Abstract
Background
Alternative therapeutic options are needed for patients with systemic lupus erythematosus (SLE) not adequately controlled with or intolerant to traditional treatments. This study evaluated the efficacy of Acthar® Gel (ACTH(1-39)) for reducing active SLE severity among patients receiving underlying conventional maintenance therapies.
Methods
Ten females (mean age = 49 yrs, disease duration = 7 yrs, Systemic Lupus Erythematosus Disease Activity Index-2000 [SLEDAI-2 K] = 10) currently on maintenance self-administered ACTH(1–39) gel 1 mL (80 U/mL) for 7–15 days and were assessed weekly for 28 days. Outcome measures included Physician and Patient Global Assessments, SLEDAI-2 K, Lupus Quality of Life scale, Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-Fatigue) scale, erythrocyte sedimentation rate, and C-reactive protein. Student’s t-test compared data obtained at days 7, 14, and 28 with those from baseline.
Results
The primary endpoint of SLEDAI-2 K improvement was reached at all observation times (p < 0.05) and statistically significant improvements were observed for most other parameters. No treatment-related serious or unexpected adverse events were observed.
Conclusions
The trial results reveal that among SLE patients in need of therapeutic alternatives, ACTH(1-39) gel may provide significant disease activity reduction.
Introduction
Systemic lupus erythematosus (SLE), a complex, chronic autoimmune disease with multi-organ involvement in vital organs and tissues such as kidneys, brain, heart, dermatologic manifestations, and blood, is characterized by heterogeneity of clinical manifestations, disease severity, and unpredictable disease flares. 1 During SLE flares, a clinically significant and measurable increase in disease activity in one or more organ systems that involves new or worse clinical signs and symptoms and/or laboratory measurements occurs, and a change in treatment is considered. 2 Effective management of chronically active disease and disease flare is critical to reduce the risk of accumulated organ and tissue damage over time, reduce the morbidity and mortality associated with end-organ damage, and reduce the economic burden of SLE on patients and improve patient quality of life.1,3–5
Organ and tissue injury and clinical manifestations in SLE derive from genetic, epigenetic, environmental, hormonal, and immune-regulatory factors. 6 The pathogenesis of SLE involves multiple abnormalities in the immune system, including hyper-activation and altered signaling among B and T cells, monocytes/macrophages, dendritic cells, and neutrophils.6,7 Auto-reactive B and T cells are activated by endogenous nuclear antigens released by apoptotic cells, resulting in secretion of pro-inflammatory cytokines and B cell production of autoantibodies through T cell-dependent and T cell-independent pathways. 8 Immune complex formation heightens and maintains the inflammatory response as immune complexes are deposited in tissues and complement and macrophages are activated, causing inflammation and leading to tissue injury.7,8 Additionally, immune complexes taken up by plasmacytoid dendritic cells interact with toll-like receptors resulting in production of the pro-inflammatory cytokine interferon alpha (IFNα). 7 Early diagnosis and effective treatment are essential in SLE as many patients experience chronically active disease or relapsing remitting disease with flares of inflammation that may lead to organ damage as a result of these processes.
Currently FDA approved medications for treatment of lupus are adrenocorticotropic hormone (ACTH(1-39) gel; H.P. Acthar® Gel, repository corticotropin injection, Questcor Pharmaceuticals, Inc., Hayward, CA, USA), prednisone, hydroxychloroquine, and belimumab. Multiple other immunosuppressants have been used also to reduce disease severity and for steroid sparing effects, but there remains a significant unmet need for flare management and active disease control among SLE patients.
Proopiomelanocortin (POMC) produces ACTH in extra-pituitary sites including the skin, which is the largest body organ. 9 Also, ACTH can stimulate local (extra-adrenal) glucocorticoids synthesis, including cortisol, which would contribute to the down-regulation of autoimmune responses.10,11 Corticotropin stimulating factor (CRF) is a stress-integrating peptide that acts as a stimulator of secretion of ACTH that can indirectly modulate immune system function through immunosuppression. 12 CRF plays a key role in tissue inflammation among patients with autoimmune and inflammatory diseases, including SLE.
ACTH(1-39) is a long-acting formulation of the full sequence ACTH(1–39) that may include other proopiomelanocortin peptides. H.P. Acthar® Gel has steroidogenic mechanisms of action through endogenous production of cortisol and is reported to have immunomodulatory properties facilitated through interactions with melanocortin receptors found in immune effector cells and other tissues. SLE is an approved use of ACTH(1–39) gel that was granted when the product was first approved in 1952. 13 Despite FDA approval, many physicians treating SLE are unaware of ACTH(1–39) gel as a treatment option approved by the FDA and clinical data are limited. Thus, ACTH(1–39) gel is a unique, approved therapeutic option for SLE.
ACTH(1–39) gel has been reported to be effective in the treatment of nephrotic syndrome, opsoclonus-myoclonus, and acute exacerbations in multiple sclerosis, dermatomyositis, and polymyositis, with suggested immune-modulating mechanisms of action.14–18 ACTH(1–39) is one of several melanocortin peptides and may have mechanisms of action beyond steroidogenesis, including anti-inflammatory and immunomodulatory effects, consistent with the broader melanocortin system. There are five melanocortin receptors (MCRs) belonging to the superfamily of G protein-coupled receptors and distributed throughout the body, including in the central nervous system and on immune cells.19,20 MCRs differ in affinity for melanocortin peptides, and ACTH(1–39) is reported to bind to all five receptors with high affinity.19,20 Inhibition of nuclear factor-κB (NF-κB) signaling is a key anti-inflammatory mechanism of melanocortin peptides.21,22 NF-κB controls the expression of many genes, including pro-inflammatory cytokines, cytokine receptors, chemokines, growth factors, and adhesion molecules, and modulation of NF-κB is a main mechanism behind the widespread and powerful effects of melanocortins on inflammatory pathways. 19
This current study examines the efficacy and safety of ACTH(1–39) gel for reducing the severity of active flares (defined as periods of increased disease activity as measured by changes in Systemic Lupus Erythematosus Disease Activity Index-2000 [SLEDAI-2 K] score) among ten patients with SLE who were receiving conventional SLE treatment regimens.
Methods
Study population
Men and women aged 18–75 who met American College of Rheumatology (ACR) criteria for SLE and who presented with chronic, moderately to severely active disease, and a disease flare while receiving standard treatments were eligible for participation. Patients were recruited from a single site in Lansing, MI, USA. All participants provided written informed consent and were free to discontinue treatment at any time. The study protocol was approved by an Investigational Research Board/Independent Human Research Ethics Committee and carried out in accordance with Good Clinical Practice guidelines. The study was conducted in accordance with the Declaration of Helsinki 1975, revised Hong Kong 1989.
Study design
In this single-site, open-label trial, which was conducted from November 2012 to May 2013, patients received 1 mL (80 U/mL) of ACTH(1–39) gel by subcutaneous injection. The first dose was given at the investigator site and subjects were educated on aseptic subcutaneous injection technique for daily home injections for all remaining doses. Patients received daily ACTH(1–39) gel over 10 days with an optional five-day rescue period for partial or non-responders (defined as having a SLEDAI-2 K score above 6) at the principal investigator’s discretion based on his clinical assessment following the day 10 dose. All patients were assessed weekly from baseline (day 0) through week 4 (day 28). Screening for eligibility was completed within one week of baseline (day 0). Completion of all study measures followed by the first dose of ACTH(1–39) gel occurred on day 0. Adverse events were assessed at each weekly patient contact, and patients could report adverse events at any time during the study.
Eligible patients met the ACR criteria for SLE and lupus flare. Patients had to fulfill at least four of the 11 ACR classification criteria for SLE, including a history of antinuclear antibody (ANA) positivity. Patients had a diagnosis of SLE with chronic disease activity requiring ongoing treatment or observation for ≥8 weeks prior to study screening. Patients had to have received either a stable dose of prednisone (or equivalent) ≤20 mg/day from at least four weeks prior to signing the informed consent or had received azathioprine, antimalarials, mycophenolate mofetil/mycophenolic acid, or weekly administration of oral or subcutaneous methotrexate at a stable dose for a minimum of eight weeks prior to signing informed consent. At screening and baseline (day 0) patients met SLE flare criteria, including a SLEDAI-2 K score ≥6 points, at least 1 on the British Isles Lupus Assessment Group Index (BILAG) A organ system score or 2 B organ system scores, and a Physician Global Assessment score ≥1 on a 0–3 visual analog scale. Female patients of childbearing age had to use an effective method of birth control and avoid pregnancy from screening through 90 days after the final dose of ACTH(1–39) gel.
Exclusion criteria included concurrent enrollment in any other clinical study with an investigational product within four weeks of baseline or within five half-lives of the investigational product used in that clinical study, whichever is longer. Patients receiving any new oral prednisone therapy (or equivalent) or any change in current oral prednisone dose (or equivalent) any time from four weeks prior to signing the consent form were excluded. Additional exclusion criteria included a known history of allergy or reaction to any component of Acthar® Gel, any live or attenuated vaccine within four weeks prior to signing the informed consent form, receipt of more than one prescribed non-steroidal anti-inflammatory drug (NSAID) at an anti-inflammatory dose within four weeks of study baseline, a diagnosis of scleroderma, osteoporosis, fungal infections, or ocular herpes simplex, or surgery within the past four weeks or planned surgery within the next four weeks. Patients with a history or presence of peptic ulcer, congestive heart failure, uncontrolled hypertension, primary adrenocortical insufficiency or adrenocortical hyperfunction, sensitivity to proteins of porcine origin, history of any type of malignancy, with the exception of basal cell carcinoma, ≤5 years before the study baseline, or known history of a primary immunodeficiency or an underlying condition, such as human immunodeficiency virus or splenectomy, that predisposes the patient to infection were excluded.
Outcome measures
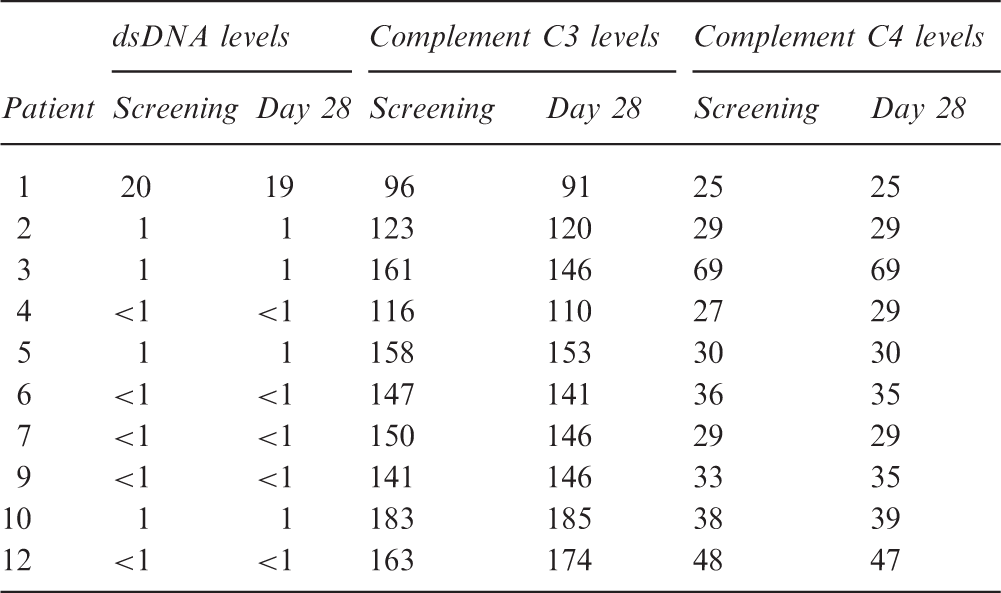
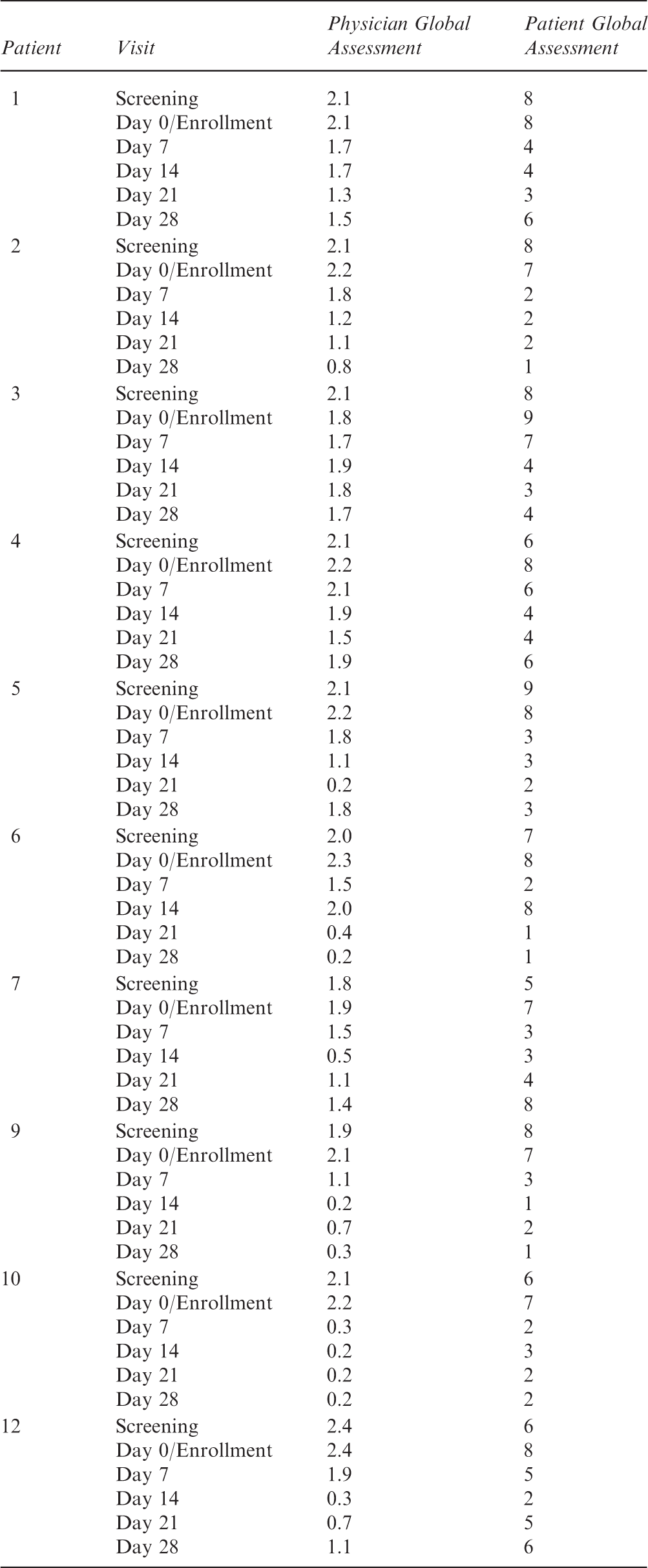
The primary study objective was to evaluate whether the addition of ACTH(1–39) gel to standard treatment of SLE reduced the intensity of flares as measured by changes from baseline in the SLEDAI-2 K score. Secondary outcome measures included Physician Global Assessment, Patient Global Assessment, Lupus Quality of Life (QoL) scale, Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-Fatigue) scale, BILAG scores, and markers of inflammation including erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). All of the measures were assessed at day 0, day 14, and day 28, and Physician Global Assessment and Patient Global Assessment were also measured at days 7 and 21. The anti-DNA and complement levels were assessed at screening and day 28, and the Lupus Flare Index was scored at screening, baseline, and each weekly visit.
Systemic Lupus Erythematosus Disease Activity Index-2000 (SLEDAI-2 K)
The SLEDAI-2 K index consists of a list of organ manifestations, each with a definition. 23 The physician completes the SLEDAI-2 K and determines whether each organ manifestation is ‘present’ or ‘absent’ in the preceding 10 days. The assessment consists of 24 lupus-related items and responses are weighted by multiplying by a particular organ’s ‘weight’. For example, renal descriptors are multiplied by four and central nervous system descriptors are multiplied by eight. The weighted organ manifestations are totaled into the final score. The SLEDAI-2 K score ranges from 0 (inactive disease) to 105. The SLEDAI-2 K is a sensitive, reliable, and valid clinical assessment of lupus disease activity.23,24
Physician Global Assessment
The Physician Global Assessment represents the overall assessment of average disease severity on a VAS scale ranging from 0 (none) to 3 (severe) disease activity over the last week. The Physician Global Assessment for a given subject was completed by the same physician whenever possible.
Patient Global Assessment
The Patient Global Assessment is a single-item question that accounts for ‘all the ways that your illness affects how you are doing’ over the previous week. Responses range from 0 (very well) to 100 (very poor) on a 100 mm VAS.
Lupus Quality of Life (QoL) scale
The Lupus QoL scale includes 16 items, each of which is scored on a seven-point scale. 25 When compared to individuals without the disease, patients with SLE have had significantly lower scores for items related to health, work, active recreation, and independence.
Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-Fatigue)
The 13-item FACIT-Fatigue scale is valid for measuring fatigue among patients included in this trial, as demonstrated by the results of a study published in 2013 that assessed the ability of that scale to measure physical or mental impairment among patients with mild-to-moderate SLE.26,27
British Isles Lupus Assessment Group Index (BILAG)
The BILAG Index is a translational index that assesses changing severity of clinical manifestations of SLE within nine systems, including general, mucocutaneous, neuropsychiatric, musculoskeletal, cardiorespiratory, gastrointestinal, ophthalmic, renal, and hematology.28,29 The BILAG Index uses ordinal scales and records disease activity across the different systems, allowing comparisons between the immediate past four weeks and the four preceding weeks. The timeframe used in the current study was two weeks (day 14) and four weeks (day 28). Disease activity is categorized into five levels from A to E. Grade A represents very active disease requiring immunosuppressive drugs and/or a prednisone dose >20 mg/day or equivalent. Grade B represents moderate disease activity requiring a lower dose of corticosteroids, topical steroids, topical immunosuppressants, antimalarials, or NSAIDs. Grade C indicates mild stable disease. Grade D implies no disease activity but the system has previously been affected. Grade E indicates no current or previous disease activity. The BILAG Index was developed based on the principle of intention-to-treat; however, the actual treatment of the patient has no bearing on the scoring index. Only the presence of active manifestations influences the scoring.
Data analysis
Paired t-tests were used to assess the change from day 0 to each of the follow-up visits for each of the dependent variables, with the exception of BILAG which is a qualitative measurement.
A response was defined as >4-point reduction from baseline in SLEDAI-2 K score along with no worsening from baseline in Physician Global Assessment and Patient Global Assessment scores AND no new BILAG A organ domain score or no more than one new BILAG B organ score compared with baseline.
Results
Diagnostic criteria, duration of disease, and previous and concomitant medications

Patients had to meet ≥4 of the 11 ACR criteria to meet the standards of diagnosis. The 11 criteria include: discoid rash, hematologic disorder, immunologic disorder, malar rash, neurologic disorder, non-erosive arthritis, oral ulcers, photosensitivity, pleuritis or pericarditis, positive antinuclear antibody, and renal disorder. 31
BID, twice daily; IM, intramuscular; IV, intravenous; PRN, as needed; QD, once daily; SQ, subcutaneous; TID, three times daily.
Efficacy of ACTH(1–39) gel for treatment of systemic lupus erythematosus
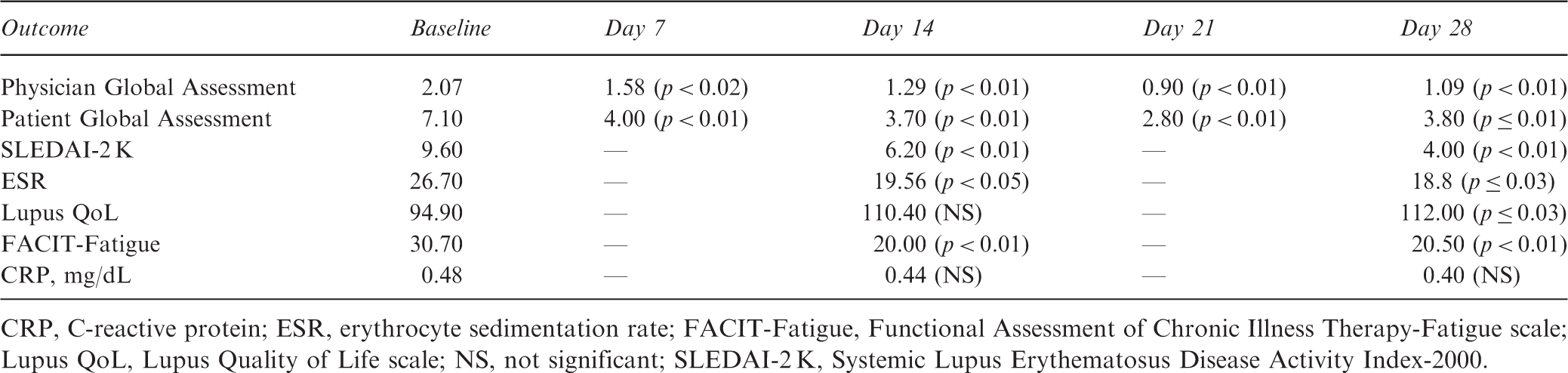
CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; FACIT-Fatigue, Functional Assessment of Chronic Illness Therapy-Fatigue scale; Lupus QoL, Lupus Quality of Life scale; NS, not significant; SLEDAI-2 K, Systemic Lupus Erythematosus Disease Activity Index-2000.
DNA and complement C3 and C4 levels
Lupus Flare Index outcomes
Among all of the patients, ACTH(1–39) gel was well-tolerated and no treatment-related serious or unexpected adverse events were observed. There were no changes in blood pressure, body temperature, or blood glucose levels in any of the patients during this 28-day study. Bilateral edema was present in the legs/ankles of one patient; however, it was no longer present two weeks after the end of treatment with ACTH(1-39). One patient reported a sinus infection during this trial that was resolved with one round of antibiotic treatment. No other adverse events were reported or observed.
Discussion
Although these patients had moderately to severely active SLE and were undergoing treatment with traditional therapeutic agents, following treatment with ACTH(1-39) gel they experienced significant improvements in all of the clinical outcome measures by the end of this study, including SLEDAI-2 K, which was the primary outcome measure. The results suggest that ACTH(1–39) gel may provide a novel anti-inflammatory and immunomodulatory treatment option with possible mechanisms of action beyond steroidogenesis.
Not only do the results obtained in this trial reveal that ACTH(1-39) gel was effective for treating patients with SLE, they also indicate that the drug was safe and well-tolerated. None of these 10 patients experienced changes in blood pressure, body temperature, or blood glucose levels, for the duration of the study.
Active lupus disease usually requires treatment with varying doses of corticosteroids, depending on severity. One study demonstrated that among women with SLE who were receiving chronic, alternate day glucocorticoid therapy with prednisone, cortisol responses to ACTH were normal. 30 Therefore, theoretically anyway, there could be comparable results among patients with SLE who are treated with corticosteroids and ACTH(1–39); however, there is no procedure available for calculating the equivalent doses. In addition, even if such a calculation was possible, the potential for variability among the relatively small number of patients who participated in this study indicates that such results would not be reliable.
The results of this study indicate that patients may also be able to benefit from treatment with ACTH(1–39). Six of these 10 patients have continued receiving ACTH(1–39) post-trial with no additional BILAG or SLEDAI-2 K measured flares. Those results will be presented once the patients are no longer receiving ACTH(1–39).
The results obtained during this trial reveal that, when treated with ACTH(1–39) gel, patients in need of therapeutic alternatives can have significant improvements in scores that indicate reduction of disease activity. Therefore, ACTH(1–39) gel could be considered as a therapeutic option for the treatment of lupus flare in patients who may be in need of treatment alternatives. A larger, long-term examination of patient responses and side effects to treatment with ACTH(1–39) gel is warranted.
Footnotes
Acknowledgments
Writing support was provided by Aric Fader, PhD, of MedVal Scientific Information Services, LLC, and funded by Questcor Pharmaceuticals, Inc. This manuscript was prepared according to the International Society for Medical Publication Professionals’ “Good Publication Practice for Communicating Company-Sponsored Medical Research: The GPP2 Guidelines.”
Conflict of interest statement
T. Montroy received a grant/research support from Questcor Pharmaceuticals, Inc.; J.J. Fiechtner also received a grant/research support from Questcor Pharmaceuticals, Inc.
Funding
Study support and writing support for this manuscript were funded by Questcor Pharmaceuticals, Inc.
