Abstract
Many studies seek to evaluate the effects of potentially harmful pregnancy exposures during specific gestational periods. We consider an observational pregnancy cohort where pregnant individuals can initiate medication usage or become exposed to a drug at various times during their pregnancy. An important statistical challenge involves how to define and estimate exposure effects when pregnancy loss or delivery can occur over time. Without proper consideration, the results of standard analysis may be vulnerable to selection bias, immortal time-bias, and time-dependent confounding. In this study, we apply the “target trials” framework of Hernán and Robins in order to define effects based on the counterfactual approach often used in causal inference. This effect is defined relative to a hypothetical randomized trial of timed pregnancy exposures where delivery may precede and thus potentially interrupt exposure initiation. We describe specific implementations of inverse probability weighting, G-computation, and Targeted Maximum Likelihood Estimation to estimate the effects of interest. We demonstrate the performance of all estimators using simulated data and show that a standard implementation of inverse probability weighting is biased. We then apply our proposed methods to a pharmacoepidemiology study to evaluate the potentially time-dependent effect of exposure to inhaled corticosteroids on birthweight in pregnant people with mild asthma.
Keywords
1. Introduction
Due to ethical concerns regarding the randomization of drug usage in pregnant individuals (including cis women and trans people), the safety of drug exposures for perinatal outcomes is typically evaluated using observational data.1,2 Researchers are often interested in establishing the potential of a drug to impede growth or produce physical or functional defects in the embryo or fetus. Notably, effects may vary by the duration of the exposure in addition to the specific gestational periods in which the exposure took place. 2 Thus, interest lies in evaluating exposure effects over the duration of the pregnancy and in identifying periods where drugs may be potentially harmful.
Over the past 35 years, major developments have improved the analysis of longitudinal exposures.3–5 In particular, in situations where time-varying confounders of a time-varying exposure may themselves be affected by previous exposure states, standard regression methods fail to estimate a parameter that is interpretable as a causative mechanism. In 1997, Robins proposed marginal structural models, 6 which are models for outcomes under an imposed (counterfactual) exposure history, as an intuitive way to address this problem. Inverse probability weighting (IPW), 4 which involves modeling the probabilities of exposure and censoring at a series of time points, 7 was presented as an accessible method for estimating the parameters of such models. G-computation3,8 is another estimation approach relying on time-dependent modeling of the outcome. More robust approaches that require modeling of the exposures, censoring states, and outcomes were subsequently developed.6,9,10 In particular, targeted maximum likelihood estimation (TMLE),11–13 which builds off of previous theory and methods,9,10 is a framework for the construction of plug-in estimators that are typically doubly robust and locally efficient.
Recently, Hernán and Robins 14 detailed the “target trials” approach, which is a cogent approach to the causal inference of longitudinal exposures that directly relates observational cohort design and exposure definitions to a hypothetical randomized controlled trial (RCT). 5 The result is that the parameters estimated in the analysis of observational data can be clearly linked to those in an RCT with possibly time-varying treatment assignments and adherence. In particular, they propose to deal with patients in the study becoming contraindicated to treatment over time by defining “treatment strategies” that assign treatment unless the contraindication is present. This allows for the definition of pragmatic treatment effects that are relatable to real clinical practice.
In this study, we propose new parameters that represent longitudinal effects of drug exposure during pregnancy using the target trials approach. Our parameters are well-defined when the outcome can be defined at all possible delivery times given the cohort structure. In particular, we define the treatment strategy “assign the drug during a specific gestational period unless delivery has already occurred.” The strategy “discontinue the drug during a specific gestational period unless delivery has already occurred” is similar. Such strategies are well-defined in the sense that they could be employed in a hypothetical RCT. We define and interpret intent-to-treat and the so-called “sustained treatment” parameters related to this approach. We propose specific implementations of IPW, G-computation, and TMLE for the estimation of such parameters and verify their performance for the intent-to-treat parameter in a simulation study. Finally, we apply these methods in an application addressing the impact of low-dose inhaled corticosteroid (ICS) during pregnancy on birthweight for pregnant individuals with mild asthma.
2. Challenges in the analysis of pregnancy exposures
Our example relates to the usage of ICSs by asthmatic individuals during pregnancy. We are interested in evaluating the gestational age-specific effect of such exposure on birthweight. For mild asthma, treatment typically alternates between low daily doses of ICSs or no controller medication 15 . During pregnancy, individuals often reduce or discontinue usage of ICS though it is not medically recommended to do so. 16 While uncontrolled asthma is known to be dangerous during pregnancy, previous investigations did not identify any safety concerns related to the use of low-dose ICS on pregnancy outcomes. 17 However, few longitudinal analyses adjusting for time-dependent confounding have been conducted. 18 To address this gap, a cohort of pregnant asthmatic individuals with deliveries between 1998 and 2008 was established from a linkage of administrative healthcare data from the province of Québec, Canada. 19
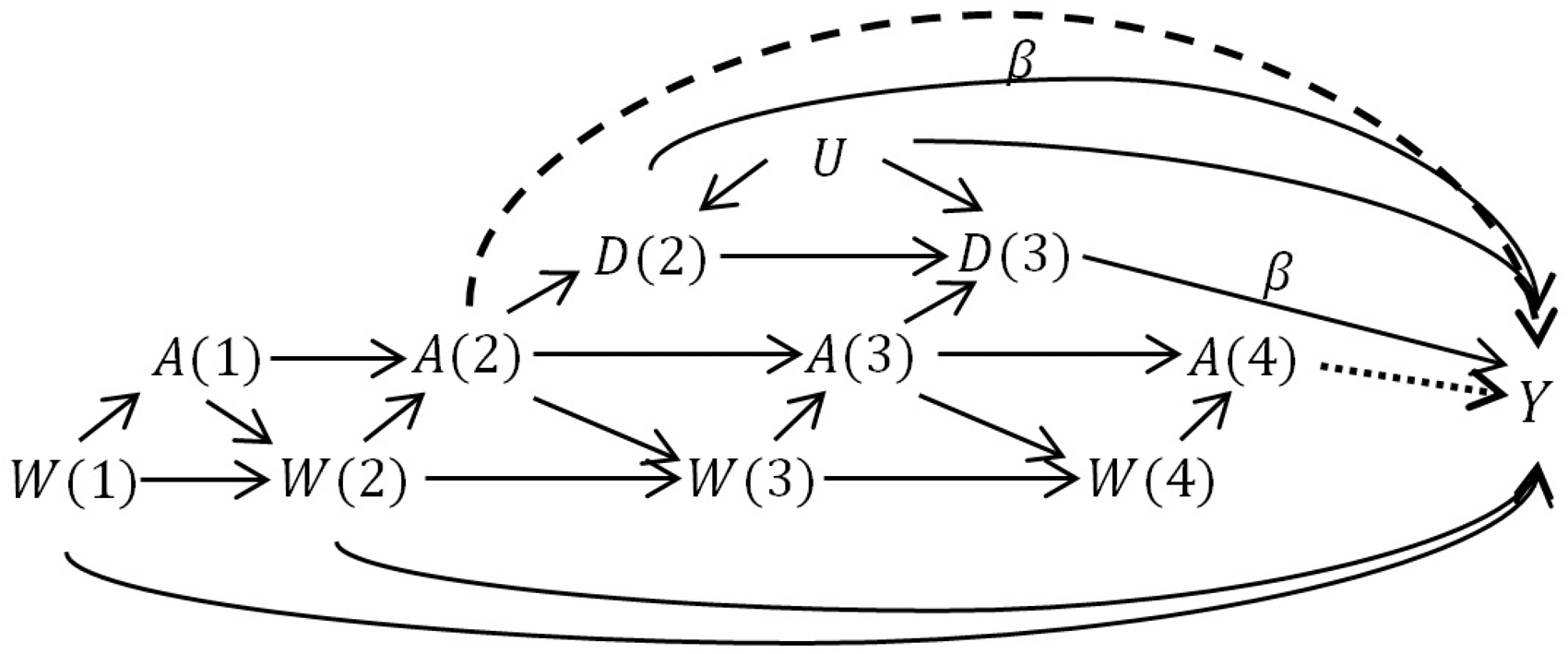
A general challenge in the analysis of time-varying drug exposures during pregnancy is that the duration of pregnancy is not the same for every person, and in fact may be related to past exposure and/or the outcome of interest when the outcome of interest is not the duration of the pregnancy itself. 20 Due to the variability in delivery times, delivery may precede (compete with) exposure at later time points, producing a violation of the standard positivity assumption that all subjects must be eligible to follow all exposure sequences of interest. 7 Thus, delivery time is both a mediator of early exposure and a contraindication/confounder for later exposure. This structure is represented in a simplified directed acyclic graph in Figure 1. The top diagram shows how the timing of delivery (e.g. early delivery: true/false) can be a mediator of early exposure to treatment and the outcome at delivery. The bottom diagram shows how, if a delivery does not occur prematurely (early delivery=false), treatment exposure can also occur in the late stages of pregnancy and potentially impact the outcome.
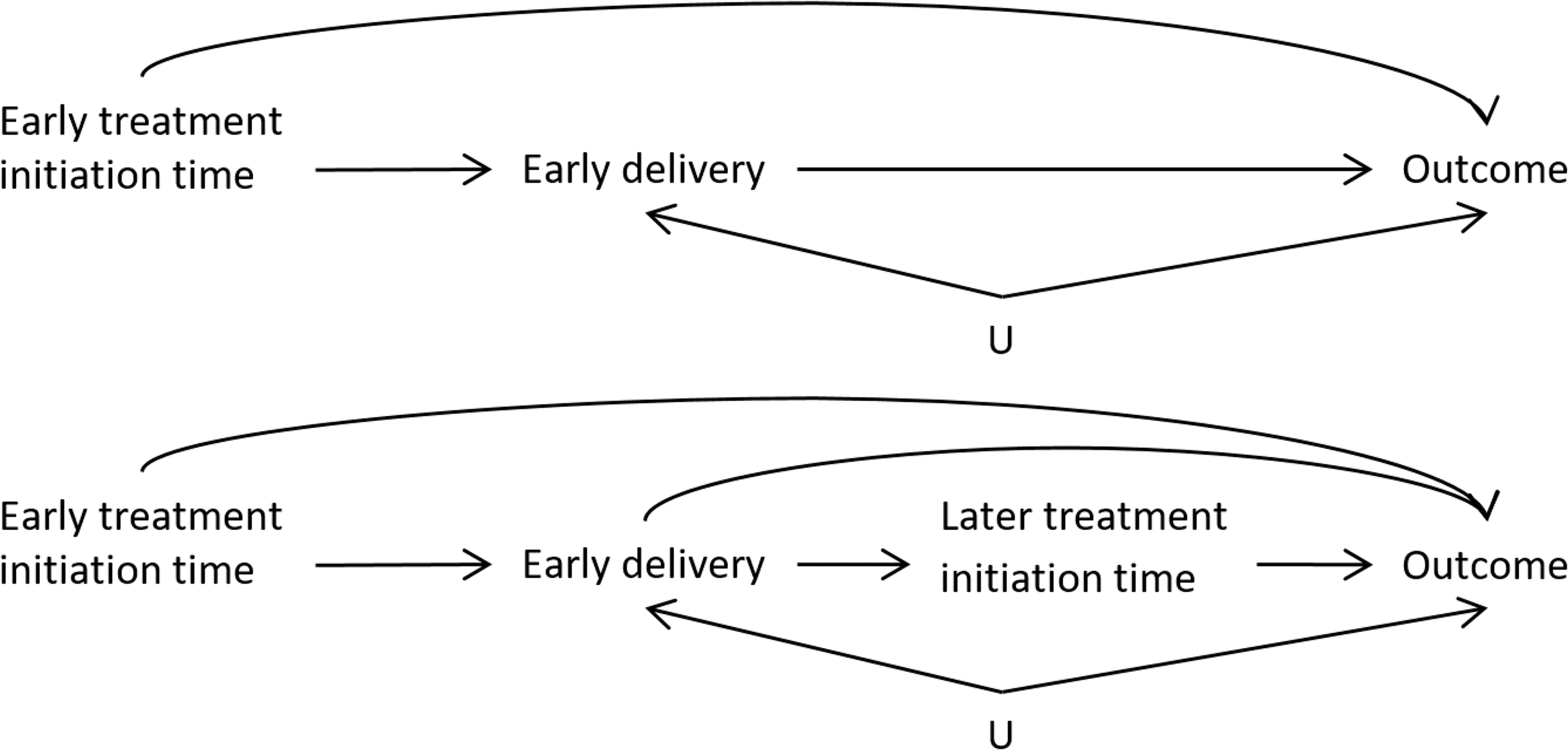
The approximate mediation structure of the hypothetical RCT where treatment initiation is randomized. (Top) The total effect of early treatment initiation may be divided into the mediated effect on early delivery and the direct effect on the outcome.
We are interested in estimating the total effect of timing-specific exposure to low-dose ICS on birthweight, which is the combination of the effect mediated by pregnancy duration and the direct effect which is not mediated by pregnancy duration. One potential way to address the problem of variable pregnancy duration is to artificially censor all pregnancies that do not reach full term. However, this approach would only estimate the direct effect on the outcome and not the total effect. Another typical approach used in pharmacoepidemiology contrasts pregnancies that were “ever exposed” with those “never exposed.” In addition to not addressing the time-dependent nature of the effects of exposure, this approach is subject to immortal time bias, 21 exposure misclassification, and unadjusted time-dependent confounder bias. 2
The target trial
The general target trials approach 14 involves the elaboration of an ideal RCT (target trial) that relates to the same scientific question as the observational study to be conducted. One must specify, for example, eligibility criteria, the baseline (when randomization occurs), treatment assignment groups, the follow-up period, and outcomes. To avoid selection bias in the RCT analysis, all subjects in the study would be retained, regardless of health or pregnancy status after baseline. The RCT parameters of interest, such as the intent-to-treat contrast, are then defined. Once the target trial is defined, the cohort design and analysis must be undertaken using the same definitions as the RCT, or as close as possible. The analysis of the observational data is then defined to estimate the same parameters as in the target trial.
For our example, we consider the hypothetical RCT that enrolls participants at conception. Each participant is then assigned a randomly selected start-time of ICS treatment. For example, we may enroll individuals with mild asthma at conception, all indicated for low-dose ICS, stop their current ICS usage, and assign them to a time to (re)start ICS. We consider four treatment arms with different start-times—conception, 13 weeks gestational age (beginning of second trimester), 28 weeks (beginning of third trimester), and 37 weeks—and a fifth control group that is assigned to never initiate. We also simplify reality so that no pregnancies terminate before 13 weeks and all will have a measurable outcome at delivery, such as weight at birth. In our RCT, it is possible that a delivery will occur before 37 weeks (11% globally) or even before 28 weeks (roughly 6
What are the options for the analysis of such an RCT if the outcome is binary, such as low birth weight, or continuous, such as birthweight? Analyses of RCTs often include the per-protocol and the intent-to-treat (ITT) approaches. In this section we discuss the potential for bias under a naive per-protocol analysis, the interpretation of the ITT effect in the described target trial, and the sustained treatment strategy parameter as an alternative to the naive per-protocol approach.
2.1 The fallacy of the naive per-protocol analysis
The naive per-protocol analysis involves the removal of all subjects who did not follow the assigned treatment protocol and estimates the treatment effect by making unadjusted comparisons between those remaining in each treatment arm. As is well-known, this type of analysis may be highly biased with respect to treatment efficacy if the reasons for deviation from the protocol are related to the outcome. 23 Some authors have alternatively proposed to use measured time-dependent covariates to adjust for non-adherence post-randomization, in order to estimate the average effect of following the protocol.24–27 This approach requires that all relevant covariates are measured in order to adjust for confounding of the time-dependent protocol adherence. However, even if we did measure these covariates, we would not be able to apply this method in our RCT as it relies on the “positivity” assumption that all participants could have hypothetically followed any treatment protocol, conditional on their health status at each time point. 7 In our case, pregnancies cannot become exposed to treatment after delivery, so we violate the assumption that any unit (pregnancy) could have followed any protocol.
In our context, a simple per-protocol analysis may also cause bias even in the absence of confounding. Consider an RCT that randomizes pregnant people 1:1 at conception either to initiating the medication at 37 weeks or control (never initiate). For illustration, suppose also that 10% of deliveries occur before 37 weeks. The outcome in this analysis is low birth weight (LBW) (<2500 g). Suppose that the medication has no biological effect on the outcome but that the 10% of early deliveries have a probability of LBW of 50% while full-term births have a probability of 5%. The initiation of treatment was randomized so half of the individuals with full-term births took the medication as assigned. However, due to early delivery, none of the early-term deliveries were exposed, despite 50% of them having been randomized to treatment. The simple per-protocol analysis would show that, because all early deliveries are included in the unexposed group regardless of their assignment, more unexposed pregnancies resulted in LBW so that the study may incorrectly conclude that medication was beneficial (Table 1, top). Note that in this simple case where no treatment arm involves initiating treatment in the second trimester, we can perform the per-protocol analysis in the subset of full-term pregnancies and get the correct null result, but this will not be the case in general.
Expected proportions (
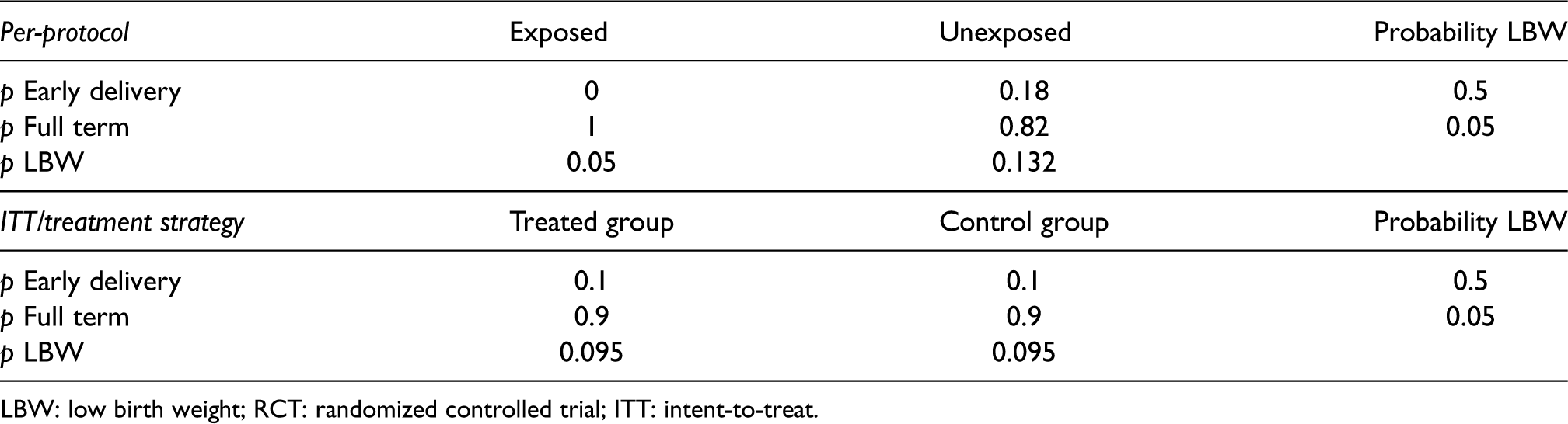
LBW: low birth weight; RCT: randomized controlled trial; ITT: intent-to-treat.
Summary of notation used for the observed and counterfactual data and the intent-to-treat (ITT) parameter of interest.
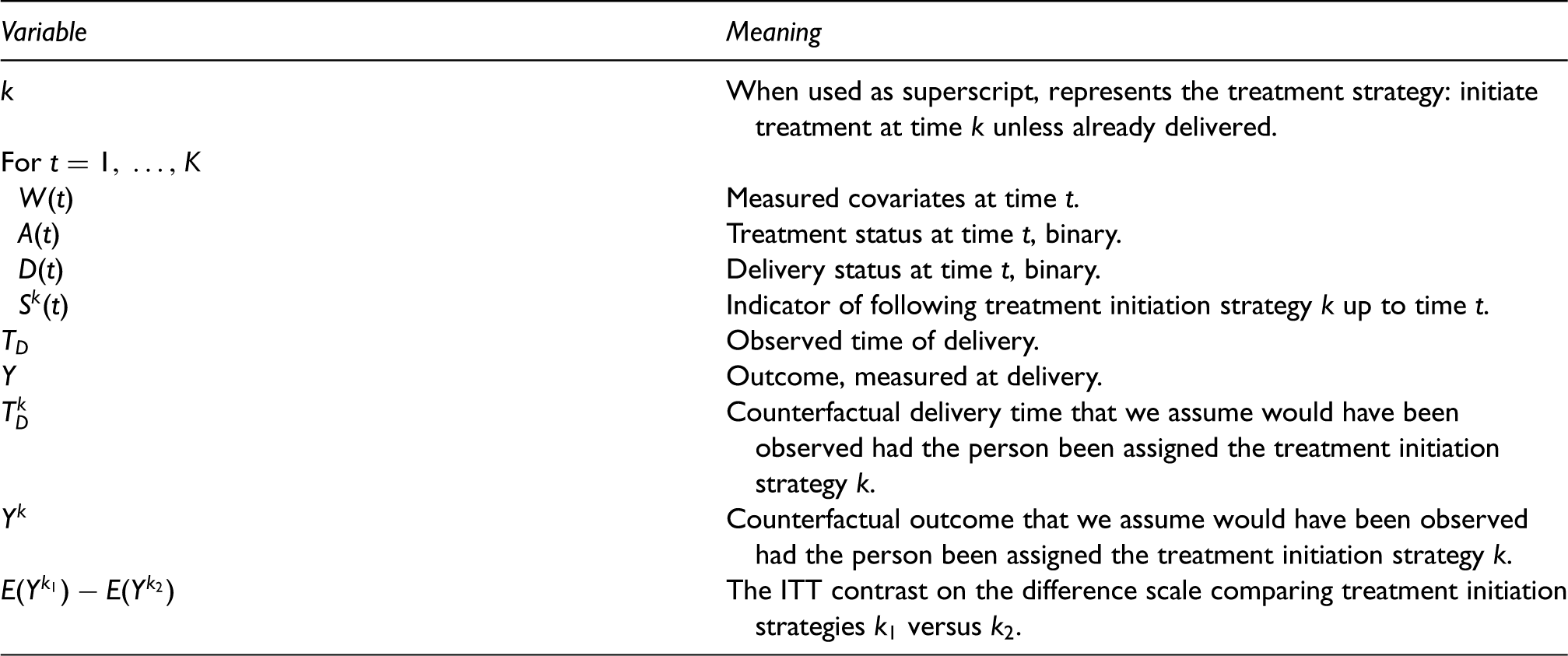
2.2 The treatment strategy approach: ITT and sustained treatment effects
Assume that the outcome is definable in all subjects regardless of delivery time and survival post-delivery. In an ITT analysis, we would aggregate the outcomes of all participants assigned to each arm regardless of whether they followed the protocol of starting treatment at the assigned time. For example, a woman assigned to initiate treatment at 37 weeks but delivers at 28 weeks would still be included in the “start at 37 weeks” group. We could then estimate the effects of being assigned to initiate treatment at each time point by comparing the means or proportions of the outcome in participants assigned to treatment initiation at week 0 versus 13, at week 13 versus 28, at week 28 versus 37, and at week 37 versus control, respectively. In the lower section of Table 1 describing the simple example in the last section, we see that the ITT approach gives the correct conclusion that there is no effect of treatment started at 37 weeks.
The ITT analysis estimates the total effect of the “treatment strategy” of assigning a treatment start-time on the outcome. If the treatment may be initiated early on in pregnancy, this effect includes the component that is a result of potentially shifting the time of delivery, as shown in Figure 1. Thus, it is possible that even if there is no direct effect of the treatment on the outcome, if the treatment has an effect on early delivery and if early delivery has an impact on the outcome, there will be a non-null effect of treatment on the outcome. It is also possible that early delivery and certain outcomes share common causes
In the simple example of Table 1, we saw that performing the analysis with only full-term deliveries gave the correct (null) answer when we were only interested in the effect of late-term exposure. When we are interested in the effects of early-term exposure, performing the full-term subgroup analysis will not be a viable strategy. If early delivery is affected by previous treatment, the full-term analysis will ignore the component of the effect of early treatment that passes through the mediator early delivery (Figure 1, top). In addition, if there is an unadjusted common cause of early delivery and the outcome (
In the next section, we define the ITT parameter under the treatment strategy approach in an observational study. The sustained treatment parameter is similar and defined in the Supplemental Materials. These parameters correspond to those we defined in the target trial.
3. Observational data and parameters of interest
In order to follow the target trials approach, the specifications of the ideal RCT must be used in the cohort design and analysis. For example, subjects in the observational study must have the same entrance criteria and prospective follow-up as in the RCT. If the observational data are collected for administrative purposes, as in our case, then this would involve a secondary selection of subjects “entering” the study when eligible and limiting the analysis to the resulting subset of person-time. If we consider weekly time-points
Thus we consider an observational study where we have sampled independent and identically distributed data of the form
Finally, we can define the variable
3.1 ITT parameter and identifiability assumptions
We are interested in defining the treatment effect corresponding to the ITT analysis (i.e. ITT parameter) in the hypothetical RCT
5
. In order to define this parameter in the context of observational data, we first define the potential outcome
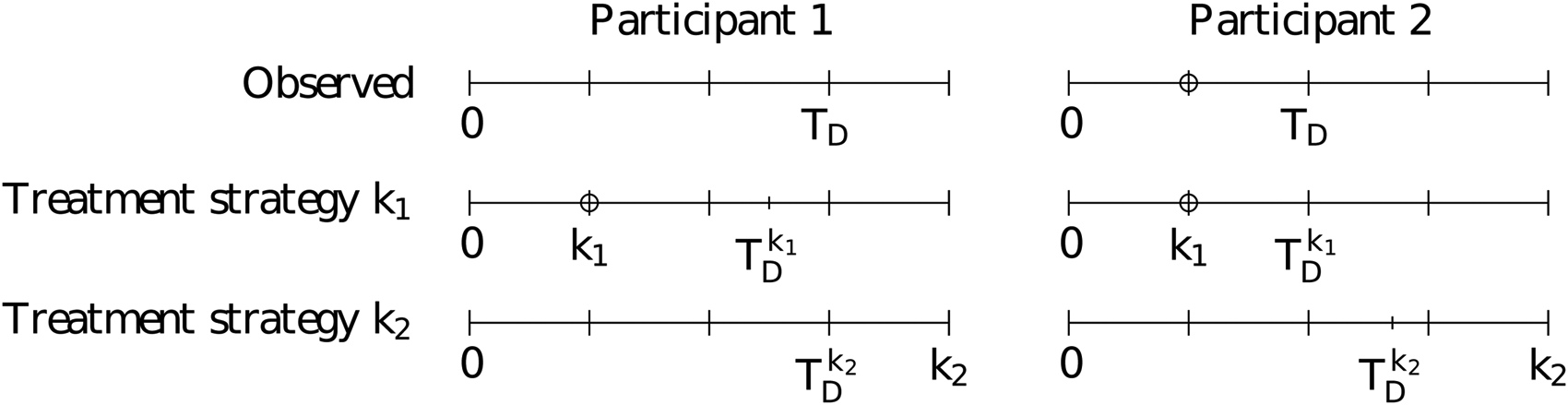
Observed and counterfactual timelines for two example study participants. The top row represents the observed delivery time while the second and third rows represent the counterfactual times under treatment strategies
Note that the potential outcomes defined above are different than the potential outcomes defined as “the outcome that an individual would have had had they started taking the treatment at time
In order to estimate the ITT effect from the observational data, we require additional assumptions. Firstly, we require “consistency,” meaning that if we observe that an individual initiated treatment at time
4. Estimation methods for observational data
In this section, we provide the algorithms to estimate the ITT parameter using IPW, G-computation, and TMLE. Corresponding estimators for the sustained treatment effect are given in the Supplemental Materials.
4.1 IPW
In a standard context where we would be interested in estimating the effect of a time-varying treatment, IPW involves the estimation of the conditional probability of treatment at each time point. The extension of IPW in this context involves the estimation of conditional probabilities of following the treatment strategy. The probability that must be estimated for each time 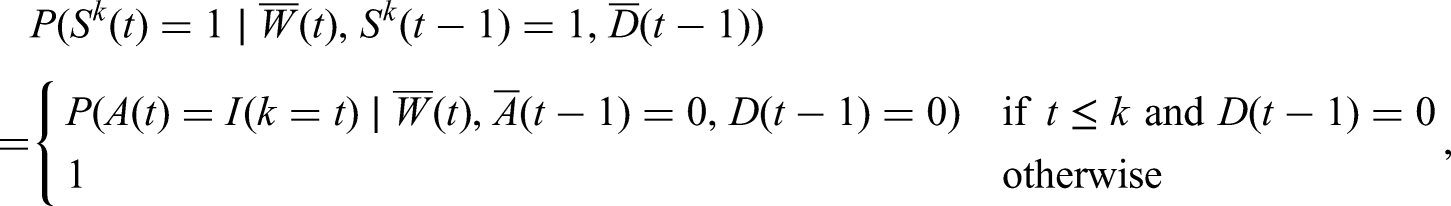
To understand this probability at a given time
We thus see that we only need to estimate the probability of treatment for times prior to or equal to
Once these probabilities are estimated, the IPW calculation for the effect of treatment initiation at 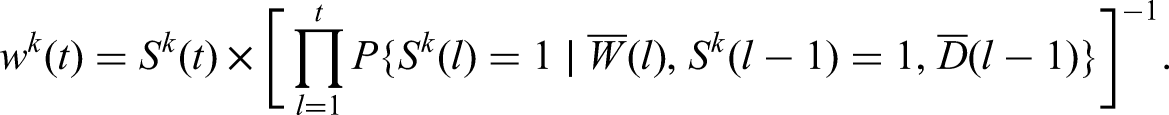
The populational means or risks
4.2 G-computation
G-computation 3 can also be used to estimate the effects of time-dependent treatments.8,31 Where IPW uses estimates of the probabilities of treatment at each time to estimate the target parameter, G-computation requires estimates of components related to the longitudinal outcome process. Here, we adapt G-computation to estimate the ITT parameter in our setting. The derivation and connection to a standard longitudinal procedure 31 are given in the Supplemental Materials.
We need to estimate nested expectations of the outcome conditional on previously following the treatment strategy. We first define the expectation of the outcome, called
Formally, we denote the nested expectations as 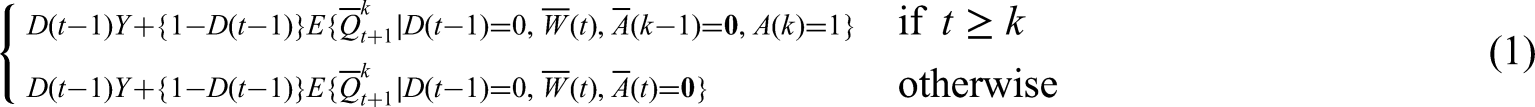
The above equation shows that if delivery has occurred by
We may model the expectations in equation (1) by regressing the predictions
The estimation is iterative: take the vector of the predictions of
4.3 TMLE
TMLE11,12 uses the information from the probability of treatment weights to adjust the components
Suppose that we only consider two time points so that our data are of the form
For the first component of the G-computation used in the TMLE, we estimate the expectation part of equation (1) by regressing
The benefits of using TMLE include double robustness, local efficiency, and the possible integration of machine-learning for the components that need to be estimated. Double robustness here means that if all of the estimators for the probabilities of treatment
5. Simulation study
We conducted a simulation study in order to evaluate these three estimators implemented under the treatment strategy approach for estimating longitudinal effects during pregnancy and to compare with a standard implementation of IPW. We simulated independent and identically distributed observational data of the form
The data-generating structure in the simulation study. In scenario 1, odds ratio
Our goal was to estimate effects of exposure in the time intervals 2-3, 3-4, and 4+, corresponding to the ITT parameters
For each of 1000 repetitions, we drew
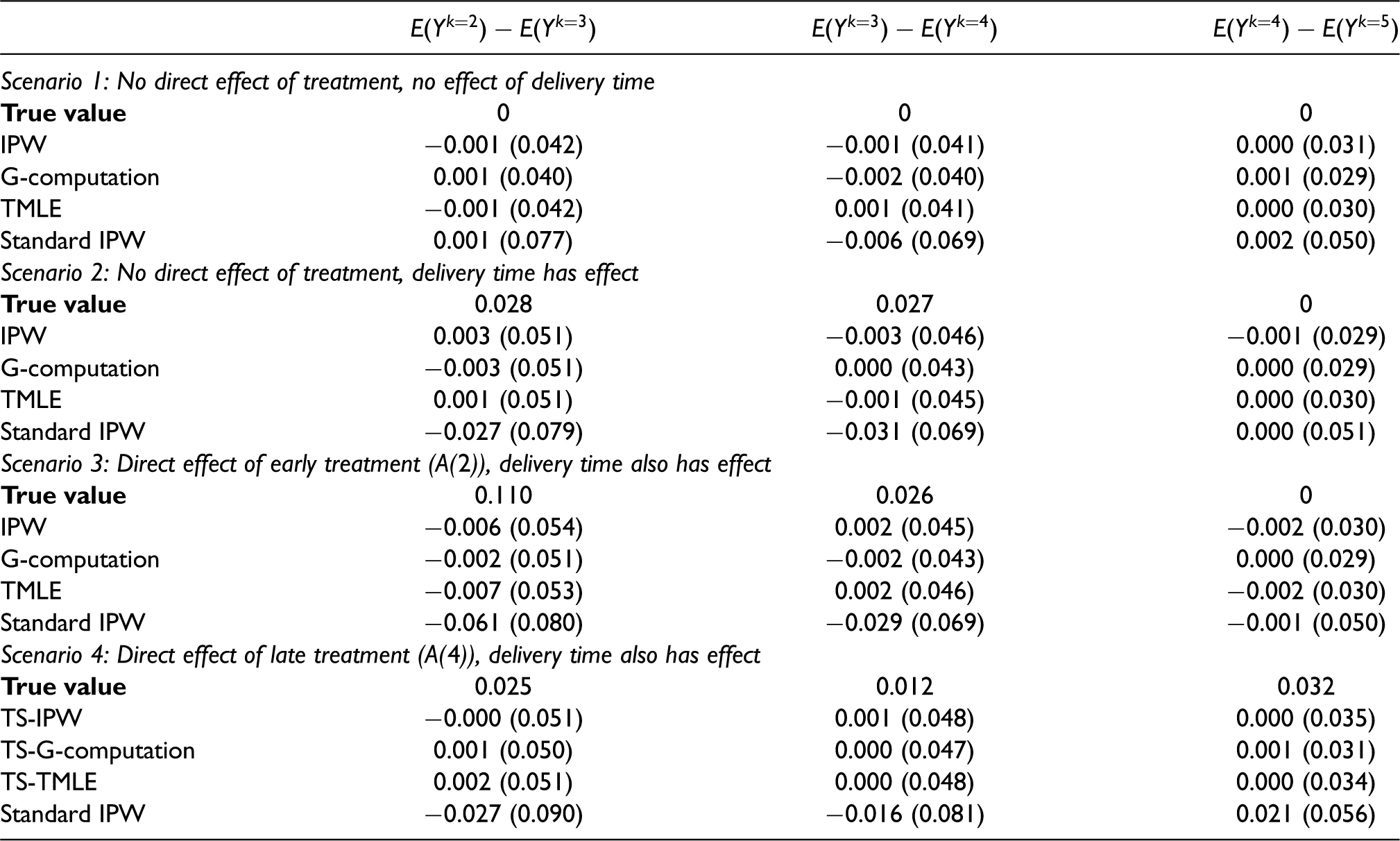
The simulation results are presented in Table 3. In all four scenarios, the treatment strategy approaches were unbiased with similar standard errors. In the first scenario, there was no direct or indirect effect of treatment, so the true total effect of treatment at every time point was null. A standard implementation of IPW could be biased in this scenario because subsetting on the collider
Bias and Monte Carlo standard errors for each estimator in the simulation study. Bias here is defined as the estimate - true value. 1000 repetitions of 1000 independent draws were taken. All estimators are for the ITT treatment strategy parameter except for “standard IPW.”
6. Application
In this application, we aim to evaluate the time-period-specific effects of exposure to ICS versus no controller medication on birthweight in a population of pregnant individuals with mild asthma in the year prior to pregnancy. A limitation of this cohort is that, because it excludes all pregnancies that terminated prior to 20 weeks, this approach cannot directly estimate effects prior to 20 weeks gestation. 33 We thus define our target trial as recruiting individuals at 20 weeks pregnancy who had not yet been exposed to ICS during the pregnancy and were diagnosed with mild asthma prior to pregnancy. In the hypothetical RCT, participants are then randomized to start ICS at 20, 26, 32, or 38 weeks and followed until the end of pregnancy, at which point the outcome, birthweight, is measured. The ITT effect is the effect of interest.
In our observational cohort, we subsetted to all individuals identified as having mild asthma who did not take ICS during the first trimester (prior to 12 weeks; this retained those who became exposed close to baseline at 20 weeks). We evaluated low-dose ICS usage at 20, 26, 32, and 38 weeks, denoted
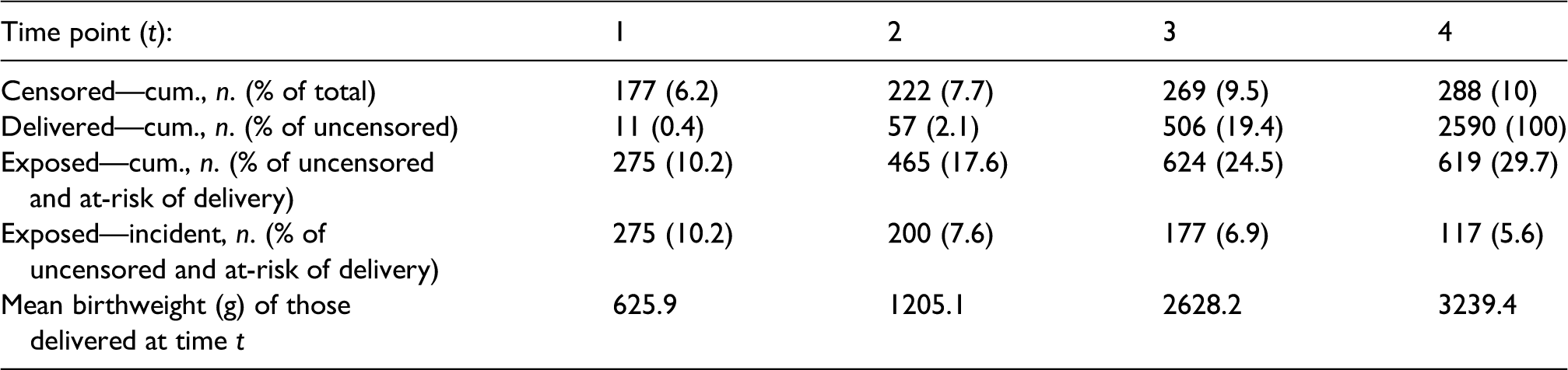
The cohort includes 2878 pregnancies. The baseline summary statistics of this cohort have been published elsewhere. 18 Table 4 provides summary statistics for the number of censored pregnancies, distribution of deliveries, and exposure states over the four time points. Notably, only 10% of the cohort was censored by 38 weeks and roughly 80% of deliveries occurred after 38 weeks. There were also between 117 and 275 new exposures among those at-risk of delivering (i.e. uncensored and still pregnant), giving us a decent sample size to estimate each exposure model for IPW and TMLE. Finally, we can see that birthweight was strongly associated with delivery time.
Censoring, deliveries, exposure to low-dose ICS, and mean birthweight over time in the pregnancy cohort of individuals with mild asthma.
We applied our IPW, G-computation, TMLE, and “standard” IPW as described previously to estimate the ITT parameters representing the effects of exposure at 20, 26, 32, and 38 weeks. We also used a naive linear regression model with only uncensored subjects, adjusting for exposure and confounders at all time points, and ignoring delivery time. For this model, all covariates were assigned a value of zero after the delivery time. Figure 4 presents the estimates and confidence intervals of the four time-specific effects for each estimator. The point estimates for all methods suggested a beneficial effect of ICS for the contrast
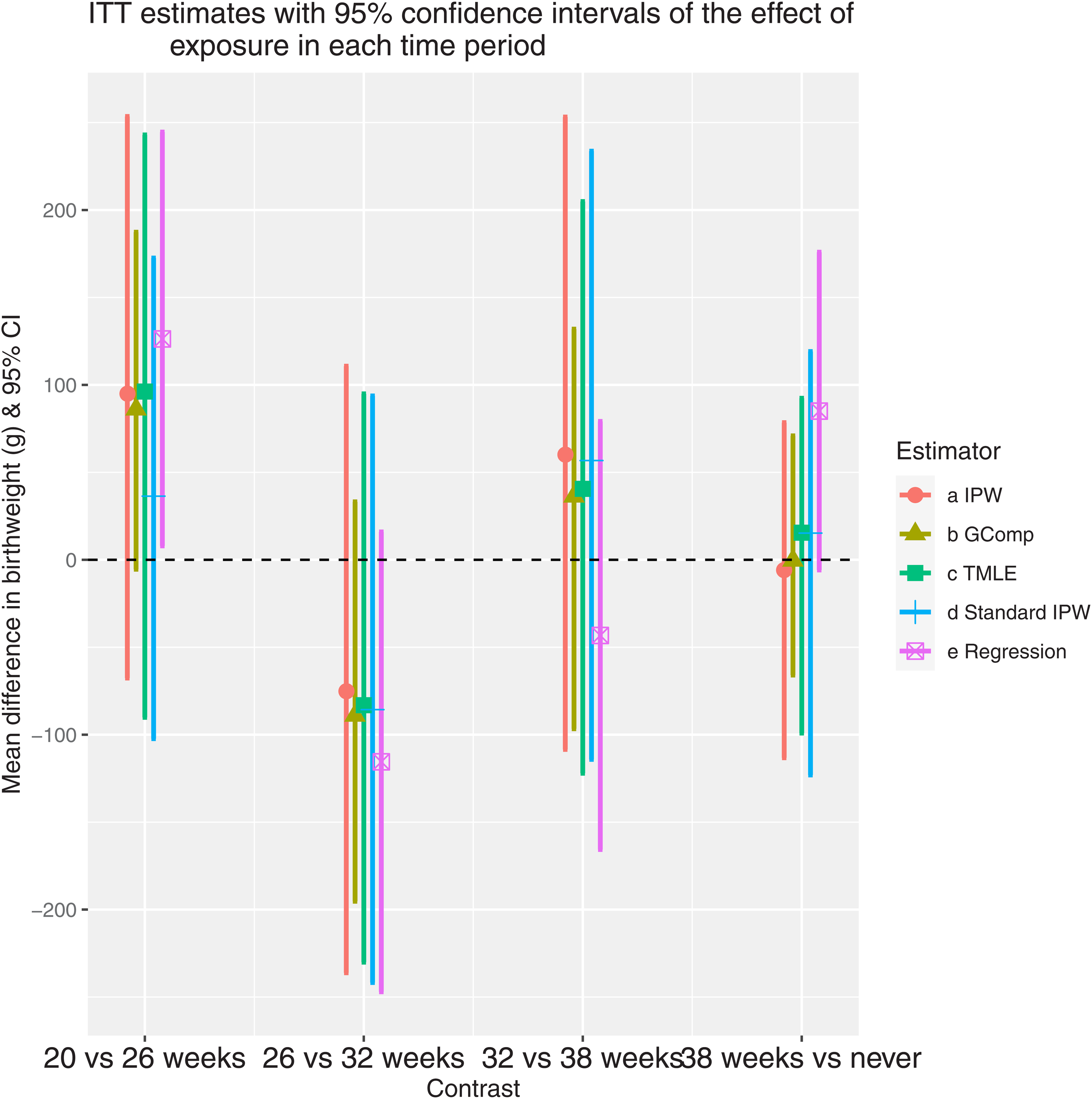
ITT estimates with 95% confidence intervals of the effect of exposure in each time period. The contrasts on the
In this paper, we have proposed new parameters, based on the target trials approach, that represent total gestational age-specific effects of exposure during pregnancy when the outcome is binary or continuous. When the outcome of interest is gestational age at birth, a more standard survival analysis for time-dependent exposures is appropriate. 4 The target trials approach avoids immortal time-bias and exposure misclassification by defining a common baseline and entrance criteria and by prospectively evaluating exposure status rather than grouping participants retrospectively according to a summary of their pregnancy exposures. We also avoid excluding eligible participants due to post-baseline health or pregnancy status by defining contrasts between treatment strategies that can be adhered to regardless of health or pregnancy outcomes during follow-up. This contributes to the definition of the contrast of interest and minimizes selection bias in the estimation.
The total effect that we defined includes the mediated portion of the effect that is the result of medication usage affecting delivery time. Because delivery time is strongly connected with birth outcomes, 34 the total effect of medication exposure is of interest when evaluating medication safety. However, it may also be possible to perform mediation analysis to estimate the component of the effect that is mediated through delivery time and the direct effect of medication exposure on the outcome that is not related to changing delivery time. This would, however, require adjustment for all confounders of delivery time and a method that can incorporate time-varying exposures and mediator. 35
We used a simple numerical example and directed acyclic graphs to demonstrate that naive per-protocol analysis and analyses that subset on full-term births are not appropriate for evaluating gestational age-specific effects of exposure. Other work has criticized methods that do not incorporate the time-varying nature of exposure.21,2 Our proposed parameters and methods allow for the estimation of the total effect of initiating treatment at different times (the ITT effect) and of initiating and sustaining treatment at different times (the sustained treatment effect).
We demonstrated the construction of an IPW estimator, a G-computation estimator, and a TMLE for the estimation of the ITT and sustained treatment effects. Each of these estimators requires the modeling of nuisance quantities, such as the probability of exposure at each time point and the nested conditional expectations of the outcome. Because of the added complication of variable delivery time, these quantities must be decomposed into random and constant components in order to propose reasonable modeling strategies. While the modeling or estimation of the random parts is flexible, standard semiparametric theory places restrictions on the regularity conditions and rates of convergence for the different types of estimators. In particular, among these, only TMLE can formally incorporate nonparametric modeling of these components, though some convergence rate restrictions apply. 36
When designing an observational study, it is necessary to measure all confounders of the initiation time and outcome in order to fulfill the exchangeability assumption for the ITT parameter. If we are interested in estimating the sustained treatment parameter, we must also adjust for all confounders of post-initiation adherence. It is not necessary to measure all confounders of delivery time and outcome. This last fact was addressed in the simulation study where we included an unmeasured confounder of early delivery times and the outcome that had no impact on the unbiasedness of the proposed estimators. However, we also showed that a “standard” IPW implementation that censors early deliveries was substantially biased.
Finally, we demonstrated the application of our methods to evaluate the safety of inhaled corticosteroids during pregnancy, taking birthweight as the outcome of interest. While all confidence intervals for the proposed methods included the null, point estimates suggested a potential benefit of ICS at 20 weeks with evidence that much of the effect is mediated by the delivery time. We would typically expect that ICS use would lead to better control of asthma symptoms and reduce the risk of premature delivery, corresponding to a positive indirect effect through delivery time. Though we adjusted for asthma control indicators, there may have been residual confounding where individuals with more severe symptoms were more likely to use ICS, and this may have attenuated the estimate of the positive effect.
An important limitation of the applicability of these methods is that the outcome must be defined and measurable for all time points under study where delivery or pregnancy loss is possible. When the study baseline is prior to the second trimester this may be problematic as many outcomes, including birthweight, are not definable for early pregnancy losses. If we restrict the cohort to only include pregnancies that continue past a given threshold (e.g. baseline at 20 weeks as in the application) and thus restrict the delivery times under study to times where the outcome can be defined, we can evaluate exposure effects after the threshold. However, in such a restricted cohort, selection bias may hinder the estimation of exposure effects prior to the threshold.37,33 Some potential solutions to dealing with outcomes that cannot be defined for early pregnancy loss were proposed by Chiu et al. 38 in the context of an RCT evaluating fertility therapies. One possibility is to define composite outcomes that include the competing event of “no live birth,” though this doesn’t explicitly consider causal pathways. A second option incorporates separable direct and indirect effects where treatments are conceptually decomposed into the treatment component that impacts the occurrence of live birth and the component that impacts the primary outcome. 38 A third option is to define exposure effects in the principal stratum of pregnancies that would always result in live birth, regardless of treatment.39,40,38 However, because it is difficult to identify pregnancy initiation and very early pregnancy losses, cohorts restricted to pregnancies that cross a time threshold are typical and selection bias omnipresent for the estimation of early exposure effects during pregnancy, 41 though this will impact RCTs as well.
These parameters and methodological extensions are important contributions to the literature as they overcome the limitations of many standard methods used to evaluate the time-dependent impact of medication exposure during pregnancy using observational data. The parameters are interpretable and estimation is flexible, with multiple modeling options available. This work also allowed for insight into the specific assumptions that are required for consistent estimation of the parameters. Understanding these assumptions is important when designing studies and interpreting the results of the application of these methods. The uptake of such methods may thus facilitate more robust evaluations of medication safety during pregnancy using observational data.
Supplemental Material
sj-pdf-1-smm-10.1177_09622802211065158 - Supplemental material for A potential outcomes approach to defining and estimating gestational age-specific exposure effects during pregnancy
Supplemental material, sj-pdf-1-smm-10.1177_09622802211065158 for A potential outcomes approach to defining and estimating gestational age-specific exposure effects during pregnancy by Mireille E Schnitzer, Steve Ferreira Guerra, Cristina Longo, Lucie Blais and Robert W Platt in Statistical Methods in Medical Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research [Operating Grant to LB, New Investigator Award and Canada Research Chair to MES] and by the Natural Sciences and Engineering Research Council of Canada [Alexander Graham Bell Canada Graduate Scholarship Doctoral award to SFG]. CL is a Marie-Curie RESPIRE-3 Fellow. RWP holds the Albert Boehringer I Chair in Pharmacoepidemiology at McGill University.
Supplemental material and code
Supplemental materials describing the sustained treatment effect parameter and containing details of the proposed estimators for both parameters are available online. Code
42
implementing the data generation and estimation in the simulation study is available at ![]() .
.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
