Abstract
A fresh osteochondral allograft is one of the most effective treatments for cartilage defects of the knee. Despite the clinical success, fresh osteochondral allografts have great limitations in relation to the short storage time that cartilage tissues can be well-preserved. Fresh osteochondral grafts are generally stored in culture medium at 4°C. While the viability of articular cartilage stored in culture medium is significantly diminished within 1 week, appropriate serology testing to minimize the chances for the disease transmission requires a minimum of 2 weeks. (–)-Epigallocatechin-3-O-gallate (EGCG) has differential effects on the proliferation of cancer and normal cells, thus a cytotoxic effect on various cancer cells, but a cytopreservative effect on normal cells. Therefore, a storage solution containing EGCG might extend the storage duration of articular cartilages. Rabbit osteochondral allografts were performed with osteochondral grafts stored at 4°C in culture medium containing EGCG for 2 weeks and then the clinical effects were examined with macroscopic and histological assessment after 4 weeks. The cartilaginous structure of an osteochondral graft stored with EGCG was well-preserved with high cell viability and glycosaminoglycan (GAG) content of the extracellular matrix (ECM). After an osteochondral allograft, the implanted osteochondral grafts stored with EGCG also provided a significantly better retention of the articular cartilage with viability and metabolic activity. These data suggest that EGCG can be an effective storage agent that allows long-term preservation of articular cartilage under cold storage conditions.
Introduction
Articular cartilage injury occurs frequently, but it cannot be self-regenerated due to its limited intrinsic healing capacity (9,13). The limited ability of articular cartilage to repair after injury has led to the investigation of new therapeutic methods to enhance osteochondral regeneration (27,30,32,33). Recently, orthopedic surgeries using fresh osteochondral allografts have become popular for the treatment of articular cartilage lesions. Numerous studies demonstrate good outcomes of fresh osteochondral allografts (1,5,11). Despite the clinical success, fresh osteochondral allografts have great limitations in relation to the fact that cartilage properties are not retained with extended storage. The viability of cartilage decreases significantly as time passes after harvesting (21). Articular cartilage was initially stored by freezing, but many studies found that freezing intact articular cartilage, even in the presence of cytopreservative agents or with controlled-rate freezing techniques, resulted in either complete or near complete loss of chondrocyte viability (23,25). Therefore, articular cartilage has been traditionally stored in lactate Ringer's solution at 4°C and culture media has been used as a storage solution recently because many studies reported that culture media provides significantly better preservation of the cartilage with viability and metabolic activity (2, 35). However, extensive studies have reported that articular cartilage stored in culture medium generally tended to lose its properties within 7–14 days (4,29).
Green tea polyphenols are phytochemicals with various bioactivities (20,31,36). (–)-Epigallocatechin-3-O-gallate (EGCG), the major polyphenolic constituent of green tea, has anticancer, antimutagenic, and antimicrobial effects. Apart from these biological activities, EGCG has beneficial preservative effects on mammalian cells and tissues that were examined in order to design a cell-or tissue-preserving solution at physiological temperature (17,24,37). Human articular cartilages stored in culture medium containing EGCG were well-preserved with high biochemical and biomechanical properties in in vitro studies. To investigate the clinical effects of EGCG storage on articular cartilage, rabbit osteochondral allografts were performed using articular cartilage stored in a culture medium containing EGCG for 2 weeks and the quality of implanted articular cartilage was examined after 4 weeks.
Materials and Methods
Animal and Surgery
All procedures received prior approval from Osaka City University Graduate School of Medicine and the Institutional Review Board of Institute for Frontier Medical Sciences, Kyoto University and all subjects provided their written informed consent.
Eighteen mature male Japanese white rabbits (Shimizu Laboratory Supplies Co., Ltd., Kyoto, Japan), weighing an average of 3 kg, were used in this study. The osteochondral plugs, which were 5 mm in diameter and consisted of articular cartilage with a 10-mm-thick portion of underlying subchondral bone, were harvested from the knee under sterile conditions. Continuous irrigation was used to minimize thermal damage to the cartilage and bone during the harvesting of the plugs. The plugs were rinsed three times with phosphate-buffered saline (PBS, pH 7.4) to remove the marrow elements. Each plug was then immediately placed into an individual sterile 50-ml culture tube containing 20 ml storage solution [serum-free RPMI-1640 medium (Sigma-Aldrich Co., St. Louis, MO) with 1% antibiotic-antimycotic solution (including 10,000 units penicillin, 10 mg streptomycin, and 25 μg amphotericin B/ml, Sigma-Aldrich Co.)] without or with 1 mM EGCG (TEAVIGO™, DSM Nutritional Products Ltd., Basel, Switzerland) and kept at 4°C for 2 weeks without changing the storage solution. The stored cartilage grafts were transplanted into another rabbit knee with the same size defects with cartilage plugs (Fig. 1B). The negative controls were not transplanted after harvesting of the plugs while the positive controls were transplanted with fresh cartilage plugs without preservation. Six individual knees were used for each experimental group. After surgery, the rabbits were allowed to move freely within their cages for 4 weeks. After 4 weeks, the rabbits were sacrificed and their entire knees were dissected and photographed. Each knee was decalcified and then sectioned sagittally for histological assessment.

Experimental scheme of this study. Osteochondral plugs were procured from the rabbit knee, transferred to storage solution without or with EGCG, and kept at 4°C. For the in vitro study, harvested osteochondral plugs were stored for various lengths of time and different concentrations of EGCG and then a histological analysis was performed (A). Osteochondral plugs for osteochondral allografts were stored with either 20 ml of a storage solution without or with EGCG at 4°C for 2 weeks. At the end of each storage period, the osteochondral grafts were transplanted into the knee defects formed by the removal of osteochondral plugs. After 4 weeks, the entire knees were dissected and then a macroscopic and histological assessment was performed as described in Materials and Methods (B).
Cartilage Specimen Collection and Cold Preservation
Before the osteochondral allografts, rabbit osteochondral plugs were preserved under various storage conditions of time and dosage to determine the most effective storage condition for surgery (Fig. 1A). The plugs were harvested by the same methods described above. On the receipt of the osteochondral plugs, the specimens were placed into either 20 ml of a storage solution with 0, 0.1, 1, 10 mM EGCG and then stored at 4°C for 1, 2, and 4 weeks without changing the storage solution. At the end of each storage period, the specimens were rinsed three times with PBS solution and then a histochemical analysis was performed as described below.
Histological Analysis
Each osteochondral specimen was fixed with 2% glutaraldehyde, 2% paraformaldehyde, and 0.2% CaCl2, followed by embedding in paraffin. The tissue blocks were cut into 5-μm sections and stained with either Safranin-O/fast green or toluidine blue for evaluating GAG content and distribution. The prepared sections were examined using an optical/fluorescence microscope (Biozero-8000, Keyence, Osaka, Japan).
Results
Histological Analysis of Osteochondral Graft
Before the osteochondral allograft, a histological examination with Safranin-O staining was done on specimens of the osteochondral grafts preserved over various storage times and EGCG concentrations. The general pattern of Safranin-O staining results was that the specimens stored with EGCG stained positively with Safranin-O, showing the abundant presence of GAG, which was comparable to that of fresh specimens (Fig. 2). In contrast, preservation without EGCG resulted in an appreciable decrease in the positivity for Safranin-O. With storage in culture medium without EGCG, evidence of severe deterioration was seen in the cartilaginous structure within 1 week; many empty lacunae were present and cells had pyknotic nuclei and a loss of cytoplasm. The peripheral region of each cartilage had a higher cell density and stained more intensely for GAG in comparison to the central region. The cells in the lacunas of fresh and EGCG-preserved cartilages were well-maintained with the original chondrocytic phenotype in comparison to those preserved without EGCG. In the various storage conditions, the osteochondral plugs stored with 1 mM EGCG were appreciably well-maintained with a high cell density and normal GAG content for up to 2 weeks. Therefore, the storage condition of the osteochondral grafts used for osteochondral allograft was determined to be stored with 1 mM EGCG for 2 weeks. The results with human cartilages performed with same methods in a previous study was also that cartilage tissues stored with 1 mM EGCG were effectively well-maintained for up to 2 weeks.
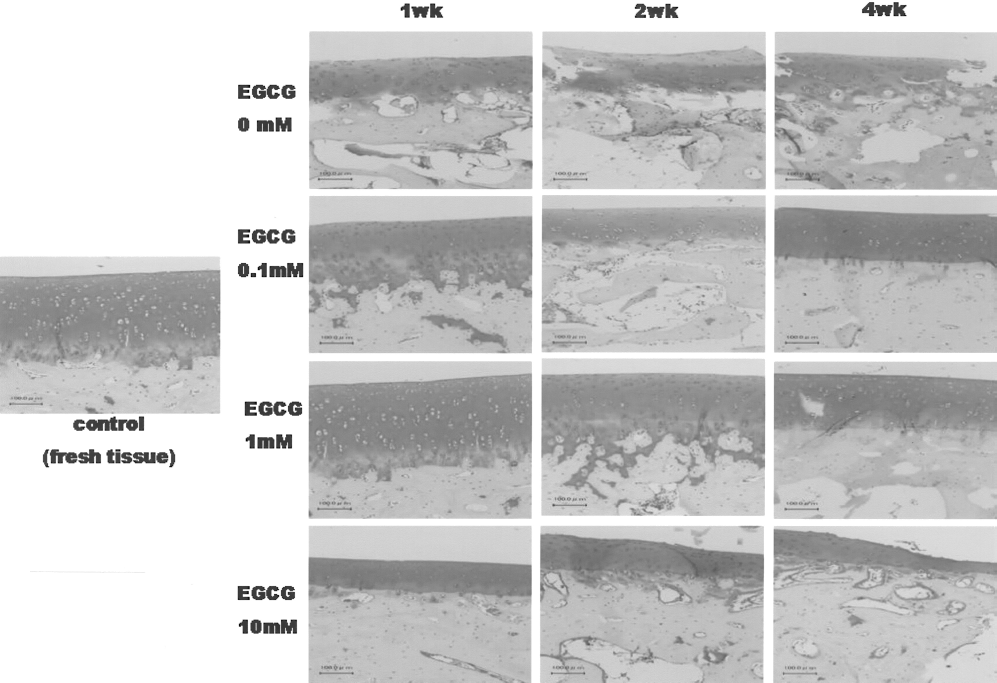
A histological analysis of rabbit articular cartilages cold preserved for various lengths of time and in different concentrations of EGCG. Specimens of osteochondral grafts were stained with Safranin-O/fast green for the evaluation of GAG content and distribution (original magnification, 100×). The control represents fresh tissue without any preservation. The photographs shown in this figure are representative of four independent experiments, showing similar results.
Macroscopic Observation
Four weeks after surgery, the subchondral bone region of the defect in the negative controls little healed centripetally, starting at the lateral borders of the defects (Fig. 3). However, the cartilage tissue was not seen yet. The defect area was clearly distinguishable from the surrounding cartilage. The level of the repair tissue surface was markedly lower than the surrounding joint cartilage and the rough surface showed a slightly tan color. The margins of the defects in the other groups were slightly recognizable, but it was generally difficult to clearly discern the boundary between the host and the transplanted osteochondral graft. In the case of storage with EGCG, the transplanted site was more distinguishable because the color of osteochondral grafts stored with EGCG changed to dark brown after storage for 2 weeks. There was no visible difference between the positive control and storage group without EGCG. There were no inflammatory symptoms in any of the experimental groups.

Macroscopic appearance of defects after 4 weeks from osteochondral allografts with different treatments. Osteochondral allografts were performed with grafts stored in culture media for 2 weeks without (C) or with EGCG (D). In addition, the control defects that were left empty (A) and those transplanted with a fresh graft (B) are also shown.
Histological Assessment of Fresh Osteochondral Allograft
A histological examination of the Safranin-O and toluidine blue-stained specimens of the sagittally sectioned knees was done after 4 weeks. The negative control that had a defect without any treatment after harvesting the osteochondral plugs was not level with the surrounding cartilage. The specimens stored with EGCG stained positively with Safranin-O and toluidine blue, showing the abundant presence of GAG, which was comparable to that of the positive control engrafted with a fresh osteochondral plug (Figs. 4 and 5). In contrast, preservation without EGCG resulted in an appreciable decrease in the positive staining for Safranin-O and toluidine blue. The cartilage tissues of the osteochondral grafts in the positive controls and storage with EGCG had higher cell density and stained more intensely for GAG in comparison to that in negative controls and storage without EGCG. The cells in the lacunas of EGCG-preserved cartilages and the positive controls were well-maintained with an original chondrocytic phenotype in comparison to those preserved without EGCG and the negative controls. The results of the Safranin-O and toluidine blue staining had the almost same results as expected.

Histological assessment of ECM structures in osteochondral defects. Osteochondral allografts were performed with grafts preserved without (A) or with (B) EGCG for 2 weeks and fresh grafts without any preservation (D). The negative control (C) was a defect without any treatment. Sagittal sections of the osteochondral defects 4 weeks after the osteochondral allografts were stained with Safranin-O/fast green for the evaluation of GAG content and distribution (original magnification, 20×). The photographs shown in this figure are representative of six independent experiments, showing similar results.

Histological assessment of ECM structures in osteochondral defects. Osteochondral allografts were performed with grafts preserved without (A) or with (B) EGCG for 2 weeks and fresh grafts without any preservation (D). The negative control (C) represents a defect without any treatment. Sagittal sections of osteochondral defects 4 weeks after the osteochondral allografts were stained with toluidine blue for the evaluation of GAG content and distribution (original magnification, 20×). The photographs shown in this figure are representative of six independent experiments, and they showed similar results.
Discussion
This study showed that articular cartilage stored with EGCG solution maintained its ECM properties and the beneficial storage effect of EGCG was also observed on the clinical level. The capacity of articular cartilage for repair is limited. Injuries of the articular cartilage that do not penetrate the subchondral bone do not heal and usually progress to the degeneration of the articular surface. A short-lived tissue response has been observed, but it fails to provide sufficient cells and matrix to repair even small defects (22). This limited ability of articular cartilage to repair after injury has led to investigation of new therapeutic methods to enhance osteochondral regeneration and one of them is a fresh osteochondral allograft. While the interest in fresh osteochondral allografts has increased remarkably because of good clinical outcomes (12,34), there are significant practical limitations because the surgical implantation must be performed within 1 week of the graft harvest to maximize its viability (26). However, the appropriate serology testing required to minimize the chances for disease transmission requires a minimum of 2 weeks. Prolongation of the storage period has been of long-standing interest in the storage of articular cartilage grafts. Lactate Ringer's solution had been traditionally used for the storage of fresh osteochondral allografts; however, this storage media lacks the essential nutrients for sustained metabolic function of the chondrocytes (3,7). Lactate Ringer's solution is elemental in constituency, with physiological concentrations of sodium, potassium, chloride, and bicarbonate, but it contains no nutrients to support the chondrocyte metabolism. A culture medium preserves better chondrocyte viability and metabolic activity and it is therefore preferred over Lactate Ringer's solution (3,19). In an effort to provide nutrients and prolong chondrocyte viability, the storage solution has been changed to a standard culture medium containing amino acids, glucose, and inorganic salts. In most tissue banks, the tissue is now stored in culture medium prior to transplantation.
EGCG has been reported to have different effects on the proliferation of cancer and normal cells. It has a cytotoxic effect on various cancer cells, but a hibernation effect on normal cells (8). Therefore, the cytopreservative effects were previously examined on mammalian cells and tissues, including human articular cartilages, to design a cell- or tissue-preserving medium at physiological temperature (14–18). In this study, fresh osteochondral allografts were performed using osteochondral grafts preserved in culture medium with or without EGCG to investigate the clinical effects of each preservation method.
Storage of osteochondral grafts with EGCG enabled cartilaginous structures to be well-preserved and delayed the denaturation of the extracellular matrices (Fig. 2). With storage in culture medium without EGCG, there was a significant decline in the viability of the cartilaginous structure within 1 week; increased disorganization, hypocellularity, pyknotic nuclei, and empty lacunae were seen in the histological specimens. The most effective storage concentration of EGCG was 1 mM. The cartilaginous structure of osteochondral grafts stored with 1 mM EGCG was well-maintained for up to 2 weeks, comparable to that of fresh grafts. Osteochondral grafts were stored without media change in this study because media change introduces a potential breach of sterility and previous studies demonstrated that it offered no benefit in comparison to not changing the storage solution (10). Macroscopic observation showed that the cartilage tissues were not seen in the negative control group until 4 weeks after the osteochondral allografts; however, the osteochondral defects of the other groups were almost fully healed (Fig. 3). In the case of storage with EGCG, the color of osteochondral graft was changed to dark brown because of the EGCG, but this is apparently not serious because the operated region of osteochondral allograft was not seen on the exterior. An immune response is a serious concern with transplantation. In this study, there were no significant inflammatory symptoms observed macroscopically in any of the groups because it seems that there were few capillaries in the osteochondral grafts. The histology from the 4-week study demonstrated that the storage of an osteochondral graft with EGCG was also effective on the clinical treatment for osteochondral defects (Figs. 4 and 5). The implanted osteochondral grafts maintained their cartilaginous structures, with high cell viability and GAG content of ECM. In this 4-week follow-up, the cartilaginous structure of an osteochondral graft stored with EGCG was seen to be well-maintained rather than that of positive control because there appeared to be a histoincompatibile rejection of the chondrocytes between the host and the graft within the positive controls. In addition, cold storage and EGCG have been reported to effectively decrease the inflammatory response (6,28).
The results of this study showed that the storage of osteochondral grafts in culture medium containing EGCG provides enhanced clinical treatment for osteochondral defects in comparison to storage in culture medium alone. The application of EGCG for the storage of osteochondral grafts may be useful for fresh osteochondral allografts. This finding has important clinical implications because it suggests that the storage of osteochondral grafts has considerable relevance to the treatment of defects in human cartilage and can provide the basis for the development of repair technology on articular cartilages. Further research with a greater long-term follow-up and human clinical study will be necessary to establish the optimal orthopedic surgery.
