Abstract
Objective
Systemic lupus erythematosus (SLE) is a complex autoimmune condition with diverse manifestations. Childhood-onset SLE (cSLE) presents more severely than adult-onset disease with a higher incidence of severe neurologic or renal manifestations and lower estimated survival rates. There is limited literature on the range of severe gastrointestinal manifestations at presentation in cSLE, and this lack of information results in underappreciation of these potentially life-threatening complications and little guidance regarding management of these patients.
Methods
We reviewed cases of patients with cSLE and severe GI manifestations at presentation diagnosed at our institution and additionally provide a comprehensive review of existing cases in the literature to discuss presenting symptoms, other clinical features, longitudinal course, treatment, and long-term outcomes.
Results
We identified six cases of cSLE with primary, severe GI symptoms at time of diagnosis at our institution and an additional 25 patients who presented similarly in the literature. Severe GI manifestations included protein-losing enteropathy, thoracoabdominal aortitis, severe pancreatitis, lupus enteritis or mesenteric vasculitis, and intestinal pseudo-obstruction. Delays in diagnosis affected several patients, and 26% of patients required surgical intervention. Many patients required intensive immunomodulatory treatment in addition to prolonged bowel rest and parenteral nutrition. Long-term outcomes of GI manifestations varied overall, however, most patients at our institution achieved clinical remission.
Conclusions
Understanding these rare cases of severe GI manifestations in cSLE can aid clinicians in prompt diagnosis and collaborative management, improving clinical outcomes for this vulnerable population.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune condition with heterogeneous manifestations. 1 It is estimated that the incidence and prevalence of SLE in North America is 23/100,000 people per year and 241/100,000 people respectively with 10%–20% of cases being childhood-onset SLE (cSLE).2,3 Despite similarities in laboratory findings, pathophysiology, and management, cSLE has a more severe disease presentation and course, with lower estimated survival rates and a two to three times higher incidence of death from sepsis or renal failure than adult-onset SLE.4–7 Although severe hematologic, renal, cardiac, and neurologic manifestations at presentation of cSLE have been well documented, there is limited literature on gastrointestinal (GI) manifestations at presentation in this population; this rarity of data may delay diagnosis and treatment, leading to serious complications.
GI manifestations in cSLE range from mild symptoms to severe life-threatening complications including lupus enteropathy, aortitis, bowel ischemia, intestinal perforation, and necrotizing or hemorrhagic pancreatitis. The pathophysiology of these complications is suspected to be related to small vessel vasculitis or vasculopathy, but large vessel vasculitis has also been described. Although approximately up to one-third of individuals develop GI symptoms at one point in their cSLE course, GI complaints are rarely the initial presenting symptom. Previous cohort studies and reviews have estimated that GI manifestations are the presenting symptom in 5–19% of cSLE cases.8–12 These cases prove to be challenging diagnostic dilemmas given the extensive number of pediatric conditions that present with non-specific GI complaints and the limited literature describing the prevalence and manifestations of severe GI symptoms at presentation in cSLE.
Methods
With approval from our Institutional Review Board, a retrospective chart review of patients with a diagnosis of SLE as per SLICC 2012 and EULAR/ACR 2019 classification criteria whose disease presentation included severe GI disease was completed. Patient characteristics, clinical course, and outcomes were abstracted. A thorough review of the literature using Pubmed for case reviews or case series of cSLE with significant GI features at diagnosis was performed with the collected articles critically reviewed. Articles were appraised and included in the final analysis if the following criteria were met: (1) new diagnosis cSLE before age 18 and (2) severe GI complications at the time of diagnosis. Severe GI disease was defined as either life-threatening or impacting treatment decisions for induction immunomodulation therapy.
Our initial search identified 89 articles which were screened to ensure they met our inclusion criteria. Of the 89 initial articles, 18 met criteria for our case series. Articles or patients were excluded if patients were over the age of 18 years at the time of SLE diagnosis, GI symptoms were not present at initial presentation, GI symptoms were deemed to not be severe, or if there was insufficient information provided about SLE diagnosis or GI course.
Results
Institutional cohort
Clinical summary of cSLE patients with severe GI manifestations (institutional cohort).
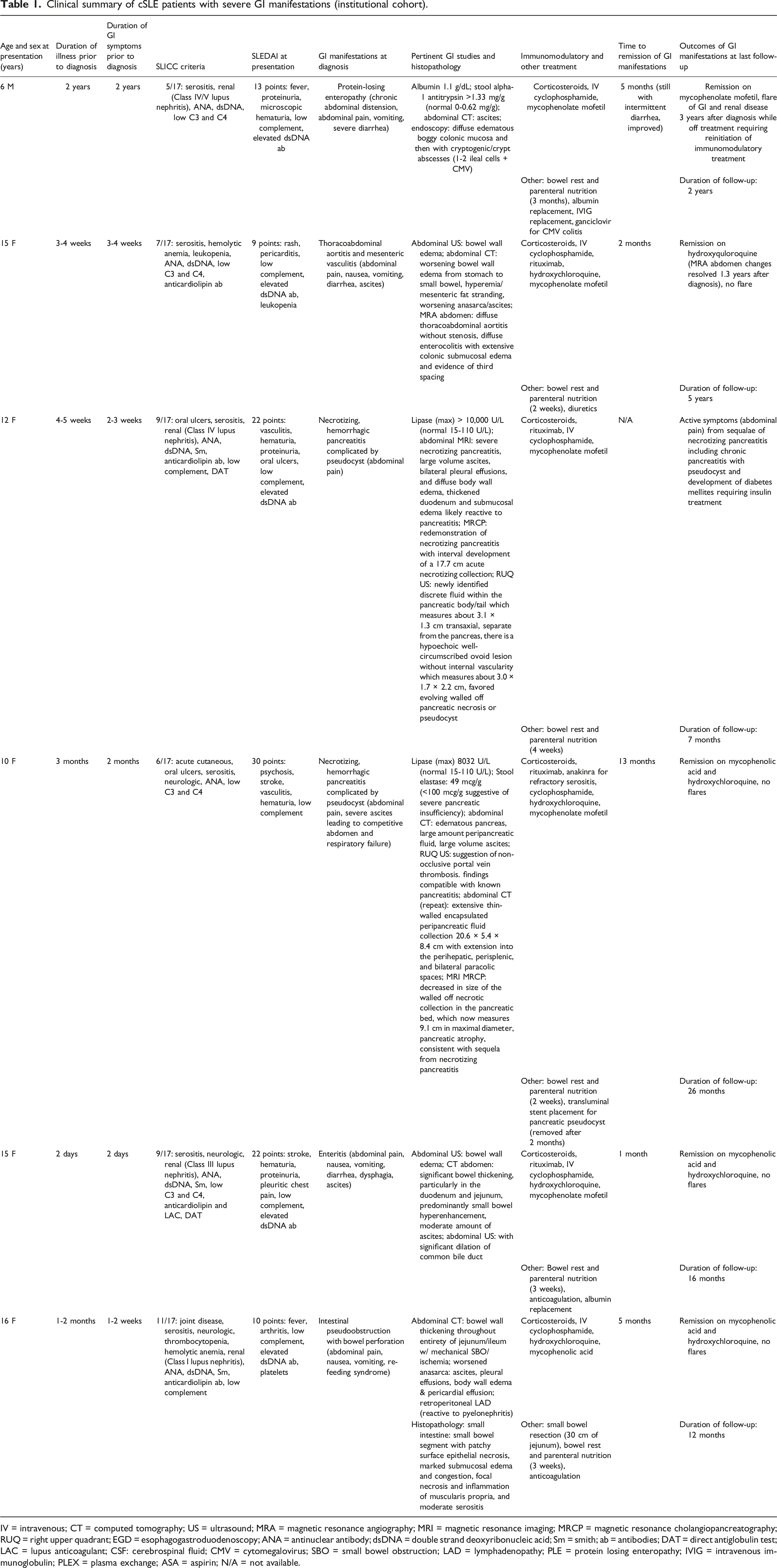
IV = intravenous; CT = computed tomography; US = ultrasound; MRA = magnetic resonance angiography; MRI = magnetic resonance imaging; MRCP = magnetic resonance cholangiopancreatography; RUQ = right upper quadrant; EGD = esophagogastroduodenoscopy; ANA = antinuclear antibody; dsDNA = double strand deoxyribonucleic acid; Sm = smith; ab = antibodies; DAT = direct antiglobulin test; LAC = lupus anticoagulant; CSF: cerebrospinal fluid; CMV = cytomegalovirus; SBO = small bowel obstruction; LAD = lymphadenopathy; PLE = protein losing enteropathy; IVIG = intravenous immunoglobulin; PLEX = plasma exchange; ASA = aspirin; N/A = not available.
Protein-losing enteropathy
Patient 1 is a 6-year-old male with a history of progressively worsening non-painful abdominal distension for 2 years who presented with 1 month of acute-on-chronic abdominal distension associated with pain, emesis, diarrhea and fever. On admission, initial exam revealed anasarca with associated periarticular swelling, and initial evaluation was notable for profound hypoalbuminemia 1.1 g/dL, elevated stool alpha-1 antitrypsin >1.33 mg/g (normal <0.62 mg/g), and computed tomography of abdomen and pelvis (CTAP) with bilateral pleural effusions and significant ascites. Additionally, he was found to have hypocomplementemia with positive ANA, anti-dsDNA and anti-RNP. His urinalysis showed elevated protein and microscopic hematuria with kidney biopsy demonstrating Class IV/V lupus nephritis.
Given disease severity, he was treated with intravenous and oral corticosteroids and monthly cyclophosphamide infusions for induction. He required prolonged bowel rest and parenteral nutrition for 3 months along with frequent albumin replacement. His initial hospital course was further complicated by microscopic GI bleed (fecal occult blood test was positive although stool was never grossly bloody), and endoscopic biopsies ultimately revealed crypt abscesses in addition to CMV positivity. The patient was treated with a 14-day course of ganciclovir for CMV colitis. After discharge, his abdominal distension and hypoalbuminemia resolved after 5 months of continued immunomodulatory therapy.
Thoracoabdominal aortitis
Patient 2 is a 15-year-old female admitted with 1 week of emesis, diarrhea, and abdominal pain. Initial evaluation was remarkable for acute kidney injury (Cr 1.42 mg/dL) responsive to intravenous hydration. CTAP demonstrated diffuse small bowel wall thickening, stranding of mesentery, and small intraperitoneal ascites with trace pericardial effusion which was attributed to infectious inflammatory response. She received supportive care and was discharged after 24 hours with improved symptoms. Two weeks later, she was readmitted due to recurrence and worsening of her GI symptoms. A repeat CTAP at this time showed worsening small bowel wall edema, extensive hyperemia, mesenteric fat stranding, ascites, and anasarca with pericardial and pleural effusions. Further evaluation revealed hypocomplementemia, autoimmune hemolytic anemia, leukopenia, positive ANA and autoantibodies (dsDNA, SSA Ro52/60, SSB and anti-cardiolipin IgM). Magnetic resonance angiography (MRA) of her abdomen demonstrated diffuse thoracoabdominal aortitis without visceral artery stenosis, diffuse enterocolitis with extensive colonic submucosal edema, and persistent third-spacing.
Given significant large vessel vasculitis, her induction therapy consisted of both intravenous and oral corticosteroids with rituximab and monthly cyclophosphamide. In addition, she required prolonged bowel rest and parenteral nutrition for 2 weeks. She had clinical remission of GI symptoms after 2 months of induction treatment. She was later transitioned to maintenance therapy with hydroxychloroquine and mycophenolate after completing a 6 month course of cyclophosphamide. MRA changes resolved after 1 year post-diagnosis. She is currently on hydroxychloroquine monotherapy without any reported flares of GI disease.
Severe pancreatitis
Patient 3 is a 12-year-old female with a history of gastritis (resolved and off medications) who presented to the hospital with 2 weeks of progressive, severe abdominal pain without weight loss, nausea, vomiting, or diarrhea. She also endorsed several weeks of fatigue, painful oral ulcers, left knee pain with swelling and a tender red rash on her palms. On admission, she was found to have evidence of severe necrotizing pancreatitis on magnetic resonance imaging (MRI) of the abdomen with lipase peak >10,000 U/L. Other imaging findings included large ascites, bilateral pleural effusions and diffuse body wall edema. She became critically ill with respiratory failure, pulmonary hemorrhage, and Capnocytophagia septicemia. She was found to have positive ANA and autoantibodies (DAT, anti-dsDNA, anti-Smith, anti-RNP, anti-SSA-Ro60, anti-Scl-70, anti-cardiolipin and anti-MPO) as well as hematuria and proteinuria with kidney biopsy confirming Class IV lupus nephritis.
Patient’s induction treatment included intravenous and oral corticosteroids, rituximab, and intravenous cyclophosphamide. She was placed on bowel rest with parenteral nutrition for approximately 4 weeks. Her course was further complicated by large pseudocyst measuring 17.7 × 6.4 × 10.7 cm that was managed conservatively with close monitoring. Genetic testing was notable for a heterozygous pathogenic mutation in cystic fibrosis transmembrane conductance regulator (CFTR). Clinical remission was achieved, and patient was discharged after a 2 month hospitalization. Maintenance therapy with mycophenolate mofetil was then initiated, and she also continued on hydroxychloroquine. Six months after discharge, she was re-admitted for 2 days due to abdominal pain. Lipase was normal with CTAP showing improvement of pseudocyst to 6.1 × 3.1 × 4.8 cm. An incidental finding of Nutcracker syndrome was reported from CTAP. She continues to have mild, intermittent abdominal pain and nausea attributed to her chronic pancreatitis.
Patient 4 is a 10-year-old female with a similar course. She presented to an outside hospital for 3 months of weight loss, fever, fatigue, malar rash, oral ulcers, and arthralgia and was ultimately diagnosed with SLE given hypocomplementemia, hemolytic anemia, leukopenia, and +ANA. She had improvement with intravenous and oral corticosteroids, hydroxychloroquine, and azathioprine, however, was readmitted 4 weeks later for severe abdominal pain. She required intubation due to respiratory failure secondary to competitive abdomen and was transferred to our institution for further care. Evaluation revealed necrotizing, hemorrhagic pancreatitis with anasarca and ascites with peak lipase 8032 U/L (normal 15-110 U/L). Her hospital course was complicated by neuropsychiatric lupus, non-occlusive portal vein thrombosis, and development of a large pancreatic pseudocyst measuring 20.6 × 5.4 × 8.4 cm (Figure 1). CT abdomen and pelvis. (A) Axial and (B) coronal views demonstrating large pancreatic pseudocyst (20.6 × 5.4 × 8.4 cm).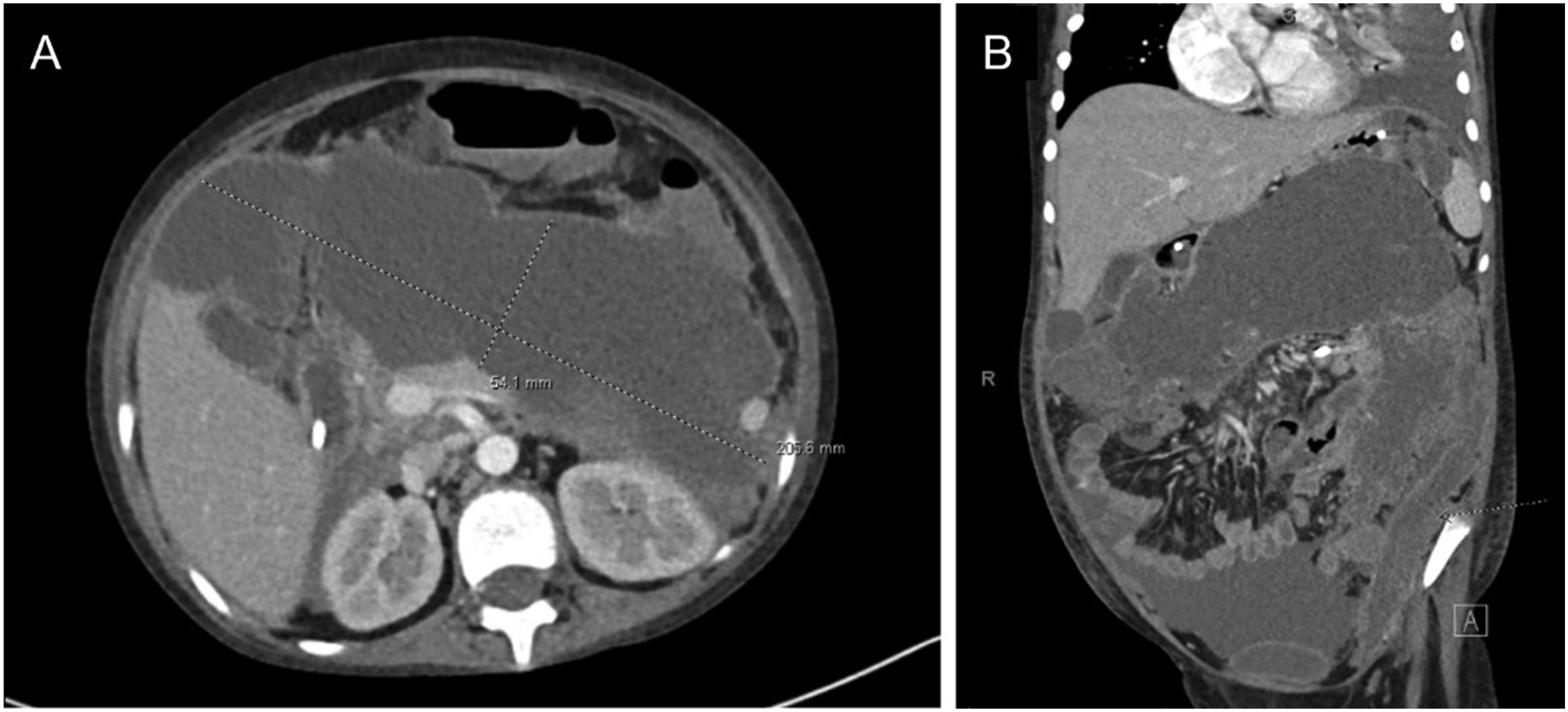
Induction therapy included intravenous and oral corticosteroids, anakinra for severe serositis, rituximab, and monthly intravenous cyclophosphamide. She required bowel rest and parenteral nutrition for 2 weeks. Her pancreatic pseudocyst persisted for up to a year after initial diagnosis, and an Axios stent was placed to allow for a conduit from the pseudocyst to her GI tract. This stent was removed 2 months later without recurrence of pancreatitis or pseudocyst. She is currently in remission on hydroxychloroquine and mycophenolate as maintenance therapy without disease flare.
Lupus enteritis or mesenteric vasculitis
Patient 5 is a 14-year-old female who presented with 2 days of acute, severe, and diffuse abdominal pain with associated nausea, vomiting, and dysphagia. CTAP revealed significant bowel thickening, particularly affecting the duodenum and jejunum, and a moderate amount of ascites consistent with enteritis (Figure 2). Further evaluation revealed that she had hypocomplementemia with positive ANA and autoantibodies (DAT, anti-dsDNA, anti-Smith, and anti-phospholipid). Antibodies to RNP, SSA, Scl-70 as well as myeloperoxidase (MPO) were also positive. Patient had microscopic hematuria with significant proteinuria. Kidney biopsy later revealed Class III lupus nephritis. Her clinical course was further complicated by micro-ischemic strokes. CT abdomen and pelvis. (A) Axial and (B) coronal views demonstrating extensive bowel wall thickening and edema primarily involving the proximal small bowel.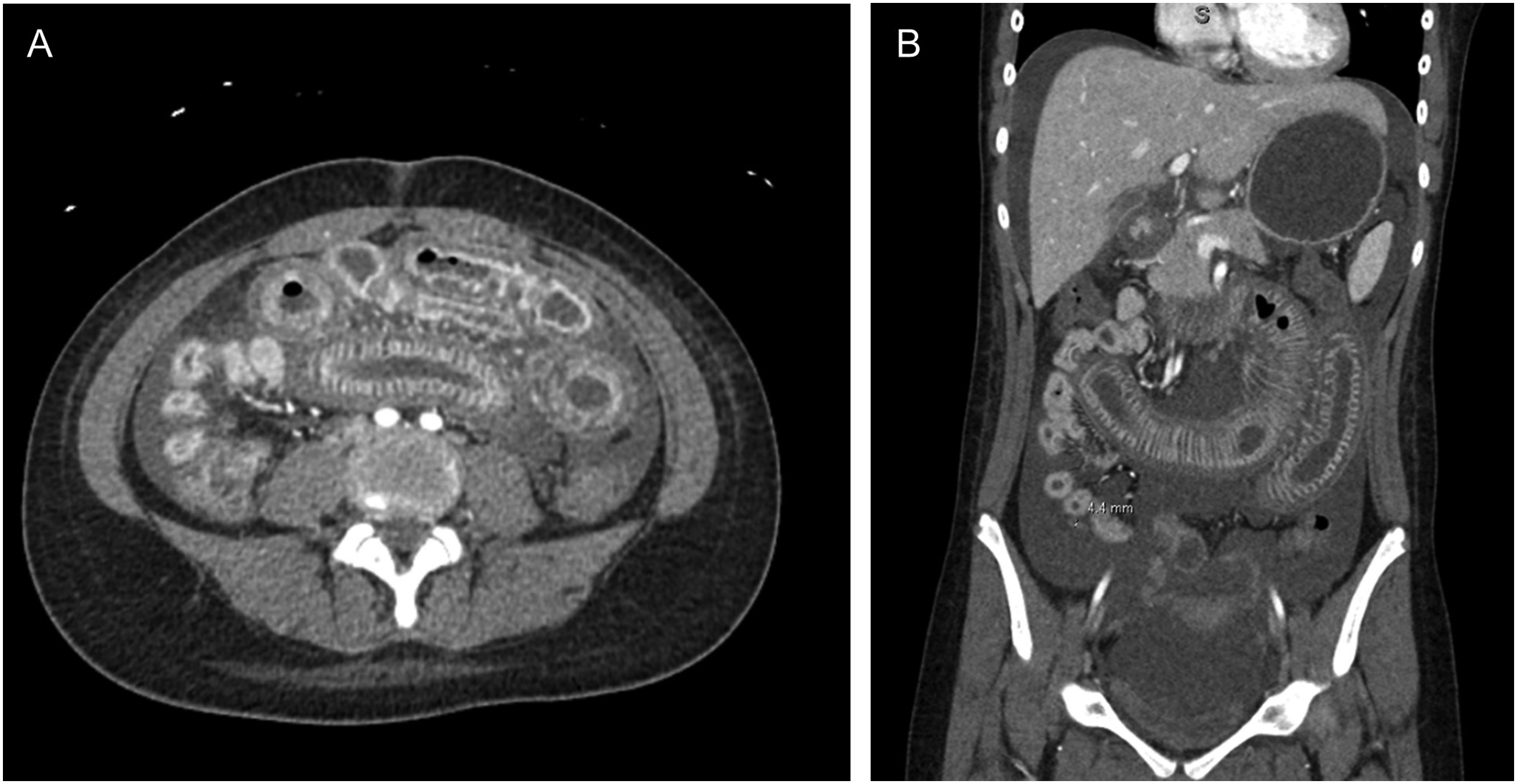
Due to her severity of disease, induction therapy was started with both intravenous and oral corticosteroids, rituximab, and monthly intravenous cyclophosphamide. She was placed on bowel rest and parenteral nutrition for 3 weeks with slow transition to enteral nutrition. Early advancement of diet was complicated by hematochezia. Bowel ultrasounds were used serially to assess for bowel wall edema and enhancement while enteral nutrition was advanced. She had complete resolution of her GI symptoms and imaging findings 1 month post-treatment. She remains on hydroxychloroquine and mycophenolate as maintenance with sustained clinical remission.
Intestinal pseudo-obstruction
Patient 6 is a 16-year-old female who developed 2 weeks of progressively worsening abdominal pain, emesis, and presenting in compensated shock. Her disease features were initially attributed to multi-drug resistant E. coli pyelonephritis. CTAP revealed pyelonephritis as well as areas of focal proximal small bowel thickening consistent with enteritis. Laboratory findings were notable for severe anemia and thrombocytopenia. She was placed on bowel rest and started on antibiotics with temporary improvement in symptoms. Following advancement of her diet, her abdominal pain and vomiting recurred with increased severity. Repeat CTAP revealed increased bowel wall thickening throughout entirety of jejunum and ileum with evidence of mechanical small bowel obstruction and ischemia. Imaging also showed ascites, pleural effusions, body wall edema, and a pericardial effusion (Figure 3). She underwent multiple exploratory laparotomies resulting in resection of 33 cm of jejunum due to ischemic injury (Figure 4). CT of Abdomen and Pelvis. (A) Axial and (B) sagittal views demonstrating severe bowel wall thickening throughout entirety of jejunum and ileum with a transition to decompressed right lower quadrant small bowel, demonstrative of mechanical small bowel obstruction with bowel wall thickening suggestive of ischemia. Stark contrast in ischemic versus perfused bowel during initial exploratory laparotomy where 33 cm of jejunum was resected.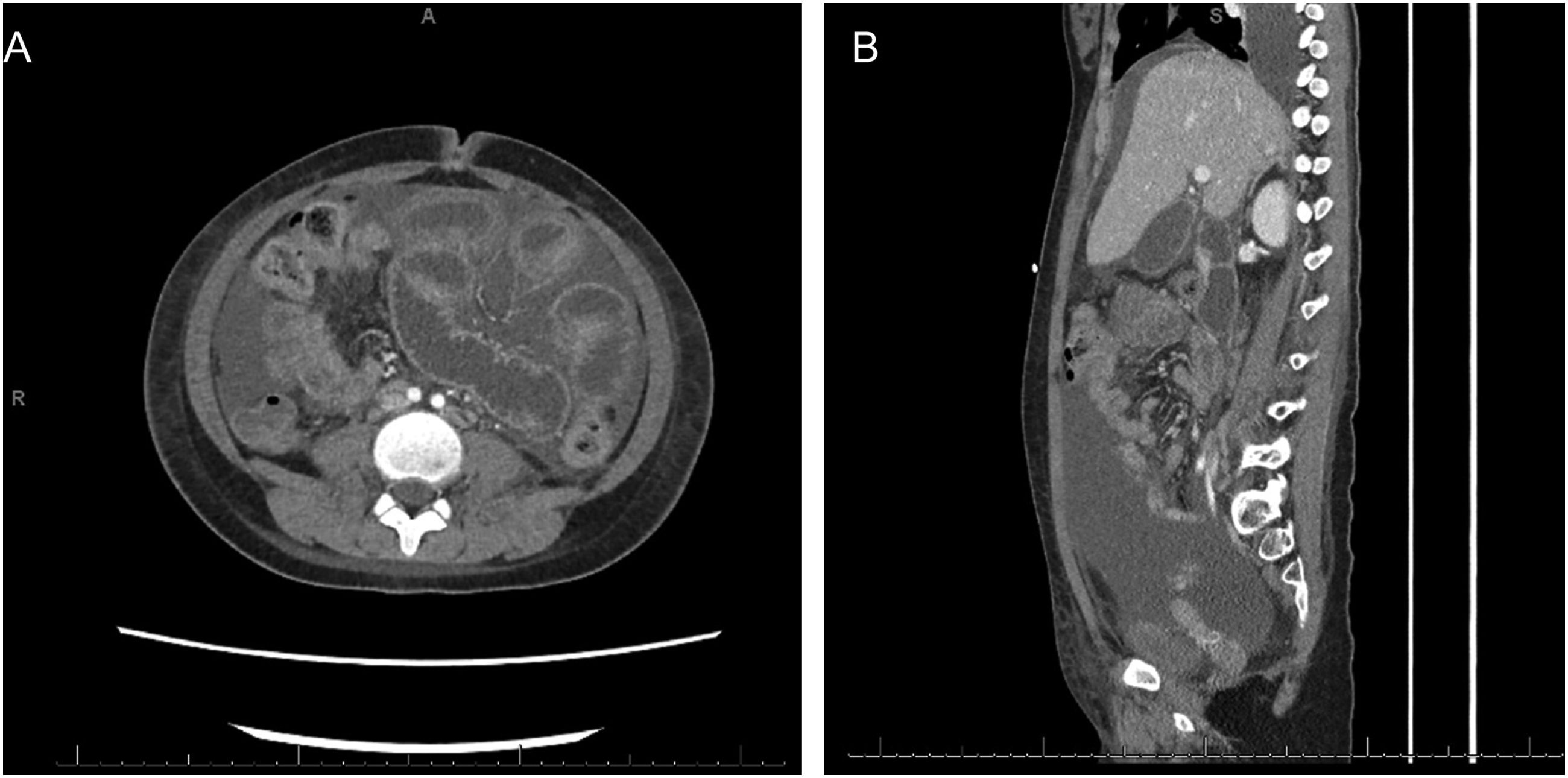
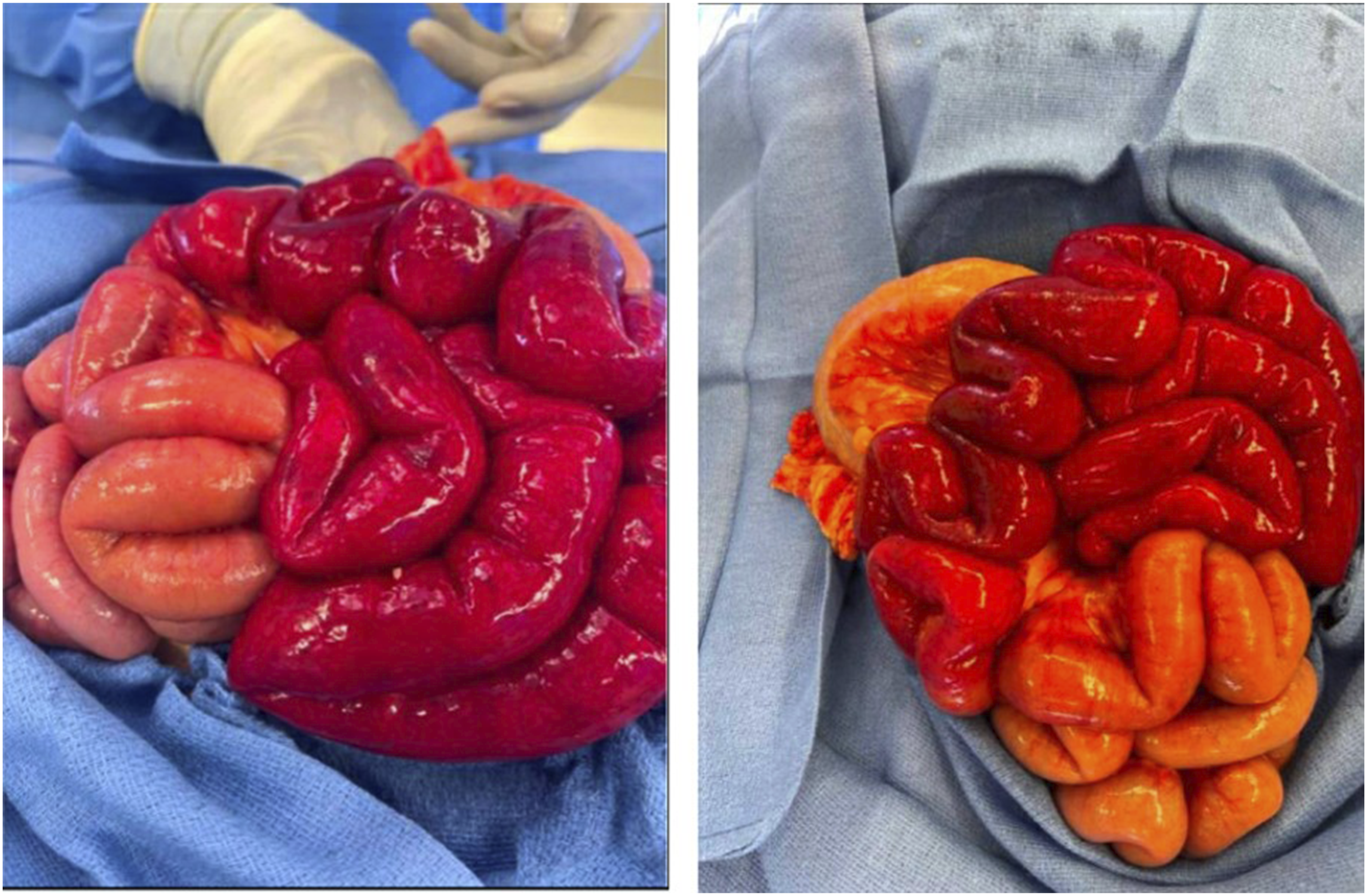
Further review of the history revealed pallor, chronic arthralgia, behavioral and cognitive symptoms concerning for psychiatric manifestations. Additional testing verified diagnosis of cSLE, with proteinuria, microscopic hematuria, hypocomplementemia, ANA positivity and positive autoantibodies (anti-dsDNA, anti-Smith, and anti-phospholipid). Kidney biopsy showed Class I lupus nephritis. A diagnosis of neuropsychiatric lupus was made with imaging findings of micro-ischemic strokes and cerebrospinal fluid abnormalities.
Induction immunotherapy included intravenous and oral corticosteroid along with monthly intravenous cyclophosphamide. Bowel rest and parenteral nutrition was started for a total of 3 weeks. Early advancement of diet resulted in recurrent abdominal pain and diarrhea. Complete remission was achieved after 5 months of therapy. She remains in remission with hydroxychloroquine and mycophenolic acid as her maintenance agents.
In summary, our institutional cohort was composed of primarily female patients (83%, 5/6) who presented at an average age of 12 years. Within our cohort, the most reported symptoms prior to cSLE diagnosis were abdominal pain and emesis (83%) while diarrhea was seen in half the patients. The duration of GI symptoms prior to cSLE diagnosis ranged from 2 days to 2 years. Surgical intervention was seen in two patients including Axios stent placement and exploratory laparotomy with bowel resection.
Literature review cohort
Clinical summary of cSLE cases with severe GI manifestation reported in the literature (Literature Review Cohort).
IV = intravenous; CT = computed tomography; US = ultrasound; MRA = magnetic resonance angiography; MRI = magnetic resonance imaging; MRCP = magnetic resonance cholangiopancreatography; RUQ = right upper quadrant; EGD = esophagogastroduodenoscopy; ANA = antinuclear antibody; dsDNA = double strand deoxyribonucleic acid; Sm = smith; ab = antibodies; DAT = direct antiglobulin test; LAC = lupus anticoagulant; CSF: cerebrospinal fluid; CMV = cytomegalovirus; SBO = small bowel obstruction; LAD = lymphadenopathy; PLE = protein losing enteropathy; IVIG = intravenous immunoglobulin; PLEX = plasma exchange; ASA = aspirin; N/A = not available.
Discussion
Several cohort studies and reviews have explored GI manifestations in cSLE, both at the initial presentation and throughout the clinical course. Although literature within the pediatric population is limited, this research suggests 19%–27.5% of patients with cSLE develop GI system involvement overall.9,10 Of these patients with GI involvement, between one- to two-thirds experience these symptoms at their initial presentation.9–11 Most studies report abdominal pain as the most common GI symptom,8,9,11 but specific data on the frequency and outcomes of severe GI manifestations in children with SLE is lacking. In a French multicenter cohort study describing the initial presentations of 155 patients with cSLE, Bader-Meunier et al. reported 8 severe GI events including episodes of pancreatitis and intestinal myositis with ileus. 8 Additionally, in a retrospective case–control study comparing cSLE patients with and without mesenteric vasculitis, Zhu et al. found that cSLE patients with mesenteric vasculitis had high SLEDAI-2 K scores at onset. 33 These children were also more likely to have damage to other organs including kidneys, serous cavity, and lungs and more likely to receive intensive immunomodulatory therapy with cyclophosphamide when compared to cSLE patients without mesenteric vasculitis. 33 Most of the patients with mesenteric vasculitis in this cohort achieved remission of their GI symptoms within 1 month of treatment with low relapse rates, however, their overall recovery was worse compared to cSLE patients without mesenteric vasculitis. 33
At our institution, 6 cases of the 714 total cases of cSLE (0.84%) evaluated over 23 years presented with primary GI symptoms with severe complications. Through our literature search, we identified an additional 25 patients who presented similarly. Consistent with the literature, females made up most of our combined sample size with a female to male ratio of 6.75:1 and mean age of presentation of 13 years. 1 The most common symptom at time of presentation in both groups were abdominal pain, emesis, or diarrhea. Abdominal complaints are a common occurrence in pediatric patients with a reported total of 4.2 million encounters for abdominal pain across the country from 2004 to 2011 with 2 million of those being at emergency centers (EC). 34 While abdominal pain, emesis, and diarrhea in conjunction with additional finding such as dysuria, right lower quadrant pain, peritonitis or guarding help guide the diagnostic work up, alone they are non-specific symptoms that are more often attributable to other causes including viral processes or functional GI disorders. Unfortunately, misdiagnosis of abdominal complaints is not a rare occurrence as several studies have reported such cases. Naiditch et al. found that of the 816 pediatric patients who underwent appendectomy from 2007 to 2010, 4.8% of them were initially misdiagnosed with viral gastroenteritis or constipation, while Reynold et al. report that 39% of patients seen at their EC were diagnosed with “abdominal pain” and discharged home without a clear etiology.35,36 Patients within our institutional cohort were diagnosed with acute gastroenteritis, appendicitis, hypothyroidism, and pyelonephritis prior to cSLE diagnosis contributing to delays in diagnosis. Time to diagnosis ranged from 2 days to 2 years in our institutional cohort, and 3 days to 3 years in cases identified from our literature search.
We also found that a significant portion of our combined cohort (26%, 8/31) underwent surgical intervention. Of these cases, 7 patients underwent emergent exploratory laparotomies with 4 requiring resections of bowel due to bowel necrosis or perforation, 4 undergoing appendectomy, and 1 requiring transluminal stent placement for a large pancreatic pseudocyst. Surgical interventions were pursued primarily due to concerns for acute abdomen as determined by physical exam. In a study completed by Shoeler et al., they found 1% of patients presenting to their EC with an abdominal complaint underwent a surgical procedure. 37 In comparison, patients within our cohort underwent surgical procedures at a much higher rate due to severity of their symptoms without a clear etiology. The severity of these presentations highlights the importance of making prompt diagnosis to expedite treatment of cSLE to prevent such invasive interventions and severe complications.
Although GI complaints were the most prominent symptom for these patients at the time of diagnosis, all patients had other features highly suspicious for cSLE including mucocutaneous, joint, cardiopulmonary, and hematologic manifestations. In our cohort, 62% of patients had renal and/or neurologic manifestations, two major organs which indicate a severe disease process, and this was similarly noted in the published cases (60%, 15/25). Besides typical autoantibodies associated with cSLE including anti-dsDNA and anti-Sm antibodies, our institutional cohort of patients exhibited various other autoantibodies including anti-SSA Ro52/60 (67%), anti-SSB (33%), anti-RNP (67%), anti-Scl70 (33%), and anti-MPO (33%) antibodies. Many of these patients required significant immunosuppression as induction therapy for cSLE including agents such as cyclophosphamide and rituximab in addition to corticosteroids. All patients in our series received supportive therapies including prolonged bowel rest and parenteral nutrition to prevent bowel ischemia and perforation. One patient with chronic pancreatitis complicated by pseudocyst required transluminal stent placement which highlights the importance of multi-specialty care to limit morbidity and preserve a high quality of life for these children.
Lastly, all patients at our institution, besides one (Patient 3) who has shortest length of follow-up, achieved complete remission of cSLE (SLEDAI-2K of 0) and complete resolution of GI manifestations between one to thirteen months after initial diagnosis. Patient 1 had a flare of SLE while he was off all medications requiring re-initiation of immunomodulatory treatment. Long-term outcomes in the published cases were often not well defined or discussed in case reports, however available data shows 15 out of 25 patients had remission without flares (60%), 6 had at least one documented flare or readmission to the hospital for GI symptoms (24%), and 4 passed away (16%).
Conclusions
Childhood-onset SLE is a complex autoimmune condition characterized by immune dysregulation with a wide array of clinical symptoms. Diagnosis of cSLE can be challenging and even more so when patients present atypically with predominant GI symptoms leading to delays in diagnosis and treatment. This delay results in possibly preventable progression of disease burden and severe clinical complications. For this reason, it is important that providers maintain a broad differential that considers cSLE when facing GI symptoms in pediatric patients and thoroughly assess for other signs or laboratory abnormalities which may suggest a systemic process including renal or hematologic manifestations. Patients with cSLE and severe GI manifestations also benefit from multi-specialty care with close collaboration between rheumatologists and gastroenterologists.
Footnotes
Author contributions
Drs. James Orozco, Jessica Nguyen, Sara M. M. Yasrebi, Marnie Blalock, and Sara L. Grisales contributed to the conception and design of the report, reviewed the course data, performed literature review and appraisal, drafted the initial manuscript, and critically reviewed and revised the manuscript. Drs. Craig L. Jensen and Marietta M. DeGuzman contributed to the conception and design of the report and critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data Availability Statement
All authors confirm that the data supporting the findings of this study are available within the article.
