Abstract
Initiatives that collect and share genomic data to advance health research are widespread and accelerating. Commercial interests in these efforts, while vital, may erode public trust and willingness to provide personal genomic data, upon which these initiatives depend. Understanding public attitudes towards providing genomic data for health research in the context of commercial involvement is critical. A PRISMA-guided search of six online academic databases identified 113 quantitative and qualitative studies using primary data pertaining to public attitudes towards commercial actors in the management, collection, access, and use of biobank and genomic data. The presence of commercial interests yields interrelated public concerns around consent, privacy and data security, trust in science and scientists, benefit sharing, and the ownership and control of health data. Carefully considered regulatory and data governance and access policies are therefore required to maintain public trust and support for genomic health initiatives.
Advances in gene sequencing, bioinformatics and big data technologies, accompanied by falling costs, have spurred ever-expanding collections of human genomic data serving to advance the promise of personalised medicine and hastened medical breakthroughs (Hulsen et al., 2019; Scott et al., 2012). Research into the genomic aetiology of disease relies on large-scale standardised heterogeneous datasets. These provide sufficient statistical power to reduce the likelihood of detecting genetic associations that are spurious and non-reproducible (Hasin et al., 2017). Starting at the turn of the century, one type of genomic data repository, the biobank, emerged as the dominant research infrastructure in advancing the genomics research agenda. Biobanks served as gateways between researcher and biospecimen donor. Crucial to unravelling the biology of complex disease, biobanks provided researchers with access to curated collections of high-quality annotated genomic data and biospecimens enhanced with relevant clinical, health and lifestyle information.
Much of the literature on the ethical, legal and social implications associated with the collection, storage, use and sharing of genomic data for health research has focused specifically on biobanks. This literature highlights that trust in data being managed in accordance with public expectations underlies public willingness to donate to and to consent for personal genomic data being incorporated into biobanks (Critchley and Nicol, 2017). Specifically, this requires that the rights, interests and confidentiality of donors are protected. In situations where donor knowledge or understanding is lacking or incomplete, for instance, when faced with the decision to consent to involvement in advanced medical research, trust functions as a heuristic that allows individuals to simplify complex decisions, especially where risk may be difficult to quantify (Critchley and Nicol, 2011; Cummings, 2014). Indeed, research has consistently identified the importance of trust in the public support of biobanks, willingness to donate biospecimens and comfort with sharing personal genetic data (Bearth and Siegrist, 2020; Critchley et al., 2015; Dive et al., 2020; Kettis-Lindblad et al., 2005; Milne et al., 2019).
The operational landscape of modern biobanking is in flux and the ramifications for public trust may be significant. Notably, biobanks are increasingly forging commercial relationships and collaborating in domestic and global sharing networks to enhance competitiveness, long-term sustainability and accelerate research outputs. Moreover, as gene sequencing technology becomes more efficient and data storage capacity increases, there is reduced need to maintain expensive centralised tissue repositories, raising questions as to whether biobanks are necessary. Data, rather than tissue, are the valuable commodity (Chalmers et al., 2016).
While a sizable body of research paints a complex picture of public attitudes towards biobanking, the majority has been conducted where biobanks have stored tissue as well as data, and where biobanks have operated as independent entities and in academic or not-for-profit contexts. Empirical research focused on exploring the extent of public concerns around commercial activities or relationships forged by genomic data repositories established for health research is scarcer. Discerning which elements of commercialisation are of concern to the public within the context of collection, storage, use and sharing of genomic data for health research is vital in crafting organisational and regulatory frameworks that can attempt to allay these concerns and help maintain public participation and support. The aim of this review then is to clarify what is already known and to further understand the effects of commercial involvement on public attitudes, trust and participation rates in the collection, use and sharing of genomic data by biobanks and genomic data repositories for health research.
1. Background
Economic sustainability remains an ongoing challenge for many biobanking initiatives (Chalmers et al., 2016). As an enterprise, biobanks are costly, requiring staff with significant training and expertise, and specialised equipment, infrastructure and consumables for sample collection, processing and storage (Coppola et al., 2019). Biobanks are commonly operated under the auspices of hospitals or medical research or academic institutions where funding has historically been provided by government (Chalmers et al., 2016). Initial investment focus has frequently been towards seed funding rather than commitment to the significant long-term operational costs which has seen public funding commitments to many biobank initiatives diminish over time (Albert et al., 2014). This has left public biobank funding vulnerable to policy shifts, reprioritisation and a progressively competitive research funding environment.
Due to these pressures it is broadly accepted that the long-term viability of biobanks, including those that receive public funding, will be reliant upon partnerships with industry and other commercial activities to increase financial security, defray operational costs and provide the capital necessary to bring novel therapeutic products to market (Caulfield et al., 2014a; Nicol et al., 2016). Efforts to commercialise publicly funded research has surged in recent decades, driven by government policies in market-oriented economies that frame commercialisation as both a social good and an economic imperative (Caulfield and Ogbogu, 2015). The benefits of commercialisation, it is argued, include increased efficiencies in the translation of knowledge to products, the ability to fund more expensive product development than can be afforded by publicly funded organisations and, more broadly, generation of wealth and concomitant economic stimulus through the advancement of the biotechnology sector (Caulfield et al., 2012). Yet the involvement of commercial actors in public sector biobanking adds another layer to the already complex ethical, legal and social challenges inherent in their operation. Careful navigation is required to ensure that the industry fairly delivers for the public good.
Commercialisation in public sector biobanking covers a broad range of activities. Commercialisation may refer to an end-user paying for access to biospecimens, data, or the knowledge or products derived from biobank resources; an operating partner providing funding; or the formation of ongoing public–private partnerships (Caulfield et al., 2014b). Different commercialisation activities are likely to affect public attitudes and trust in different ways. One particular concern is that the inclusion of a commercial partner creates tension between medical research broadly serving the public good and any discoveries being paywalled to protect and advance private interests (Nicol and Critchley, 2012). Another is the risk that commercial incentives may lead to perceived or actual conflicts of interest, threatening research independence and scientific integrity. Furthermore, the presence of commercial interests can shift the participation decision dynamic, undermining trust by making more salient donor concerns around consent, control, ownership, exploitation, privacy and personal risks (Caulfield et al., 2014b).
Empirical research into the impacts of commercialisation on public trust in biobanking and the sharing of genomic data has consistently identified what Critchley et al. (2021) term a ‘commercialisation effect’ where reported trust and an individual’s willingness to share their data with private entities is significantly lower relative to public entities (e.g. Critchley et al., 2015; Goodman et al., 2017; Nicol et al., 2016; Vidgen et al., 2020). Accepting that the future of public biobanks and genomic data repositories will inevitably involve commercialisation, either through fees for access or through formal public–private partnerships or industry research funding, it is increasingly important to understand what the empirical literature says about the extent of commercialisation effects on public trust, and to investigate what research may exist on how public trust and support may be maintained.
In recent years several related literature reviews have been published with a focus on attitudes towards biobanking (Domaradzki and Pawlikowski, 2019), the sharing of health data for research (Kalkman et al., 2019), privacy and genetic information (Clayton et al., 2018), and genomic data sharing (Shabani et al., 2014) but none have focused specifically on the commercialisation effect on public attitudes in relation to biobanks and genomic data. Maintaining public trust in the presence of commercialisation is therefore an important but arguably under-studied aspect of biobank governance.
2. Method
Inclusion criteria
A PRISMA (Moher et al., 2009) guided search strategy was formulated to encompass original research in both the academic and grey literature. The prespecified criteria for inclusion in the review was that the research:
(a) was original and featured a collection of primary data from the public, patients or other laypeople but excluded experts and other professional stakeholders. Reviews and meta-analyses were excluded;
(b) related to attitudes and opinions towards the sharing or accessing of genomic, genetic or biobank data with commercial entities or the involvement of commercial entities in the collection, generation or management of this data;
(c) was focused on data sharing for public health research rather than diagnostic clinical purposes;
(d) included all research methodologies (e.g. focus groups, deliberative events, interviews, surveys);
(e) was available in English.
Search strategy
The systematic review was undertaken in October 2020 using the PubMed, Web of Science, EBSCO and Scopus academic databases. The results were bolstered by supplementary searches of Google Scholar and the CORE database, an index of open access research from academic and institutional repositories, and journals that include grey literature. As most research into public attitudes towards biobanking and genomic data sharing has occurred within the past two decades, no restrictions on publication year were imposed. It was decided that a deliberately broad search strategy would be employed despite inflating the number of false positives returned. For the academic databases the following terms were used in a title and abstract search:
i. (‘care giver*’ OR participant* OR caregiver OR carer OR citizen OR clinician OR community OR consumer OR donor OR layp* OR parent OR patient OR populat* OR public OR stakeholder)
ii. AND
[‘perceived benefit*’ OR ‘perceived motivation*’ OR ‘perceived risk*’ OR ‘risk perception’ OR accept* OR attitud* OR attribution OR behav* OR belief OR comfort* OR consent OR donation OR expectation OR goal OR intention OR knowledge OR norm OR opinion OR oppos* OR participation OR perception OR preference OR reaction* OR resist* OR stereotype OR support* OR trust* OR understanding OR value OR view OR willing*]
iii. AND
[‘direct to consumer’ OR ‘for profit’ OR ‘private public’ OR ‘public private’ OR ‘state owned’ OR biotech* OR commercial* OR company OR consumerization OR denational* OR dtc OR entrepren* OR government OR industry OR national OR pharmaceut* OR privatisation OR translation]
iv. AND
[‘big data’ OR ‘health data’ OR ‘health information’ OR ‘human tissue’ OR ‘medical data’ OR ‘medical records’ OR ‘research result’ OR ‘stem cell*’ OR dna OR embryo OR gene OR genetic OR genom*]
v. AND
[‘human research’ OR ‘open science’ OR ‘scientific research’ OR ‘use of * resources’ OR access OR diagnosis OR fund* OR share OR sharing]
vi. AND
[‘gene* bank’ OR ‘tissue bank’ OR biobank OR governance OR regulation OR repository OR researcher OR scientist OR university]
vii. AND
[‘case study’ OR ‘control condition*’ OR ‘control group*’ OR ‘control trial*’ OR ‘focus group*’ OR ‘mixed method*’ OR experiment* OR interview OR questionnaire OR rct OR survey OR poll]
This returned 1433 records from PubMed, 4369 records from Web of Science, 11,134 records from EBSCO and 3473 records from Scopus. For both Google Scholar and CORE, the first 500 records as returned by their relevance algorithms were included. After de-duplication, a total of 14,325 records were screened. The majority (13,274) were manually rejected by title. The remaining records (1051) were independently judged by two authors for relevance based on title and abstract. After this secondary screening, 147 records remained for a full text evaluation of which 103 were deemed to contain primary research that explored public or patient views and attitudes involving the collecting, accessing, sharing, or management of biobank or genomic data with commercial entities. Disagreements on eligibility were resolved by a third author. The eligible articles were subjected to ‘reference harvesting’ which yielded an additional nine papers, increasing the tally of papers included to 113 (see Figure 1). A full list of references is available in the supplemental materials.
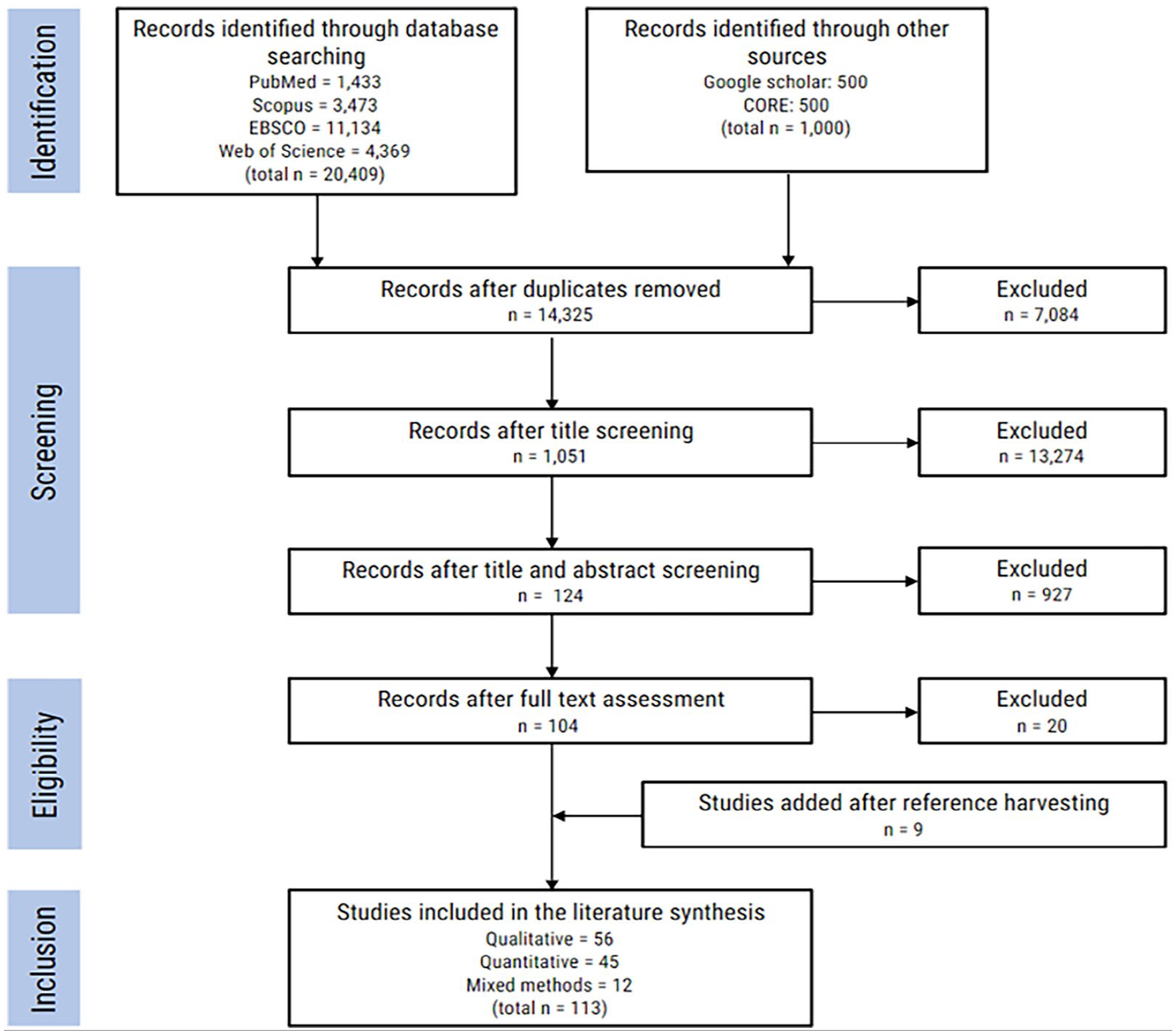
PRISMA flow chart of study triage and selection.
3. Results
The growth of biobanking globally over the past two or more decades has seen mirrored growth in scholarly interest of public attitudes towards biobanking and genomic data sharing with the number of eligible publications trending upwards across time (see Figure 2). The bulk of the eligible studies were from peer-reviewed journals (n = 104, 92%) but also included a small number of commissioned reports (n = 3, 2.7%), conference papers (n = 3, 2.7%) and unpublished manuscripts (n = 3, 2.7%).
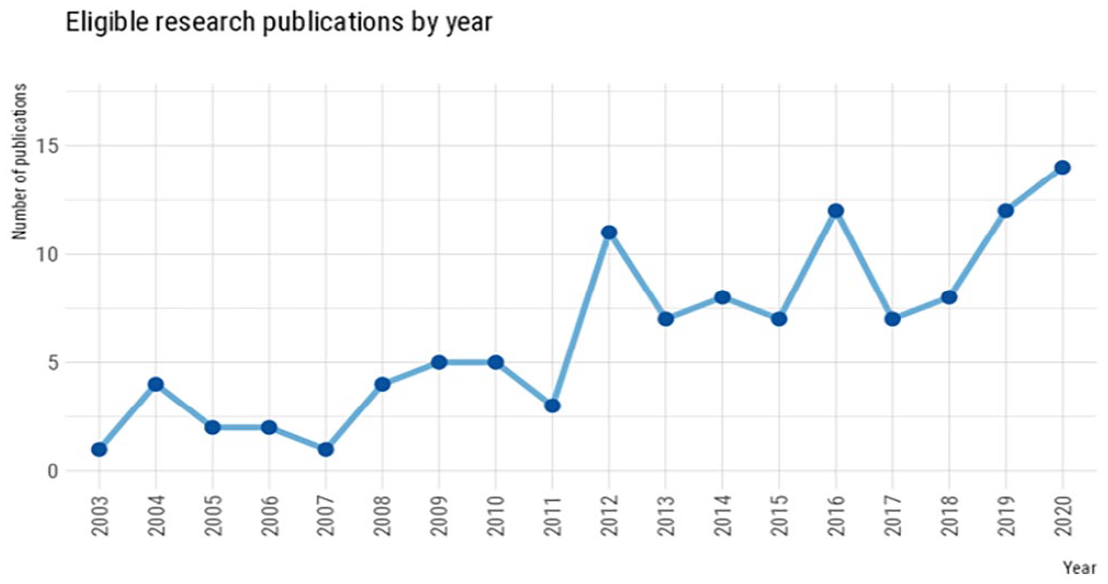
Eligible research publication by year.
Location of research
Eligible studies contained cohorts recruited across 36 countries. Eighteen (16%) of the studies included multinational cohorts. While research in this field is an increasingly global endeavour, just six countries were represented in 75% of the eligible studies. As shown in Table 1, these countries were the United States, the United Kingdom, Australia, Canada, Germany and the Netherlands.
Origins of the study cohorts.
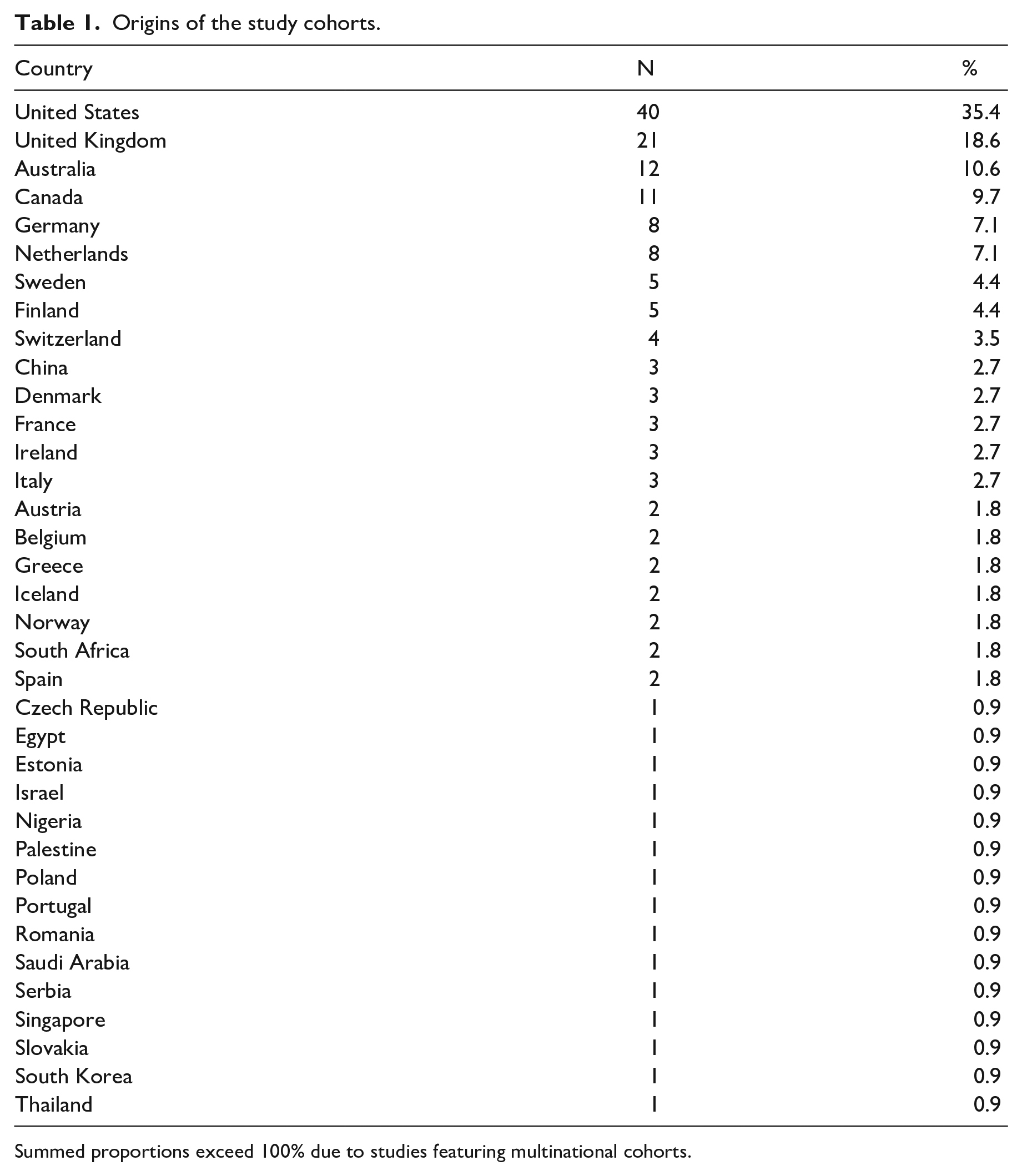
Summed proportions exceed 100% due to studies featuring multinational cohorts.
Study designs
Surveys were the most common research methodology employed, accounting for more than half of the eligible studies (n = 63, 55.8%), followed by focus groups (n = 29, 25.7%), interviews (n = 22, 19.5%), deliberative workshops/citizen juries/public dialogues (n = 9, 8%) and a solitary study employed a Delphi panel (0.9%). Of the included studies, six (5.3%) incorporated an experimental design, utilising randomised scenarios or vignettes (Antommaria et al., 2018; Critchley et al., 2015; Gornick et al., 2014; Weidman et al., 2019), a qualitative story deck (Passmore et al., 2019) or choice-modelling approaches such as discrete choice analysis (Hapgood et al., 2004).
Descriptions of the methods of participant recruitment in the studies varied in the level of detail provided. Just over a fifth of the studies (n = 24, 21.2%) recruited members of the public using a randomised method (e.g. random digit dialling (RDD) or random sampling of the electoral roll), while 20.3% (n = 23) of the studies used a non-random method (e.g. convenience sampling, purposive sampling or social media adverts). Other studies purchased panels from market research agencies (n = 16, 14.2%) or used crowdsourced task completion platforms (e.g. Amazon MTurk, n = 3, 2.7%). A sizable number of studies recruited from specific target populations including patients (n = 28, 24.8%) or relatives of patients (n = 5, 4.4%), biospecimen or biobank donors (n = 10, 8.9%), existing medical research projects (n = 5, 4.4%) and health care organisations (n = 5, 4.4%).
The variety of research on biobank participation and health data sharing captured by the broad parameters of the review is diverse. It spans numerous topics related to biobanking and genomic data sharing (e.g. trust, networks, moral concerns, consent and privacy), donation contexts (e.g. population or disease-specific biobanks), nationalities, cohort types and sample sizes, research methodologies, and peer-reviewed and non-peer-reviewed work. While the heterogeneity of the eligible papers rules out meaningful quantitative analysis it provides a foundation for extracting general themes and concerns around trust in biobanks and genomic repositories, and data sharing in the context of commercial involvement. It is worth noting that few (n = 8, 7.1%) of the eligible articles had a primary research focus on the effects of commercialisation on public trust and willingness to provide genomic or biobank data, and only two studies contrasted specific governance policies to measure their influence on genomic data sharing decisions (Briscoe et al., 2020; Middleton et al., 2019). Instead, commercialisation was frequently incorporated as a factor within the broader context of biobank operations and governance. Consequently, while many papers reported public concerns around commercialisation, it was rarely explored in-depth. Furthermore, in focus group and semi-structured interview studies where commercialisation concerns were reported as a theme, it was not always stated whether these concerns had been initially raised by the researchers or by the study participants.
The research review identified a range of issues where the existence of commercial involvement and/or interests provokes community apprehensions about biobank and genomic research participation. Categorised broadly, these issues relate to the organisational type and downstream uses of the data, informed consent, benefit sharing, privacy, confidentiality and data security, ownership and control of the data, funding sources and function creep.
Organisational contexts and downstream uses
It is evident that individuals’ decisions to participate in biobanks or otherwise share their genetic data will be influenced by organisation type and downstream uses of the data. Indeed, measuring trust in the organisations (or the researchers working for the organisations) collecting the donated materials or using the data derived from them was a frequent avenue of research, particularly in papers that employed public opinion surveys. A total of 23 studies (20.4%) included a contrast between public and for-profit organisations (e.g. Critchley et al., 2015, 2017; Dive et al., 2020; Goodman et al., 2017; Kaufman et al., 2009; Nicol and Critchley, 2012; Pawlikowski and Sak, 2015; Tupasela and Snell, 2012; Tupasela et al., 2010; Voigt et al., 2020). Some studies included not only a public/private contrast but a domestic/international dimension as well (e.g. Critchley et al., 2017; Goodman et al., 2017; Nicol and Critchley, 2012; Tupasela and Snell, 2012; Vidgen et al., 2020). Different researchers used conceptually distinct response scales intended to gauge a respondent’s (hypothetical) willingness to provide their data to repositories, such as trust (e.g. Critchley et al., 2015; Master et al., 2013) and comfort (Barnes et al., 2020).
An example of this research by Nicol et al. (2016) in Australia, compared trust in biobanks in five different contexts: domestic public biobanks, domestic biotechnology company biobanks, international biotechnology company biobanks, domestic public biobanks which allow access to domestic company biobanks, and domestic public biobanks which allow access to international public biobanks. The results showed that public trust was significantly higher for domestic public biobanks than all other biobanks (p < .001), and significantly lower for international company biobanks than all other biobanks (p < .001). Trust significantly decreased when a domestic public biobank allowed external access to domestic and international commercial biobanks; however, this arrangement evoked higher trust compared with the two purely commercial biobanks. The authors concluded that while commercialisation was deleterious to public trust in biobanking organisations, this public scepticism could be, in part, offset through independent governance and transparency.
Similarly, Vidgen et al. (2020) compared the public’s willingness to share genomic data from clinical testing that was anonymous or identifiable in an assortment of organisational contexts. They found that willingness to share genomic data decreased markedly when the downstream user was a commercial company compared with not-for-profit organisations. Furthermore, willingness to share identifiable genomic data was lower compared with anonymised data.
An RDD Computer Assisted Telephone Interview (CATI) survey of 750 Australians again found that commercial operations greatly lowered hypothetical willingness to donate. Most (94%) were willing to donate a biological sample if the biobank was a public research institution, decreasing to 57% if it was funded by a pharmaceutical company and 59% if it was operated by a biotechnology corporation (Dive et al., 2020). This pattern was repeated globally among samples of the public and patients. For instance, among male Irish urology patients (n = 259), 89% agreed that they would like donated biological samples be used for non-profit research compared with 39% for for-profit research (Fitzpatrick et al., 2009). In a survey (n = 151) of Swiss senior citizens, 75% were willing to donate genomic data to public research institutions but only 15% if the institution was private for-profit (45% were willing to donate to private not-for-profit institutions; Mählmann et al., 2016). In a Canadian survey of patients previously involved in medical research, 87% would be comfortable sharing with non-profit organisations and 53% with for-profit organisations (Barnes et al., 2020).
The qualitative research studies allowed greater exploration of the topic and frequently permitted respondents to convey a more nuanced and often pragmatic view of commercialisation. For instance, while participants in a deliberative democracy event were concerned about the prospect of commercialisation of biobanks, they felt it was permissible where necessary to foster medical research (McWhirter et al., 2014). Similarly, in a UK focus group, concerns raised by some about pharmaceutical companies profiting from donated biological samples were rationalised by others as necessary to fund further research (Lewis et al., 2013). In an interview study of those who had donated their personal genomic, health and clinical data for unrestricted data sharing to the US Personal Genome Project (N = 35), few interviewees (n = 4, 11%) raised concerns about their data being used commercially for private gain (Zarate et al., 2016).
In terms of where research using donated data should be conducted, without exception, participants were more comfortable with research being carried out in their home country than overseas (Dive et al., 2020; Goodman et al., 2017). Studies examining the interaction between research type (commercial vs non-commercial) and location (domestic vs foreign) found that off-shore commercial research causes the greatest concern (Critchley et al., 2017; Nicol and Critchley, 2012; Pawlikowski and Sak, 2015). Tupasela and Snell (2012) reported that Finns were more concerned about the distinction between domestic/international research than the distinction between public/private research with 78% comfortable with commercial use of tissue samples by local biotechnology and pharmaceutical companies.
In a survey exploration of how genomic database governance policies affected the US public’s reported willingness to provide their genomic data, Briscoe et al. (2020) reported that prohibiting the data from being sold, rented or shared with others increased participation willingness in 69.8% of respondents. Conversely, a policy of selling access to the data to pharmaceutical companies decreased willingness to participate in 67.2% of respondents. It is worth noting that the policy of selling data to pharmaceutical companies was the tested policy that provided the strongest disincentive to participate.
Informed consent
The presence of commercial interests in biobanking and data sharing impacts upon the public’s consent preferences for research participation and control of individual data. While research has been divided on public preferences for models of consent and many biobanks operate using a broad consent model (Master et al., 2012), the evidence suggests that more restrictive consent choices are preferred when commercial interests are involved (Tomlinson et al., 2015). In a Canadian study testing willingness to provide blanket consent across a range of scenarios, Tomlinson et al. (2015) demonstrated that 68% of respondents would be willing to donate tissue and medical information to a biobank and provide blanket consent for any research allowed. However, that figure dropped to 55.2% when the research could be used to develop patents and earn profits for commercial companies, a slight but statistically significant decrease. Importantly, however, this scenario was contextualised with information that ‘most new drugs used to treat or prevent disease come from commercial companies’ (p. 4), which may explain why more than half the respondents were comfortable providing broad consent to research that would benefit commercial interests.
In a focus group study of the opinions on large-scale genomic data and biospecimen sharing using a sample of patients with rare diseases across 16 European Union countries (n = 52), most were opposed to a broad consent model of data sharing, preferring an ‘opt-in’ approach in the absence of knowledge about research goals and citing concerns around the management, third-party access to and security of the database (McCormack et al., 2016).
Privacy, confidentiality and data security
Privacy concerns were frequently canvassed by investigators. The broad results reflect that issues surrounding privacy and data security are of high salience for biobanking participants. The definition of privacy, where it was defined, varied across the research but was sometimes conflated with overlapping concerns around confidentiality and data security.
In an Australian population survey, Critchley et al. (2017) sought to determine the relative importance of a variety of public expectations of biobanks. Privacy, operationalised as protecting ‘the identity of participants by not releasing information that could identify them’ (p. 675), was a primary concern. An expectation of privacy was valued, and statistically more important, than almost all other concerns (excepting from ensuring that research was ethically evaluated) including potential health benefit outcomes. This was further reinforced in the qualitative component of the study. Privacy concerns were the second-most raised topic by respondents whose concerns were for privacy being protected generally. Personal information being protected from access by specific commercial industries such as health insurance companies was also important. These findings indicate that privacy is another aspect by which the presence of commercial interests affects the calculus of participation decisions. However, despite privacy being a key concern among respondents, earlier research found that as trust in the biobanking organisation increased, so too did participants’ willingness to yield on privacy (Critchley et al., 2015). A US study found residual privacy concerns, defined as worry that an unauthorised person may access personal information despite organisational privacy assurances, were a powerful predictor of the willingness to donate to biobanks (De Vries et al., 2016). This finding was echoed in pan-European research where respondents who were concerned about privacy were significantly less likely to participate in biobank research (Gaskell et al., 2013).
In a large-scale US survey (N = 13,000), 90% of respondents who had sought medical treatment for themselves or a dependent from a health care network agreed that the privacy of their health information was important, while 64% agreed that the privacy of their information was something they worried about (Sanderson et al., 2017). Similarly, a representative population survey (N = 4659) found 90% of respondents were concerned about privacy in the context of biobanking and 56% were concerned about researchers having access to their information. Again, in this study privacy concerns were linked to willingness to participate in biobanking but only among participants who were promised lower (US$50) versus higher (US$200) compensation amounts, or among participants who were not offered the return of individual research results (Kaufman et al., 2009). This indicates that privacy concerns may be tempered through the promise of monetary compensation or other benefits.
In a descriptive study of female breast cancer patients, 83% of those surveyed believed genomic data should be protected, but few participants (13%) held significant concerns regarding the privacy of their data. The study measured privacy concerns in the context of discrimination by insurance or employers (43% and 28% concerned, respectively). Concerns relating to the security and privacy of data were heightened when research was being conducted by pharmaceutical companies (44% concerned) or government agencies (37% concerned) opposed to research institutions (20% concerned) (Rogith et al., 2014).
Distrust in the health insurance (Trinidad et al., 2010) and pharmaceutical industries (Van Hoof and Moens, 2018) as well as government (Lemke et al., 2010; Rogith et al., 2014) to protect donor data were commonly cited concerns. Other privacy and confidentiality concerns include data access by third parties (Bearth and Siegrist, 2020; Joly et al., 2015; Zarate et al., 2016), re-identification of genetic data (McCormack et al., 2016; Tai et al., 2018; Zarate et al., 2016), identity theft (Joly et al., 2015; Oliver et al., 2012) and fear of genetic discrimination by employers or health insurers (Botkin et al., 2014). It should be noted that some studies highlighted that privacy concerns around the sharing of genetic data were not always limited to concern for the self, but also of kin who could be identified through the data of a blood relative (Haddow et al., 2008; Weidman et al., 2019).
Benefit sharing
That the commercial benefits flowing from genetic health research form a public good rather than serving private interests was a recurrent theme. In an Australian public survey, Nicol and Critchley (2012) explored who should benefit from the use of publicly funded biobank resources including in the context of commercial involvement. In general, participants opposed the notion that donors be compensated directly. However, more people were likely to support direct compensation when biobank resources were utilised by a commercial entity, in contrast to a publicly funded research organisation or the biobank itself. Participants broadly agreed that research organisations should pay for access to biobank resources and were significantly more likely to agree that commercial entities pay compared with non-commercial entities. There was significantly stronger agreement that international accessors pay than domestic ones. In terms of who should benefit from research undertaken by commercial companies using public biobank resources, there was strong agreement that any treatments developed should be freely available to donors as well as affordable to citizens and the developing world.
Participants in a deliberative democracy event arrived at a broader view on benefit sharing, noting that investment in biobanking had economic benefits in the creation of jobs and research opportunities and lifted the profile of the local medical research industry (McWhirter et al., 2014). In other qualitative research, Haddow et al. (2007) reported that Scottish public and patient apprehension was not solely about profit-making on a publicly funded genetic database, with respondents having different opinions on what should be done with commercial profits. For patients, there was a level of pragmatism, accepting the commercialisation of research on the proviso that promised health benefits were delivered. By contrast, for the public tolerance of commercial involvement it was predicated upon profits being reinvested back into the community via continued research investment or channelled into the public health system. Similarly, the idea that commercial profits should be used to provide broader community benefit was supported in a representative US survey, where 62.1% of respondents believed that profits should only be used to support future research, while a smaller proportion (40.5%) agreed that profits should be shared with the public via a charitable medical trust (Spector-Bagdady et al., 2018). Indeed, the idea that commercial profits should generate some form of broad societal benefit was echoed by respondents in other studies (Beskow and Dean, 2008; Joly et al., 2015; van Schalkwyk et al., 2012). For example, participants in a Norwegian focus group acknowledged the necessity of commercialising donated biological material, but opposed the disproportionate exploitation of a communal resource for private gain (Steinsbekk et al., 2013). Participants were concerned about fairness, and that access to the benefits flowing from a public resource should be just.
There are mixed findings on whether people want or expect direct payment in exchange for their participation in biobanking and genomic health data research that could be used commercially. Among Norwegians, Steinsbekk et al. (2013) reported a moral objection among focus group participants to direct compensation for biospecimen donation to biobanks. In research contrasting European and Chinese expectations, the idea of payment was broadly rejected but reimbursement of costs was supported by focus group participants in both cohorts (Starkbaum et al., 2014). In a US survey, Briscoe et al. (2020) reported that half their survey respondents would provide their genomic data in exchange for payment, 11.7% would provide it altruistically and 37.8% would refuse to provide it even if compensation were offered. Likewise, in Australian research Nicol and Critchley (2012) reported that a large proportion of survey respondents supported a reciprocity arrangement of payment for biobank participation, especially in the context of commercial use of their data.
Ownership and control
Issues around ownership and control could be separated into two distinct categories: first, public preferences regarding the ownership and management of the organisations collecting biospecimens themselves; and second, preferences regarding ownership and control of the samples and data collected, including their downstream uses. Qualitative responses from an Australian survey indicated that 14 out of 16 respondents raised the issue of ownership, asserting that genetic biobanks should be in public hands (Critchley et al., 2017). Similarly, that genetic biobanks be publicly owned and managed, or at least be subject to public ethical oversight, was also proposed by participants in a Scottish focus group study (Haddow et al., 2007). Concern about genetic data being in private hands was illustrated by an Icelandic focus group participant who noted that when the privately managed Icelandic national DNA database became insolvent it was acquired by an American company and then later resold to a Chinese pharmaceutical company (McCormack et al., 2016).
Other research raised concerns regarding the intellectual property rights of discoveries using donated genetic data, with some focus group participants expressing the view that genetic information should not be patentable (Trinidad et al., 2010). Indeed, public concerns towards the patenting of information derived from DNA was recurrent in several other studies (Critchley et al., 2017; Levitt and Weldon, 2005; Shickle et al., 2003; Sterckx et al., 2016). In a Dutch study that sought to investigate perceptions of ownership of stored excised tissue 38% of patients believed they retained ownership of the tissue and 43% believed that the DNA stored in the tissue was their possession. The perception of ownership of the excised issue was mostly driven due to the DNA contained in the sample (Vermeulen et al., 2011).
Funding source
Relatively few studies included consideration of the role of funding source when gauging willingness to participate in biobank research. Funding arrangements can be complex, where public biobanks can be funded by private monies and private biobanks may be funded by public monies. In a survey of Canadian leukaemia patients on entrusting health information to biobanks, Master et al. (2013) reported that 40.4% of patients had a great deal of trust in university researchers that were publicly funded compared with 16.3% of patients who held a great deal of trust towards university researchers that were funded by industry. Frye et al. (2015) reported that 87% of survey participants regarded funding source as an important concern when deciding on whether to donate DNA to a bipolar disorder biobank. However, Critchley et al. (2015) in examining interaction effects of public and private biobanks and public and private funding found that funding source did not significantly influence trust when the biobank was public. Public biobanks were typically afforded a high level of public trust and when individuals consider participation, trust in the organisation conducting the research is of greater importance than the source of their funding.
Kettis-Lindblad et al. (2005) reported that only 18.7% of Swedish survey participants would be influenced by funding source when deciding to donate a blood sample for research purposes that could potentially be linked back to them. In this study, most participants were willing to defer to the judgement of the institution’s ethics committee. This highlights the importance of governance, where the public may suspend their natural scepticism of commercially driven research provided there is appropriate and independent ethical oversight.
In further qualitative evidence, the consensus among groups in a Canadian deliberative democracy event was that the governing body of a biobank should remain independent of the funding sources (Burgess et al., 2008). A similar Australian study (McWhirter et al., 2014) reported that biobank funding from commercial sources was deemed acceptable so long as it did not vest them with control. The same participants endorsed the idea that it was better to have a commercially funded biobank than none, but this was contingent on the research funder not having ownership or direct control over the biobank.
Function creep and unauthorised access
The public recognises that their genomic data can hold significant commercial and forensic value. One cause for concern was function creep where data provided to publicly managed repositories for health research under established terms of use and regulatory safeguards were vulnerable to future changes in government policy and legislation after successful lobbying for access by commercial third parties such as insurance or law enforcement (Hassan et al., 2020; Jones et al., 2019; Kaufman et al., 2009). For some, it was not necessary that change to access and safeguards actually occur. Suspicion that it could occur in future was sufficient enough to trigger withdrawal from biobank research (Broekstra et al., 2020). To counter the potential for function creep, participants in a UK public workshop opined the ‘need for robust governance, data anonymization, data security, and greater transparency in data use’ (Jones et al., 2019: 3).
4. Discussion
This diverse review of the literature highlighted that the broad public attitudes and expectation around the provision and custodianship of genomic and other health-related data in the context of commercialisation are complex, yet largely coherent and consistent. Despite this, significant knowledge gaps remain. The broad parameters of the review allowed for the inclusion of many papers. It should be noted, however, that while all included papers contained research findings relevant to commercial involvement in the collection, access or use of biobank and genomic data, comparatively few provided novel advancement of knowledge of commercialisation effects in a substantial way. This reinforces the notion that despite its acknowledged importance to the long-term sustainability of biobanking and genomic data sharing repositories via public behaviour, the underpinnings of commercialisation concerns remain under-researched. The relative lack of substantive research focusing specifically on commercialisation effects was unsurprising and is not a reflection of the quality of the research papers included. Rather it is the outcome, as previously highlighted, that the investigation and understanding of commercialisation effects was seldom the primary focus of the research and that commercial concerns were often incorporated among broader aspects of biobank and genomic repository governance. In terms of the quantitative surveys, many of the included studies consisted of a simple public versus company comparison. Likewise, for the qualitative studies, commercialisation findings were often limited to a handful of quoted comments from participants on general unease of commercial access to their data with little exploration regarding the source of the unease or how it might be moderated.
A universal and expected finding across the research is that the public is considerably more comfortable with and more hypothetically willing to participate in research conducted by, or entrust their data to, publicly operated biobanks and genomic repositories than those that are operated privately. Likewise, allowing commercial entities to access the data held by public repositories also results in the diminishment of trust. Taken as a whole, the empirical evidence reinforces the notion that a significant proportion of the public view participation in biobank and other health research via provision of their data in relatively binary terms, with a divide drawn between research perceived to advance the public good and that which is perceived to benefit specific interests. Overall, the public remains wary of commercial entities accessing their genomic data, oft citing an incongruence between the perceived profit-making motive of commercial entities and the desire to contribute to research that widely benefits public health. However, commercial access to data being regulated via a more trusted public research institution may be one avenue to temper public concerns around commercial involvement. Likewise, commercial interests funding research that is conducted by public bodies was similarly shown to protect against the erosion of trust (Critchley et al., 2015).
The review demonstrates that public concerns around commercialisation are wide-ranging, permeating through manifold factors influencing the public’s participation decisions. The presence of commercial interests impacts upon the perception of the benefits that flow from the research and to whom, the sensitivity of the data, the risk to one’s interests where the data are shared, preferences for consent and withdrawal, the level of control retained over one’s data, level of transparency and oversight, in addition to heightening privacy fears, and diminishing expectations that data would remain confidential and secure. However, despite these fears the review also revealed public acknowledgement and understanding that it is commercial enterprise that often drives the discovery of new drugs and treatments and which can provide the significant funding required in bringing these to market.
While there is a good understanding of the nature of public concerns around commercial access to genomic data, one notable gap was the dearth of research focusing specifically on data governance and regulatory mechanisms that might temper this public reluctance to provide genomic data where commercial parties will have access. Given that research has demonstrated that among the most powerful predictors of willingness to provide genomic information to repositories is a prohibition on selling the data or providing it to commercial entities (Briscoe et al., 2020), understanding which governance mechanisms might be employed to increase public acceptance of commercial access is critical. In extension, research focusing on how to reduce the perception of risk and personal harm specific to commercial access and governance and/or legislative measures that can reduce the fear of function creep in genomic databases is also worthy of investigation. The review also revealed that individuals can be pragmatic regarding commercial access to their data, willing to compromise on their concerns if they believe the research will deliver beneficial results.
Another gap that emerged is a lack of research that has explored the relationship between individual differences and the commercialisation effect. In terms of public demographics, there is little evidence about which factors, if any, may drive commercialisation concerns around genomic data access. Understanding why commercialisation concerns may differ across demographic groups and potentially lead to underrepresentation of populations in genomic repositories is important. Particularly if differences are manifested from a genetic basis (e.g. ethnicity, sex) as the heterogeneity of the research data is paramount (Popejoy and Fullerton, 2016).
The findings of the review indicate that individuals’ decisions to provide their personal data for health research are complex, being contingent upon a range of factors, and are particularly influenced by the prospect of commercial access to the data. While broad genomic data sharing policies are being promoted to drive research efficiency and to gain the maximum benefit and utility from a public resource, care needs to be taken around the conditions of access and governance mechanisms to ensure that benefits to commercial entities and the public are in harmony.
Supplemental Material
sj-docx-1-pus-10.1177_09636625241230864 – Supplemental material for A systematic literature review of the ‘commercialisation effect’ on public attitudes towards biobank and genomic data repositories
Supplemental material, sj-docx-1-pus-10.1177_09636625241230864 for A systematic literature review of the ‘commercialisation effect’ on public attitudes towards biobank and genomic data repositories by Jarrod Walshe, Brad Elphinstone, Dianne Nicol and Mark Taylor in Public Understanding of Science
Footnotes
Acknowledgements
This article is in memory of Professor Christine Critchley who developed and initially led the project, of which this article is a part, prior to her sudden passing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This paper was funded by an Australian Government Medical Research Futures Fund Grant #GHFMESLI000005.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
