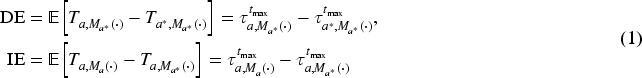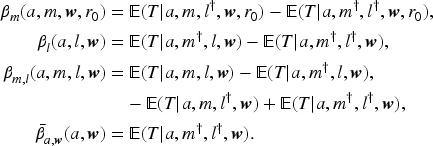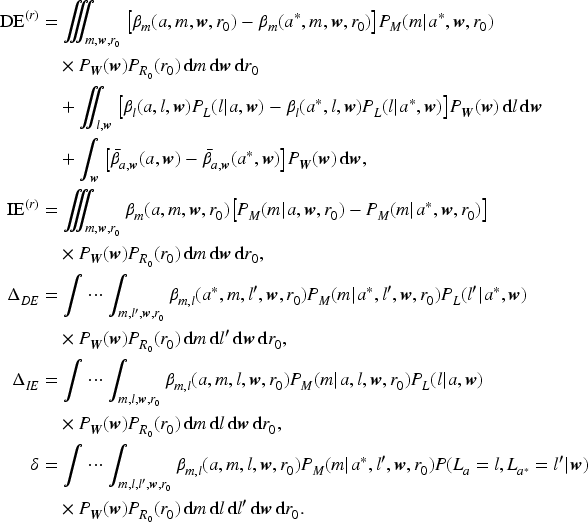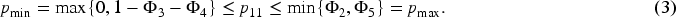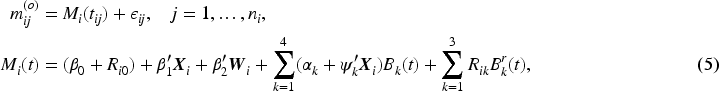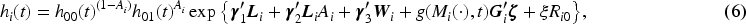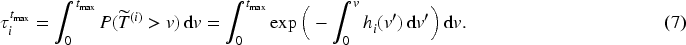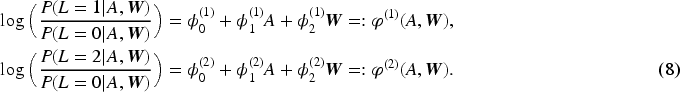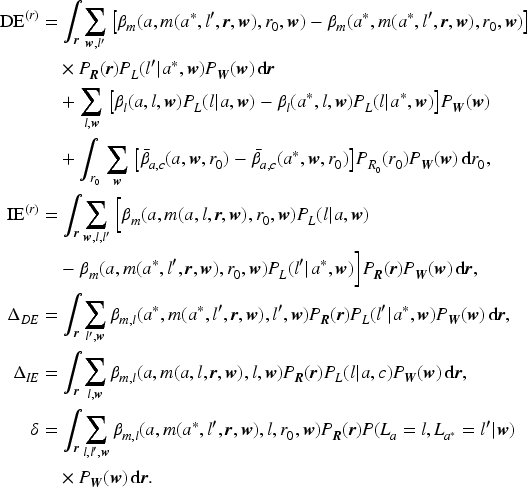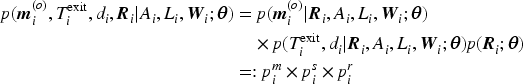Abstract
In interventional health studies, causal mediation analysis can be employed to investigate mechanisms through which the intervention affects the targeted health outcome. Identifying direct and indirect effects from empirical data become complicated, however, when a confounder of the mediator-outcome association is itself affected by the treatment. Here, we investigate identification of mediational effects under such post-treatment confounding in a setting with a longitudinal mediator, time-to-event outcome and an ordinal treatment-dependent confounder. If the treatment affects the treatment-dependent confounder only in one direction (monotonicity), we show that the mediational effects are identified up to stratum-specific sensitivity parameters and derive their empirical non-parametric expressions. The feasibility of the monotonicity assumption can be assessed using empirical data, based on restrictions on the marginal distributions of counterfactuals of the treatment-dependent confounder. In an empirical analysis, we use data from the Finnish Diabetes Prevention Study to assess the extent to which the effect of a lifestyle intervention on avoiding type 2 diabetes is mediated through weight reduction in a high-risk population, with other health-related changes acting as treatment-dependent confounders. We avoid pitfalls related to post-treatment conditioning by treating the mediator as a functional entity and defining the time-to-event outcome as a restricted disease-free time.
Keywords
Introduction
Lifestyle choices play an important role in the prevention of type 2 diabetes (T2D). As T2D causes major health and economic burdens globally, lifestyle interventions to reduce its incidence in high-risk populations are an area of active research.1–4 Studies of such interventions typically follow a cohort of individuals over time and aim at collecting information on biomarkers and health status evaluations at several follow-up visits.5–7 In countries with extensive health registers, data gathered at follow-up visits can be augmented with time-to-event outcomes retrieved from the registers.
Intervention studies are often based on experimental designs where study participants are assigned to treatment and control groups and the target of inference is the causal effect of an intervention on a specific health response. A more elaborate question involves understanding the extent to which the treatment effect is mediated through an intermediate variable. For example, potential mediating mechanisms of liraglutide treatment on cardiovascular and chronic kidney disease through changes in clinical biomarkers have been previously investigated in people with diabetes.8,9
Mediating mechanisms can be addressed within the causal mediation analysis framework, where the total effect of treatment is decomposed into direct and indirect (i.e. mediated) effects.10,11 The estimands of these causal effects can be defined in terms of counterfactuals, that is expected outcomes in hypothetical scenarios where the treatment and the mediator are intervened upon to set them at certain values. 12 Under non-trivial conditional independence assumptions, the estimands can be non-parametrically identified from empirical data. 13 Studies using a lifestyle intervention as the treatment and a clinical risk factor as the mediator, however, are especially prone to violations of the identifiability assumptions, because the intervention often induces behavioural changes that affect the response through both the intended mediator and other mechanisms. The behavioural change then acts as a treatment-dependent confounder, that is a variable that confounds the mediator–outcome relationship while also lying on a causal path between the treatment and outcome. In such situations the standard independence assumptions do not suffice to identify natural mediational effects non-parametrically. 14
Tchetgen Tchetgen and Vanderweele 15 showed that identification can be retained even under treatment-dependent confounding by further assumptions such as monotonicity of the treatment effect on a binary treatment-dependent confounder. Here, monotonicity means that the treatment can have only a positive (or only a negative) effect on the treatment-dependent confounder. Other assumptions retaining point identification include independence of counterfactuals of the treatment-dependent confounder or absence of an additive interaction between the mediator and the treatment-dependent confounder.15,16 Without any additional assumptions, the mediational effects under an observed discrete treatment-dependent confounder are still partially identifiable. This means that lower and upper bounds for the estimates can be obtained. 17
Further methodological issues arise when study participants are followed over time and the mediator is a longitudinal process. While causal mediation analysis has been extended to settings with longitudinal mediators,18–20 most literature treats the mediator as a vector-valued entity containing successive measurements of the mediating variable. Such an approach results in high dimensionality if the number of repeated measurements per individual is large and also poses challenges in handling uneven measurement intervals and missing values. Some authors have instead applied functional regression to represent the mediator trajectory as a function.21–23 Treating the mediator as a functional entity avoids the problem of dimensionality and enables flexible use of its full history at the time the response is evaluated. The repeated measurements in the longitudinal setting can also be used to extract information about individual-level latent properties. For example, Zheng and Liu 24 relaxed the assumption of no unmeasured mediator–outcome confounding under a longitudinal mediator and a time-to-event outcome by employing a joint modelling framework to estimate and control a common random effect reflecting an unobserved confounder between the two.
Time-to-event outcomes pose additional challenges in the causal inference framework. 25 A particular issue arises when causal estimands are defined by measures that condition on prior survival, such as the hazard function. If there exist latent variables affecting survival, conditioning opens a backdoor path from the treatment to future survival through the latent variables. This issue can be addressed by defining the response as an unconditional measure, such as the restricted mean survival time (RMST), that is the mean event-free time within a preset time period.23,26 If a significant portion of individuals do not experience the event during the study, RMST has the additional benefit over the mean survival time of allowing the time period to be chosen so that it remains robust to misspecification of the unobserved tail of the event-time distribution. In addition, if it is considered possible to have a zero risk for the event, the true mean survival time would be infinite, whereas RMST would remain constrained to the chosen clinically relevant time period.
In this study, we address identification of mediational causal effects in interventional studies with treatment-dependent confounding. We extend the previously presented monotonicity assumption 15 to a trichotomous treatment-dependent confounder and show that this results in expressions identifiable up to a stratum-specific sensitivity parameter. Our approach is similar to the partial identification in Miles et al. 17 but imposes restrictions on the unobserved joint distribution of the counterfactuals of the treatment-dependent confounder, leading to a necessary condition for their marginals. As the marginals can be estimated from observed data, the feasibility of the monotonicity assumption can be empirically assessed. As an application, we consider the effect of an intensive lifestyle intervention on T2D incidence among a high-risk population, based on the Finnish Diabetes Prevention Study (DPS). 27 The aim of the empirical analysis is to quantify the extent to which the effect of the intervention on T2D-free time is mediated through it inducing weight loss. We apply functional regression to represent the body mass index (BMI) trajectory as a functional entity and use a joint modelling framework to control potential latent confounding between the BMI trajectory and T2D incidence.
The paper is structured is as follows. Section 2 presents the empirical problem motivating this study. Section 3 defines the targeted causal estimands, specifies conditions for their identification, and describes the methods we propose to use for their estimation. Sections 4 and 5 present the results of the empirical application and discuss the results along with further considerations of the used methodology and its possible limitations.
Data sources and motivation
The aim of the Finnish DPS is to assess the effectiveness of an intensive lifestyle intervention in preventing and delaying T2D onset in a high-risk population.27–29 The study cohort was enrolled between 1993 and 1998 and originally consisted of 522 individuals. The eligibility criteria required the participants to be overweight (
A previous analysis of the DPS data showed a 40% lower hazard for T2D and greater weight loss in the intervention group during the first 13 years of follow-up and also showed greater improvements in their lifestyle compared with the control group, particularly in dietary intakes. 31 Moreover, previous analyses have found associations between the lifestyle intervention, physical activity, nutritional components and diabetes incidence in the DPS cohort.32,33
In this study, we aim to quantify the effect of the lifestyle intervention on the prevention of T2D mediated through weight loss. Since obesity is one of the most prominent risk factors for T2D, it is of interest to assess the extent to which the success of the lifestyle intervention can be attributed to its ability to reduce body weight in people with overweight or obesity. Previous literature on lifestyle intervention studies has suggested a so-called legacy effect, wherein the intervention’s impact on T2D incidence persist long after the intervention ends and the obtained group differences in risk factors have diminished. As summarised by Wilding, 34 such results have been reported in the major T2D prevention trials, including the DPS, 31 a study conducted in China, 35 and the Diabetes Prevention Program in the USA. 36 These findings motivated us to consider the change in the BMI during the early phase of the intervention as the effective mediator. Moreover, we apply a three-year time window because the majority of differences in BMI between the two groups occurred during this period. We will use the restricted survival time as the outcome measure, with the maximum time as 15 years, reflecting a clinically relevant time horizon. The outcome is thus interpreted as the number of healthy (i.e. T2D-free) years during the first 15 years after intervention onset.
In addition to weight reduction, the DPS intervention aimed at moderate physical activity and healthy nutritional composition measured by intakes of total fats, saturated fats and fibre. 28 These lifestyle factors can be assumed to influence the study participants’ BMI trajectories and also T2D incidence through mechanisms other than weight loss, thus rendering them potential treatment-dependent confounders. We created a summary variable to represent individuals’ lifestyle choices influenced by the intervention, combining total physical activity and the dietary intake components. The amount of total physical activity was measured by self-reports at every study visit and the components of dietary intake by three-day food diaries prior to the study visits for the first three (in addition to baseline). All variables from each study visit were standardised with respect to their baseline means and standard deviations and the lifestyle score was computed as the mean over the standardised variables across the three post-baseline study visits. The lifestyle score was then categorised into three levels based on its baseline distribution. As the distribution was nearly Gaussian, the tertiles were used as cut-points for this categorisation. Constructing the lifestyle score in this way ensures its ordinal interpretation in the sense that belonging to a higher category implies on average higher levels in each of the four lifestyle variables.
Causal mediation analysis requires controlling for any factors confounding the relationships between the treatment, mediator and outcome. We considered age, sex, smoking status and the baseline lifestyle score as potential confounding baseline variables. Age at baseline was categorised as <45, 45 to 59 and
Methods
In this section we present the proposed methodological framework. Sections 3.1 and 3.2 outline the assumed causal model and the estimation targets. In Section 3.3 we give assumptions sufficient to identify the causal estimands from empirical data and present the resulting expressions for the direct and indirect effects. In Sections 3.4 and 3.5 we define the parametric models and describe the strategy for their estimation.
Causal estimands
Let
We use the RMST,
If there is interaction between the treatment and the mediator, the interpretation of mediational effects depends on the choice of the levels at which the fixed variables are held in each case. The definitions (1) lead to interpreting the DE as the pure direct effect and the IE as the total indirect effect, as discussed by VanderWeele.38,39 For ease of notation, we hereafter denote the mediator trajectory and its realisation often simply by
The causal model is represented graphically by the directed acyclic graph (DAG) shown in Figure 1, describing the assumed causal structure of the relevant variables involved. The main interest lies in the interplay between the treatment
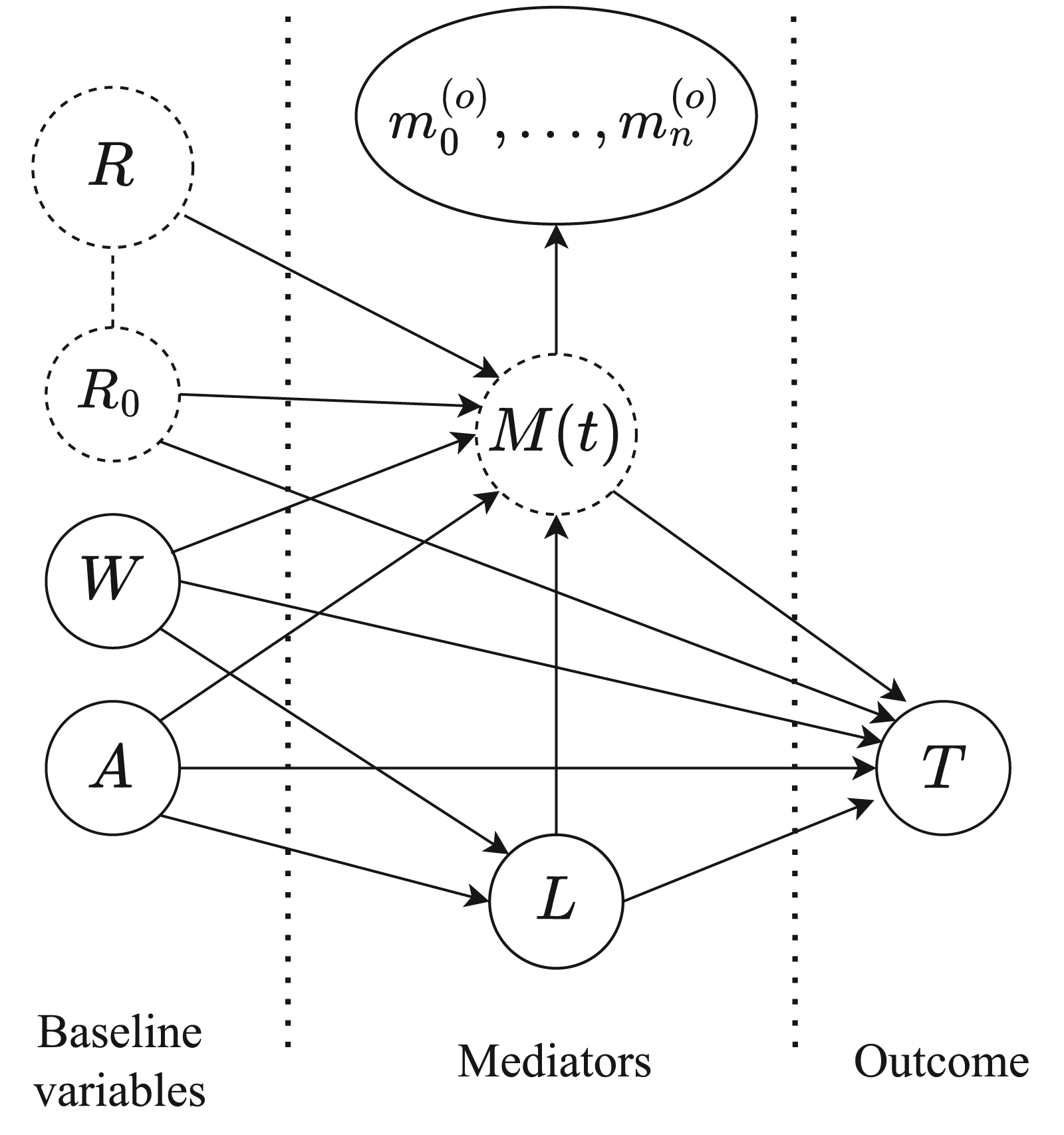
Directed acyclic graph describing the assumed causal mechanism within an individual. The effect of treatment
We assume that individual-specific random effects (
We interpret the DAG in the framework of nonparametric structural equations models (NPSEM). 11 The directed arrows imply a causal ordering between the variables, and an absence of an arrow between two variables implies no direct causal relationship between them. Importantly, the absence of any bidirectional arrows implies an assumption that any randomness affecting one variable in the graph is independent of the randomness affecting any other variable.
Under the NPSEM framework, the following conditional independence assumptions are implied by the causal DAG of Figure 1:
The first three assumptions translate to no unmeasured confounding between
Let
Tchetgen Tchetgen and VanderWeele showed that assuming no additive interaction of
We now extend the monotonicity assumption to a trichotomous 
where the marginal probabilities
For a fixed
If
Under the identifying assumptions of Section 3.3, the DE and IE can be expressed in terms of the observed data. Although the effects and their corresponding empirical expressions were derived nonparametrically, the components forming the empirical expressions were estimated parametrically. In this section we describe the parametric models we used to estimate the terms in the expressions (2) for the DE and IE.
Mediator trajectory
We assumed a linear mixed model for the mediator trajectory. The underlying true mediator was assumed to be a smooth trajectory
We assumed a parametric proportional hazards model for the time-to-event outcome with separate piecewise-constant baseline hazards for the two treatment groups. Denoting the baseline hazard functions as
The choice of the functional form of
The RMST for an individual
We used multinomial logistic regression to model the dependence of the trichotomous treatment-dependent confounder
The above model includes only the main effects of each predictor and could thus be considered relatively inflexible. As the research question is concerned with the mediating mechanism of the treatment, we also considered models with interaction terms between the treatment and the baseline covariates to allow the treatment effect on the treatment-dependent confounder to differ among the baseline covariate strata. Model comparison was carried out to determine whether any of the more flexible models should be favoured against model (8).
Since the underlying BMI trajectory as the mediator is stripped of the stochastic error terms, its distribution conditionally on the covariates and treatment is determined by the distribution of the random effects. Integrating an arbitrary functional
With the definitions of mediational causal effects in (1), the assumed parametric models imply the following formulae for the terms determining the DE and IE:
Estimation
We employed a joint modelling framework to estimate the parametric models of the mediator trajectory and the time-to-event outcome. Since these models rely on only the observed values of the treatment-dependent confounder, the model for the treatment-dependent confounder was estimated separately. Pareto smoothed importance sampling leave-one-out cross-validation 41 was used to compare the treatment-dependent confounder model against its more flexible variations.
A joint model for the longitudinal and time-to-event outcomes comprises specifying submodels for both outcomes and linking them via some association structure, thus allowing incorporating any information shared between the two outcomes.42–45 The association structure was here induced by including a function of the longitudinal mediator trajectory into the linear predictor of the survival submodel and also assuming a random effect which is shared between the mediator trajectory and the time-to-event outcome, enabling adjustment for a latent confounder.
We adopted a Bayesian framework to estimate all parametric models. Let
For all regression parameters, we assumed relatively uninformative normal priors with mean zero and standard deviation
The models were run using four parallel chains with 2000 burn-in iterations and 2000 sampling iterations each, resulting in 8000 Markov chain Monte Carlo (MCMC) draws from the posterior distribution of the model parameters and the convergence was assessed by the Gelman–Rubin
Application
The DPS originally followed a cohort of 522 individuals, with 265 randomised to the intervention group and 257 to the control group. After excluding 19 individuals with missing baseline covariate values, the final sample consisted of 503 individuals with 254 people in the intervention group and 249 in the control group. The median number of clinical visits per person was 11 (interquartile range 6–13) in the intervention group and 9 (IQR 4–13) in the control group. The intervention group contributed a total of 3505 person years during which 166 T2D cases were observed with a RMST of 11.2 years. The control group contributed a total of 2865 person years and 168 T2D cases with a RMST of 9.5 years.
The study endpoint was the diagnosis of T2D, either ascertained at any of the study visits or inferred from the register data. Since the clinical study visits were considered the more reliable source, we used the first diagnosis made at the study visits as the primary endpoint and considered the register data only after their last study visit for each individual. Figure 2(a) shows the cumulative incidences of T2D from both sources. The clinical risk set refers to the number of individuals in the risk set having not yet made their last study visit, whereas the register risk set is the number of individuals in the risk set being followed through the drug registers. The cumulative incidence curves based on the study visits and the drug registers grow reasonably closely in proportion to the number of individuals, implying that any bias due to uneven sensitivity of T2D detection was unlikely.
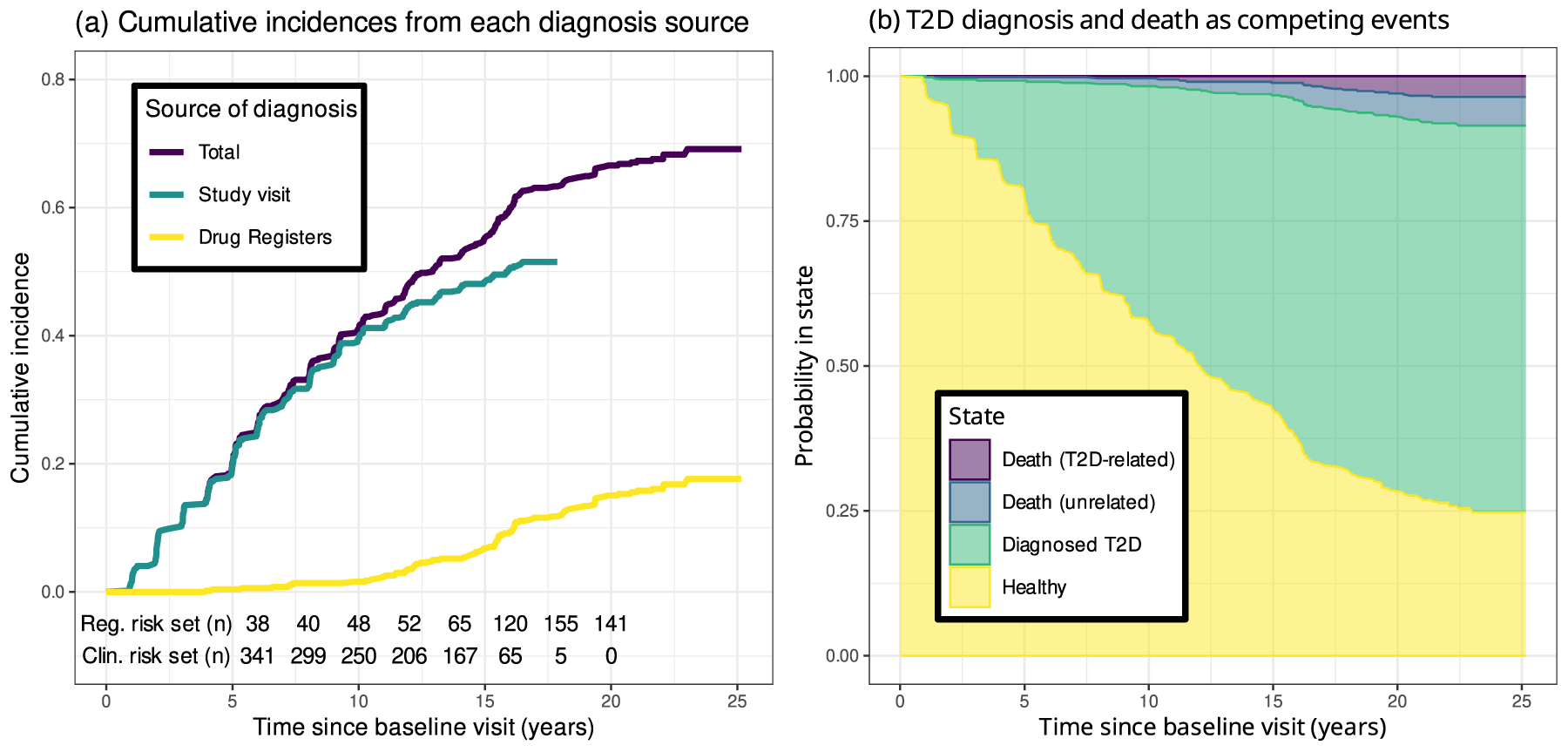
(a) Cumulative incidence of type 2 diabetes (T2D) diagnoses due to both sources of diagnosis. Underneath the curves, the number of individuals being followed through each source is shown. (b) Probabilities of reaching the three competing (absorbing) states as functions of time. T2D-related deaths include cases in which the cause of death is potentially associated with T2D, namely cardiovascular and cerebrovascular complications.
For each study participant, the follow-up started at the baseline visit and terminated at the event of T2D diagnosis, death or end of follow-up at the end of 2018. The dates and causes of deaths were obtained from the Finnish Cause of Death Register. Figure 2(b) shows the cumulative risks for the competing events of T2D diagnosis, death with a potentially T2D-related cause, that is cardiovascular and cerebrovascular complications, and death from other causes. As the proportion of potentially T2D-related deaths appears negligible, treating all deaths as uninformative right censorings was deemed justified. With the availability of the register data after the clinical follow-up, deaths were the only source of censoring in the data.
Figure 3(a) shows the observed and estimated mean BMI trajectories for the two treatment groups. Figure 3(b) to (d) display the Kaplan–Meier curves and estimated survival functions from model (6), illustrating how accurately the model reproduces the observed dependence of T2D survival on the treatment, change in BMI over the first three years, and the lifestyle score. In these plots, the cumulative change in BMI over the first three years was categorised into tertiles of the individual-level point estimates obtained from the model. The survival functions were then computed as the means of the estimated survival functions for each of the three groups. These plots are based on the model using the three-year legacy parameterisation
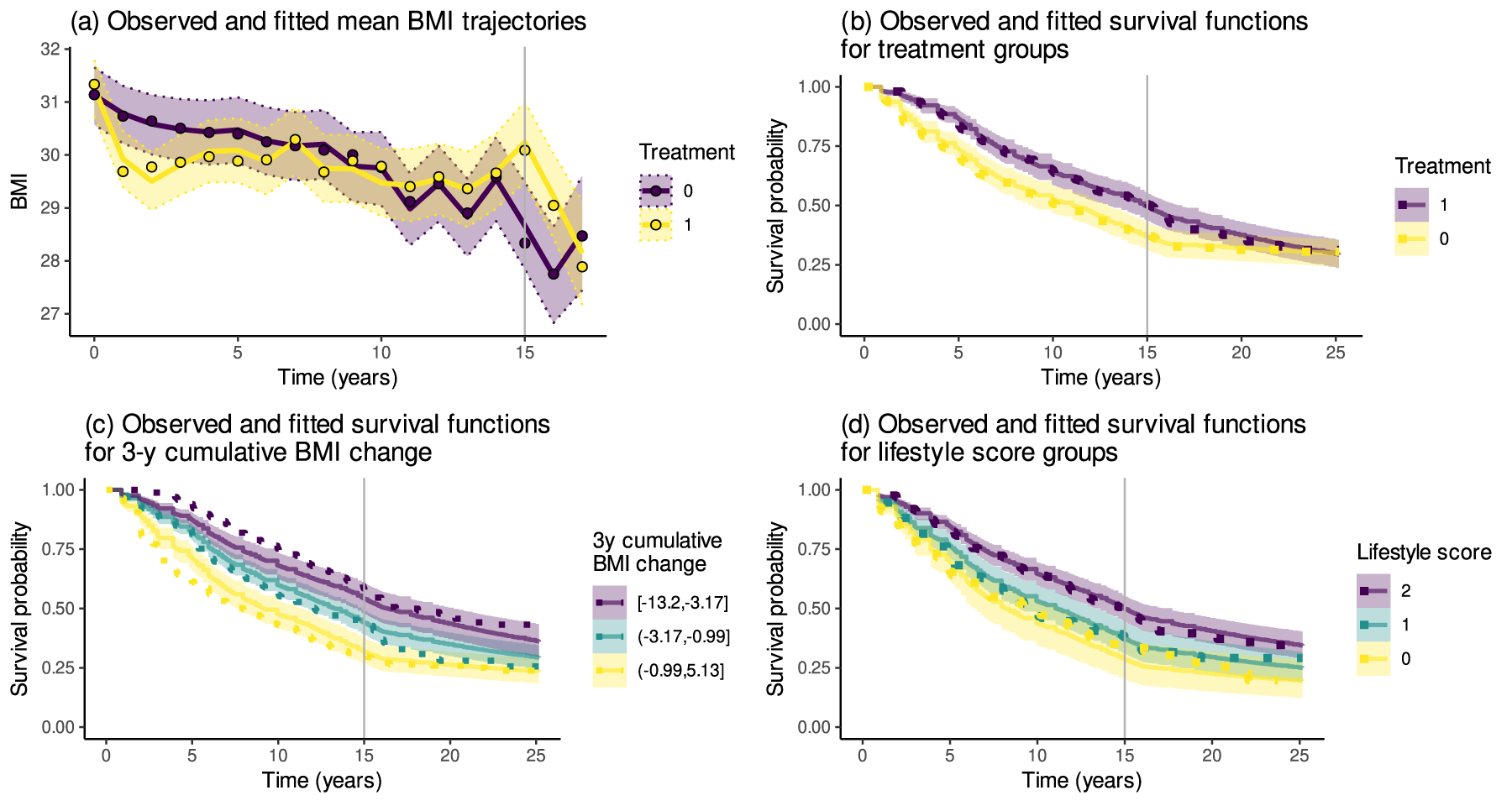
(a) The observed (dots) and estimated (solid lines) mean trajectories of body mass index (BMI) in the intervention (1) and control (0) groups with the
The mean BMI trajectory in the intervention group (Figure 3(a)) exhibits a decrease in the early years since the start of the intervention, after which the mean trajectory gradually rebounds close to the trajectory of the control group. The estimated curves replicate the observed average trajectories reasonably well, although the shape showed slight differences in the intervention group over the first two years. The Kaplan–Meier curves indicate a lower T2D risk for individuals in the intervention group (Figure 3(b)), in the higher lifestyle score groups (Figure 3(d)) or having decreased their BMI more over the first three years (Figure 3(c)). The estimated survival functions agree closely with the Kaplan–Meier curves. However, with the early BMI change, the differences in the estimated survival functions are less pronounced than those suggested by the Kaplan–Meier curves, which might indicate some lack of fit in the model with respect to the relationship between the early BMI change and T2D hazard.
In addition to model (8) of the treatment-dependent confounder, we considered more flexible models including interactions of the treatment with each baseline covariate all at the same time or each one separately. The model comparison showed no discernible difference between the model’s performances and so we chose the most parsimonious one, that is, model (8) (Section S1 in the online Supplemental Material).
The monotonicity condition under the selected model was assessed from the empirical data by estimating the marginal probabilities for the two counterfactual lifestyle scores (
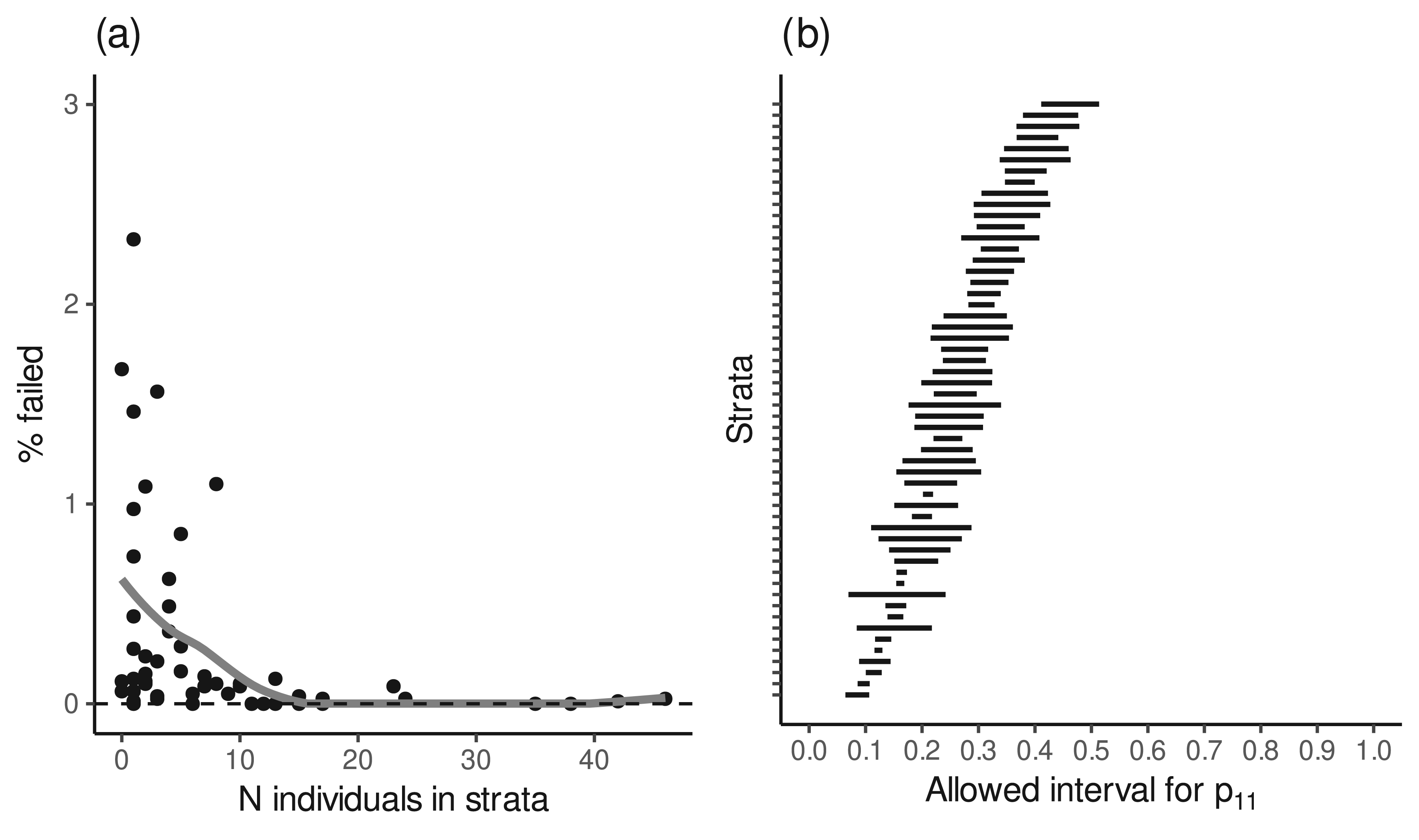
(a) Proportions of Markov chain Monte Carlo samples failing the monotonicity condition in the 54 strata defined by the covariates (age, sex, smoking and baseline lifestyle score) under the selected model. The largest proportions of failures occurred in strata with low numbers of individuals. (b) The average posterior boundaries within which the free parameter in the joint distribution of the two counterfactual treatment-dependent confounders (
Figure 4(b) shows, for each stratum, the average boundaries within which the probability
We also investigated the agreement of the step monotonicity assumption with the data. Under the step monotonicity assumption, 3279 (
Table 1 shows the posterior means and
The posterior means and 95% credible intervals (in parentheses) of the
,
and
under the two alternative functionals and different choices of stratum-specific sensitivity parameters
(see equation (4)); min/max (choosing the parameter to minimise/maximise the mediational effects separately in each covariate stratum; 0.5 (choosing
in all strata).
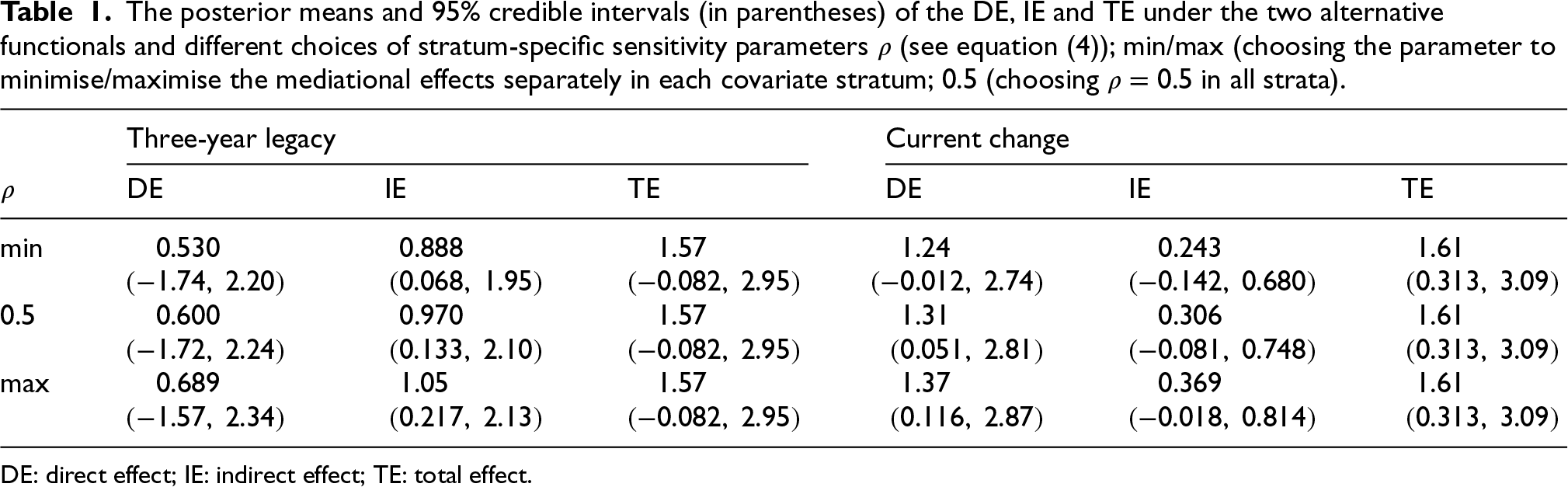
The posterior means and 95% credible intervals (in parentheses) of the

DE: direct effect; IE: indirect effect; TE: total effect.
Under the three-year legacy parameterisation, the estimated indirect treatment effect, that is the effect mediated through the change in BMI, amounts to roughly one year of additional time without T2D over the 15 years since the treatment onset (
The current change parameterisation resulted in a more pronounced DE of
As it could be considered a plausible a priori assumption, that the effects of the intervention cannot be negative, the results are also shown by discarding posterior draws (
The posterior means and
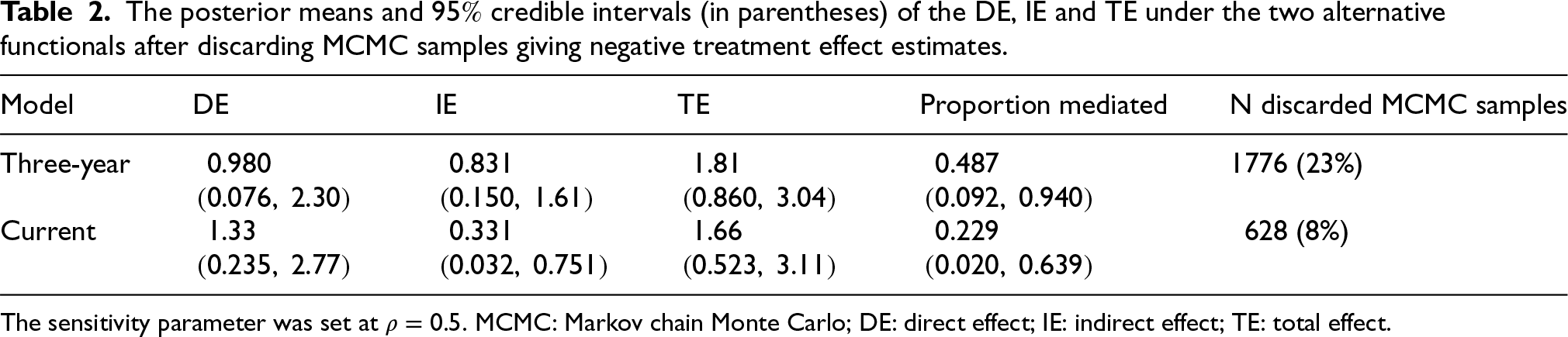
The sensitivity parameter was set at
For sensitivity analyses, we used the approach of Miles et al.
17
to find lower and upper bounds for the estimates relaxing the monotonicity assumption, that is, optimising the expressions with respect to a joint probability matrix
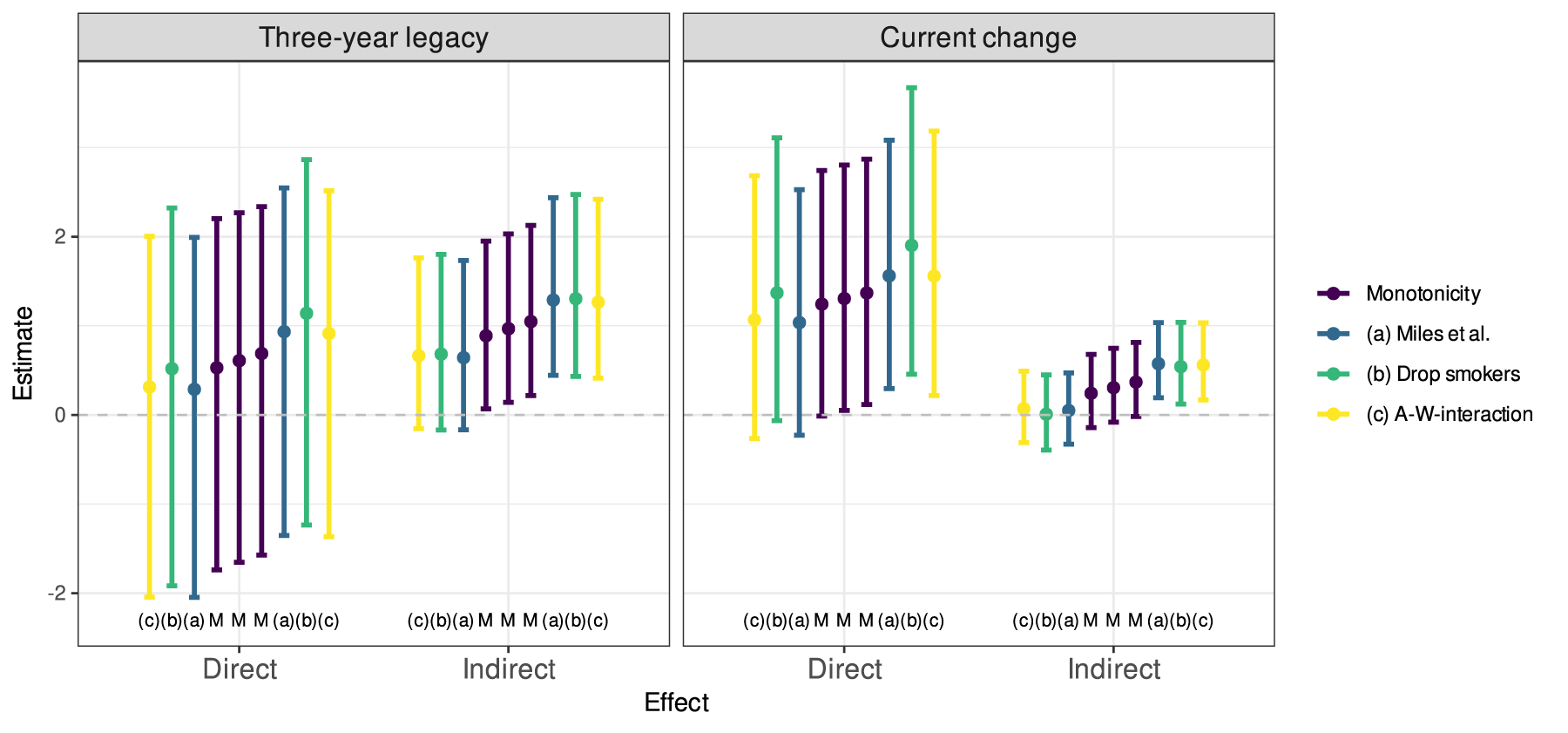
The lower and upper bounds for the estimates of direct and indirect effects and their
Interestingly, the estimated direct effects appear to be higher when current smokers are excluded from the data while the indirect effects are largely unchanged. This suggests that the treatment might be less favourable for smokers than the others. The total effect estimates using the simple estimator based on areas under Kaplan–Meier curves resulted in a TE of
We addressed causal mediation in the case of a longitudinal mediator and a time-to-event outcome in a randomised lifestyle intervention study, where some of the lifestyle changes acted as treatment-dependent confounders. Such situations may arise in interventional studies aimed at reducing the risk for an adverse health outcome through lifestyle changes. When the interest lies in the treatment effect mediated through changes in a biological risk factor, such as body weight in the current study, other lifestyle changes will act as treatment-dependent confounders, complicating the identification of mediational effects. The methods described here can be applied to address such situations in order to elucidate mechanisms by which treatments affect health outcomes.
Previously, Tchetgen Tchetgen and Vanderweele
15
showed that if the effect of treatment on a binary-valued treatment-dependent confounder is monotonic, mediational effects can be identified from empirical data. We here extended their approach to an ordinal trichotomous case, where a single sensitivity parameter (for each baseline covariate stratum) needs to be specified to identify the mediational effects. The sensitivity parameter determines the joint probability distribution of the two counterfactuals,
Other approaches to deal with treatment-dependent confounding have been introduced. Identification can be retained if the counterfactuals of the treatment-dependent confounder are assumed to be independent or if one is a deterministic function of the other, 16 or if there is no additive interaction between the treatment and the treatment-dependent confounder. 15 Another option would be to switch the estimation target from natural direct and indirect effects to their interventional analogues, which are identifiable even in the presence of treatment-dependent confounders but do not, in general, share the same interpretation as the natural effects.53–55
We treated the longitudinal mediator (BMI) as a functional entity, that is a smooth function describing the underlying trajectory which the mediator follows and of which observations endowed with stochastic deviations were made over time. The contribution of the stochastic deviations to the association between the mediator and the time-to-event outcome was assumed to be negligible, hence promoting the underlying function itself as the effective mediator. An implicit assumption was that the structure given for the trajectory function is flexible enough to capture the longitudinal process to a relevant extent. By defining the mediator as a functional entity, we avoided the practical difficulties dealing with a high-dimensional mediator, as well as the conceptual challenges that might arise from having to control for post-treatment values of the mediator. Similar approaches treating longitudinally measured mediators as functional entities have been previously investigated in the mediation analysis literature.21–23
We chose the restricted survival time as the time-to-event outcome to avoid issues arising from unmeasured heterogeneity under outcomes defined conditionally on previous survival. 40 For instance, if there were an unobserved genetic factor influencing T2D risk, conditioning on survival at any post-treatment time would induce a selection bias, potentially invalidating the analysis. In addition, as one third of the study participants did not develop T2D, a substantial tail proportion of the event time distribution remained unobserved. This may introduce the risk of misspecifying the parametric shape of the tail distribution. To address this, we chose the restriction time as 15 years since the baseline to represent a clinically meaningful time period providing reasonable number of follow-up visits and T2D events.
We used a joint modelling framework to estimate the parametric models. The association structure was induced by incorporating the latent mediator trajectory in the survival submodel and also specifying a shared random effects structure.42,45 As the mediator was considered a latent quantity, individual-specific mediator trajectories needed to be estimated and the uncertainty regarding the estimation appropriately propagated to the estimates of the causal effects. This propagation of uncertainty was straightforwardly handled in the Bayesian set-up of computations.
Incorporating the mediator into the survival submodel required selecting a functional of its trajectory to serve as a linear predictor. The indirect effect then represents the influence mediated through the property captured by the chosen functional. In our analysis, we considered two functionals, three-year legacy and current change, chosen a priori. Alternatively, one could apply some model selection procedure to identify the best-performing functional from a set of candidates. For example, within the joint modelling literature, Mauff et al. 56 proposed an approach to determine an optimal weighting function for a cumulative effect measure.
From the causal perspective, shared random effects between the longitudinal and survival submodels represent latent confounding between the mediator and the outcome. The joint modelling framework can thus be employed to account for such unmeasured confounding. 24 We here used the random intercept of the mediator trajectory as a shared random effect to reflect a latent property of the individual that may influence both the mediator trajectory and the time-to-event outcome. In our empirical application this might be, for example, an unmeasured metabolism-related genetic factor.
Estimating the mediator trajectories and shared random effect relied on the relatively large number of repeated measurements of the mediator. With a small number of repeated measurements, it may not be possible to estimate the mediator trajectory accurately enough to justify, for example, the current change parameterisation. In such cases, it would be necessary to use simpler functionals that can be estimated reliably from the available data. For example, the three-year legacy parameterisation could be feasible with even a few repeated measurements over the early follow-up.
The primary goal of our empirical application was to decompose the treatment effect on the restricted mean T2D-free time into the indirect (mediated through weight reduction) and direct (all other mechanisms) effects. Using the three-year legacy parameterisation, the estimated IE translated to one year of additional time without T2D over the 15 years after the start of follow-up, with a
Assuming that the treatment effects cannot be negative had little impact on the estimates under the current change parameterisation, and the proportion mediated was estimated to be
The choice of the legacy parameterisation was motivated by previous literature demonstrating the persistence of the intervention effects on T2D risk long after the discontinuation of the active treatment and diminishing of the acquired group differences in the clinical risk factors. 34 We used the cumulative change at three years as a time-constant predictor in the survival submodel starting from the onset of treatment. We justify this by noting that the individual trajectories are determined by the observed baseline covariates and the random effects and can thus be interpreted to exist at the baseline, even though they may be learned only by observing the trajectory unfold over time. In this context, the weight reduction during the first three years was interpreted as a surrogate for some biological process that responds quickly to the initiation of the lifestyle intervention and the induced behavioural changes. This underlying biological process was then assumed to manifest through the subsequent weight reduction and be the true causal mechanism linking weight reduction to a decrease in T2D risk.
The DE and IE were identified up to sensitivity parameters controlling the probabilities (
Our empirical analysis has some potential limitations. First, as is always the case in causal analyses, the validity of inferences relies on the untestable assumption of no unmeasured confounding. We used age, sex, smoking and the baseline lifestyle score as baseline covariates and used a shared random effects structure in the joint model to include a latent confounder of the BMI trajectory and T2D incidence. Randomisation addresses confounding involving the treatment. However, it is possible that some important confounders between the treatment-dependent confounder, mediator and outcome were not accounted for. Second, the lifestyle score serving as the treatment-dependent confounder was constructed somewhat crudely by summarising four variables capturing lifestyle changes across the first three follow-up visits. Because this confounder plays a key role in the causal mechanism, any imprecision in its measurement may bias the resulting causal effect estimates. Our decision to use a three-level categorisation reflected a compromise between measurement accuracy and limiting the dimensionality of the unidentified parts of the causal model. Third, the generalisability of the results is limited. The DPS inclusion criteria selected volunteers who were already overweight, had developed impaired glucose tolerance and were between 40 and 65 years of age at the screening visit but had not yet been diagnosed with T2D. As such, the DPS cohort represents a selected population, that is, individuals at a high risk of T2D who had managed to avoid the disease until a relatively old age.
In conclusion, we investigated causal mediation in longitudinal intervention studies with a time-to-event outcome in the presence of an ordinal treatment-dependent confounder. Foremost, we showed that assuming monotonicity of the treatment effect on a trichotomous ordinal treatment-dependent confounder, the DE and IE can be identified up to stratum-specific scalar sensitivity parameters. The time-to-event outcome was defined as a restricted survival time to avoid issues pertaining to measures conditioning on prior survival. To overcome challenges with a high-dimensional mediator, we treated the longitudinal mediator as a functional entity and employed a joint modelling framework to control for possible unobserved confounding between the mediator and the outcome. The methodology was applied to decompose the effect of a lifestyle intervention on restricted T2D-free time into an IE through weight reduction and a DE involving other mechanisms. We found some evidence suggesting the existence of a clinically significant IE through weight reduction, however, the magnitude of the estimated IE depended considerably on the assumed effective form of the mediator. When using the weight change over the first three years as the mediator, the IE accounted for a large fraction of the TE. Conversely, when considering the current weight change since baseline as the mediator, the DE was substantially larger than the indirect one. The results remained similar in sensitivity analyses relaxing the monotonicity, indicating robustness to violations of this assumption.
Supplemental Material
sj-pdf-1-smm-10.1177_09622802261418211 - Supplemental material for Mediation analysis in longitudinal intervention studies with an ordinal treatment-dependent confounder
Supplemental material, sj-pdf-1-smm-10.1177_09622802261418211 for Mediation analysis in longitudinal intervention studies with an ordinal treatment-dependent confounder by Mikko Valtanen, Tommi Härkänen, Matti Uusitupa, Jaakko Tuomilehto, Jaana Lindström and Kari Auranen in Statistical Methods in Medical Research
Supplemental Material
sj-zip-2-smm-10.1177_09622802261418211 - Supplemental material for Mediation analysis in longitudinal intervention studies with an ordinal treatment-dependent confounder
Supplemental material, sj-zip-2-smm-10.1177_09622802261418211 for Mediation analysis in longitudinal intervention studies with an ordinal treatment-dependent confounder by Mikko Valtanen, Tommi Härkänen, Matti Uusitupa, Jaakko Tuomilehto, Jaana Lindström and Kari Auranen in Statistical Methods in Medical Research
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MV was financially supported by the EXACTUS programme in the University of Turku Graduate School (UTUGS).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JT owns shares in Orion Pharma, Oriola, Aktivolabs LTD and Digostics LTD. The other authors declare no conflict of interests.
Data availability
Supplemental materials
Supplemental materials are available online.
Appendix A: Identification of causal effects
The assumed causal DAG (Figure 1 in the main text) implies the following conditional independencies:
In addition, we make the consistency assumption stating that the observed outcome for an individual with given treatment, mediator and treatment-dependent confounder is equivalent to the potential outcome we would have observed, had the individual been assigned those values for the treatment, mediator and intermediate confounder. This means, for example, that
In what follows, we first derive the expressions for the expectation of the nested counterfactual
Invoking the assumptions 1–5 along with the consistency assumption (denoted by ‘c’) and using factorisation of probabilities (denoted by ‘f’), the expression for
Following the derivation given in the supplement of Tchetgen Tchetgen and Vanderweele,
15
by applying the additive decomposition for
Estimates of the causal effects can be obtained by estimating the components comprising the empirical formula. In the presence of (non-informative) censoring, the correct estimation of the expectations of the restricted survival time requires that the censoring is dealt with in the estimation step. Similarly, the drop-out in the longitudinal process is required to be at most missing at random in order to enable unbiased estimation of the mediator trajectories.
Appendix B: Ordinal treatment-dependent confounder with K values
Assume
Let
Appendix C: Step monotonicity
In this section, we describe a special case of the monotonicity assumption referred to as step monotonicity. We show that under step monotonicity, the joint probability of the counterfactuals of the treatment-dependent counfounder is identified and its consistency with the observed data can be empirically assessed. Let
Denote the row totals corresponding to the marginal probabilities