Abstract
Pilot and feasibility studies are routinely used to determine whether a definitive trial should be pursued; however, the methodologies used to assess feasibility endpoints are often basic and are rarely informed by the requirements of the planned future trial. We propose a new method for analyzing feasibility outcomes which can incorporate relationships between endpoints, utilize a preliminary study design for a future trial and allow for multiple types of feasibility endpoints. The approach specifies a Joint Feasibility Space (JFS) which is the combination of feasibility outcomes that would render a future trial feasible. We estimate the probability of being in the JFS using Bayesian methods and use simulation to create a decision rule based on frequentist operating characteristics. We compare our approach to other general-purpose methods in the literature with simulation and show that our approach has approximately the same performance when analyzing a single feasibility endpoint but is more efficient with more than one endpoint. Feasibility endpoints should be the focus of pilot and feasibility studies. The analyses of these endpoints deserve more attention than they are given, and we have provided a new, effective method their assessment.
Introduction
Pilot and feasibility studies play an important role in research; they serve as proving grounds for ideas and when well-designed generate useful information for planning future randomized controlled trials (RCTs). These studies are ubiquitous, with a PubMed search for 2023 returning over 30,000 results for the terms “pilot study” or “feasibility study,” with no doubt many more studies that are unpublished. However, there has been a lack of clarity in the literature about the definition of, and differences between, pilot and feasibility studies. After conducting a Delphi study and literature review, Eldridge et al. 1 concluded that feasibility studies, which encompass pilot studies, are concerned with three questions: “whether something can be done, should we proceed with it, and if so, how?” To address these questions, feasibility studies collect many types of data, including estimates of preliminary efficacy, and measures of feasibility related to both the intervention and design of a future trial. Common feasibility measures focused on the intervention include acceptability to participants, demand for the intervention in the general population and related measures. 2 Feasibility endpoints related to the design of a future trial include recruitment rate, retention and measures of adherence. In our examples we focus on feasibility endpoints related to the design of a future trial, and specifically, how researchers can use these outcomes to inform their decision of whether to proceed to a larger trial, however, we note that our approach can incorporate measures related to the implementation as long as they are quantitatively assessed. For trials collecting multiple types of endpoints a mixed methods framework can be utilized with an overall decision on whether to proceed determined by if each part of the study was determined to be feasible. 2
Despite the widespread use and importance of feasibility studies, their analysis plans are often basic. The Consolidated Standard of Reporting Trials (CONSORT) extension to pilot and feasibility studies 3 identified the feasibility of a future RCT as the primary aim of a pilot study. Nevertheless, many studies continue to treat other endpoints, typically preliminary estimates of efficacy, as the primary endpoint. In a recent survey of pilots for cluster RCTs only 56% had feasibility as their primary outcome. 4 Additionally, many pilot and feasibility studies use hypothesis testing of efficacy outcomes as their primary outcome; a 2010 survey found 81% 5 of pilots used hypothesis tests and a more recent 2022 survey of one hundred pilots in rehabilitation research found 92% intended to evaluate efficacy with a hypothesis test. 6 It is likely that researchers want to assess efficacy because those are the outcomes that are clinically meaningful. It is also possible that many researchers conducting early-stage studies do not have statistical support and may be most comfortable with hypothesis tests. Additionally, there has been a relative lack of methodological research concerning the best way to assess feasibility outcomes, or to determine an appropriate sample size for a pilot study based on feasibility outcomes, especially when there is more than one outcome driving “feasibility.” This combination of factors has contributed to basic methods being the norm in pilot and feasibility studies.
One of the most common approaches for assessing feasibility is to compare the outcome from each feasibility endpoint to a threshold, for example, comparing the observed retention rate to a predefined threshold of 0.80. Examples in recent articles using this approach abound: “Fidelity to the intervention manual at least 75% of activities in each session completed at a satisfactory/superior level”,
7
“feasibility criteria were set …recruitment
Avery et al. 11 proposed a variation of the threshold approach that explicitly defines what values are close enough. Their approach uses three tiers depending on the strength of the feasibility outcome: Red: Do not proceed, Amber: Potentially proceed based on possible trial improvements and Green: Proceed to a future trial. The method is referred to as a traffic light procedure and the decision is explicitly made if the point estimate falls in the Red or Green zone. If the estimate is in the Amber zone the decision depends on whether the study team believes this value can be improved in a subsequent trial (e.g. if retention missed the target rate, participant surveys might identify ways to improve retention). The adoption of this traffic light procedure has increased from 12% of trials surveyed in 2015 to 62% of trials surveyed in 2016 12 indicating a desire in the research community to better evaluate feasibility endpoints. The advantage of this approach, the Amber zone, is also its major limitation. The importance of the Amber zone rests on whether feasibility parameters can be improved which will not always be the case and there is concern that researchers may be optimistic in evaluating whether a criterion can be improved.
The traffic light procedure was further formalized by Lewis et al.
13
as a combination of a point estimate and a hypothesis test. The decision to proceed in the Amber zone is based on a
This approach was presented in terms of Type I error control and power; however, the method depends on both a point estimate, which is not constrained by the Type I error rate, and a hypothesis test, which is. If the null hypothesis is true, then the
In this paper, we propose a new method for assessing the feasibility of one or more quantitative endpoints. Our approach focuses on the probability that the set of feasibility measures are jointly feasible, and we compare our approach to other common methods both descriptively and using simulation. Notably, the method allows for trade-offs by incorporating functional relationships between multiple endpoints. We highlight this with examples including retention and recruitment rates, two of the most commonly collected feasibility endpoints. The motivation for the development of this method came from our experience designing pilot studies and attempting to create decision rules for multiple feasibility endpoints that had good operating characteristics, specifically a high probability of determining a future trial is feasible when it is and a low probability of determining a future trial is feasible when it is not.
Motivating example
We recently worked on the design of a pilot study for adults with Down syndrome (DS) and their caregivers. The intervention provides exercise sessions for the person with DS and counsels the caregiver in managing behavioral and neuropsychiatric symptoms associated with dementia, a common diagnosis in a person aging with DS. The study (KL2TR002367-08S1) is currently in pre-enrollment and will have co-primary aims, but for ease of the example we focus on only one, lessening the burden on the caregivers. In both the pilot and the future RCT the Modified Caregiver Strain Index (MCSI) 14 will be used as the primary evaluation of caregiver burden. The pilot will recruit N = 20 caregivers and will be a 12-week intervention which mirrors the future RCT. The feasibility outcomes include recruitment and retention rate and our goal in designing the study was to have good operating characteristics for our decision based on these criteria.
The remainder of the manuscript is structured as follows, in Section 2 we discuss preliminary plans for a future trial and how this can lead to more realistic assessments of feasibility, in Section 3 we discuss Bayesian estimation of feasibility, and frequentist operating characteristics. In Section 4 we compare our approach to other methods and in Section 5 we summarize the proposed methodology. All simulations and plots were completed using R version 4.2.1 or higher 15 and the code is available on GitHub.
Pre-planning and joint assessment
The questions a pilot study seeks to address (whether a future trial can be done, should we proceed, and if so, how?) depend on the specific future trial researchers hope to conduct. Consider a feasibility study with a stated goal of achieving at least 70% retention and a recruitment rate of 5 participants per month. An obvious, and typically unaddressed, question is why meeting or exceeding these thresholds would convince us that a future trial is feasible. When reasons are given for the thresholds, they are usually based on similarity to past trials. However, unless the planned RCT is extremely similar to the past trial, in required sample size, length of trial, participant burden, effectiveness, etc., there is little reason to believe we would need the same feasibility thresholds. Suppose the thresholds of 70% retention and 5 participants per month were based on what was achieved in a past RCT that targeted an intervention with an effect size of 0.50; if the intervention in a future RCT has a larger or smaller effect, then the thresholds will either be conservative or liberal because the required sample size will change. The only way to assess whether a future study will be feasible, is to have at least a rough outline of the planned design and required sample size depending on a set of assumptions (e.g. effect size, attrition rate, etc.). Then the feasibility of the assumptions that go into the design and power analysis of an RCT can be assessed using data from the pilot study. Outside of internal feasibility studies, developing a preliminary plan for a future RCT is rare but should be the standard for all feasibility trials. And even in internal pilot studies there is room for improvement in utilizing these plans to inform the pilot analyses; a 2019 review of 57 internal pilots funded by the National Institute for Health Care Research in the United Kingdom found that “while objectives were clear for the trials … none provided a specific rationale for choosing the progression criteria.” 16 We believe a feasibility study cannot answer the questions it seeks to, unless the assessment of the feasibility endpoints is directly related to the planned future trial.
For our proposed pilot in patients with DS and their caregivers we conducted a preliminary design and power analysis for the planned RCT. The MCSI will be used to assess the impact on caregivers; however, there is no known clinically meaningful change in MCSI. Sorensen et al.
17
conducted a meta-analysis on the effectiveness of interventions for caregivers and found that the effectiveness varied greatly depending on the type of intervention (e.g. Respite/daycare, Psychotherapy, etc.) with the smallest effect size of 0.01 and the largest of 0.62 (our proposed trial is similar to the interventions with larger estimated effects). For our examples, we proceed with a moderate effect size of
Relationships between pilot outcomes
Multiple feasibility criteria are usually collected in pilot studies but are typically analyzed independently. Lewis at al. recently proposed a hypothesis test for feasibility parameters, and when discussing multiple outcomes proposed that “feasibility criteria should be considered separately, and hence overall progression would be determined by the worst-performing criterion.”
13
While this approach may be reasonable in certain situations, even if it is likely conservative, there are situations where feasibility criteria are functionally related, and it would be appealing to directly incorporate these relationships. Two of the most frequently reported feasibility outcomes, recruitment, and retention rate, are related through the required sample size of a future trial. Given a preliminary sample size estimate for a future trial (NF) assuming no attrition, an assumed retention rate (ret.) and an enrollment length (L), the required recruitment rate is given by the following formula:

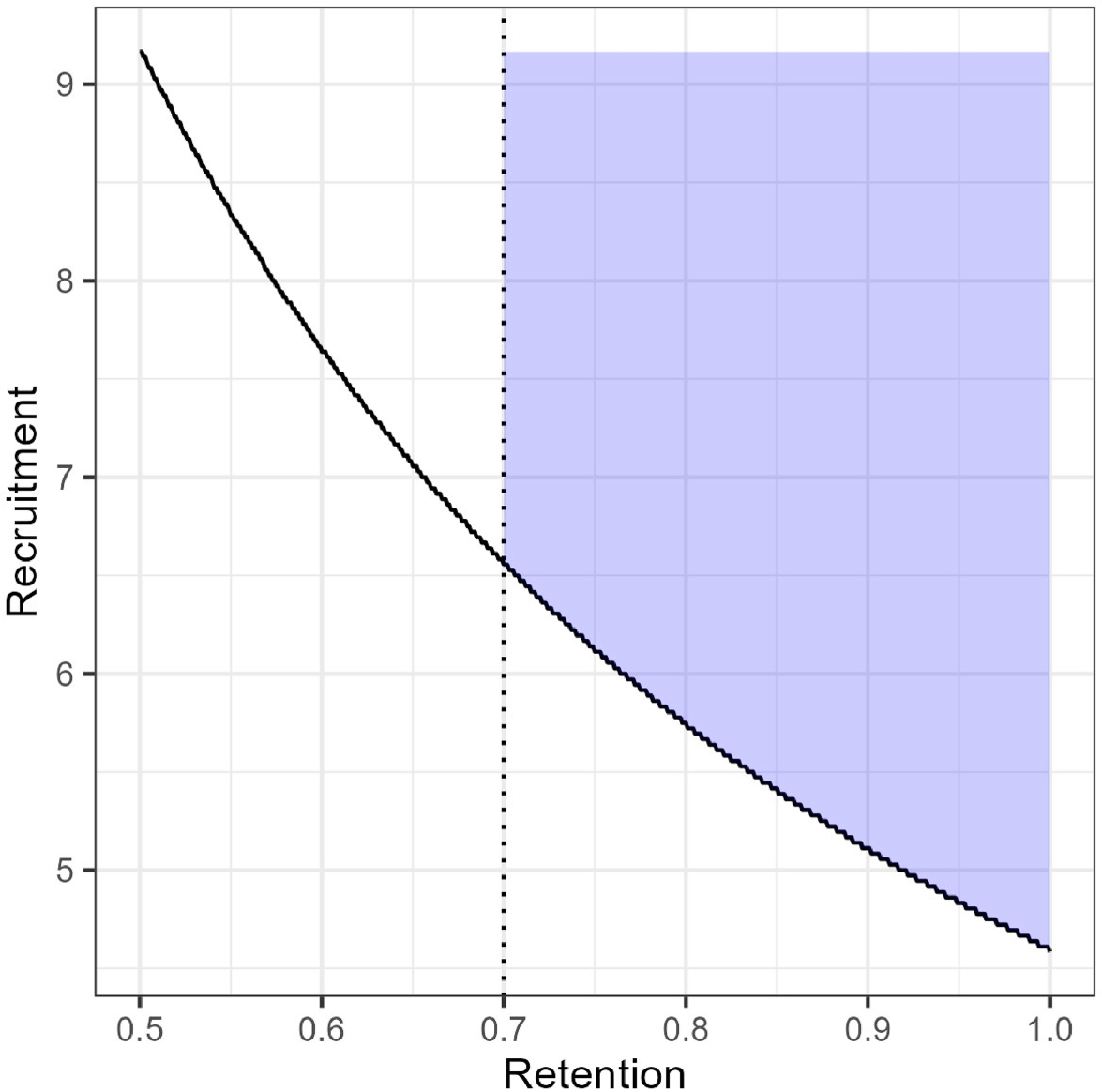
Joint feasibility space (JFS) for our proposed pilot constrained to require retention of at least 0.70.
While some parameter combinations might be feasible, they may not be acceptable either to the research team or to potential funders. A study with true retention rate of 50% and recruitment of 10 participants per month would indicate a future trial would be feasible, however, high attrition may indicate serious problems with the intervention that would limit its real-world efficacy. The JFS can be constrained based on these subjective reasons; Figure 1 shows the JFS constrained to require retention of at least 70% which is what we used in our study of DS patients and their caregivers. We also note that there is no requirement that feasibility criteria have a functional relationship, for example if we also measure acceptability (e.g. as a proportion) and wanted to proceed if the acceptability parameter were at least 0.70, that could be combined with our measures of retention and recruitment. The JFS in that case would be a quarter cylindrical shape in three dimensions (e.g. recruitment on the
Let
For our example, let
We will use weakly informative prior distributions for these examples; a Beta(
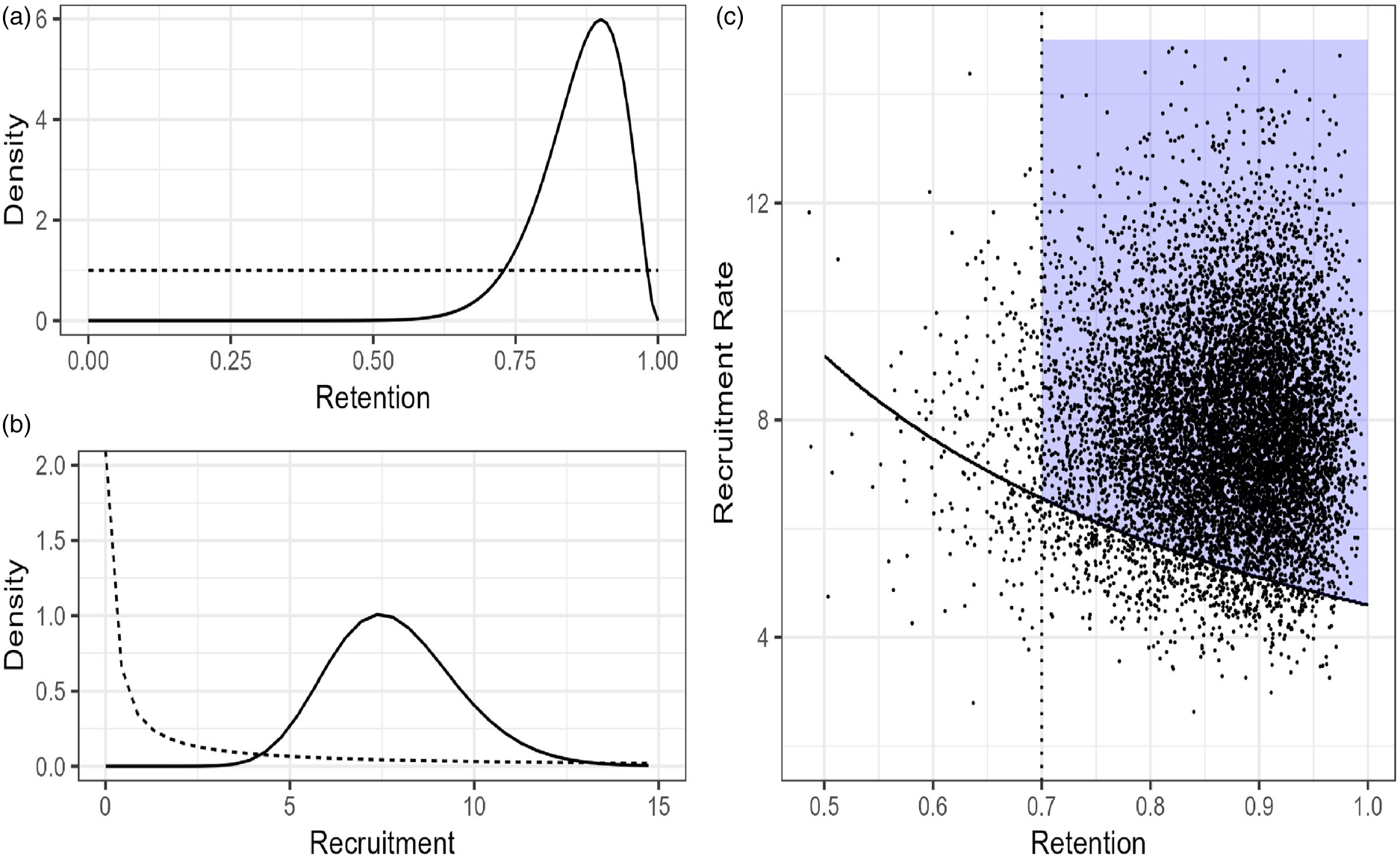
(a) Prior (dashed) and posterior (solid) distributions for Retention rate. (b) Prior and posterior distributions for Recruitment rate. (c) 10,000 draws from the joint posterior distribution overlaid on the JFS.
Given that our pilot has not been completed, we simulated hypothetical retention and recruitment data, shown in Table 1. We plan to enroll participants in cohorts and track the number of weeks of total active enrollment and convert this to a monthly rate. The simulated data in Table 1 shows it took 11 weeks, just over 2.5 months, to fully enroll the 20 participants, for a monthly recruitment rate of 7.9 participants per month. Recruitment data was simulated weekly with a rate parameter of 5.75/4.33, the posterior probability was calculated for weekly data and then converted to monthly. Data could have been simulated (or with real data, collected) as a monthly rate; however, the prior would be stronger in that case relative to the data and would need to be adjusted. We plan to calculate retention as the total number of individuals who complete both pre- and post-testing and for this simulated data set, we observed 18 out of 20 participants (90%) complete all visits.
Simulated feasibility data.

To estimate the probability of being in the JFS, we generated pairs of observations from the joint posterior distribution and estimated the probability as the number of observations in the JFS divided by the total number of simulations (Figure 2(c)). With our simulated sample Pr(
We want to maximize the probability of proceeding when a future trial is feasible and minimize the probability when it is not. We propose to evaluate the observed probability of being in the JFS based on a cut point derived from frequentist operating characteristics. We will identify a cut point that will be exceeded at a high rate when a future trial is feasible and at as low a rate as possible when a future trial is infeasible.
Typically, the design of a definitive RCT rests on assumptions about a set of parameter values. For example, a submission for an RCT to a funding agency may specify assumptions for recruitment, retention, adherence, acceptability, etc. Based on these values, a specific sample size is required which drives many aspects of the trial, including timeline and budget. If these assumptions are met, then the trial should be sufficiently powered to assess its primary aim. We focus on the decision at this set of assumptions and denote the feasible set of parameters values as
Returning to our example, based on our preliminary power analysis, a feasible set of parameter values is given by
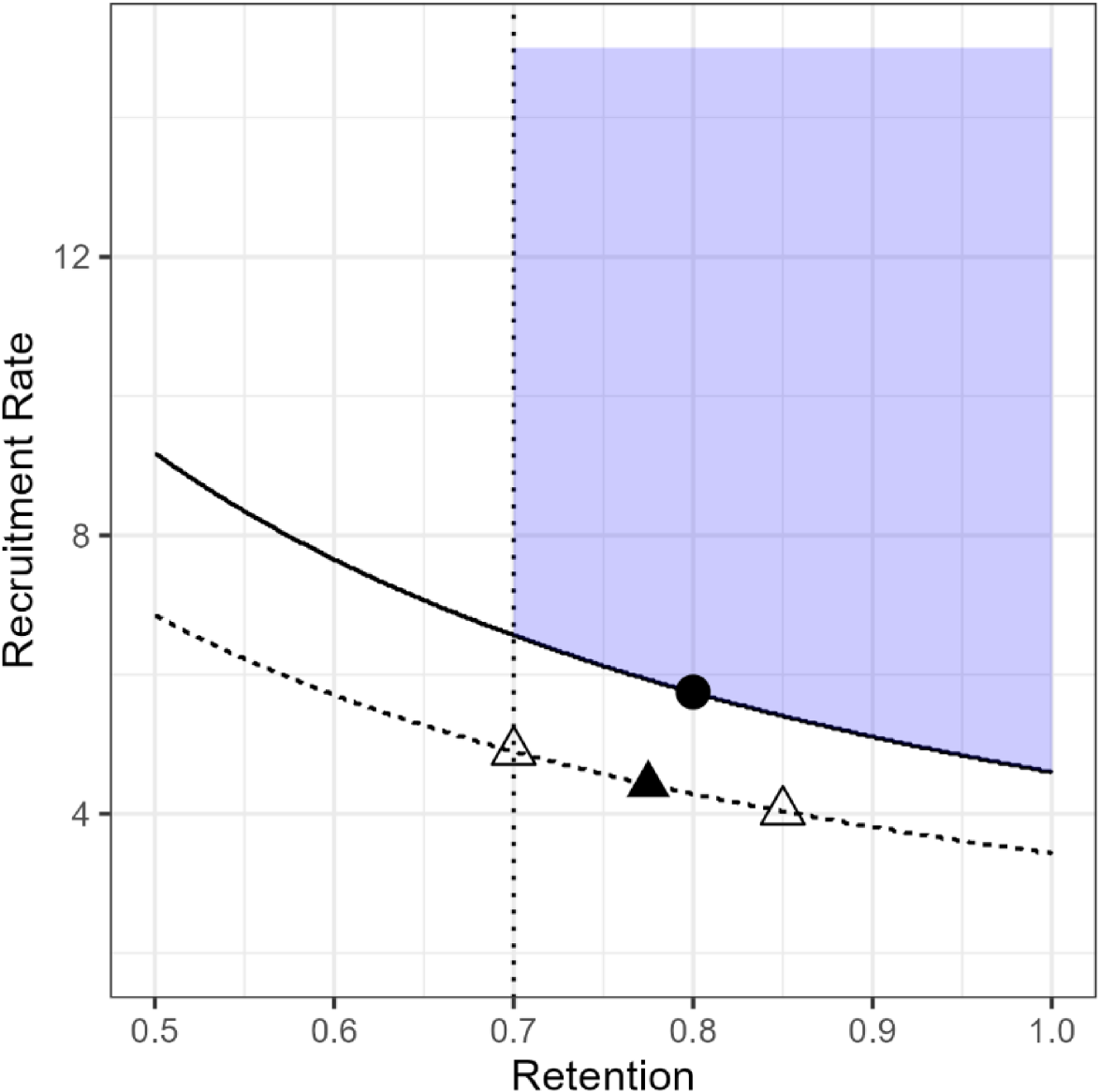
Condition
Given these scenarios we can simulate pilot data sets with these characteristics and estimate the probabilities of being in the JFS. For each combination we generated 2000 unique data sets and for each data set estimated the joint posterior probability, allowing for trade-offs, with simulation. Given the small sample sizes and fact that condition F is only on the border of the JFS the overall probabilities are skewed toward 0; however, the sampling distribution of the posterior probabilities under F was much less skewed than the other conditions. The median probability for
If the operating characteristics of the decision rules are unacceptable then either the sample size can be increased or the distance between the
Our proposed approach to evaluating one or more feasibility criteria involves several steps and is listed here for reference:
Define the boundary of the JFS and prior distributions. Define scenarios for when we should proceed ( Use simulation to identify a probability cut point to discriminate between Assess sensitivity of the chosen points. After data has been collected estimate Pr( If Pr( If Pr(
For the simulation study we chose to focus only on proportions (e.g. retention rate) and to simulate the data from binomial distributions, and for multiple endpoints we simulated them independently. We did this for three reasons (1) many studies only include proportions as feasibility outcomes, even recruitment is often calculated as enrolled/screened with a target rate and we wanted to compare our procedure under this common scenario; (2) the hypothesis test method proposed by Lewis et al. can only include proportions and we wanted to include this method given its quick adoption in the research community (over 200 citations since being published in 2021); and (3) we did not include functional relationships between endpoints because this could bias the results in favor of our procedure.
We conducted two simulation studies. The first focused on a specific example which is similar to our pilot study and used retention values of
We compared our approach to three different methods, the threshold approach, an approach we refer to as the Tolerance method and the hypothesis test proposed by Lewis et al. The threshold approach specifies a target rate (T) for each endpoint and if the sample point estimate meets or exceeds the target
Thresholds are routinely listed as part of a decision rule; however, we recognize that researchers will attempt to modify feasibility criteria that are close to the threshold and will therefore still choose to proceed even if the threshold is not strictly met. The tolerance method we propose is an attempt to formalize this common practice. We specify a threshold but will proceed if (
The final comparator was the hypothesis test proposed by Lewis et al.
13
For each endpoint the researcher specifies a “Green lower limit” (
Comparison of JFS with other methods.
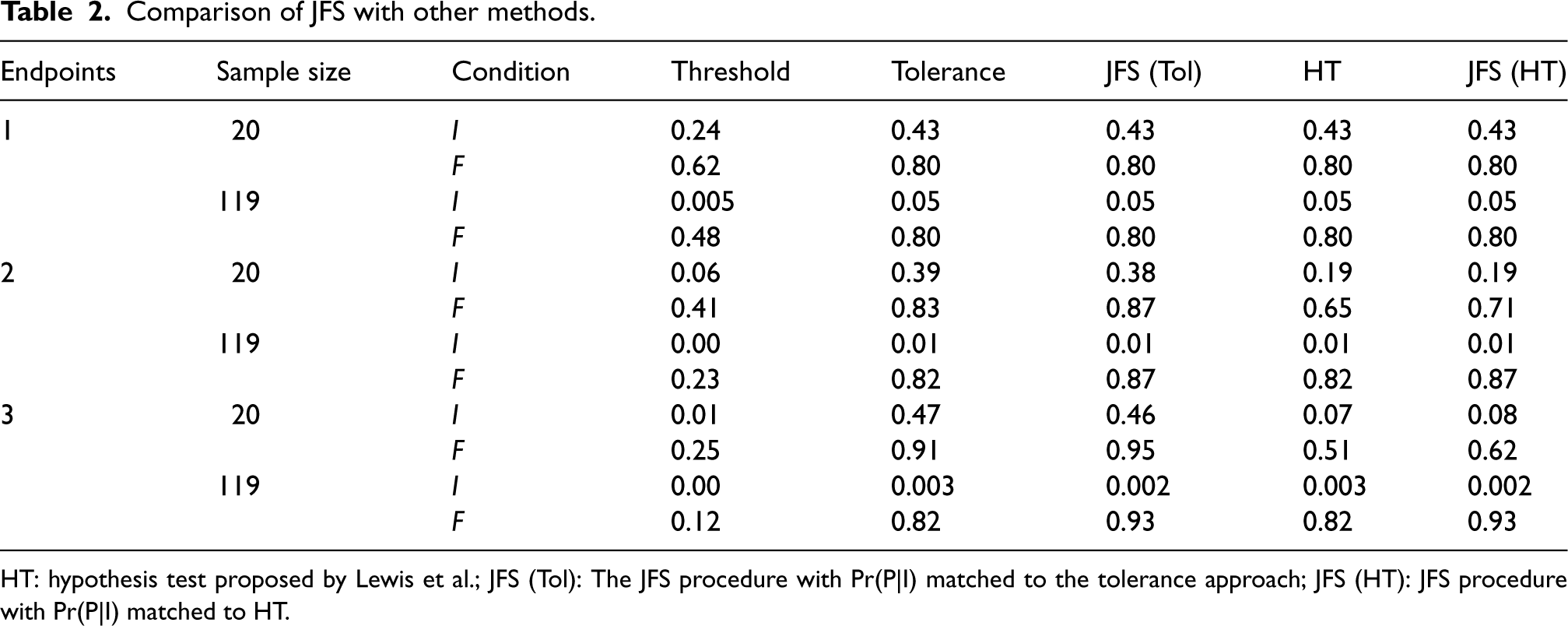
HT: hypothesis test proposed by Lewis et al.; JFS (Tol): The JFS procedure with Pr(P|I) matched to the tolerance approach; JFS (HT): JFS procedure with Pr(P|I) matched to HT.
Comparison of JFS with other methods.

HT: hypothesis test proposed by Lewis et al.; JFS (Tol): The JFS procedure with Pr(P|I) matched to the tolerance approach; JFS (HT): JFS procedure with Pr(P|I) matched to HT.
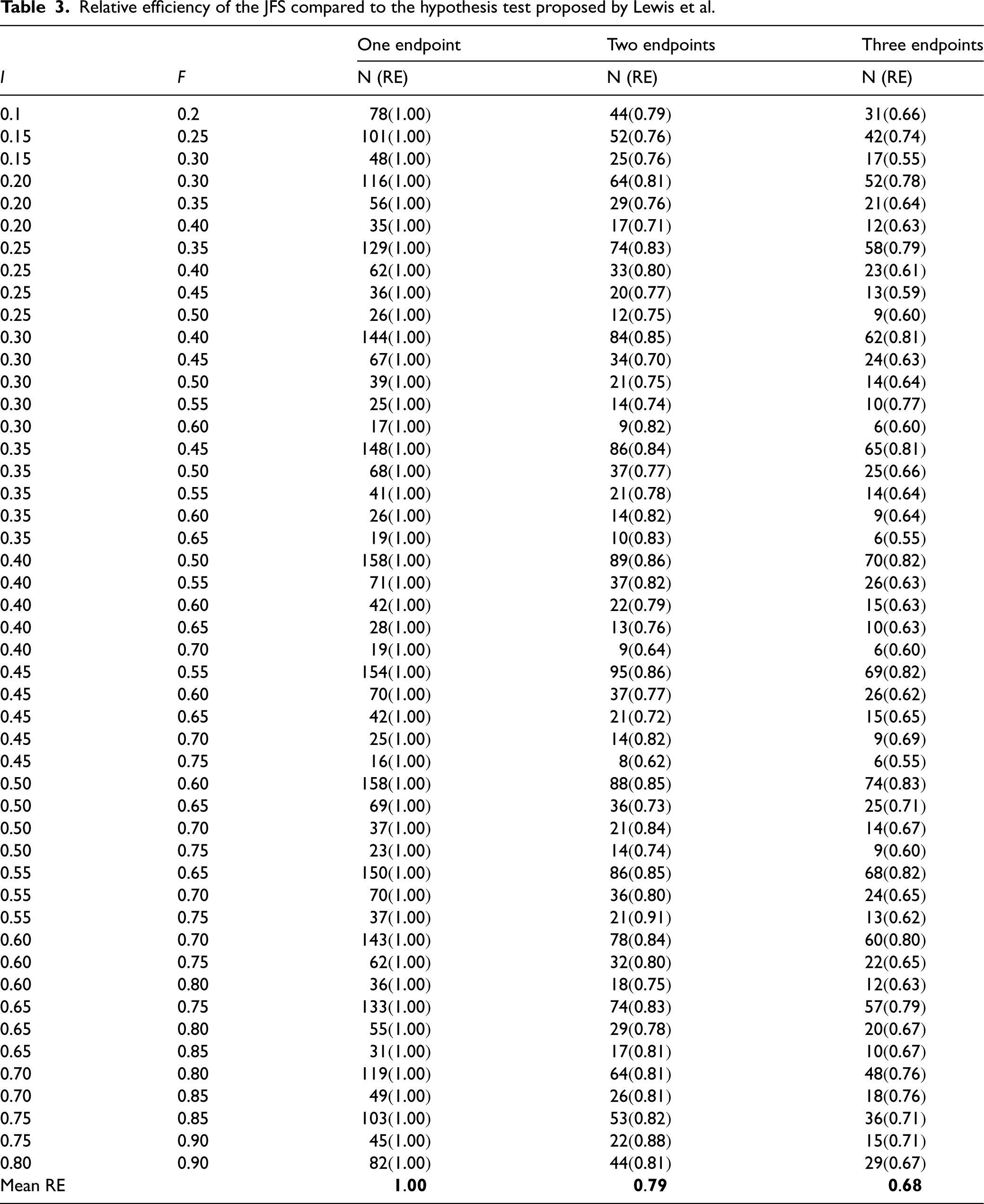
Relative efficiency of the JFS compared to the hypothesis test proposed by Lewis et al.
The decision rules for each approach are:
Threshold: Proceed if point estimate ≥ 0 Tolerance: Proceed if point estimate ≥ 0 Hypothesis test: Proceed if point estimate ≥ 0 JFS: Proceed if estimated Pr(θ ∈ JFS) ≥ cut point.
The threshold, tolerance, and hypothesis test methods cannot always achieve the specified performance, that is,
The simulation results are presented in Table 2. As expected, the Threshold approach performed poorly; despite a strict threshold being reported in many pilot and feasibility studies we doubt they are rigorously enforced, and this simulation provides further evidence that they should not be. The only advantage of the Threshold procedure is that it has the best control of proceeding when we should not; however, in early-stage studies a higher rate of erroneously proceeding is typically accepted given that tight control of this error rate would lead to effective interventions being abandoned early. The tolerance and hypothesis test approaches have similar, often identical, operating characteristics. In the sample size and effect combinations we examined this was because of the discreteness of the underlying binomial random variable. For example, with
For a single endpoint, the three measures have approximately identical operating characteristics; however, as the number of feasibility endpoints increased so did differences in performance. With two feasibility endpoints we were able to match the Pr(P|
To determine whether these results would hold in more general settings, we conducted another simulation study to estimate the required sample size to achieve specific progression probabilities. Table 3 shows the combinations of
We used the
Well-conducted pilot studies are critical to ensure that follow-up trials are feasible. Commonly used methods for assessing feasibility criteria are often simplistic and fail to account for potential relationships between the outcomes. These issues can lead to poor decision making at the end of a feasibility study. In this manuscript, we have presented a new procedure to assess multiple, possible related, feasibility endpoints. Through simulations we have shown that our procedure outperforms widely used alternative methods by achieving a higher probability of proceeding when we should while maintaining an equivalent probability of proceeding when we should not in specific examples and showed that in general our procedure is more efficient than the hypothesis test approach. A key advantage of the JFS procedure is that it allows for specific functional relationships between two or more feasibility criteria, these simply change the boundaries of the feasibility space and additional relationships beyond retention and recruitment can be incorporated if they exist. An additional advantage of our approach is the ability to include feasibility endpoints of many types, for example, proportions, rates, means, etc., without requiring specific combinations of endpoints. We have provided sample size estimates for one, two, and three binomial feasibility endpoints for many combinations of
A key limitation of this study is that other methods have been proposed that were not included in the simulation studies. Wilson et al. 19 proposed a Bayesian decision theoretic approach based on the traffic light procedure that allowed for trade-offs between acceptable error rates for incorrect decisions (e.g. choosing not to proceed when the parameter is in the green zone). This approach was shown to have good performance. However, it is computationally expensive, and it requires the specification of a loss function (a piecewise loss function was used in the examples). The elicitation of a loss function is a difficult, and often unfamiliar, problem, and this is likely to be exacerbated in the setting of a pilot study. Another interesting method proposed by Wilson et al. 10 constructed a hypothesis test based on the power of a future trial for pilot studies collecting recruitment, adherence and follow-up rates (what we have called retention), and was extended to incorporate estimation of the standard deviation. The hypothesis test also incorporated an assumed relationship between adherence and effectiveness and allowed for randomness of recruitment and observation in defining the power and operating characteristics of the future trial. The major limitation of this approach, and the reason it was not included in the simulation studies is that it is only applicable for a pilot study collecting a subset of recruitment, adherence and follow-up rates as the feasibility endpoints. These are common feasibility outcomes; however, pilot studies collect many types of endpoints, including measures of acceptability, safety, compliance, cost and others. A cursory look at published pilot studies will show that it is uncommon for the key feasibility criteria to only include a subset of recruitment, retention, and adherence. Our proposed method is meant to be a general solution and can incorporate as many endpoints as needed, and these can be rates, counts, proportions etc. In our simulations we chose to focus on comparing our method to another general-purpose solution, the hypothesis test from Lewis et al. However, the hypothesis test from Wilson et al. may be more appropriate for trials that collect a subset of recruitment, adherence, and follow-up rates. Further simulation studies would be needed to compare the operating characteristics of our proposed approach to the test proposed by Wilson et al. or other methods under specific scenarios.
Another limitation of our approach is the computational complexity. A probability cut point must be defined, based on simulation, and outside of simple scenarios the posterior probability of
We note that some of the required sample sizes in Table 3 are extremely small, and even though the simulations showed good operating characteristics we would not feel confident in the results from a pilot with
Supplemental Material
sj-docx-1-smm-10.1177_09622802241311219 - Supplemental material for Jointly assessing multiple endpoints in pilot and feasibility studies
Supplemental material, sj-docx-1-smm-10.1177_09622802241311219 for Jointly assessing multiple endpoints in pilot and feasibility studies by Robert N Montgomery, Amy E Bodde and Eric D Vidoni in Medical Research
Footnotes
Acknowledgements
We thank the reviewers for their helpful comments and corrections which have sharpened the focus and clarity of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institute on Aging of the National Institutes of Health under award number R03AG073932. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
