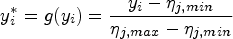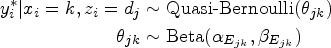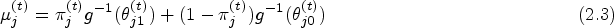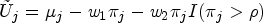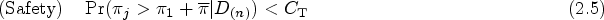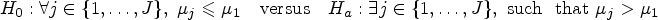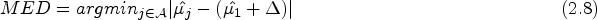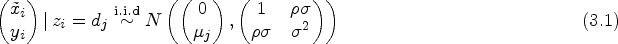Abstract
Most existing dose-ranging study designs focus on assessing the dose–efficacy relationship and identifying the minimum effective dose. There is an increasing interest in optimizing the dose based on the benefit–risk tradeoff. We propose a Bayesian quasi-likelihood dose-ranging design that jointly considers safety and efficacy to simultaneously identify the minimum effective dose and the maximum utility dose to optimize the benefit–risk tradeoff. The binary toxicity endpoint is modeled using a beta-binomial model. The efficacy endpoint is modeled using the quasi-likelihood approach to accommodate various types of data (e.g. binary, ordinal or continuous) without imposing any parametric assumptions on the dose–response curve. Our design utilizes a utility function as a measure of benefit–risk tradeoff and adaptively assign patients to doses based on the doses’ likelihood of being the minimum effective dose and maximum utility dose. The design takes a group-sequential approach. At each interim, the doses that are deemed overly toxic or futile are dropped. At the end of the trial, we use posterior probability criteria to assess the strength of the dose–response relationship for establishing the proof-of-concept. If the proof-of-concept is established, we identify the minimum effective dose and maximum utility dose. Our simulation study shows that compared with some existing designs, the Bayesian quasi-likelihood dose-ranging design is robust and yields competitive performance in establishing proof-of-concept and selecting the minimum effective dose. Moreover, it includes an additional feature for further maximum utility dose selection.
Introduction
Phase
Several adaptive designs have been developed for dose-ranging trials.2,3 Compared with conventional one-stage designs, adaptive dose-ranging trial designs are more flexible and efficient, as they allow for prospectively planned modifications of one or more aspects of the design based on interim data from the trial participants.
4
Examples of interim modifications include adding or dropping doses, adaptively randomizing patients to beneficial doses, and selecting the dose–response model while accounting for model uncertainty. Berry et al.
5
proposed an adaptive dose-ranging trial design based on a normal dynamic linear model to sequentially assign doses to new patients. Dragalin et al.
6
proposed an adaptive dose-ranging trial design based on the
There is increasing interest in incorporating benefit–risk consideration into the assessment and selection of doses in dose-ranging trials. In 2009, the U.S. Food and Drug Administration (FDA) released guidelines for performing benefit–risk assessments of new drug and biological products.
12
Recently, the FDA launched Project Optimus to reform the dose optimization and dose selection paradigm in oncology drug development.
13
Although Project Optimus focuses on oncology, its central message, that is, that dose selection should be based on a benefit–risk assessment, is also applicable to other therapeutic areas. Another important reason for considering toxicity in dose-ranging trials is that the doses selected for dose-ranging trials are often based on very limited phase I data (e.g. data from six patients treated at one dose). There is large uncertainty on the safety of the doses: a dose regarded as safe in a phase
In addition, dose-ranging studies sometimes can have non-continuous efficacy endpoints, such as binary response. With the increasing availability and variety of biomarkers, there is a growing number of trials based on ordinal efficacy endpoints14–19 or semi-continuous bounded endpoints, such as a composite score.20–23 It’s desirable to devise a design that can accommodate diverse types of efficacy endpoints such as binary, ordinal, semi-continuous, and continuous endpoints. For example, Pinheiro et al. 24 extended MCP-Mod to handle binary, count, or time-to-event endpoints.
In this article, we propose a Bayesian quasi-likelihood dose-ranging (BQD) design that can handle various types of efficacy endpoints and explicitly incorporates benefit–risk assessment into the decisions of dose assignment and selection. Our design employs a quasi-likelihood methodology as a unified framework to accommodate binary, ordinal, semi-continuous, and continuous endpoints. We utilize a utility function as a measure of benefit–risk tradeoff and to adaptively assign patients to doses based on the doses’ estimates of utility. The design takes a group-sequential approach. At each interim, the doses that are deemed overly toxic or futile are dropped. At the end of the trial, we use posterior probability criteria to assess the strength of the dose–response relationship (i.e. to establish the PoC). If the PoC is established, we identify both the MED and maximum utility dose (MUD) that maximizes the benefit–risk tradeoff.
Compared to existing methods, BQD has several appealing features. First, it does not make any parametric assumptions about the shapes of dose–toxicity and dose–efficacy curves and thus is robust. For comparison, MCP-Mod considers a set of prespecified candidate dose–efficacy models and uses model selection to improve the robustness of the design. However, as our simulation study showed, when the true dose–efficacy model is not in the candidate model set, the performance of MCP-Mod is subject to the influence of model misspecification. Second, BQD provides a uniformed framework to handle binary, ordinal, semi-continuous, and continuous endpoints. In contrast, most existing methods handle only one type of endpoint. Third, BQD explicitly accounts for benefit–risk assessment using utility and enables the selection of both the MED and MUD, whereas most existing methods focus on only efficacy and the MED. Last, owing to the use of a quasi-likelihood methodology with closed-form posteriors, the computation of BQD is easy and fast, which is important for application.
The remainder of this article is organized as follows. Section 2 introduces the probability model for safety and efficacy, the benefit–risk tradeoff, and the trial design. In Section 3, we use simulation to evaluate the operating characteristics of the BQD design. Section 4 gives a summary with concluding remarks.
Method
Probability model
Consider a dose-ranging trial with
We assume that
We next consider modeling the conditional distribution of
Let
Our design uses a utility function to measure the benefit–risk tradeoff between efficacy and toxicity. One simple and intuitive utility function is given by
In addition to defining the benefit–risk tradeoff based on the marginal probability of toxicity and efficacy, such as (2.4), a more general approach involves assigning a utility score to each possible combination of patient-level toxicity and efficacy outcomes. The desirability of
To safeguard patients from futility and/or a toxic dose, we define that a dose is admissible only if it satisfies the following safety and efficacy criteria:
The BQD design takes a group sequential approach and consists of In the first stage, equally randomize For On the basis of the final data, assess whether a significant dose–response relationship exists to establish the PoC. If the PoC is established, select the MED and MUD; otherwise, claim that there is no significant dose–response relationship.
Below, we first describe the method for establishing the PoC and selecting the MED and MUD and then describe the procedure of adaptive randomization.
At the final analysis, we first assess the dose-response relationship as a PoC based on the following null and alternative hypotheses:
Specifically, we select the MED as the dose whose posterior mean of
We now discuss the adaptive randomization rule in step 2 of the BQD design. Given the interim data
To ensure reasonable power for establishing the PoC, it is important to assign a sufficient but not excessive number of patients to the control. Let
When implementing the BQD design, a practical consideration is the determination of the total sample size and interim times. The total sample size (i.e.
Setup
We used simulation to evaluate the operating characteristics of the BQD design and compared them with those of a conventional analysis of variance (ANOVA) approach
2
and MCP-Mod.
7
MCP-Mod is based on five dose-efficacy models, including
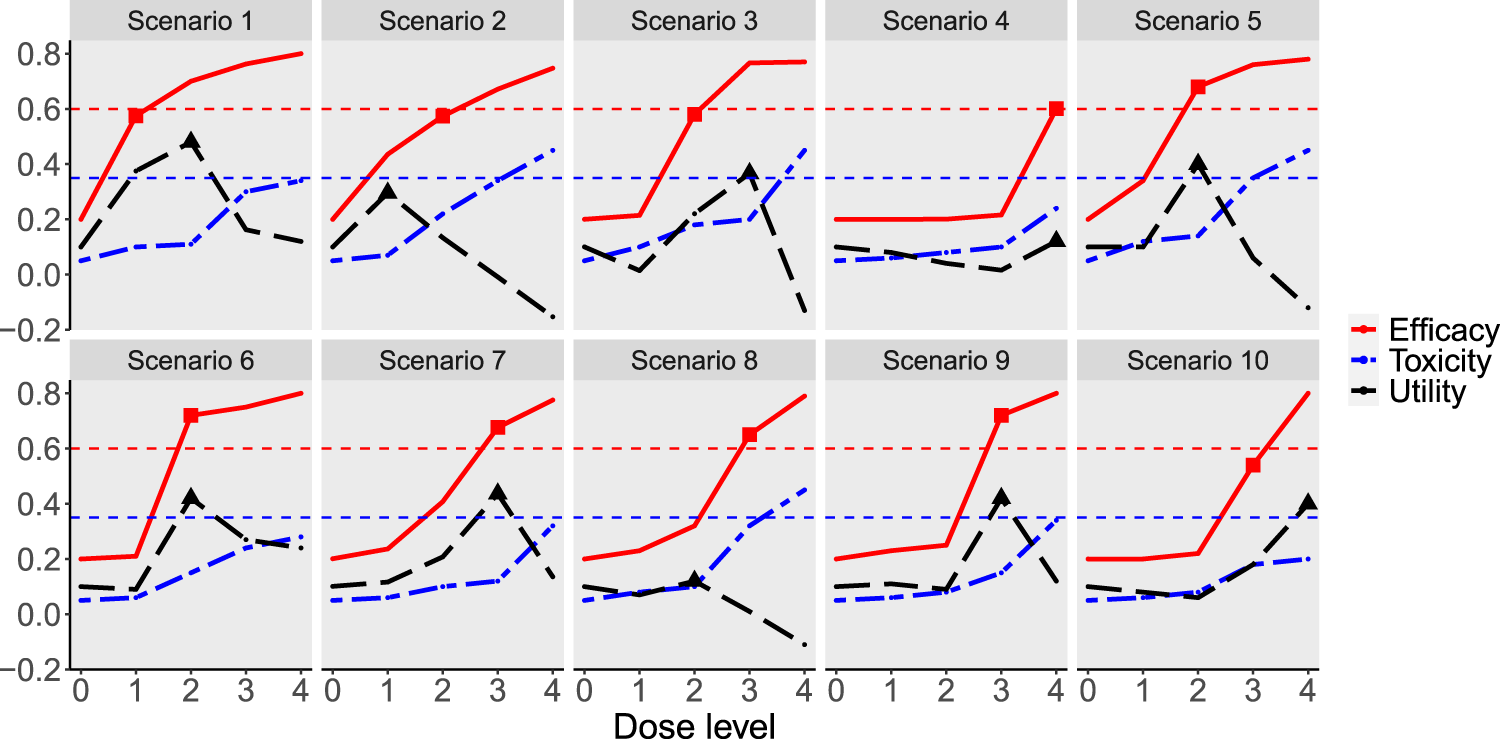
Simulation scenarios for the continuous endpoint. The solid red line is the dose–efficacy curve. The dash-dotted blue line is the dose–toxicity curve. The dashed black line is the dose–utility curve. The horizontal dashed red line corresponds to the target efficacy, and the horizontal dashed blue line is the maximum acceptable toxicity rate. The minimum effective dose is marked by a square (
Simulation scenarios for the continuous efficacy endpoint.
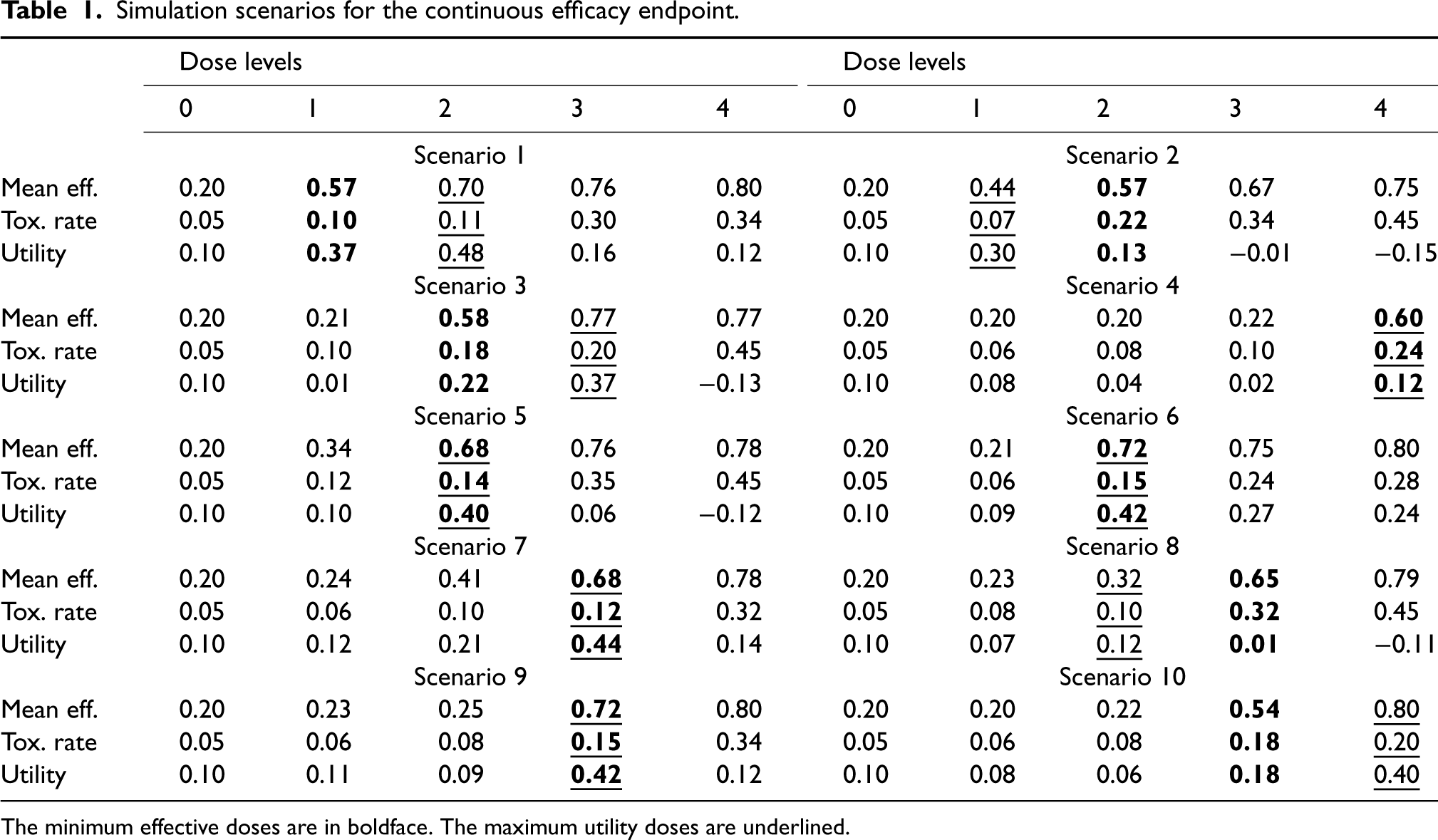
The minimum effective doses are in boldface. The maximum utility doses are underlined.
To facilitate the comparison of the three methods, we calibrated
For the ordinal efficacy endpoint, we assumed that
Simulation scenarios for the ordinal efficacy endpoint.
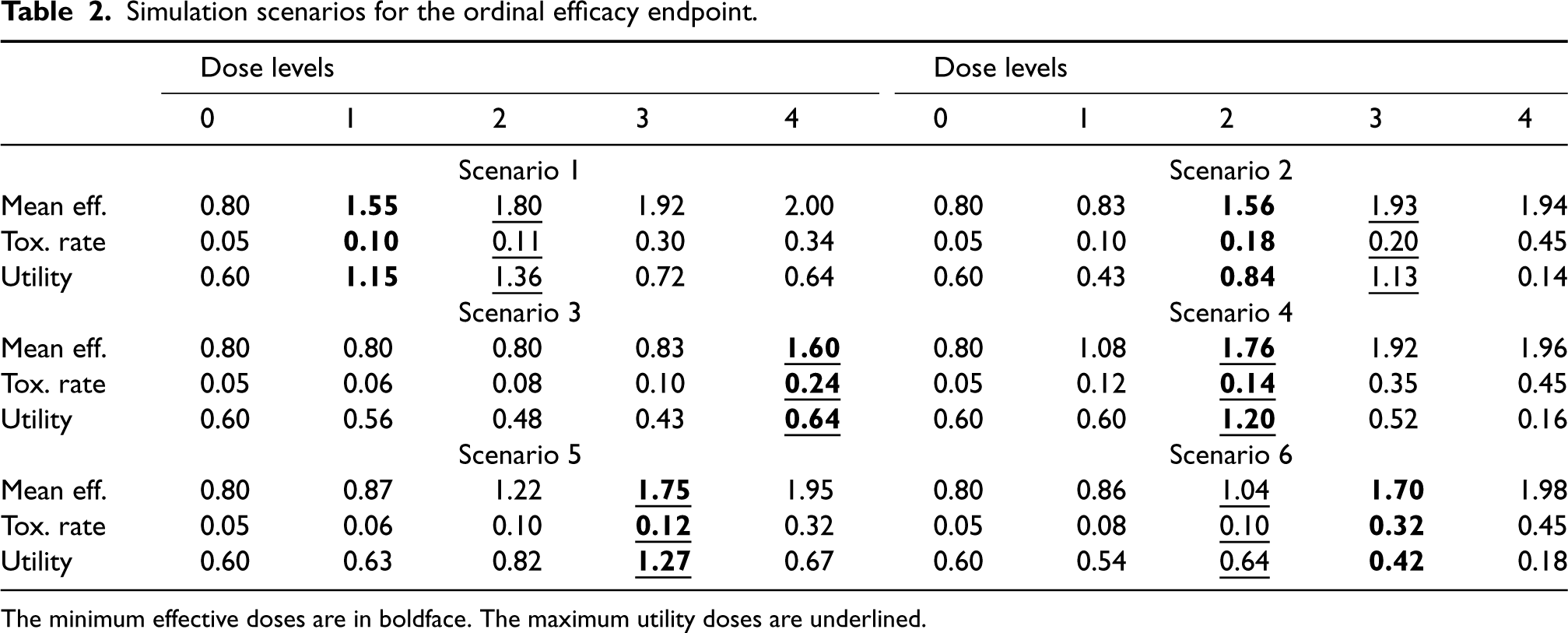
The minimum effective doses are in boldface. The maximum utility doses are underlined.
For the binary efficacy endpoint, we generated
Simulation scenarios for the binary efficacy endpoint.
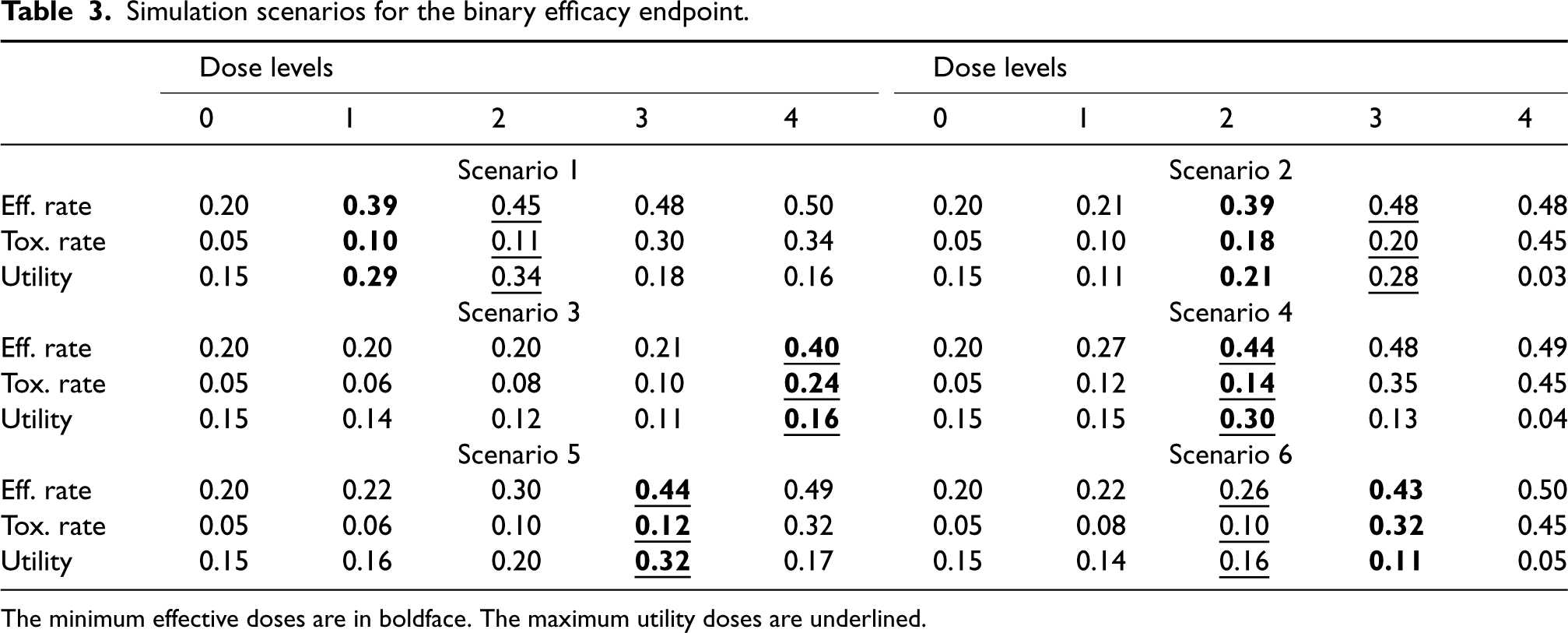
The minimum effective doses are in boldface. The maximum utility doses are underlined.
Notice that, both MCP-Mod and ANOVA consider only efficacy and the target MED and were used as comparators to evaluate the performance of BQD in MED selection for continuous, ordinal and binary efficacy endpoint. There is no existing dose-ranging method targeting MUD. We will thus only present the simulation results in MUD selection for our proposed method to demonstrate the advantage of the BQD design to select multiple target doses. We believe that this MUD selection step can have great potential to be further developed in dose-ranging designs.
We performed 10,000 simulations for each scenario. The operating characteristics of the designs were evaluated in three aspects: (1) the power to detect the dose-response (i.e. the power for the PoC); (2) the correct selection of the MED and MUD; and (3) patient allocation to treatment arms. Specifically, for the second aspect, we will evaluate the percentage of correct selection of the MED (
Results for continuous efficacy endpoint
Figure 2 shows the simulation results for the continuous efficacy endpoint. Figure 2(A) displays the power for establishing the PoC. The power BQD had for establishing the PoC was similar to that of MCP-Mod and up to
Simulation results for the continuous efficacy endpoint. (A) The power for establishing the PoC. (B) The percentages of the correct selection of the MED (
In terms of the percentage of correct selection of the MED (
In terms of patient allocation, BQD generally outperformed MCP-Mod and ANOVA, allocating higher percentages of patients to the MED and MUD arms (Figure 2(D)). Because ANOVA and MCP-Mod utilize equal randomization to five arms, the percentages of patients they allocate to the MED and MUD arms are both fixed at
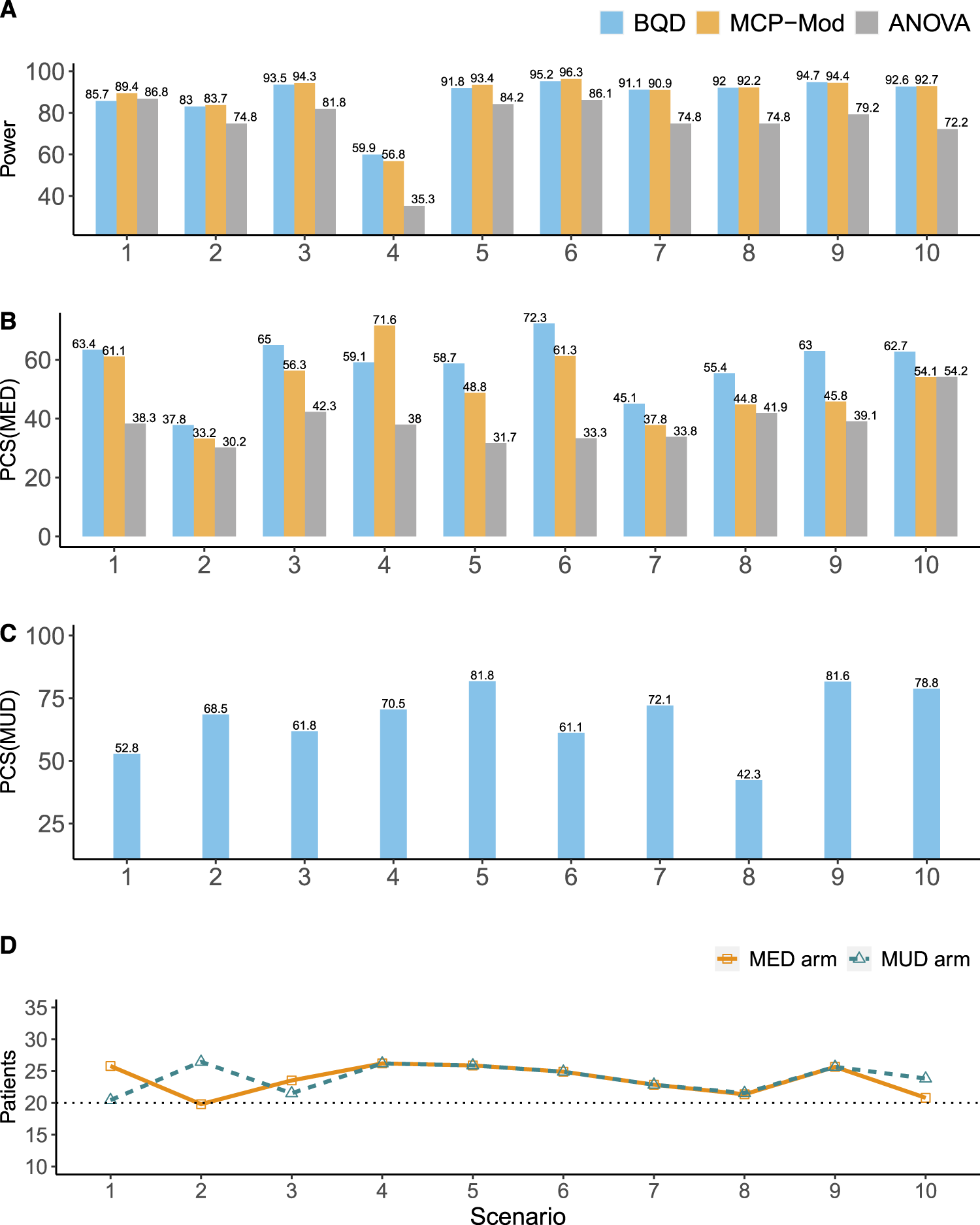
Figure 3 shows the simulation results for the ordinal efficacy endpoint. Similar to the results for the continuous endpoint, the power BQD had for establishing the PoC was similar to that of MCP-Mod, and both BQD and MCP-Mod outperformed ANOVA (Figure 3(A)). In terms of selecting the MED (see Figure 3(B)), ANOVA had the worst performance with the lowest
Simulation results for the ordinal efficacy endpoint. (A) The power for establishing the PoC. (B) The percentages of the correct selection of the MED (
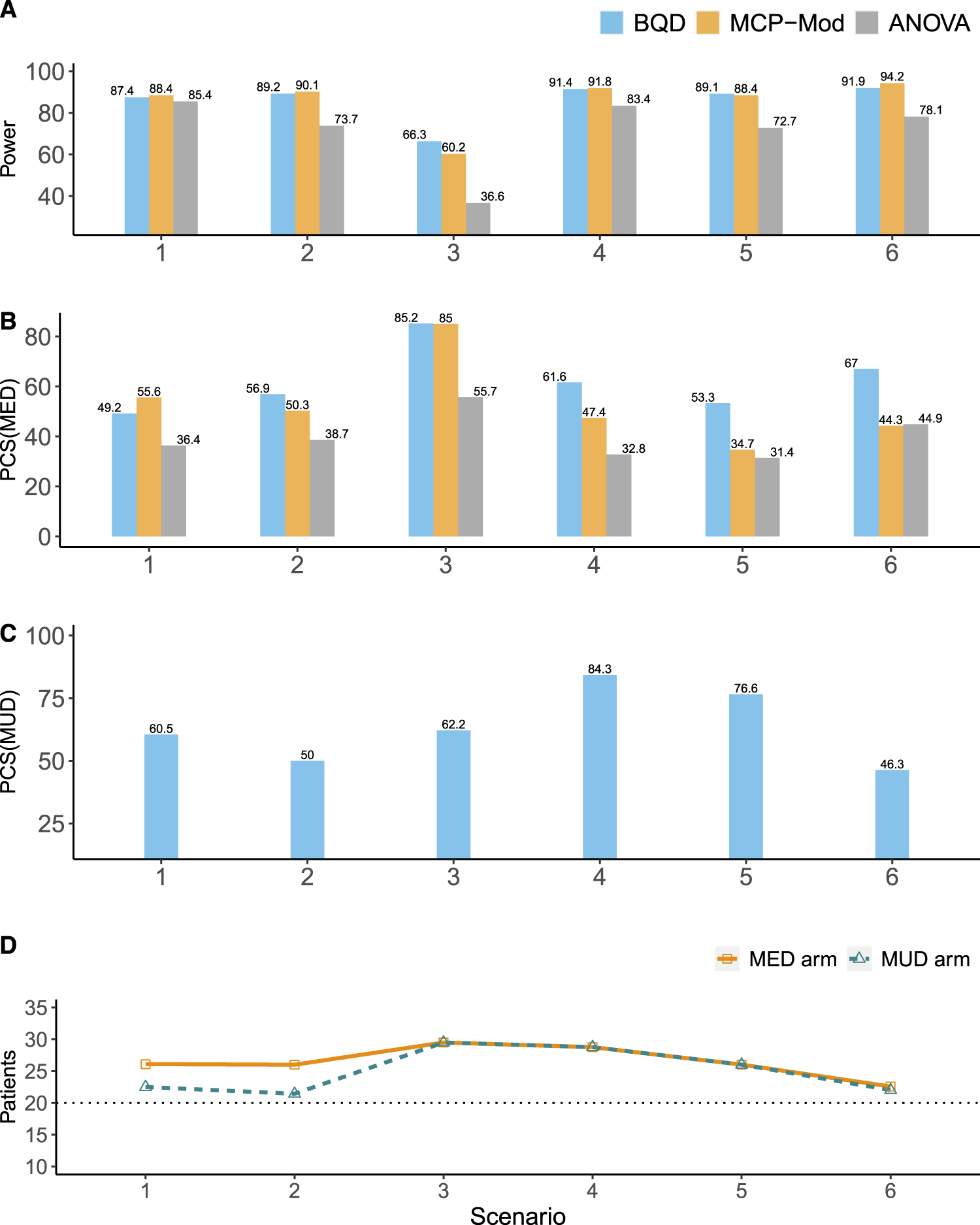
The results for the binary efficacy endpoint are generally similar to those for the continuous and ordinal endpoints, but the advantage of BQD is more pronounced. As shown in Figure 4(A) and (D), compared with MCP-Mod-B, BQD had higher power for establishing the PoC in all six scenarios. In addition, in most scenarios, BQD yielded higher selection percentages of
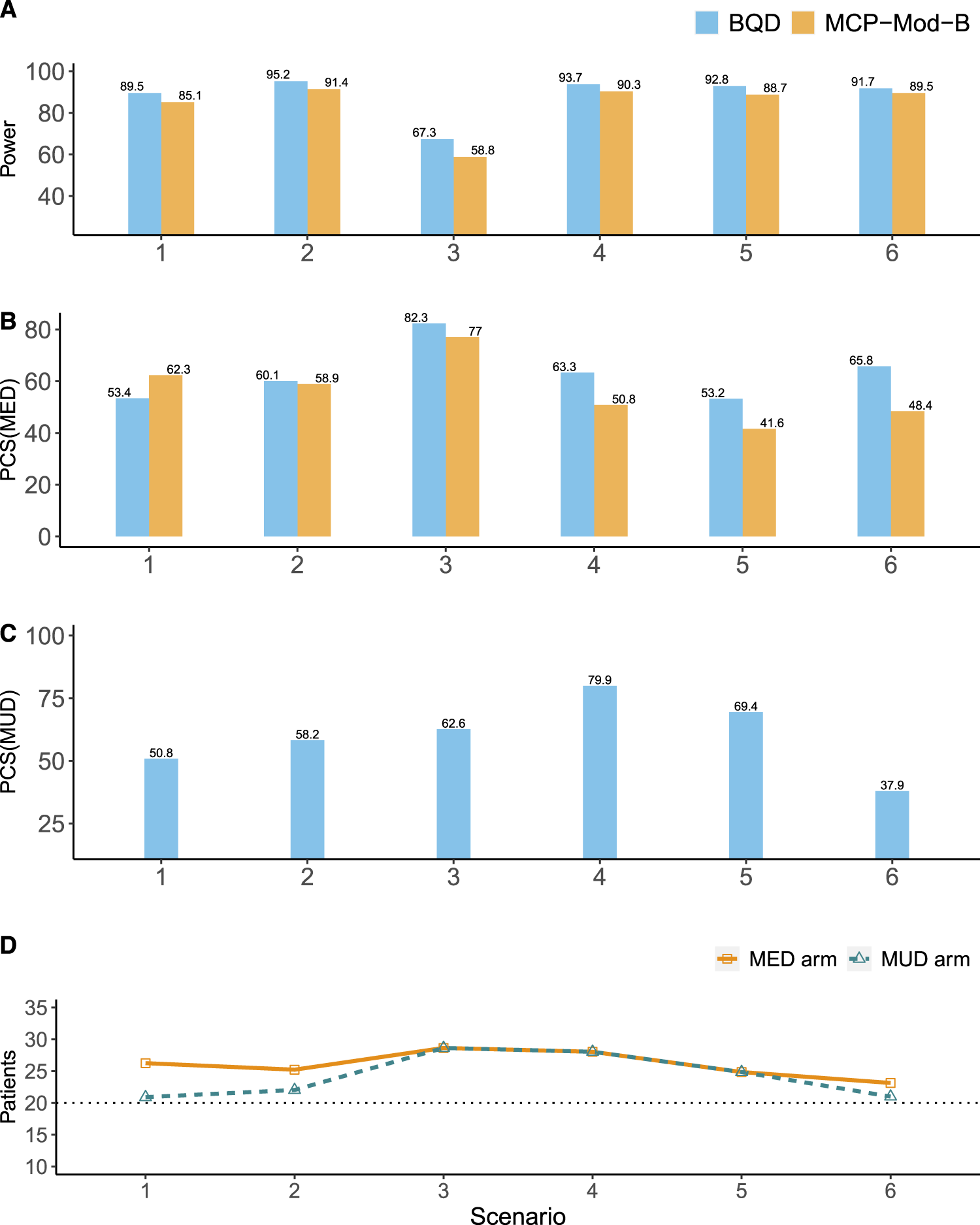
Simulation results for the binary efficacy endpoint. (A) The power for establishing the PoC. (B) The percentages of the correct selection of the MED (
We conducted sensitivity analyses for different number of dose levels (i.e.
The performance of BQD is similar to or better than that of MCP-Mod, suggesting that the quasi-Bernoulli likelihood approach is highly efficient. To further investigate its efficiency, we compared BQD (based on the quasi-Bernoulli likelihood) with a version of BQD based on the true likelihood (denoted by BQD-L). The only difference between BQD and BQD-L is that the latter uses the true likelihood; all other decision rules are the same for the two designs. Thus, BQD-L provides the upper bound of the performance. Figure S5 in the Supplemental Material shows that BQD and BQD-L have similar performance, with similar power for establishing the PoC and similar accuracy in identifying the MED and MUD, further confirming the high efficiency of the quasi-Bernoulli likelihood approach.
Discussion
We propose a Bayesian quasi-likelihood dose-ranging design that handles various types of efficacy endpoints (e.g. binary, ordinal, and semi-continuous endpoints) and explicitly incorporates benefit–risk assessment into the decision of dose assignment and selection. Our design utilizes a utility function as a measure of benefit–risk tradeoff and adaptively assigns patients to doses based on the doses’ likelihood of being the MED and MUD. The design takes a group-sequential approach. At each interim, the doses that are deemed overly toxic or futile are dropped. The simulation study shows that the BQD design is robust and has competitive performance in selecting the MED. Additionally, it includes an extra feature for further MUD selection.
The BQD design models the toxicity and efficacy at each dose locally without making any parametric assumption on the shape of dose–efficacy or dose–toxicity curves. This makes the design simple (with closed-form posteriors) and more robust than MCP-Mod. Simulation shows that this nonparametric approach does not compromise the efficiency of the BQD design. This is because the isotonic regression utilizes efficacy and toxicity data across dose levels, which improves the efficiency of estimation and decision making. In addition, the design’s adaptive randomization and Bayesian decision rules enable more efficient patient allocation and provide extra flexibility to calibrate the design to improve the operating characteristics.
To accommodate various types of efficacy endpoints and streamline posterior calculations, we adopt a hybrid approach, where we model toxicity using its true likelihood while modeling efficacy using quasi-Bernoulli likelihood. It’s crucial to emphasize that our design does not mandate the use of the hybrid approach; the true likelihood of the efficacy endpoint can be employed. However, this alternative necessitates distinct models for different types of endpoints (e.g. the Bernoulli model for binary endpoints, the normal model for continuous endpoints, and the multinomial model for categorical endpoints) and involves more complex computations. Both our simulation and previous research28,27,29 demonstrate that the quasi-likelihood is highly efficient and yields desirable operating characteristics.
The BQD design can be extended in various ways. In some trials, the efficacy endpoint is the time to the event. In this case, a quasi-Bernoulli or quasi-normal likelihood approach cannot be used. We may use a piecewise exponential model and modify the utility along the lines of Murray et al. 34 In addition, the BQD design assumes that the efficacy endpoint is quickly ascertainable for adaptive randomization and group sequential decisions. If this assumption does not hold, we may use imputation methods32,35 or likelihood approximation methods 36 to facilitate real-time decision making.
Supplemental Material
sj-pdf-1-smm-10.1177_09622802241239268 - Supplemental material for A Bayesian quasi-likelihood design for identifying the minimum effective dose and maximum utility dose in dose-ranging studies
Supplemental material, sj-pdf-1-smm-10.1177_09622802241239268 for A Bayesian quasi-likelihood design for identifying the minimum effective dose and maximum utility dose in dose-ranging studies by Feng Tian, Ruitao Lin, Li Wang and Ying Yuan in Statistical Methods in Medical Research
Footnotes
Acknowledgements
The authors would like to thank the Editor, the Associate Editor, and the two reviewers for their valuable comments and suggestions, which greatly contributed to enhancing the quality of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. Ying Yuan is partially supported by the grant from the National Cancer Institution (U01DK108328), and Bettyann Asche Murray Professorship. Ruitao Lin is partially supported by grants from the National Cancer Institute (5P30CA016672, 5P50CA221703, and 1R01CA261978).
Supplemental material
Supplemental material for this article including additional simulation details and results as well as the posterior calculation are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.