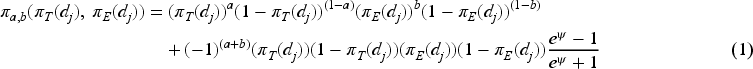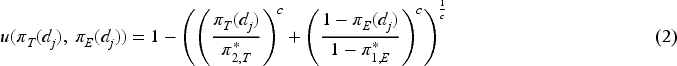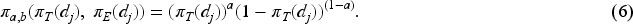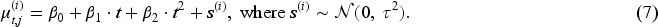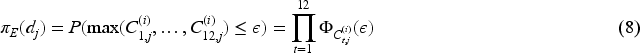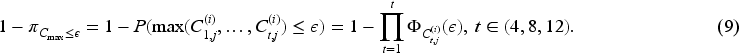Abstract
Dose-finding trials are designed to identify a safe and potentially effective drug dose and schedule during the early phase of clinical trials. Historically, Bayesian adaptive dose-escalation methods in Phase I trials in cancer have mainly focussed on toxicity endpoints rather than efficacy endpoints. This is partly because efficacy readouts are often not available soon enough for dose escalation decisions. In the last decade, ‘liquid biopsy’ technologies have been developed, which may provide a readout of treatment response much earlier than conventional endpoints. This paper develops a novel design that uses a biomarker, circulating tumour DNA (ctDNA), with toxicity and activity outcomes in dose-finding studies. We compare the proposed approach based on repeated ctDNA measurement with existing Bayesian adaptive approaches under various scenarios of dose-toxicity, dose-efficacy relationship, and trajectories of regular ctDNA values over time. Simulation results show that the proposed approach can yield significantly shorter trial duration and may improve identification of the target dose. In addition, this approach has the potential to minimise the time individual patients spend on potentially inactive trial therapies. Using two different dose-finding designs, we demonstrate that the way we incorporate biomarker information is broadly applicable across different dose-finding designs and yields notable benefit in both cases.
Introduction
Background
Early phase dose-finding trials, including both phase I and phase II studies are largely focussed on defining the dose and schedule of drug therapy for late-phase, pivotal trials. The traditional phase I oncology dose-finding paradigm based on toxicity without using efficacy has some undesirable consequences that the chosen dose may fail to consider the trade-off of toxicity and efficacy. 1 Recently, there has been a significant transformation in the paradigm of drug development in oncology to integrate Phase I and Phase II trials so that drug development process may be accelerated while potentially reducing costs.2–4
Subsequently, the objective of the dose-finding trials is to find the optimal biologic dose (OBD) of a treatment. The concept of OBD was introduced to account for efficacy in addition to toxicity. By efficacy or activity, it normally indicates a desirable clinical outcome, which may be a composite of several events that can be scored soon enough after dose administration for adaptive decision-making to be done feasibly. For instance, this could indicate a special case of ‘activity’, such as more than 50% shrinkage of a solid tumour, engraftment of a stem cell transplant, or the resolution of an infection. By toxicity, it refers to acute events or side effects after the delivery of the study treatment.
The FDA also actively seeks ways to reform dose optimization and dose selection paradigm in oncology drug development. For instance, the Oncology Center of Excellence launched an initiative known as Project Optimus, 5 emphasizing a core change in the dose selection that maximizes not only the efficacy of a drug but the safety and tolerability as well. Consequently, seamless phase I/II approaches allow for a more rapid and efficient transition in early phases. 6
Seamless phase I/II designs adaptively use the (dose, efficacy, and toxicity) data from all previous patients to make decisions and select the best dose for subsequent patients. Several methods have been proposed to achieve a balance between drug safety and efficacy via a trade-off.7–10 One of these approaches is the EffTox design, a Bayesian adaptive dose-finding design that models correlated binary efficacy and toxicity via a joint probability function. 11 The utility of a dose with associated posterior efficacy and toxicity probabilities measures its attractiveness and guides dose selection decisions. There are also designs where both toxicity and efficacy are independently accounted for.12,13
Despite the major advances in the methodology, Bayesian models for early-phase clinical trials have seen limited use in clinical practice.14–17 One of the practical impediments in adaptive clinical trials is that outcomes must be observed soon enough to apply decision rules to choose treatments for the next patients. In phase I, there are proposals to address the problem of delayed late-onset toxicity.18–22 In seamless phase I/II trial designs, there are few approaches to the problem of late-onset toxicities and efficacies. One of the existing approaches investigated a joint time to event model with delayed onset outcomes for both safety and efficacy.13,23–27 Data augmentation for delayed outcomes, which are regarded as missing data, is also considered.26–28 It seems to be more challenging in seamless phase I/II trial designs if efficacy, for instance, current clinical trials with traditional RECIST-based endpoints for efficacy, is not assessed as quickly as toxicity. The trial would then proceed at the speed determined by the efficacy outcome with the longest assessment period, and there would be an increased risk of incomplete data when one outcome is assessed and the other is waiting to be assessed. A possible solution is to use a shorter-to-evaluate activity proxy endpoint for the decision-making, where early readouts of drug response can be coupled to adaptive decision schemes. Different cases of a ‘good’ and a ‘bad’ surrogate were discussed,29,30 where the proposed trivariate binary model is supposed to utilize all the available data at any given time point.
In parallel, clinical trial designs of biomarker research in oncology have been popular. 31 Mutations in cell-free DNA are highly specific markers for cancer, and this gave rise to the term ‘circulating tumour DNA (ctDNA)’, 32 which was explored as a prognostic or predictive marker,33,34 and for cancer detection. 35 In the last decade, ‘liquid biopsy’ technologies have been developed, which allow the detection of low levels of ctDNA present in plasma. 36 In addition, there are initiatives which aim to establish international standards for ctDNA response criteria. 37 This promotes the use of ctDNA as a promising biomarker that can obtain quantitative and qualitative comprehensive tumour DNA in a minimally invasive manner. These assays could potentially be repeated at multiple time points during the course of a patient’s treatment to provide a faster and more accurate ‘real-time’ assessment of treatment response for cancer patients on a wide range of therapies. It has been shown that in some cases – early changes in ctDNA – can predict subsequent response to treatment.38–41 In addition, it has been shown in several trials that early ctDNA change in response to treatment correlates with either disease-free survival or progression-free survival.42–44 This allows for the incorporation of novel early response or activity endpoints based on liquid biopsy-directed approaches in dose-finding studies.
In this work, we build on the EffTox approach and seek to evaluate the use of a rapidly available biomarker, ctDNA, in dose-finding studies. Specifically, we propose the Biomarker-informed EffTox (BMI-EffTox) approach, which considers additional information from continuous ctDNA measurements, and compares the BMI-EffTox and EffTox approach via a comprehensive simulation. The robustness to variability of the ctDNA values and its different trajectories are evaluated by sensitivity analyses. Wages and Tait 12 introduced an early-phase method for trials investigating targeted agents, which falls into a broader class of extended model-based designs. To highlight the general ability of our proposal across various dose-finding designs, we extend our biomarker-informed approach to the Wages and Tait design. 12 We do not intend to compare the EffTox design with the Wages and Tait design, as they target different objectives and follow distinct methodologies. Instead, our goal is to highlight the benefits that the proposed biomarker-informed approach can offer to both designs, which take fundamentally different approaches to modelling.
Motivation
Figure 1 illustrates the late-onset problem in an oncology trial, where there are three cohorts of patients with three patients in each. With a 28-day treatment cycle (i.e., four weeks), a cohort of patients is recruited every treatment cycle, at
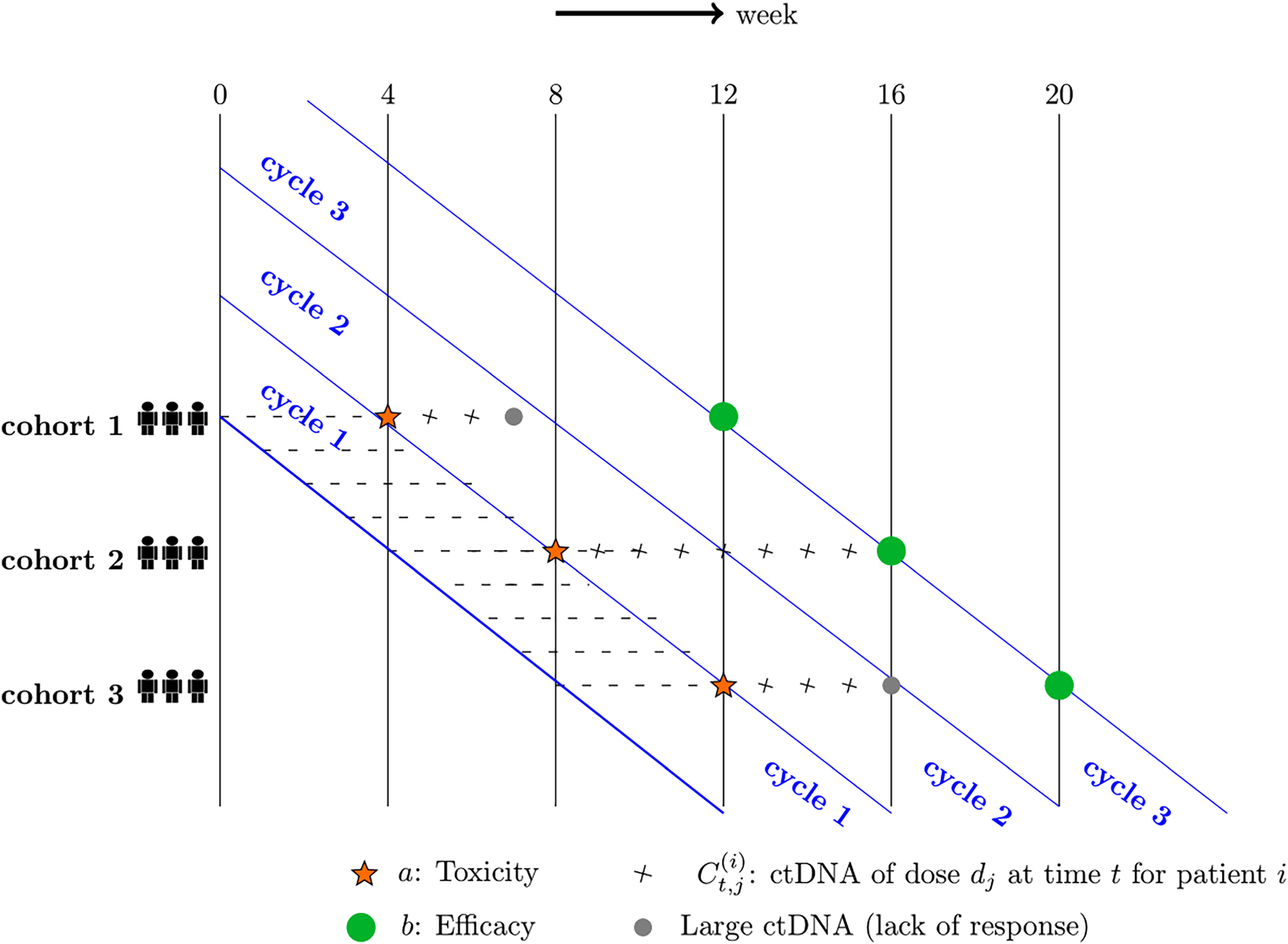
Late-onset problem in dose-finding studies.
In Figure 1, cohort 1 is firstly recruited, corresponding binary toxicity outcomes
One of the challenges of phase I/II trials arises from the fact that efficacy is not assessed immediately over a similar time horizon as toxicity and takes significantly longer to be evaluated. In the example in Figure 1, a patient with no toxicity event (i.e.,
There is more than one way to use ctDNA values before week 12, and the example in Figure 1 uses large ctDNA on an absolute scale as an indicator of ‘lack of response’. That is, if corresponding values are sufficiently large, the treatment will be deemed not useful and the patients can move to another trial/treatment before assessing efficacy endpoints. In Figure 1, ctDNA values of patients from cohort 1 and cohort 3 can be used as an indicator of ‘lack of response’ as they are large enough in this example. However, this is not the same case for patients from cohort 2. In this case, outcomes of cohort 1 can impact dose-decisions for cohort 2 and later. Thus the use of ctDNA data can help with the late-onset problem, and rapidity and efficiency of dose-finding studies can be improved.
The use of a shorter-to-evaluate activity proxy before week 12 can offer at least the following benefits: (i) The next enrolled patients can be assigned to better doses based on more information from early readouts of ctDNA; (ii) investigators can expect a faster trial; (iii) the current patients can benefit from early stopping of treatment and turn to some alternatives instead.
As our proposal of dose-finding designs builds on the EffTox approach, 11 we begin by describing it in detail first in Section 2.1. This is then followed by the introduction of the newly proposed Biomarker-Informed EffTox approach in Section 2.2. The integration of ctDNA into the Wages and Tait design 12 is described in Section 2.3. This approach employs relatively simple models and does not require prior specification for a large number of parameters.
EffTox approach
The EffTox approach is a Bayesian adaptive dose-finding design that models correlated binary efficacy and toxicity outcomes, which aims at facilitating seamless phase I/II dose-finding. It uses Bayesian models for the marginal probabilities of efficacy and toxicity at each dose and utility contours to measure the attractiveness of each dose based on the posterior probabilities of efficacy and toxicity. 45
For
To select the dose with relatively higher efficacy and lower toxicity for the next cohort of patients, utility is defined as,
A likelihood-based approach could be employed, where parameters
Following the original proposal,
11
we use a Bayesian approach to estimate the model parameters. Specifically, accumulated data about toxicity and efficacy is used to update the belief about parameters
The idea of introducing regularly available (e.g., daily, weekly or monthly) ctDNA values in dose-finding studies is achieved by regarding these early readouts as a proxy for ‘not-yet-available’ efficacy outcomes. That is, it is possible that the decisions about which dose to administer could be based on toxicity outcomes and ctDNA values when definitive efficacy outcomes are not yet observed.
There is no standard way to use ctDNA as a biomarker for the evaluation of treatment response in various contexts in oncology. We choose to use ctDNA as an early indicator of ‘lack of response’ in terms of efficacy as in Equation (5). This one-way information approach prevents being overly optimistic about efficacy of the administered dose and a dose cannot be deemed as ‘active’ based on ctDNA only. Consequently, higher doses are more likely to be explored, dose-escalation is more likely to be encouraged and patients are less likely to be stuck on an inactive treatment for a long time.
Regular ctDNA values are evaluated from week 4 onwards after finishing a complete treatment cycle and toxicity assessment at week 4 and week 8. In the case of ‘lack of response’ based on large ctDNA values, dose selection for the next cohort of patients can be made based on current data of toxicity and ctDNA values. While in the case of no such evidence, if ctDNA values are relatively small, the corresponding decision of dose selection will be based on only toxicity outcomes of the current patients. In this case, the corresponding marginal model shown in Equation (6) is the likelihood when there is only toxicity (See the proof in Appendix A):
In the example of Figure 1, seven weeks after recruitment of cohort 1, there is evidence of ‘lack of response’. Dose selection for cohort 2 patients enrolled at week 8 can be made based on toxicity
In contrast to the plug-in estimate for utility based on mean values of posterior efficacy and toxicity probabilities of the doses, we consider the posterior distribution of utility scores instead. That is,
Overall, the BMI-EffTox with additional information from ctDNA values is linked to the EffTox approach in the way that the response outcomes are replaced by early readouts of ctDNA in the case of ‘lack of response’. In the EffTox, decision-making based on utility defined in Equation (2) is conducted at week 4 if the event of toxicity happened, otherwise current patients will have to wait for efficacy outcomes until week 12 (see Figure 1). In the BMI-EffTox, selection of the OBD can occur at week 4 and week 8 in the latter case, given there is evidence of ‘lack of response’ from ctDNA. The simple replacement of the long-term, definitive efficacy data with the ctDNA data can be an effective way considering that ctDNA has been shown to be a promising biomarker.
In summary, our proposed BMI-EffTox approach for dose-finding designs is as follows:
Assign the first cohort of patients to the lowest dose level
Update the posterior distributions of the probabilities of toxicity and efficacy at all dose levels. Check the admissibility of all the doses. The trial stops if there is no admissible dose. Early stopping can occur if no dose satisfies the toxicity, efficacy, or both criteria. If there is at least one dose satisfying the admissibility criteria, assign the next cohort of patients to the dose with the maximum value of utility among admissible doses. Dose skipping is not permitted. Repeat from Step 2 to Step 4 until the trial is terminated early or the maximum sample size is achieved.

The original proposal by Wages and Tait was evaluated under the setting were both safety and activity data is available at the same time. As this setting does not allow for early ctDNA data to inform dosing decisions, we consider Wages and Tait’s approach when the late-onset problem (Section 1.2) is present. Henceforth, we will refer to the original proposal with the existence of the late-onset problem as WagesTait and its biomarker-informed version as biomarker-informed WagesTait (BMI-WagesTait).
In general, the WagesTait aims to assign each patient cohort to the dose estimated to be most efficacious among those with acceptable toxicity levels. This design assumes a functional dose-toxicity curve through a power model, which is common in the CRM literature. The skeleton
Our proposed BMI-WagesTait approach, developed using the same principle as for the BMI-EffTox designs, is outlined as follows:
Assign the first cohort of patients to the lowest dose level Identify the set of acceptable
Check the safety based on exact binomial confidence interval ( If there is at least one dose satisfying the both criteria, assign the next cohort of patients to the selected dose. Dose-skipping rule is allowed as in the original proposal. Repeat from Step 2 to Step 4 until the trial is terminated early or the maximum sample size is achieved.
Instead of defining the OBD defined based on utility, as in EffTox approach described in Section 2.1, Wages and Tait
12
employ the concept of ‘best dose’ and ‘good dose’ to evaluate the design, as introduced by Hoering et al.
48
In this framework, the best dose is defined to maximize efficacy while assuring safety, whereas a good dose is one that meets a predefined efficacy threshold and maintains safety.
Numeric evaluation
To assess the performance of the biomarker-informed approach relative to the original designs, we conduct a comprehensive simulation study across various dose-toxicity, dose-response scenarios, and potential ctDNA trajectories.
Data generation
We consider a study with four doses,
Measurements of ctDNA are evaluated every week, while decision-making is assumed to be every four weeks as in Figure 1. In this case, the duration of treatment for patients with no toxicity outcomes (
We evaluate absolute values of ctDNA for convenience in this paper, while it is also possible to evaluate the change of ctDNA values before and after the treatment on a relative scale in clinical practice. We use
In Equation (7),
For the relationship between ctDNA
where
We begin by considering this case initially to establish the maximum potential benefit of the BMI-EffTox approach. For instance, the definitive efficacy outcomes will never turn to efficacy result once early evidence from large ctDNA values indicates ‘lack of response’. The robustness of the design to the assumption of the perfect predictability of the ctDNA in Equation (8) is explored in Section 4.
Table 1 gives a summary of all relevant parameters for different approaches, which follows the practical guidelines for EffTox in the implemented Matchpoint trial. 45 For WagesTait, we primarily use the same parameters; for those that cannot be reasonably matched, we provide specific details below.
Chosen parameters for the simulation study.
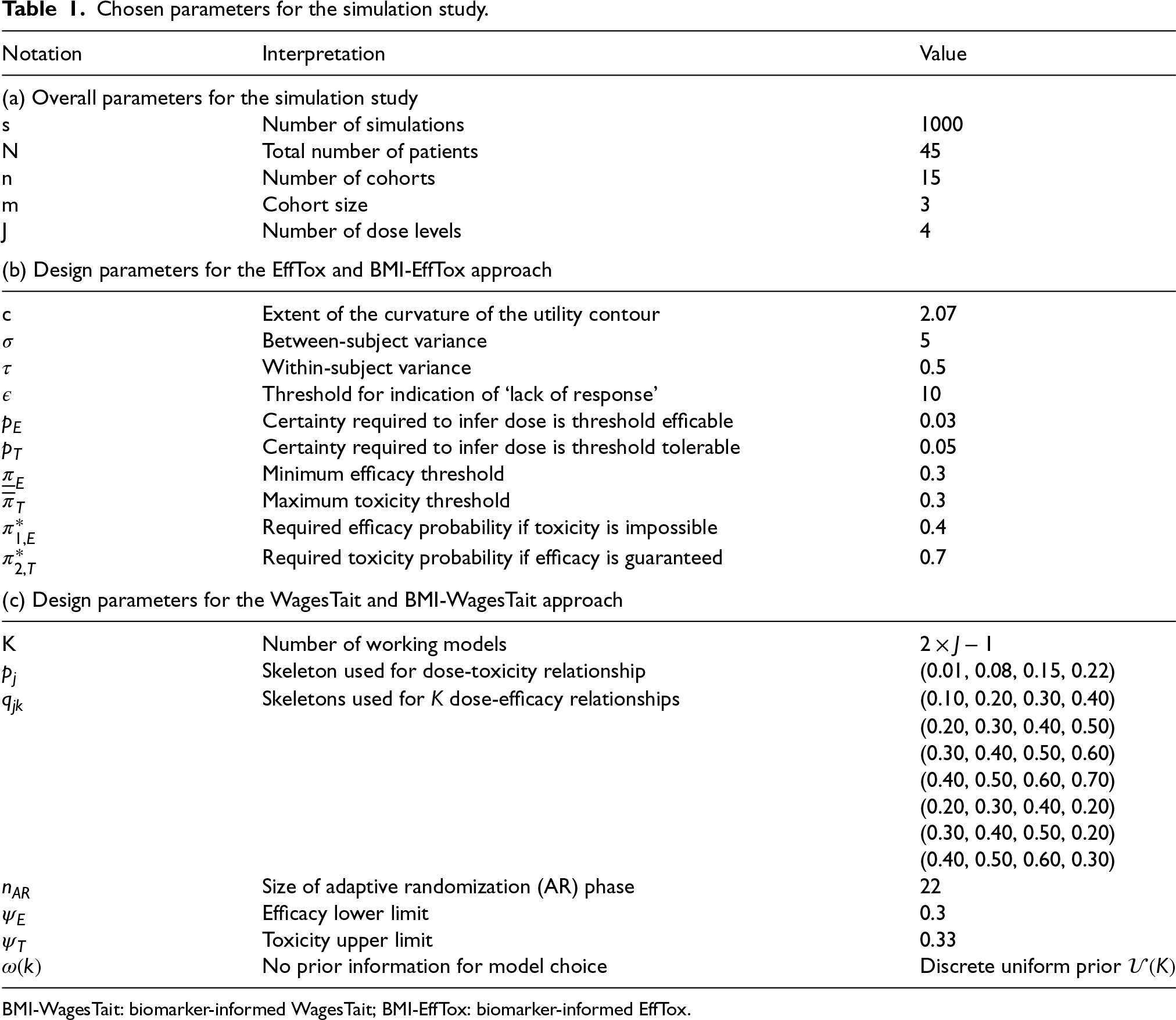
Chosen parameters for the simulation study.

BMI-WagesTait: biomarker-informed WagesTait; BMI-EffTox: biomarker-informed EffTox.
For the Efftox and BMI-EffTox approaches, the prior joint distributions for the parameter vectors of
For the WagesTait and BMI-WagesTait approaches, a normal prior with mean 0 and variance 1.34 is utilized for models for toxicity and efficacy, which can be traced back to some early work.
49
Besides, skeletons for both toxicity and efficacy need to be specified. For working models about efficacy, a prior
For the Efftox and BMI-EffTox approaches, the maximum toxicity threshold is set to
Scenarios investigated in terms of dose profiles of toxicity
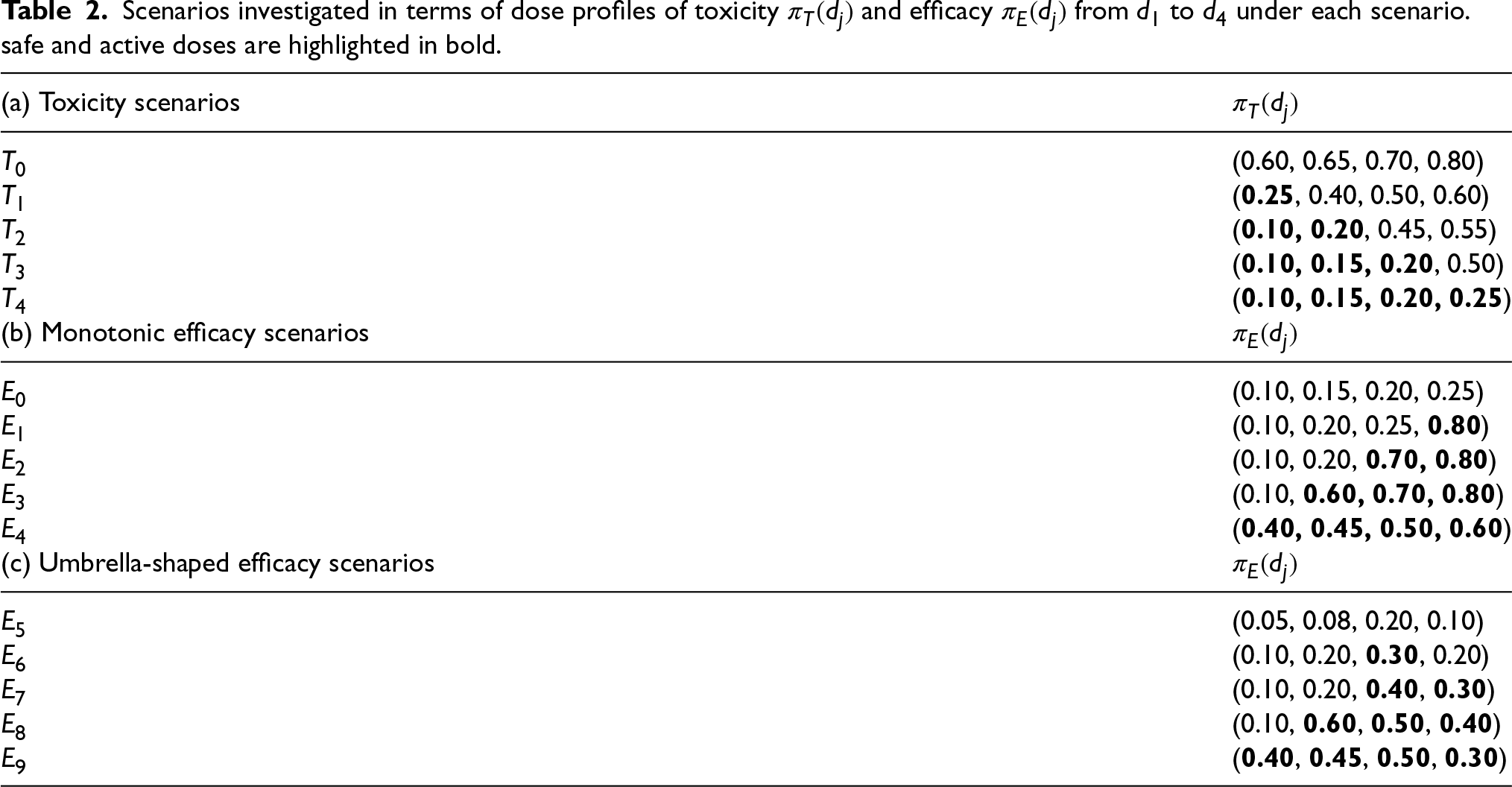
Considering both toxicity and efficacy, a specific scenario is denoted as
For data generation of
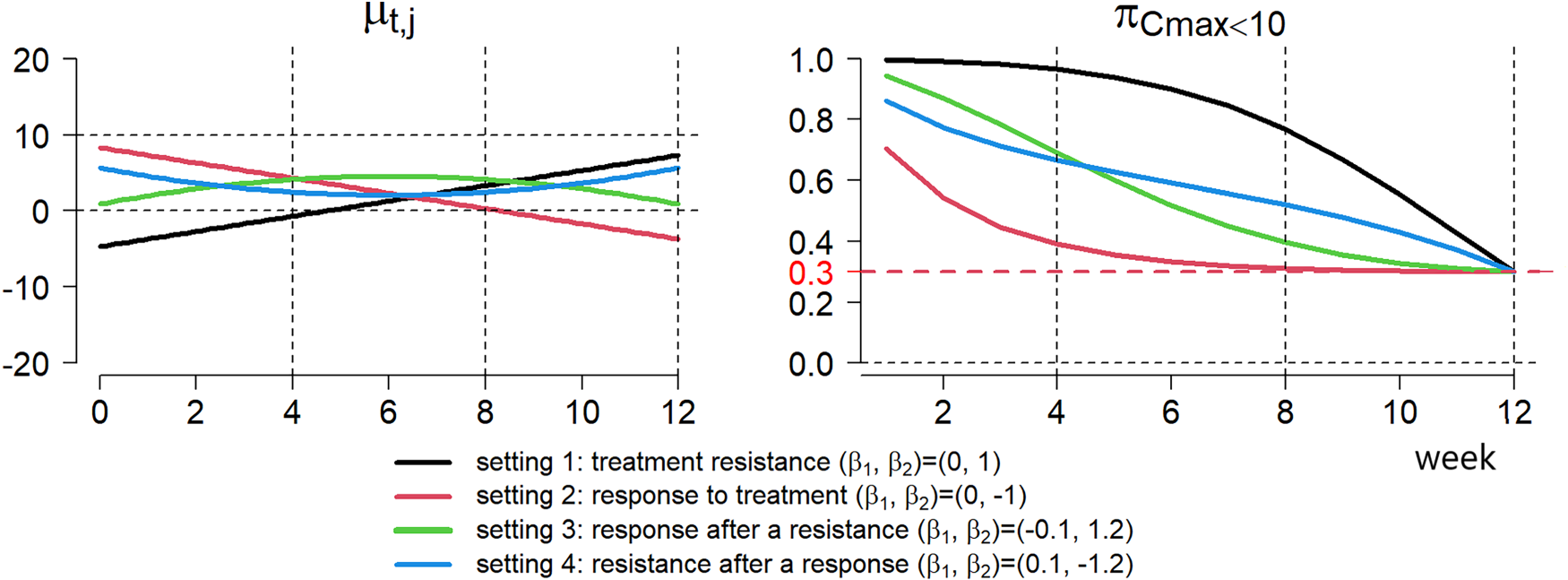
Trajectory of
Corresponding values of
The contribution of ctDNA to dose-finding studies is also intuitively reflected by the changing pattern of
To explore if decision-making for dose escalation could be improved by additional information based on ctDNA values, we focus on different metrics of the simulation results: (i) Proportion of recommendation per dose reflects the distribution of dose recommendations at the end of a trial. It also reflects how dose recommendations shift from one to another; (ii) Proportion of correct selection (PCS); (iii) Proportion of early stopping evaluates the probability that a trial ends up with an early stopping when the specified constraints defined in Equation (3) and Equation (4) are not satisfied; (iv) Duration of the treatment reflects the average number of weeks that a patient is on a given dose (see Section 1.2); (v) Proportion of toxicity reflects the possible harm on patients.
Among these metrics, the PCS and the proportion of early stopping correspond to the proportion of correct decisions, depending on whether it is a scenario with an OBD. Dose-finding studies under a scenario with an OBD would expect a higher PCS; otherwise, a higher proportion of early stopping is anticipated. Simulation results of the duration of treatment can reflect the rapidity of a trial, which is expected to be facilitated if there is no OBD. The proportion of toxicity is examined to ensure that we do not lose too much in terms of the win of efficiency of dose-finding studies.
For the WagesTait and BMI-WagesTait approaches, the PCS for both best and good dose(s) are represented. In addition to the proportion of correct decisions, the proportion of toxicity is considered. The best dose and good dose(s) for each scenario are shown in Table 5 and Table 6 in Appendix C.
Simulation results
We investigated simulation results under all combinations of scenarios of dose-toxicity and dose-response relationship shown in Table 2, which are displayed in terms of metrics introduced in Section 3.3. We consider the average value over all settings mentioned in Figure 2 for generalization .
Proportion of recommendation per dose and proportion of correct decision
Figure 3 illustrates simulation results of the average difference after introducing ctDNA in the BMI-EffTox with regard to the proportion of recommendation for scenarios under the assumption of monotonic efficacy curves as shown in Table 2b. The way to read Figure 3 is that negative values (i.e., decreases in the proportion of recommendation via the BMI-EffTox) in all doses are expected under scenarios with no OBD (i.e., grey background) as early stopping is expected in this case. While a positive value in the OBD (i.e., a larger PCS via the BMI-EffTox) is expected under scenarios with an OBD (i.e., white background). For scenarios with an OBD, recommendations are shifted from lower doses to higher doses, which are assumed to be more active. This effect is particularly observed under scenarios with
However, for scenarios with
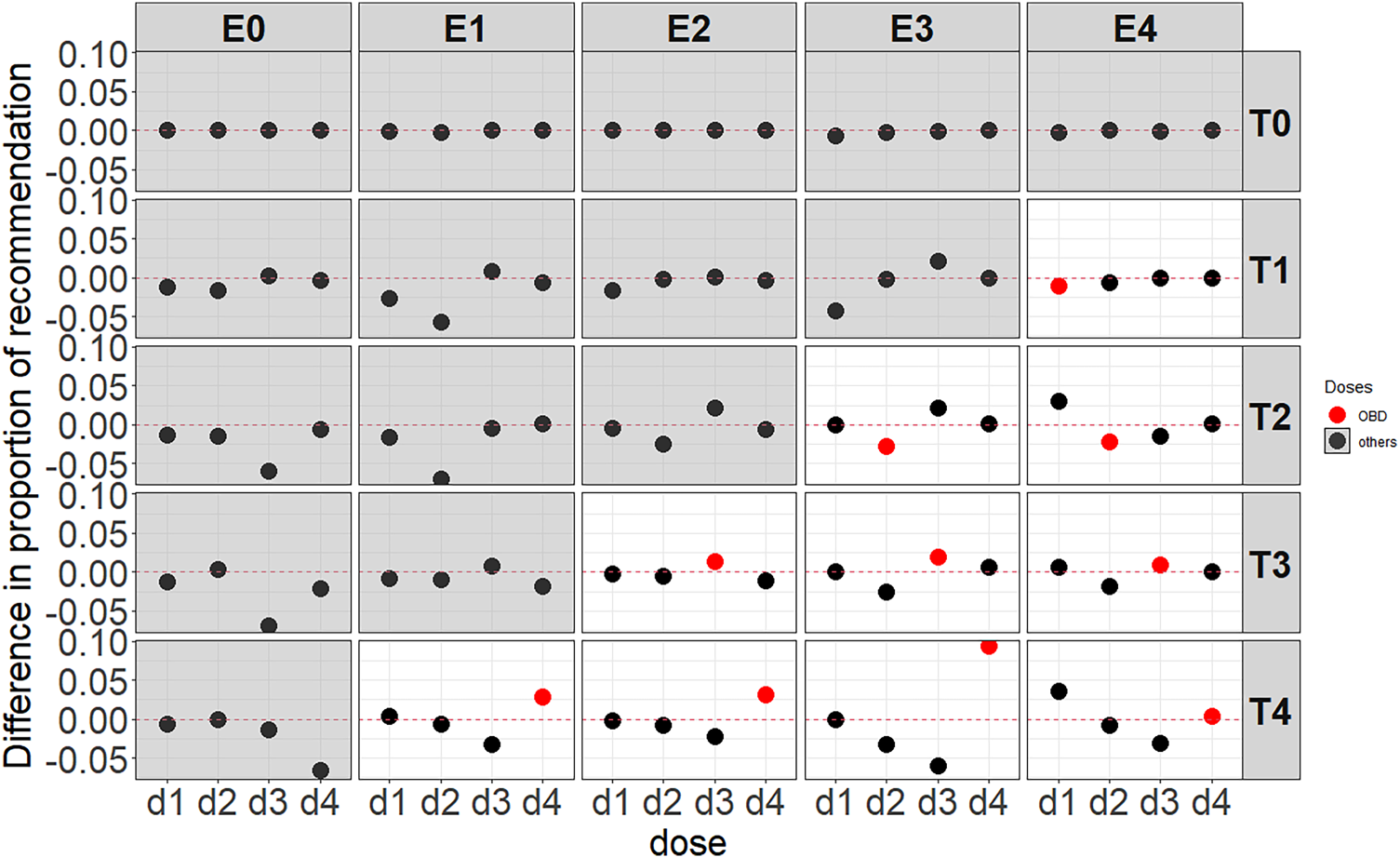
Average difference of the BMI-EffTox from the EffTox in the proportion of recommendation under monotonic efficacy scenarios with a fixed
In Figure 3, as a result of the shift of recommendations among different doses after introducing ctDNA, the PCS, which is referred to as a selection of the OBD defined in Section 2.1 is improved in most cases. This is particularly obvious when doses are not constrained too much (i.e., scenario
For scenarios without an OBD, recommendations of some doses are prevented as a result of more early stopping. In clinical practice, stopping rules are commonly imposed, such that a trial can be stopped when any of the safety constraint (i.e., Equation (3)) or efficacy constraint (i.e., Equation (4)) cannot be satisfied. A trial is more likely to stop early under the BMI-EffTox approach where there are some early readouts of ‘lack of response’ results based on large ctDNA. This desirable property leads to more early stopping under almost all of the scenarios without an OBD as shown in Figure 3 (exact values are included in Appendix E). For instance, under scenario
Simulation results in Figure 4 below illustrate the scenarios under the assumption of umbrella efficacy curves as shown in Table 2c. In scenarios where an identifiable OBD exists, most cases show an increase in dose recommendations for the OBD. The PCS under scenarios with
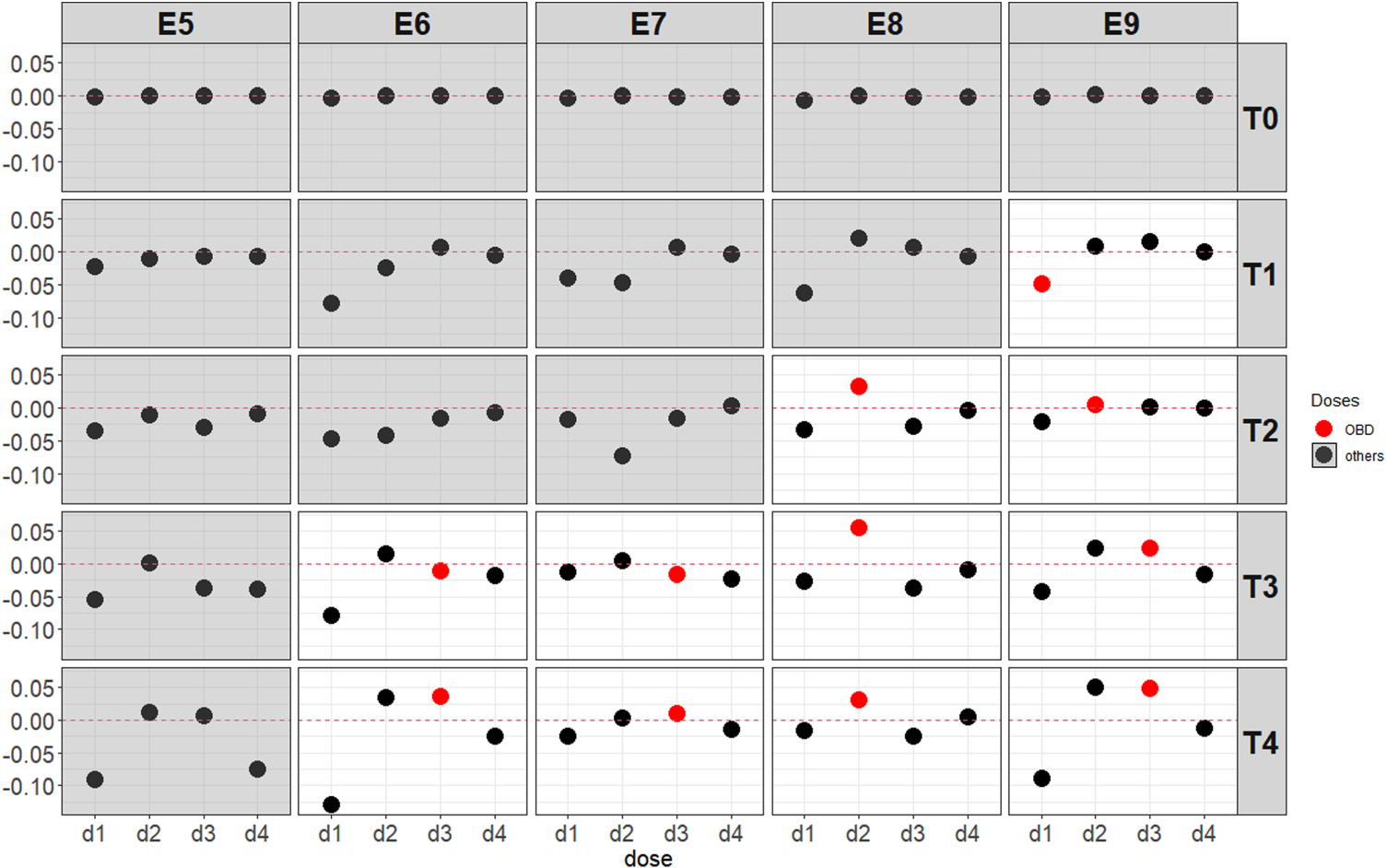
Average difference of the BMI-EffTox from the EffTox in the proportion of recommendation under umbrella-shaped efficacy scenarios with a fixed
For the EffTox approach, ctDNA encourages the dose-escalation procedures when appropriate, often leading to the recommendation of OBD by the end of the trial. This encouragement on the basis of ctDNA is more obvious under scenarios with minimal safety constraints (i.e., all doses are safe, namely, scenarios with
For the WagesTait approach, Table 3 reveals similar patterns to those observed in the EffTox versus BMI-EffTox comparison (see Figure 3 and Appendix E). That is, the BMI-WagesTait improves the proportion of correct decision under most of the cases. When there is neither best dose nor good dose(s), the BMI-WagesTait approach tends to reach early stopping more frequently than the WagesTait method. For instance, the introduction of ctDNA information leads to 5% more simulations resulting in early stopping in scenarios
Average difference of the BMI-wagesTait from the wagesTait in the proportion of correct decision (
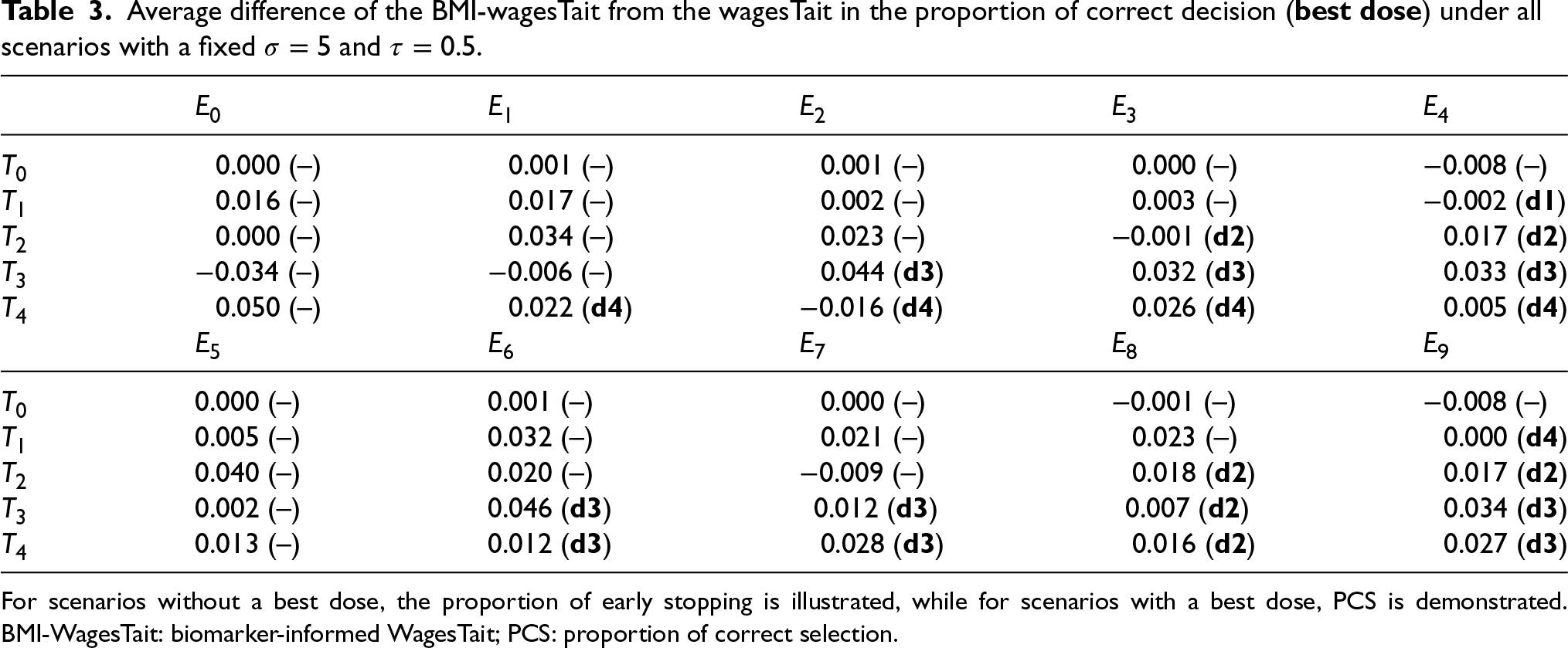
For scenarios without a best dose, the proportion of early stopping is illustrated, while for scenarios with a best dose, PCS is demonstrated. BMI-WagesTait: biomarker-informed WagesTait; PCS: proportion of correct selection.
Overall, the biomarker information supports both the EffTox and WagesTait models in a similar manner, contributing similarly to improved decision in both designs. Across different baseline designs, ctDNA leads to a maximum 8% increase in early stopping and a 10% improvement in the proportion of correct decisions (PCS) in the BMI-EffTox model. In the BMI-WagesTait model, ctDNA improves early stopping by up to 5% and PCS by up to 4.6%. Additional findings on the proportion of correct decision for good dose are provided in Appendix C, showing rather similar conclusions. We find that the advantage of incorporating ctDNA remains consistent in identifying both the best and good doses.
Duration of treatment
For the rapidity of dose-finding studies, the duration of treatment is reduced after introducing ctDNA (see Figure 5), which allows more early stopping of doses showing evidence of the ‘lack of response’ discussed above.
The duration of all safe doses is fixed at 12 weeks in the EffTox approach as discussed in Section 1.2. Figure 5 illustrates the distribution of the duration of doses under all of the investigated scenarios via the EffTox and the BMI-EffTox approaches. In general, there is a reduction of the duration of treatment in the BMI-EffTox in the cases of all doses under all investigated scenarios. This is particularly observed in doses with smaller
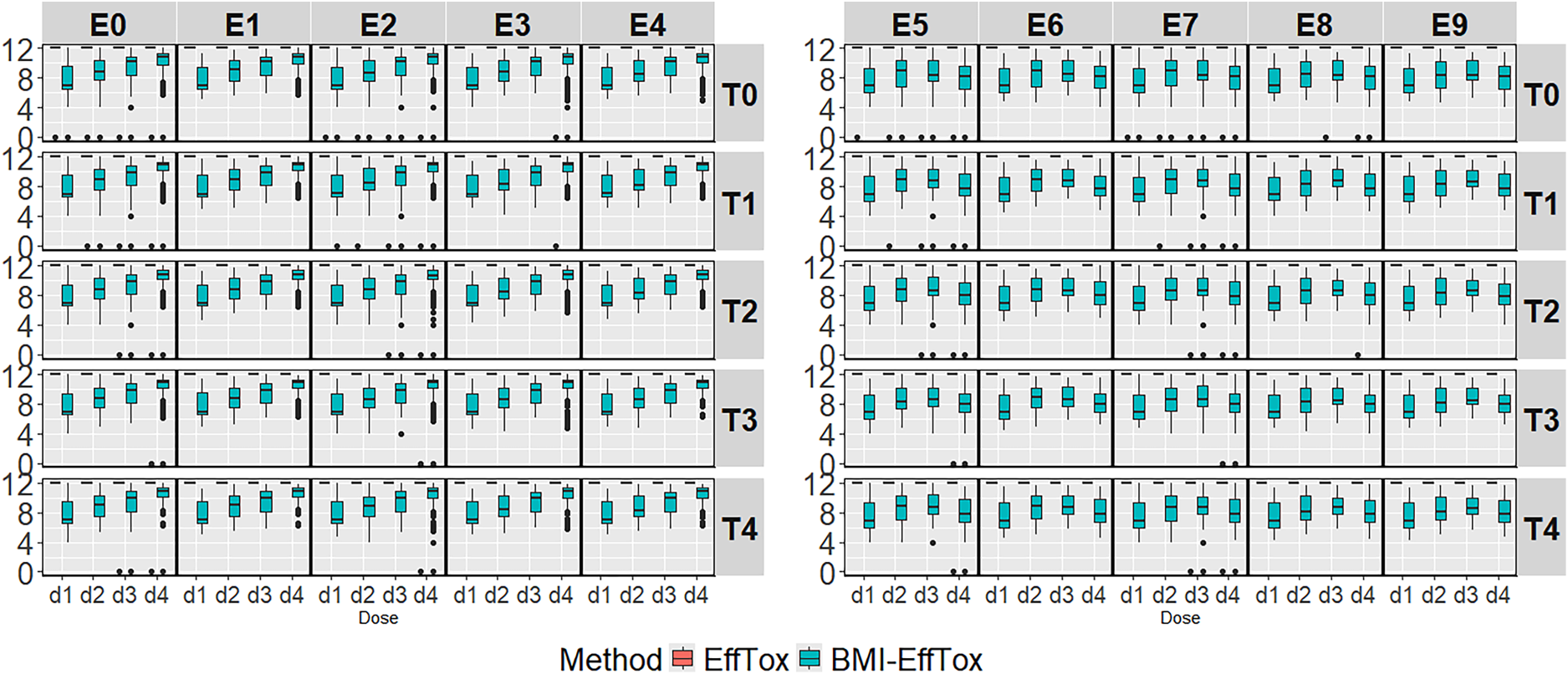
Treatment duration for the biomarker-informed EffTox (BMI-EffTox) and EffTox models across monotonic and umbrella-shaped efficacy scenarios with a fixed
The procedure of dose-escalation might be accelerated based on the results of the ‘lack of response’, and ctDNA might encourage the exploration of higher doses. However, it might happen that an active dose leads to high values of ctDNA occasionally and is mistakenly deemed as an inactive dose. Current patients on this dose might suffer from the wrong early stopping of the treatment and lose the chance of staying on effective treatment. Meanwhile, a higher dose, which is potentially more active but more toxic, might be recommended to the next cohorts.
Simulation results of the proportion of correct decisions and the duration of treatment show the gains in rapidity and efficiency in dose-finding studies. The process of dose escalation is governed by a fundamental conflict since both the therapeutic aim and the safety concern should be considered simultaneously for cancer patients. On the one hand, there is a need to go slowly in order to avoid a sudden jump from no observable toxicity to a lethal dose. For instance, a slow dose-escalation procedure is preferred to avoid an overdose if OBD is at a lower dose. On the other hand, there is a need to go rapidly so that large numbers of patients are not treated at ineffective doses.
To explore whether the encouragement of the dose-escalation procedure by the BMI-EffTox is always tolerate, simulation results of the proportion of toxicity are evaluated. Under all of the investigated scenarios, the proportion of toxicity is similar between the EffTox and the BMI-EffTox and between the WagesTait and the BMI-WagesTait as shown in Appendix G. Large increases in proportion of toxicity are rarely observed after introducing ctDNA, suggesting that ctDNA can be safely integrated into dose-finding studies.
Above, it was assumed that binary efficacy response and ctDNA are perfectly correlated to understand the largest benefit the biomarker-informed approach can attain over the original proposal. In this section, we relax this assumption that the prediction of ‘lack of response’ based on ctDNA might be either an underestimation or an overestimation of the real value of
To explore the contribution of ctDNA when relaxing the assumption of perfect predictability of ctDNA to the efficacy, we generated ctDNA based on
In Figure 6, the introduction of ctDNA in the BMI-EffTox encourages early stopping under scenarios without an OBD. While in the case of scenarios with an OBD, the PCS is improved in most cases regardless of
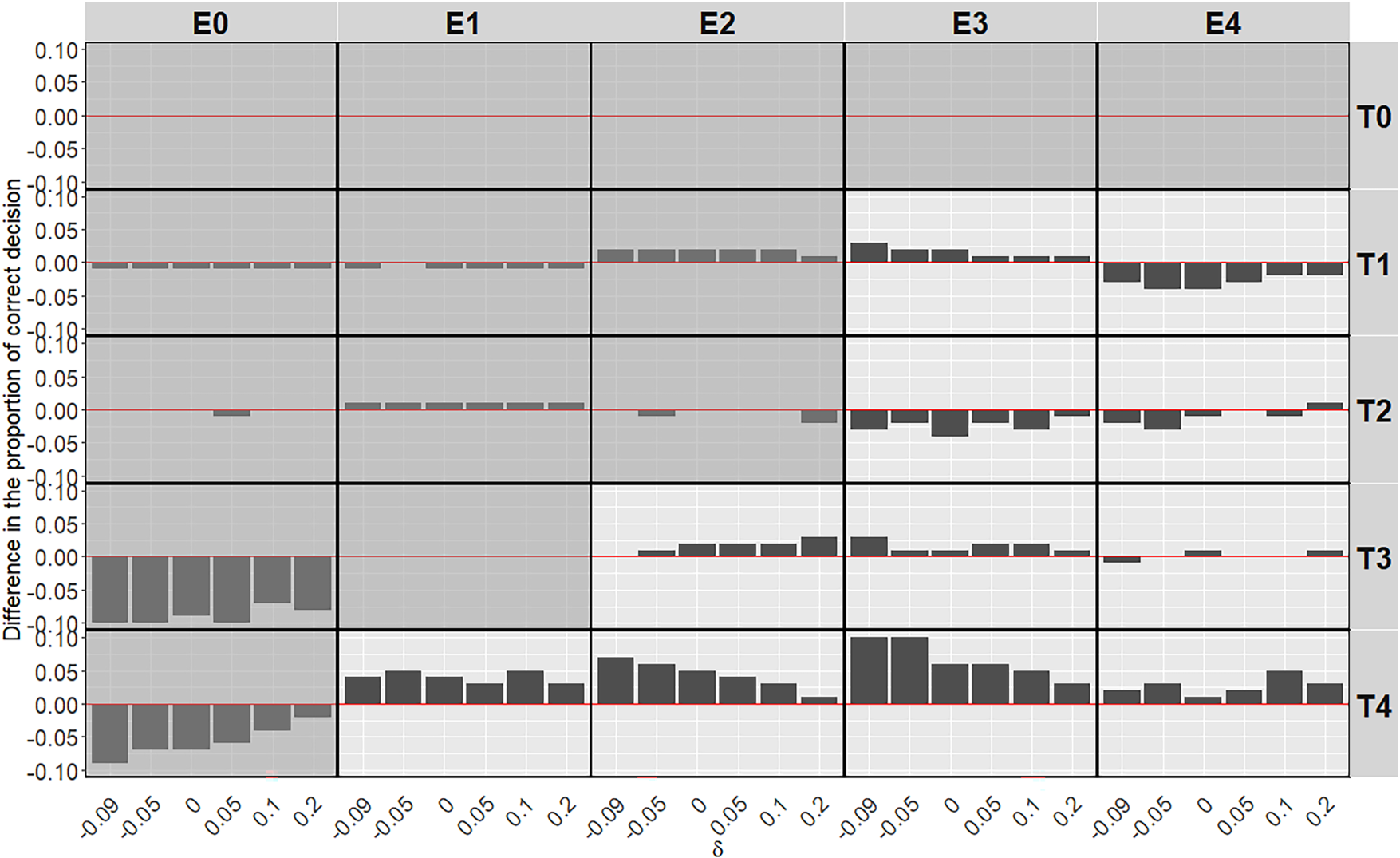
Average difference of the BMI-EffTox from the EffTox in the proportion of correct decisions under monotonic efficacy scenarios with different
For scenarios with
For scenarios with
For the proportion of early stopping under scenarios without an OBD, the benefit of more frequent early stopping is particularly obvious when there is an underestimation in
The reduction of the duration of treatment in the BMI-EffTox happens under all scenarios regardless of the value of
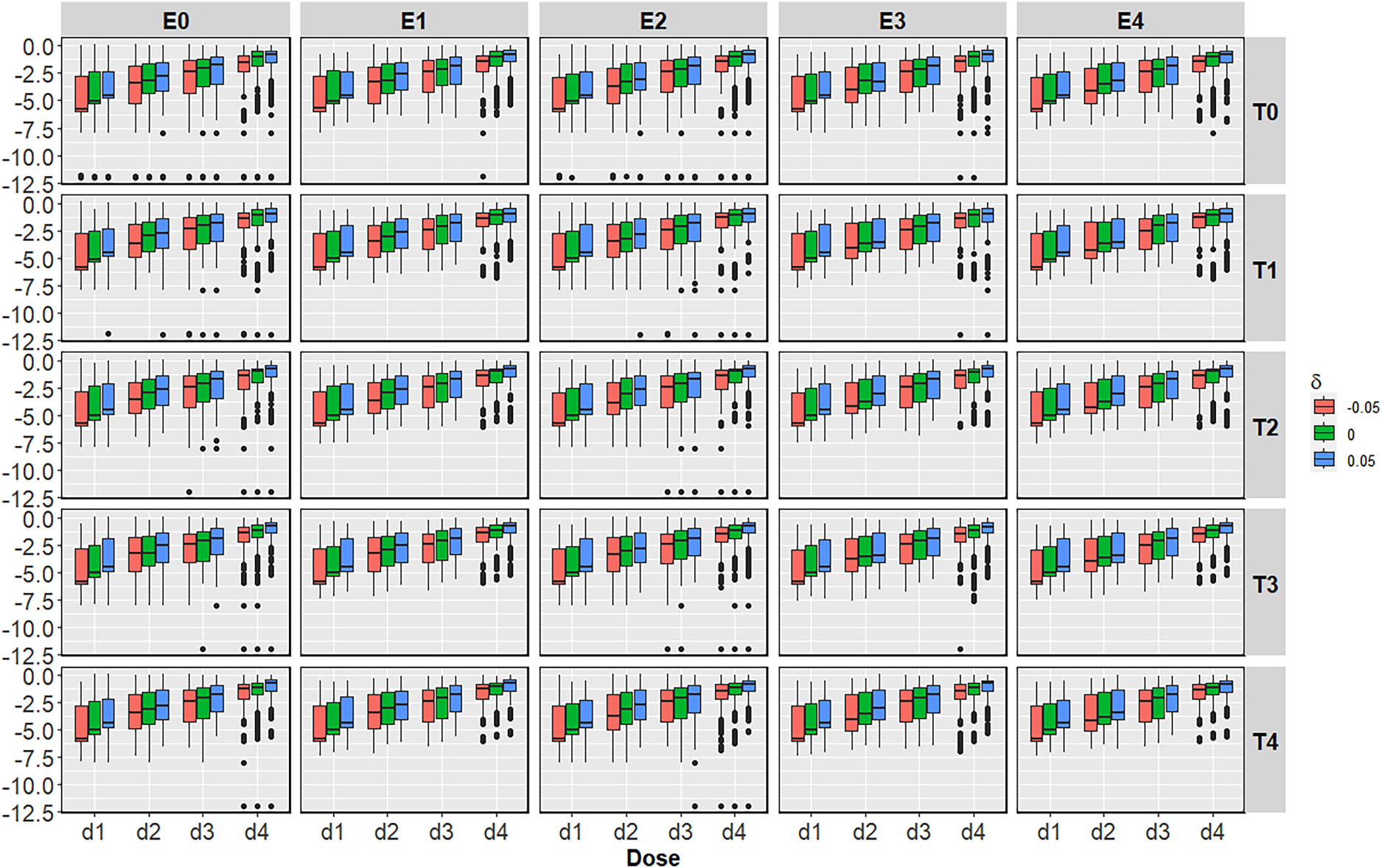
Treatment durations for the biomarker-informed EffTox (BMI-EffTox) and EffTox models under monotonic efficacy scenarios with
This study aimed to incorporate a novel efficacy biomarker, ctDNA, into dose-finding studies together with toxicity and efficacy data. The clinical context addresses the late-onset problem, with different observation windows for toxicity and efficacy outcomes. We compared the EffTox 11 and WagesTait 12 approaches with their biomarker-informed counterparts under various assumptions regarding dose-toxicity, dose-response scenarios, and possible ctDNA trajectories.
The pace of the uptake and implementation of adaptive dose-finding trial designs, such as the EffTox approach, have lagged behind advancements in the statistical literature introducing novel methodologies. Implementing the EffTox approach poses several challenges, including the specification of investigated doses, the determination of prior distributions for parameters, and the elicitation of targeted quantities from investigators and clinicians. Discussions on utility approaches and elicitation method can be found the in literature.8,50 Despite these practical challenges, our proposed method aims to utilize additional biomarker information to enhance the efficiency and speed of dose-finding studies. While we used the EffTox design as an example for incorporating biomarker information, similar ideas can be applied to any dose-finding approach that balances safety and efficacy. In response to reviewer feedback, we successfully integrated ctDNA into the WagesTait approach, which offers advantages in terms of model simplicity and ease of parameter specification. Simulation results demonstrate that the novelty of our approach lies in its independence from the actual model and its applicability to different existing methodologies.
We employed a Bayesian adaptive dose-finding trial design in the proposal. The likelihood-based approach would be more demanding in the sense of data configuration, particularly with an increasing number of parameters, as more data is required in the initial stage to initiate the model. For instance, two DLTs (dose-limiting toxicities) and two non-DLTs might be required in the first run-in stage. 51 Extensive discussions on the number of parameters and model complexity exist.52,53
Our simulation results show that additional information from ctDNA can significantly reduce the duration of treatment and increase the efficiency of trial design. In the former case, the duration of inactive doses is reduced tremendously more than that of active doses. The latter case includes the proportion of early stopping and the PCS accordingly. Other than the benefits obtained from using ctDNA, even with the encouragement of dose-escalation procedure after introducing ctDNA in the BMI-EffTox and BMI-WagesTait, there is no obvious increase in the proportion of toxicity. It shows that the incorporation of ctDNA leads to more rapid and efficient dose-escalation schemes. The improvement in rapidity and efficiency resulting from the additional ctDNA information of applies to scenarios characterized by either monotonic or umbrella efficacy curves.
We want to point out that there are some related works for similar settings, considering the impact of the level of surrogacy in the joint model. For instance, it was emphasized that the quality of surrogate markers, which is available at any given time point, is an important factor in finding an optimal dosage.29,30 Our work is more specifically targeted for the promising biomarker, ctDNA, thus different assumptions, such as the perfect predictability and the evaluation of various outcomes at different time points are distinct from related works. For this reason, a direct comparison of the performance between our approach and related work might not be sensible and fair enough.
In the case that ctDNA fails to predict efficacy outcomes perfectly, results show systematic bias corresponding to
The levels of ctDNA in different clinical contexts were not yet accurately defined, it is essential that the biology of ctDNA is identified and validated in advance. For the robustness of the proposed approach, we consider sensitivity analysis in this paper: (i) Between-subject variance of ctDNA values
Supplemental Material
sj-pdf-1-smm-10.1177_09622802251350457 - Supplemental material for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology
Supplemental material, sj-pdf-1-smm-10.1177_09622802251350457 for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology by Xijin Chen, Pavel Mozgunov, Richard D Baird and Thomas Jaki in Statistical Methods in Medical Research
Supplemental Material
sj-png-2-smm-10.1177_09622802251350457 - Supplemental material for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology
Supplemental material, sj-png-2-smm-10.1177_09622802251350457 for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology by Xijin Chen, Pavel Mozgunov, Richard D Baird and Thomas Jaki in Statistical Methods in Medical Research
Supplemental Material
sj-png-3-smm-10.1177_09622802251350457 - Supplemental material for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology
Supplemental material, sj-png-3-smm-10.1177_09622802251350457 for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology by Xijin Chen, Pavel Mozgunov, Richard D Baird and Thomas Jaki in Statistical Methods in Medical Research
Supplemental Material
sj-png-4-smm-10.1177_09622802251350457 - Supplemental material for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology
Supplemental material, sj-png-4-smm-10.1177_09622802251350457 for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology by Xijin Chen, Pavel Mozgunov, Richard D Baird and Thomas Jaki in Statistical Methods in Medical Research
Supplemental Material
sj-png-5-smm-10.1177_09622802251350457 - Supplemental material for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology
Supplemental material, sj-png-5-smm-10.1177_09622802251350457 for Using circulating tumor DNA as a novel biomarker of efficacy for dose-finding designs in oncology by Xijin Chen, Pavel Mozgunov, Richard D Baird and Thomas Jaki in Statistical Methods in Medical Research
Footnotes
Acknowledgement
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 965397. Infrastructure support is acknowledged from Cancer Research UK, the NIHR Cambridge Biomedical Research Centre (BRC-1215-20014) and Cambridge Experimental Cancer Medicine Centre. For the purpose of open access, the author has applied a Creative Commons Attribution (CC BY) license to any Author Accepted Manuscript version arising.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 965397. This report is independent research supported by the National Institute for Health and Care Research (NIHR Advanced Fellowship, Pavel Mozgunov, NIHR300576)). PM and TJ also received funding from the UK Medical Research Council (
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.