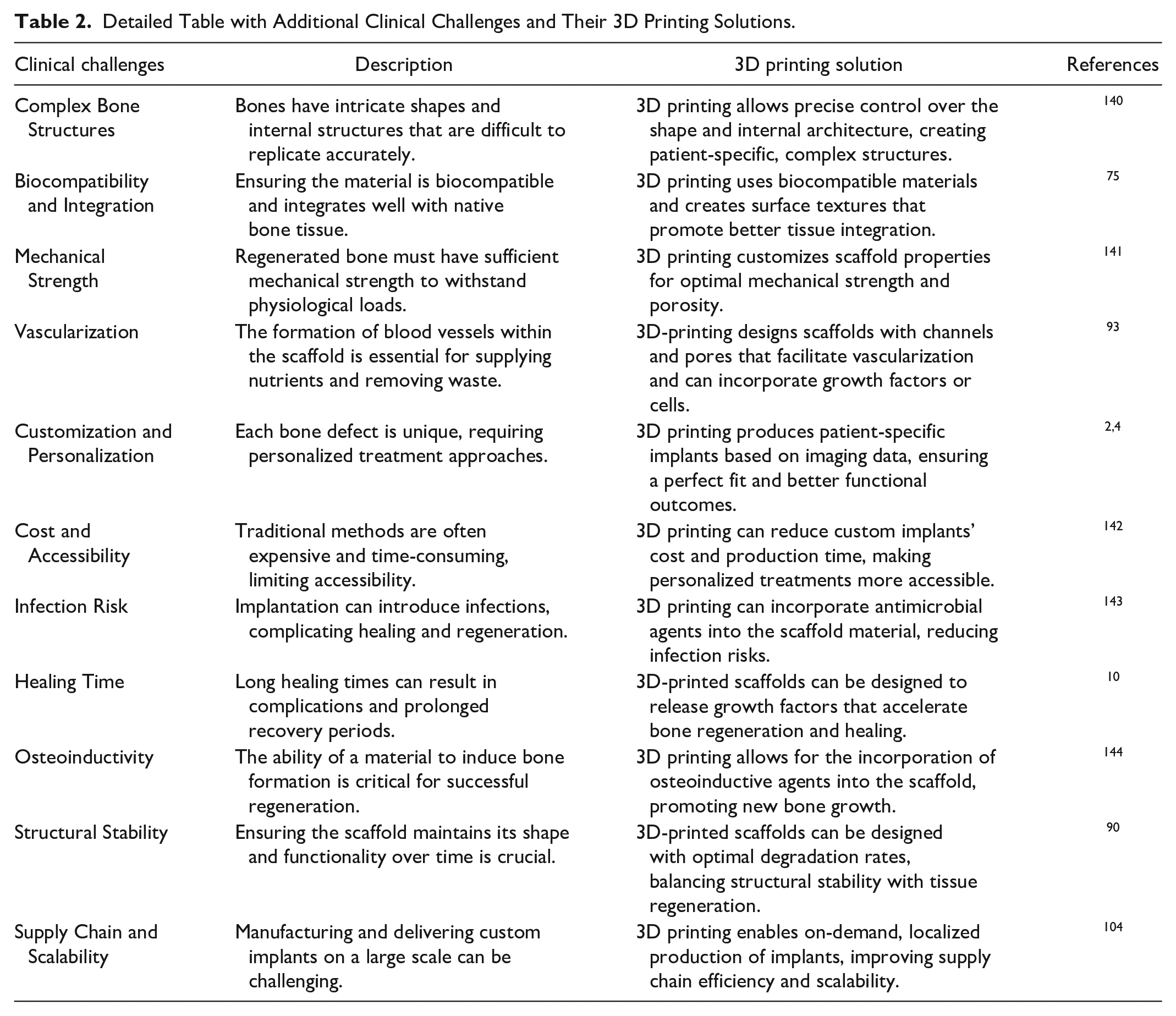
Human hepatocytes and methacrylate gelatin bio-ink were used to treat liver failure in mice. A 3D scaffold constructed stimulated liver cells’ vascularization and normal function 138 . However, scaffolding in the mouse liver damage increased albumin expression and accelerated cell proliferation by using the 3% alginate hydrogel with induced hepatocyte cells 139 . In addition, Table 2 explains the present challenges and their solution provided by 3D technology.
Detailed Table with Additional Clinical Challenges and Their 3D Printing Solutions.
Hybrid Scaffolds
Hybrid scaffolds are a promising field of study because they combine multiple materials to create improved features better suited for various tissue engineering applications. PCL/collagen scaffolds have been employed to fabricate artificial human skin145,146. The resulting material gains muscular tensile strength by combining collagen with a small amount of PCL. This makes it helpful in creating scaffolds perfect for human skin tissue engineering147,148. Recent investigations indicate that NPs are widely utilized in biomedical applications, including biosensing of metabolites, drug delivery, bioimaging, anti-biofilm, and antibacterial applications 149,150. A study utilized PCL combined with titanium oxide, which has antibacterial properties and is coated with collagen to create a wound dressing material with antibacterial capabilities103,151. A different research study utilized human endometrial stem cells (hEnSCs) by introducing them into a PCL/collagen scaffold to create an innovative structure for skin engineering 152 . Due to the endometrium’s remarkable regenerative ability, this nanofiber was suggested to stimulate angiogenesis without needing growth hormones. This indicates its potential for repairing skin tissue during wound healing 153 . A composite of poly L-lactide (PLLA) and chitosan was created to preserve the essential properties of both materials. PLLA possesses high mechanical strength but is not conducive to cell growth 154 .
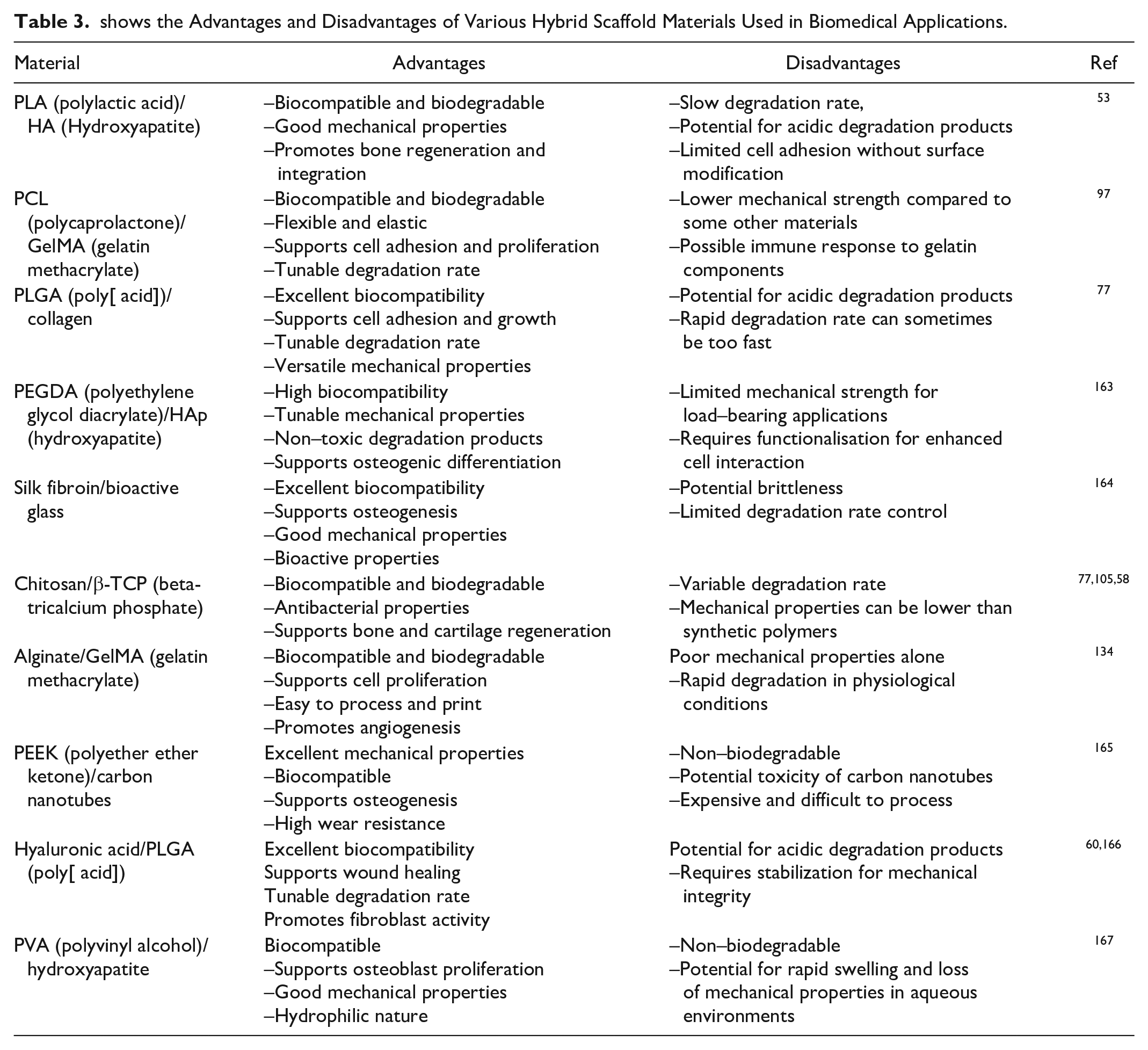
On the contrary, chitosan demonstrates favorable tissue regeneration capabilities, but it lacks sufficient strength58,105. Consequently, collagen was utilized to construct scaffolds with funnel-shaped structures with pores, enabling the cells to proliferate and establish connections beneath the surface 77 . Subsequently, the collagen funnels were positioned onto a PLLA mesh to create hybrid scaffolds. Animal experiments have demonstrated that applying these hybrid scaffolds to seed fibroblast cells can improve the repair of incisional lesions17,52,126. Combining chitosan and silk nanofibrils has been utilized to create nanocomposite scaffolds that demonstrate excellent resistance to high temperatures and impressive mechanical durability 126 . The nanocomposites share a comparable composition with the ECM, so they can produce a more sophisticated biomaterial for skin engineering 155 . An article has described a bilayer nanocomposite scaffold made from silk fibroin, gelatin, and oxidized alginate. This scaffold has a structure comparable to the ECM and has the potential to be used in regenerative medicine and skin engineering 156 . The efficacy of hybrid collagen scaffolds containing ZnO-curcumin nanocomposites was assessed to promote accelerated wound healing and minimize scarring 157 . An 80% cell viability was recorded, indicating a favorable cell growth and attachment level. Experiments conducted on live albino rats showed an increase in the expression of TGF-β3 and a notable recovery of burnt wounds without scarring 158 . A separate research project involved the creation of a nanocomposite scaffold using chitosan and PVA. This scaffold was infused with photogenic iron oxide nanoparticles (FeO NPs) to investigate their effects on diabetic wounds associated with anemia 159 . The FeO NPs were synthesized using a leaf extract obtained from Pinus densiflora. The FeO NPs exhibited favorable anti-diabetic and antioxidant effects in biological experiments and antibacterial solid capabilities 160 . The in vitro wound healing experiment demonstrated enhanced cellular proliferation by HEK 293 cells. These data indicate that this composite scaffold could be used for treating diabetic wounds, pending a thorough in vivo assessment161,162. Table 3 summarizes various hybrid polymers utilized as scaffolds for applications in wound healing and skin tissue engineering 163 .
shows the Advantages and Disadvantages of Various Hybrid Scaffold Materials Used in Biomedical Applications.
Future Perspectives
The application of 3D printing in bone tissue engineering is now undergoing extensive research and exploration. The primary benefit of this technology is its capacity to regulate the arrangement of fibers, leading to scaffolds that exhibit superior performance due to their optimized structure and function at many scales 168 . Despite the advancements in 3D printing technology, certain constraints still impede the application of tissue engineering scaffolds in practical medical settings. For instance, the selection of printing materials is restricted 169 . It is essential to consider the physiochemical qualities of the materials, such as rheology (flow behavior), wetting performance, and melting point 170 .
In addition, their biological properties should also be considered, including biodegradability, biocompatibility, and cell interaction. Internal faults may arise during printing, leading to subpar mechanical quality in the printed product 171 . To utilize 3D printing technology effectively, it is crucial to thoroughly examine ink materials regarding material selection, design of 3D structures, and function novelty. Furthermore, the printing procedure is essential to optimize the associated parameters for high-quality outcomes172,173. Although several fabrication material concerns have not yet been resolved, there is still ample opportunity to investigate beneficial approaches for bone tissue engineering. The current research on repairing infected bones needs a thorough grasp of the repair mechanism in a complicated model of infected bone defects174,175. The currently described bone analogs possess only one specific function, and creating bone analogs capable of performing multiple tasks in an integrated manner remains a significant challenge 176 . Vascularization is essential for developing organs in massive bone tissues, as they require a well-developed network of blood vessels to provide nutrients and oxygen. Regrettably, the construction of vascular networks remains a significant obstacle due to the low vascularization of bone tissue 177 .
To efficiently address these glitches, it is vital to understand the inherent structural properties of bone tissue and the natural mechanisms involved in bone tissue healing and regeneration, including the impacts and interplay of many factors in infected bone defects 178 . The design of bone tissue engineering scaffolds should aim to replicate the bionic structure of natural bone tissue. These scaffolds can be enhanced by incorporating inflammatory cytokines, ECM, and ligands. This integration allows for imitating the initial phases of bone regeneration and tissue repair179–181.
Conclusion
Infected bone injuries and defects are still a core problem for orthopedic surgeons and practitioners. Engineering technologies and biological integration are critical elements in developing bone tissues. This review concisely discusses strategies for addressing bone defects through sustainable hybrid 3D-printed scaffold materials. A fundamental requirement for bone defects is the promotion of tissue regeneration and the stopping of bone infection. Many factors are responsible for this infection therapy, which include antibacterial coatings, bioactive metal ions, anti-infective drugs, and 3D-printed scaffolds that collectively fight against bone issues and treat osteomyelitis by producing the ideal microenvironment for rapid bone growth. Moreover, essential factors like photothermal, electric, and magnetic stimuli can be enhanced to promote bone regeneration during the loading of the 3D porous scaffolds. In tissue engineering, 3D-printed scaffold technology is currently the most reasonable solution for designing scaffolds according to infected patients’ complex shape injuries.
Footnotes
Authors Contribution
A.R.K. and N.S.G. wrote the manuscript and collected all the data. F M. O.T, F. T, and R. M.Z they also interpreted the manuscript’s results. H-J. Z. and Z. J. developed the theory and reviewed the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the Researchers Supporting Project Number (RSP2024R440), King Saud University, Riyadh, Saudi Arabia.
