Abstract
The objective of this study was to determine the incidence, risk factors, outcome, and clinical significance of pericardial effusion (PE). We retrospectively analyzed outcomes of 272 pediatric patients undergoing their first hematopoietic stem cell transplantation (HSCT) from 1998 to 2016. In total, 15% (3/20) and 5.9% (15/252) of autologous and allogeneic HSCT recipients, respectively, were identified with PE. However, there was no statistically significant difference in the incidence of PE between the 2 groups. The mean age at transplantation was 11.12 ± 5.41 y. Eighteen patients developed PE at 4.13 ± 4.44 mo after HSCT. PE was confirmed by echocardiogram in all patients. Three patients presented with severe PE with cardiac tamponade and required urgent pericardiocentesis. Overall survival (OS) rates for patients who developed PE were 83.3% and 38.9% at 100 d and 3 y, respectively, after HSCT. Death was not directly attributable to PE in patients who died in the first year after HSCT. Multivariable analysis identified the following variables to be associated with OS: PE (relative risk[RR]: 3.70; 95% confidence interval [95% CI]: 1.89-7.23; P < 0.001), active disease at HSCT (RR: 1.59; 95% CI: 1.02-2.49; P < 0.001), and thalassemia (RR: 0.62; 95% CI: 0.45-0.84; P < 0.001). PE is, thus, a debilitating and significant complication of pediatric HSCT. Therefore, prospective studies are required for better determination of the etiology and optimal method of PE treatment after HSCT.
Introduction
Although exact incidence and risk factors remain unclear, the incidence of pericardial effusion (PE) associated with hematopoietic stem cell transplantation (HSCT) has been reported to vary between 0.2% and 19%. 1 –3 Notably, PE after HSCT in patients with thalassemia is associated with the conditioning regimen and iron overload. 4 PE is also a well-described manifestation of polyserositis associated with chronic graft-versus-host disease (GVHD) that may not be associated with cardiac toxicity per se. 5 –8 This is also determined in studies with different experimental designs.
Materials and Methods
Patient Characteristics
We conducted a retrospective single-center study of 300 consecutive pediatric patients undergoing HSCT between April 1998 and December 2016. Consents were obtained at the time of transplantation for retrospective analysis from all patients undergoing HSCT. Patients were excluded from this analysis if they did not receive a preparative regimen (n = 1) or if they received more than 1 transplant (n = 27). In total, 272 pediatric patients undergoing HSCT, including 23 autologous transplant recipients, were identified. Information regarding the specifics of the conditioning regimen and GVHD prophylaxis is provided in Table 1.
Clinical Characteristics of Study Patients.a,b

Abbreviations: AD, active disease; ATG, antithymocyte globulin; BM, bone marrow; BU, busulfan; CBU, cord blood unit; CML, chronic myeloid leukemia; CP, chronic phase (for CML only); CR, complete remission; FA, Fanconi anemia; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplantation; MAC, myeloablative conditioning; MDS, myelodysplastic syndrome; MSD, matched sibling donor; MUD, matched unrelated donor; NMAC, nonmyeloablative conditioning; PBSC, peripheral blood stem cell; PE, pericardial effusion; RIC, reduced-intensity conditioning; SAA, severe aplastic anemia; SE, standard error; TBI, total body irradiation.
a N = 272.
b P value of less than 0.05 was considered statistically significant.
The study group comprised 272 pediatric patients (161 boys and 111 girls) with a median age of 7.5 y (range: 0.1-20.7 y) who underwent HSCT for hematooncological malignancies, nonmalignant hematological diseases, primary immunodeficiencies, and inborn errors of metabolism. The patients received transplants from human leukocyte antigen (HLA)-matched unrelated adult donors (n = 58), HLA-identical siblings (n = 73), unrelated cord blood donors (n = 119), or haploidentical family donors (n = 2) or they underwent autologous transplantation (n = 20). All autologous and allogeneic transplant recipients received standard prophylaxis including antimycotics, virostatics, and cotrimoxazole. GVHD prophylaxis was intravenously administered with cyclosporine A, antithymocyte globulin, methotrexate, or methylprednisolone, depending on the type of transplantation.
Data Collection
Medical records were used to collate data from follow-up visits, including physical examinations and assessments of disease status, growth, and organ function. Prefreeze parameters were provided by the cord blood bank procuring the unit for transplantation, and postthaw viability assessment was performed by the Laboratory of Chang Gung Memorial Hospital.
Majority of patients were diagnosed with PE as an incidental finding of cardiomegaly on computed tomography or chest X-ray performed for other reasons. None of our patients had clinical features highly reminiscent of heart failure. PE was confirmed by echocardiogram in all the patients. Two-dimensional transthoracic echocardiography studies were reviewed and interpreted by pediatric cardiologists. PE was diagnosed if posttransplant echocardiography showed new or increased presence of a clear space between visceral and parietal pericardia, reflecting fluid accumulation. 9 None of the patients had a history of PE before their first transplant. Factors analyzed to determine the risk of PE development included age, gender, underlying disease, number and type of transplants, donor type, conditioning regimens, and disease status at HSCT.
Participants
This analysis was conducted in accordance with the Declaration of Helsinki and under the waiver for retrospective anonymized studies in accordance with the independent ethics committee of Chang Gung Memorial Hospital. Written informed consent was obtained from parents of participants or their legal representatives.
End Points and Statistical Analysis
Overall survival (OS) was defined as the length of time from HSCT to death from any cause. OS percentages and standard errors were calculated using the Kaplan–Meier method, and log-rank tests were used for group comparisons. A Cox proportional hazards regression model was used for investigating risk factors that were associated with survival. The properties of the 2 groups (PE and no-PE) were compared using the chi-square tests for categorical variables and the Mann–Whitney U test for continuous variables. The following variables were evaluated in these analyses: age, recipient gender, diagnosis of underlying disease, graft type, and disease status at transplantation. The primary end point of the study was the incidence of PE. The second statistically significant end point was the difference in OS. Crosstabs and Student’s t test were used to identify the baseline characteristics associated with this end point. Factors with P value <0.1 in univariate analysis were used in a multivariate proportional hazards Cox regression analysis. P values < 0.05 were considered statistically significant, and the hazard ratios and their 95% confidence intervals (95% CIs) were calculated. Survival end points were calculated from the date of infusion to the date of death or last follow-up. Data were analyzed with SPSS software version 20.0 for Windows (SPSS Inc., Chicago, IL).
Results
Most patients underwent HSCT for malignant disease (54%), and more than half of the patients with malignant diagnoses underwent transplantation for leukemia (43%). PE was identified in 15% (3/20) of autologous HSCT recipients and in 5.9% (15/252) of allogeneic HSCT. However, there was no statistically significant difference in the incidence of PE between autologous and allogeneic HSCT groups. Of the 18 patients with PE, 13 (72%) were males; the median age at transplantation was 12.5 y (1.3-17.5 y) and most of them were asymptomatic, with PE incidentally found on radiographs. Nine patients died (50%) during follow-up (Table 2), but none died as a direct result of PE. Majority of patients (83%) with PE were managed conservatively. Three patients (17%) with hemodynamically unstable pericardial tamponade underwent echo-guided pericardiocentesis as the immediate treatment strategy. Hemorrhagic PE was observed in 2 patients with β-thalassemia major, and serous PE was drained in 1 patient with leukemia. One of the three patients with pericardiocentesis survived to the conclusion of the follow-up. OS rates for patients who developed PE were 83.3% and 38.9% at 100 d and 3 y, respectively, after HSCT. Notably, the presence of PE had a profound negative impact on OS (P < 0.001; Fig. 1).
Clinical Characteristics of Patients with Pericardial Effusion.

Abbreviations: ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ARDS, acute respiratory distress syndrome; AWD, alive with disease; BCNU, bis-chloroethylnitrosourea; Bu, busulfan; Cy, cyclophosphamide; F, female; Flu, fludarabine; Haplo, haploidentical transplant; HD, Hodgkin disease; HSCT, hematopoietic stem cell transplantation; M, male; MAC, myeloablative conditioning; Mel, melphalan; MSD, matched sibling donor; MUD, matched unrelated donor; NB, neuroblastoma; NED, no evidence of disease; PE, pericardial effusion; RIC, reduced-intensity conditioning; TBI, total body irradiation; Topo, topotecan; UCB, unrelated donor cord blood; VP, VP-16.
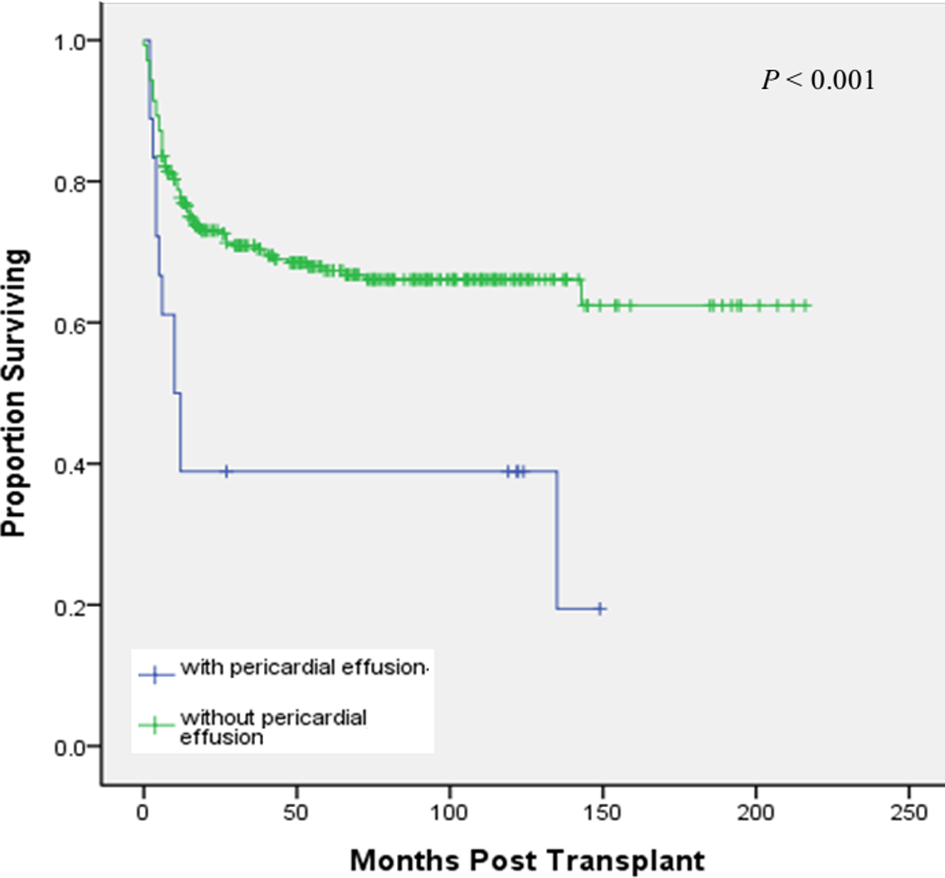
Probability of overall survival rates for patients who developed pericardial effusion.
As per the univariable analysis, PE and underlying diseases other than thalassemia were associated with OS (Table 3). Multivariable analysis identified the following variables associated with OS: PE (RR: 3.70; 95% CI: 1.89-7.23; P < 0.001), active disease at HSCT (RR: 1.59; 95% CI: 1.02-2.49; P < 0.001), and thalassemia (RR: 0.62; 95% CI: 0.45-0.84; P < 0.001; Table 4).
Univariate Analysis of Categorical Variables and Overall Survival.a
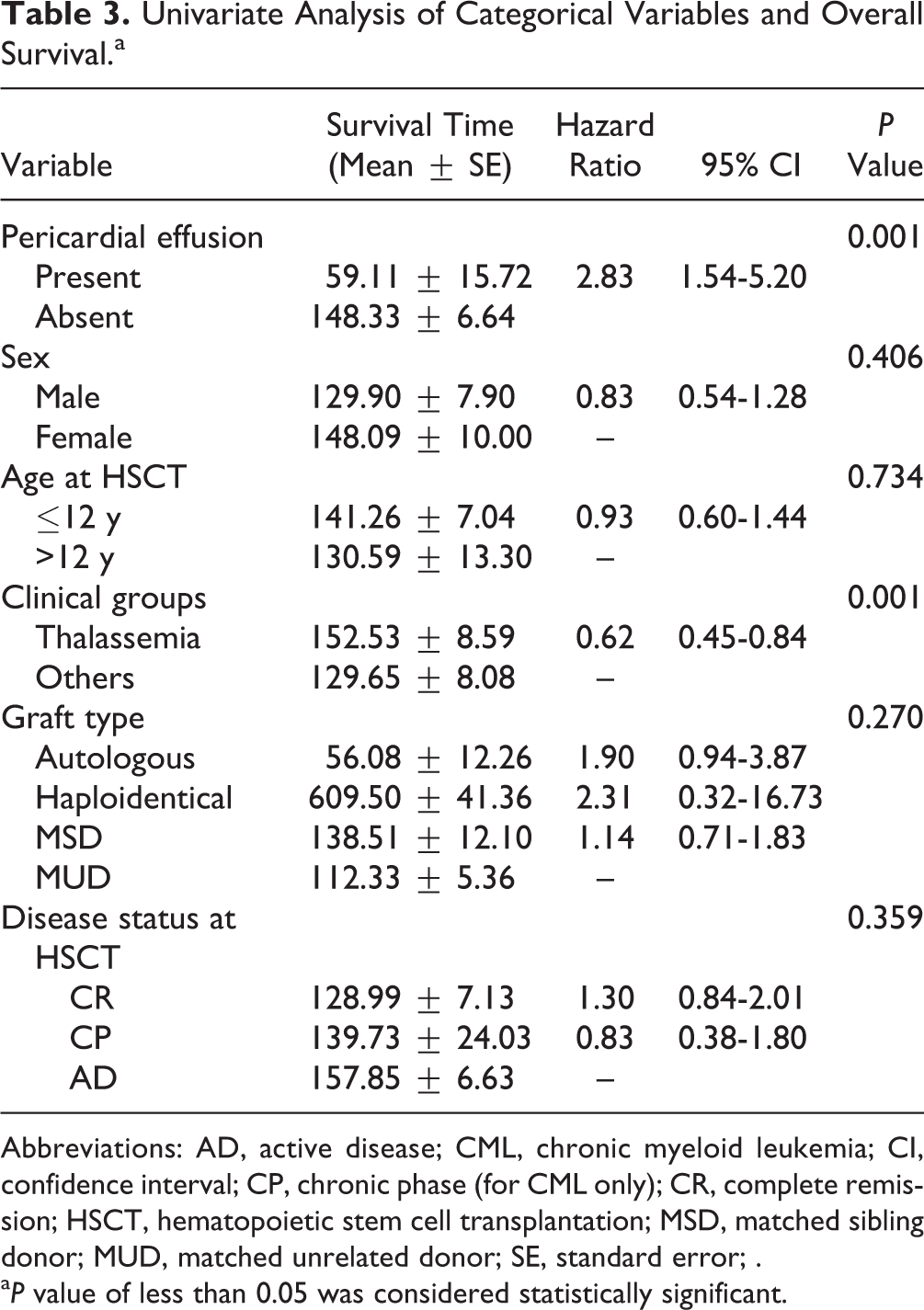
Abbreviations: AD, active disease; CML, chronic myeloid leukemia; CI, confidence interval; CP, chronic phase (for CML only); CR, complete remission; HSCT, hematopoietic stem cell transplantation; MSD, matched sibling donor; MUD, matched unrelated donor; SE, standard error; .
a P value of less than 0.05 was considered statistically significant.
Multivariate Cox Regression Analysis of Overall Survival.

Abbreviations: AD, active disease; CI, confidence interval; CP, chronic phase (for CML only); CR, complete remission; HSCT, hematopoietic stem cell transplantation; MSD, matched sibling donor; MUD, matched unrelated donor.
a P value of less than 0.05 was considered statistically significant.
Discussion
PE is a rare and potentially life-threatening complication observed in HSCT recipients. Because milder cases are clinically silent, risk factors, etiology, incidence, and treatment remain unclear. 10 Age, gender, disease risk, conditioning regimen, neutrophil engraftment, relapse, GVHD, GVHD prophylaxis, donor type, and CMV viremias have been suggested to be potential risk factors. 1 PE is often associated with known or unknown (e.g., hypothyroidism) medical condition (up to 60% of cases). 11 –13 PE after HSCT in patients with thalassemia is associated with both the conditioning regimen and iron overload. 14 Neier et al. 1 showed increased age, high-risk patients, and ablative conditioning regimens as risk factors for PE development. Other suggested etiologies of PE include infection, hemolytic uremic syndrome, and relapsed disease. 15
Ten of the 18 patients with PE underwent unrelated donor HSCT. Thirteen cases of PE were observed less than 100 d after HSCT, which was considered as the period for acute GVHD. PE appears to be more common in patients with thalassemia; however, the difference was not statistically significant. Comorbidities have no impact on transplant success but do have a negative effect on OS, indicating that survival of patients with thalassemia is determined more by comorbidities than by thalassemia per se. Iron-induced pericardial siderosis could play an important role in the development of PE; however, we were unable to investigate this issue because of the limitations of the current noninvasive techniques.
On detecting PE, the first step is to assess its size, hemodynamic importance, and possible associated diseases. The relative frequency of different causes depends on the local epidemiology, hospital setting, and diagnostic protocol that had been adopted. We acknowledge that our study has certain methodological limitations because we identified a subgroup of patients at risk of PE development. The time span of the transplantation was 18 y. However, over 80% of patients underwent transplantation within the last decade. Furthermore, some prognostic factors at diagnosis are missing, and almost a quarter of patients were diagnosed and underwent initial treatments at other institutions. PE following HSCT predicts the subsequent development of cardiac tamponade and represents a risk factor for increased morbidity and mortality. Strategies to identify PE early in patients undergoing HSCT should be implemented and could positively influence patient outcome. 16 –18
However, we were unable to analyze some potentially important variables such as the CMV serological state and killer immunoglobulin-like receptor disparity. Collectively, we acknowledge that this study is limited because of its retrospective nature and that there were no standardized time points for performing routine echocardiograms. There may have, therefore, been a substantial selection bias based on the decisions of physicians to request echocardiography.
Because of the small size of the study population, relationships between underlying diseases and preconditioning regimen could not be fully investigated. Through our observations, we would like to raise awareness of a potential association between high-risk thalassemia recipients and clinically significant PE that can contribute to morbidity and prolonged hospital stay.
Footnotes
Author Contributions
Y.-C. Wen, T.-Y. Chang, D.-Y. Tsai, and P.-K. Tsay performed the research and analyzed data. T.-H. Jaing and S.-H. Chen participated in the diagnosis, management, and recruitment of patients. T.-H. Jaing and H.-T. Chung designed the research, obtained funding, analyzed and interpreted the data, and wrote the paper. All authors read and approved the final version of the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
Human subjects were treated in accordance with the Institutional Review Board (IRB) guidelines.
Statement of Informed Consent
Consent was obtained at the time of transplantation for retrospective analysis from all patients undergoing HSCT.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work in the author’s laboratory was supported by grant from Ministry of Health and Welfare (KMRPB3F033).
