Abstract
Objectives
Myositis is an infrequent feature of SLE and may often be overlooked. We aimed to estimate the incidence of myositis in SLE, and to determine demographic and clinical factors associated with it.
Methods
Within our lupus cohort, we identified potential myositis cases using the SLICC Damage Index for muscle atrophy or weakness, the SLEDAI-2K item for myositis, and annually measured serum creatinine kinase. Cases were confirmed through chart review. We performed descriptive analyses of prevalent myositis cases as of January 2000. From that point onward, we studies patients without myositis to determine risk of incident myositis, using cohort analyses adjusted for demographic variables (age, sex, race/ethnicity).
Results
As of January 2000, there were 5 prevalent myositis cases in our SLE cohort. Among 560 SLE patients with a study visit from January 2000 onward, with no history of myositis at baseline, 5 new cases (4 females, 1 male) were identified over an average follow-up of 8.5 years (incidence 1.05 cases per 1000 person-years). There was a higher proportion of Caucasians in the non-myositis group versus myositis group, with a trend for fewer females in the myositis cases. Arthritis, Raynaud’s phenomenon, and anti-Smith antibodies were common pre-existing features, occurring in all incident myositis cases. In Cox regression analyses adjusting for age, race/ethnicity and sex, non-Caucasian patients had a markedly increased risk of developing myositis.
Conclusion
We found a low incidence of myositis in our SLE cohort. A cluster of variables, particularly non-Caucasian race/ethnicity, arthritis, Raynaud’s phenomenon, and anti-Smith antibodies were associated with risk of developing myositis in SLE. These variables may aid clinicians in identifying SLE patients at highest risk for this important complication.
In systemic lupus erythematosus (SLE) myalgias are commonly experienced but overt myositis is not a prominent feature.1–5 Some studies have suggested that SLE patients with more severe disease are most likely at risk for developing myositis, 3 and that myositis itself may be one early indicator of poor SLE outcomes. 1 Because myositis is not a well-known SLE feature, this potentially disabling feature may be overlooked. 6 The current study was designed to investigate prevalent and incident myositis cases in our SLE cohort, and to evaluate demographic and clinical factors associated with development of this condition.
Materials and methods
Population
The SLE McGill University Health Centre (MUHC) cohort enrolls unselected consecutive patients meeting the 1997 revised American College of Rheumatology or 2012 Systemic Lupus International Collaborating Clinics (SLICC) SLE classification criteria. 7 We have followed these patients with annual study visits, where information is collected on disease activity and other demographic and clinical data. For the current study, we identified all patients with a study visit from January 2000 and onward.
Demographic and clinical data
Demographic variables evaluated as potential covariates of interest included age at SLE diagnosis, sex, race/ethnicity, and SLE duration at the first assessment as of January 2000 or thereafter. Clinical variables included Raynaud’s phenomenon, documentation of arthritis or nephritis based on the 1997 modified American College of Rheumatology, ACR or Systemic Lupus International Collaborating Clinics, SLICC SLE classification criteria7,8 or the Systemic Lupus Erythematosus Disease Activity Index 2000 scores (SLEDAI-2K), and antibody serologies for anti-ds-DNA, anti-Smith, anti-RNP, anti-SSA/Ro, and anti-SSB/La. These variables were chosen on the basis of past publications suggesting possible associations of these variables with myositis in SLE, mixed connective tissue disease, and/or overlap syndromes.1,9
Laboratory testing
Our lab performs assays for these antibodies using Euroimmun immunoassay kits (Euroimmun AG, Luebec). 10 All lab tests were assessed at baseline (first visit as of Jan. 2000 or enrolment visit if later than this date) and annually up to the index date of each risk set. Normal ranges for serologies were obtained from the McGill University Health Centre laboratory and were as follows: anti-ds-DNA (0-99 IU/ml), anti-Smith (0-19 RU/ml), anti-RNP (0-19 RU/ml), anti-SSA/Ro (0-19 RU/ml), anti-SSB/La (0-19 RU/ml), CK (24-160 U/L for females, and 24-195 U/L for males).
Possible cases of myositis were identified using the SLICC-ACR Damage Index, SDI 11 for muscle weakness or atrophy, the SLEDAI-2K variable myositis, and annually measured serum creatinine kinase levels. Patients positive for any of these variables were then confirmed through chart review for results of muscle biopsies, muscle magnetic resonance imaging, electromyography, and/or neurologic consultation.
Statistical analysis
Descriptive statistics for demographic and clinical variables were calculated for all SLE non-myositis and both prevalent and incident SLE-myositis cases. Differences in proportions between groups was analysed using the Newcombe-Wilson score method. We then used Cox proportional hazards regression to generate hazard ratios (HRs) with 95% confidence intervals (CIs) for myositis incidence from January 2000 onward. Here we used time-dependent clinical variables (ever/never, up to the index time for each risk set), including nephritis (based on 1997 ACR updated SLE classification criteria), anti-RNP, anti-SSA/Ro, anti-SSB/La, and anti-dsDNA antibodies. Since arthritis, Raynaud’s phenomenon, and anti-Smith antibodies were present in all incident myositis cases, we could not evaluate these in HR models.
Written informed consent was obtained from all participants, as well as approval from the McGill University Health Centre Research Ethics Board.
Results
Patient demographics
As of January 2000, there were 5 prevalent myositis cases in our SLE cohort. Among 560 SLE patients with a study visit from January 2000 onward, with no history of myositis at baseline, 5 new cases (4 females, 1 male) were identified over an average follow-up of 8.5 years (incidence 1.05 cases per 1000 person-years).
Table 1 presents baseline demographic characteristics of the prevalent and incident myositis cases, and SLE patients who never developed myositis. There was a higher proportion of Caucasian patients in the non-myositis groups relative to both the prevalent and incident myositis patients. Based on SLE classification criteria or SLEDAI-2K, the vast majority of SLE patients with myositis (including all the incident cases) had arthritis prior to the development of myositis, but arthritis was also very common among subjects who did not develop myositis. All cases had Raynaud’s phenomenon and had been positive for anti-Smith antibodies.
Patient demographics and difference in proportions between groups.
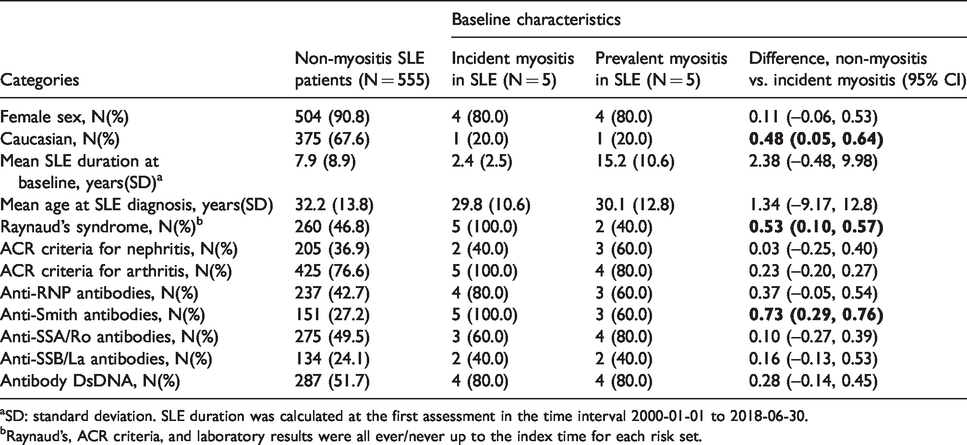
aSD: standard deviation. SLE duration was calculated at the first assessment in the time interval 2000-01-01 to 2018-06-30.
bRaynaud’s, ACR criteria, and laboratory results were all ever/never up to the index time for each risk set.
Cohort analyses
The results for the univariate and multivariate analyses are shown in Table 2. The multivariate analysis confirmed that non-Caucasian patients had an increased risk of developing myositis (adjusted HR: 10.7, 95% CI 1.17, 96.9). Since arthritis, Raynaud’s phenomenon and anti-Smith antibodies were present in all incident myositis cases, we could not evaluate these in HR models. For all other variables, confidence intervals were very wide and included the null value.
Unadjusted and partially adjusted hazard ratio, HR estimates for myositis risk in SLE.
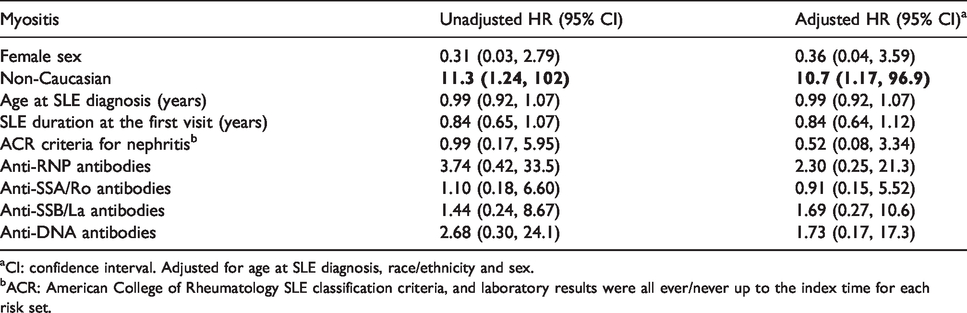
aCI: confidence interval. Adjusted for age at SLE diagnosis, race/ethnicity and sex.
bACR: American College of Rheumatology SLE classification criteria, and laboratory results were all ever/never up to the index time for each risk set.
Many clinical variables correlated with each other (Supplementary Table 1). For example, anti-Smith antibody positivity was correlated with positivity for other antibodies (anti-RNP, anti-dsDNA, anti-SSB/La, anti-SSA/Ro) in addition to ACR nephritis and Raynaud’s phenomenon. The highest correlation was between anti-Smith and anti-RNP antibodies (correlation coefficient 0.6361).
Supplementary Table 2 provides information about disease activity and medications over time in the incident cases.
Discussion
We estimated myositis incidence in SLE to be 1.05 events per 1000 person-years. Our multivariate analyses suggest that non-Caucasian race/ethnicity was strongly associated with myositis risk, and all incident cases also had arthritis, Raynaud’s phenomenon and anti-Smith antibodies.
The finding that non-Caucasian patients are more prone to developing myositis than Caucasian patients is not a prominent feature in prior literature (which is focussed on myositis prevalence, not incidence). One recently published study similarly found that there was a high proportion of non-Caucasian patients in SLE patients with myositis. 9 Nevertheless, very few other studies have measured race/ethnicity as a risk factor for myositis in SLE, and some only included racially homogenous patient populations.3,12 Our observation that non-Caucasian patients were more likely to develop myositis, could reflect the known observation that SLE patients of African, Hispanic, and Asian race are more likely to develop multiorgan involvement and have multiple positive serologies. 13 Further analyses of how phenotypic subsets of SLE might be related to race/ethnicity (and possible underlying genetic or epigenetic changes) are one direction that warrants future investigation.
Most of the myositis cases, including all of those with incident myositis, had arthritis as an SLE ACR criteria, although so did three-quarters of the non-myositis SLE patients. The suggestion of arthritis as a factor associated with myositis has been found in two cross-sectional analyses,1,9 but ours is the first to evaluate this prospectively. We also demonstrated that anti-Smith antibody positivity was common among SLE patients who developed myositis, and this antibody was also present in all our incident myositis cases. A link between anti-Smith antibodies and myositis was suggested in one study of an adult SLE population, 3 and one study of a pediatric SLE population. 14 One other publication noted a high proportion of anti-SSB/La and anti-RNP positivity. 9 We found a trend towards higher prevalence of these antibodies in our incident myositis cases.
Histologic changes seen in muscle biopsies of patients with SLE and myositis from several studies show evidence of vasculitis, vacuolar myopathy, and necrosis.1,5,9,15,16 One study emphasized correlations between SLE disease activity and deposits of immune complexes in skeletal muscle biopsies. 17 Another recent study found that a high proportion of their SLE patients with myositis had pathological changes consistent with dermatomyositis and polymyositis. 9 Given the similarities in muscle histopathology between SLE myositis patients and the histopathology of idiopathic inflammatory myopathies, one might consider the SLE-myositis patients in our cohort to represent patients with an overlap syndrome. Although we did not assess for myositis-specific autoantibodies in our analyses, we do plan to undertake this in future studies.
The strengths of our study include the relatively large prospective SLE cohort from which our cases were drawn, and the fact that we studied incident myositis cases (not prevalent, as prior studies have done), confirmed by chart review. The main limitation was the relatively low number of incident SLE-myositis cases. Given our screening tools to identify myositis cases (SDI, SLEDAI-2K, and CK), were performed only annually, it is possible that myositis cases were missed. However, this limitation is consistent with all other published studies of myositis in SLE.1,3,12 Since we followed patients for many years, we think it is unlikely that many patients with significant muscle disease would remain undetected.
A priori, we studied clinical variables on the basis of past publications suggesting possible associations of these variables with myositis in SLE, mixed connective tissue disease, and/or overlap syndromes. Many of our serologic variables correlated with clinical features, and with each other. Though on one hand this precludes the ability to define a single most predictive factor, it does suggest that the SLE phenotype most likely to develop myositis is represented by a cluster of features including seropositivity for anti-Smith and potentially other autoantibodies.
In conclusion, over an average follow-up of 8.5 years, the incidence of myositis in SLE was 1.05 cases per 1000 person-years. Because myositis is not a well-known feature of SLE, this potentially disabling feature may be overlooked in a busy clinical practice. Our study suggests that a cluster of key factors, including non-Caucasian race, arthritis, Raynaud’s phenomenon, and anti-Smith antibodies, may help clinicians in identifying SLE patients most at risk for this important manifestation.
Supplemental Material
sj-pdf-1-lup-10.1177_0961203320988587 - Supplemental material for Myositis in systemic lupus erythematosus
Supplemental material, sj-pdf-1-lup-10.1177_0961203320988587 for Myositis in systemic lupus erythematosus by Thaisa Cotton, Omid Zahedi Niaki, Boyang Zheng, Christian A Pineau, Marvin Fritzler, Evelyne Vinet, Ann E Clarke and Sasha Bernatsky in Lupus
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The McGill University Health Center Lupus Clinic research activities are funded by the Singer Family Fund for Lupus Research.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
