Abstract
An independent cross-sectional survey assessed systemic lupus erythematosus (SLE) disease and treatment burden. Variables included medication classes prescribed, disease activity, flare occurrences, treatment satisfaction, and validated measures of health-related quality of life (HRQoL), fatigue and work productivity. Of 886 eligible patients (mean age 41.3 years, 89% female), 515 completed the survey. One-third reported moderate-to-severe disease activity, and 31% had flared in the last 12 months. Higher severity of disease activity (moderate-to-severe) was associated with ≥2 medication classes prescribed and treatment regimens that included corticosteroids (CS) (both p < 0.0001). Patients receiving CS reported lower EQ-5D scores (p = 0.0019) and higher fatigue levels (p < 0.001), and both patients (p = 0.0019) and physicians (p = 0.0001) were less likely to report satisfaction with treatment regimens including CS. Among responders eligible for work (n = 456), severity of disease activity (moderate-to-severe vs. mild) was associated with unemployment (52.9% vs. 40.8%; p = 0.0189), greater impairment in work productivity (36% vs. 21%; p = 0.0003) and participation in daily activities (41% vs. 21%; p < 0.0001). This survey confirms that SLE and current treatment options substantially impair patients' health status and work productivity. Physician- and patient-reported satisfaction with current treatment regimens, despite poorly controlled disease activity, indicate they are resigned to the limitations of available SLE treatment regimens.
Keywords
Introduction
Health-related quality of life (HRQoL) of patients with systemic lupus erythematosus (SLE) has repeatedly been reported to be worse than that of the general population. 1 Clinical manifestations of SLE are multisystemic, variable in severity, 2 and include fatigue, 3 depression, 4 pain, 5 sleep disturbances and cognitive dysfunction. 6,7 These affect patients’ HRQoL comparably to or even more severely than other chronic conditions such as rheumatoid arthritis, heart failure, diabetes and Sjögren’s syndrome. 1,8,9 Compared with the general population, patients with SLE report impairment in not only physical function, but across all domains of the Medical Outcomes Survey Short Form 36 (SF-36), including social and emotional functioning. 1,10
Fatigue is one of the most prevalent symptoms of SLE, 11 and is reported even in patients with quiescent disease. 12 In an online survey of 223 patients with SLE, fatigue was reported by as many as 86%, and approximately 50% considered it to be their most disabling disease symptom. 11,13 Fatigue is multifactorial in origin and ascribed to disease activity, comorbid conditions, as well as behavioral factors. 2 Consequently, SLE patients have high rates of work disability, frequently with reduced working hours and/or changes in the nature of their work. 14,15 In the 2010 Lupus European Online (LEO) survey, 69.5% of 2188 patients with SLE reported that SLE affected their careers, with fatigue, inability to plan ahead and reduced physical health significantly associated with impaired productivity. 16
Current SLE treatment regimens include three major medication classes: corticosteroids (CS), immunosuppressants (IM), and antimalarials (AM). These therapies are frequently associated with adverse effects 17 that affect patients’ physical well-being and everyday activities, 18 and tolerability issues associated with these treatments often may be worse than the disease itself. 9 High-dose CS in particular result in morbidities such as changes in appearance, obesity, osteoporosis, hypertension, diabetes, cataracts and infections. It has been challenging to develop and confirm the efficacy of treatments for SLE. 19 Thus disease burden remains high, further aggravated by poor tolerability to current treatment regimens.
The Adelphi Disease-Specific Programmes (DSPs) are large multinational surveys of clinical practice. They are designed to capture a cross-section of ‘real-world’ data that reflects current symptom prevalence and severity, and associated treatment practices in a range of common chronic diseases, including SLE. 20 This analysis of data from the Adelphi Lupus DSP examines the treatment burden reported by SLE patients, patient and physician satisfaction with their treatment, and the impact of the disease on health status and productivity.
Methods
Disease-specific programme
Adelphi is a research-based organization that collects both quantitative and qualitative real-world data through a wide range of DSPs covering distinct disease areas; results are analyzed at national and international levels. 20
Physician-reported data
Candidate-respondent physicians are identified by local DSP fieldwork teams, who use public lists of healthcare professionals, according to predefined eligibility criteria that differ for each DSP. Identified physicians are invited to participate in the full program. Data collection from physicians includes face-to-face interviews, completion of a physician workload form documenting the total number of specific patients examined over the next 5 days (including the diagnosis in question as well as receipt of relevant medication classes). In addition patient record forms (PRFs) are completed by physicians for a number of consecutive patients who consult them with the relevant diagnosis, providing a representative ‘point-in-time’ sample.
Participating physicians and their patients are assigned anonymized study numbers to ensure confidentiality and allow appropriate linkage between data collection and analysis. Determination of patient eligibility is made solely by the physicians. As physician participation is voluntary, criteria do not require that patient samples be representative of the practice population in terms of ethnicity, socioeconomic background or age; samples are not weighted a priori to reflect underlying country populations.
Rating scales were the same for both physician- and patient-reported questionnaires for treatment satisfaction, defined as: 1. ‘satisfied’; 2. ‘not satisfied and I believe better control can be achieved’; 3. ‘not satisfied but I believe this is the best control that can be achieved’.
Patient-completed records
Patients voluntarily fill out self-completion records (PSCs) at data collection that include their assessment of disease activity as well as validated self-report instruments, including HRQoL (EQ-5D), 21 Work Productivity, Activity Impairment Index for SLE (WPAI-Lupus) 22 and Functional Assessment of Chronic Illness Therapy-Fatigue scale (FACIT-F scale). 23 Questions posed to patients focus on symptoms and severity of disease activity, impact of both disease and its treatment, compliance with therapy, reasons for treatment discontinuation and expectations of therapy. PSCs also include one or more validated HRQoL instruments, where appropriate. At no stage are responses seen or influenced by the treating physicians. Should patients choose not to complete PSCs, this does not disqualify inclusion of data recorded by the physician in the analysis.
The FACIT-F scale is a 13-item questionnaire that assesses self-reported fatigue and its impact on daily activities and function, using a 5-point Likert-type scale; 23 the total score ranges from 0–52, where greater scores represent less fatigue. Originally developed to measure fatigue associated with anemia in cancer patients, it has been validated in SLE. 24
EQ-5D (1990 version) 21 is a generic measure evaluating health status and includes five-item health status domains with a visual analogue scale (VAS). 25 Domains include: mobility, self-care, usual activities, pain/discomfort and anxiety/depression, measured on a three-level scale. The EQ-5D VAS records the respondent’s self-rated health status on a vertical 20 mm scale, graduated from 0–100 (0 = worst imaginable health status; 100 = best imaginable health status).
The WPAI questionnaire queries impairment due to SLE during the prior 7 days, and includes six items grouped into four domains. 22 Results for each domain are expressed as percentages of impairment; higher scores indicate greater impairment. The four WPAI domains include: absenteeism (percentage time missed from work); presenteeism (percentage impairment of productivity while at work); overall work impairment (combination of absenteeism and presenteeism domains); and percentage of impairment of regular daily activities such as housework, shopping, childcare, exercising, studying, etc., outside of work.
Based on responses to this self-assessment questionnaire, a subset of the patient population was categorized as ‘eligible for work’.
Survey design
This analysis is based on data extracted from the Adelphi 2010 Lupus DSP, 20 a cross-sectional survey of physicians and their SLE patients conducted between December 2009 and May 2010 in US, France, Germany, Italy, Spain and UK. Each physician completed a comprehensive PRF for five SLE patients, which together comprise the ‘overall patient population’. Physician-reported data included assessment of disease activity (by professional judgment), occurrence of flares, treatment regimens/medication classes prescribed and satisfaction with treatment.
Prescribed CS, IM and AM were recorded. Associations between use of these medication classes with disease activity, flare occurrence, treatment satisfaction, health status, productivity and employment were examined.
Statistical analyses
Statistical analyses were performed using t-test, analysis of variance (ANOVA), Mann–Whitney, Kruskal–Wallis, chi-squared and Fisher’s exact tests (dependent upon variable). ANOVA and chi-squared tests were used to determine the effects of SLE on WPAI-Lupus domains and employment status by disease activity, organ systems affected by SLE, age and flare status. Kappa analyses estimated agreement between patient and physician assessments of disease activity. Regression analyses determined variables associated with employment status, impairment of productivity and severity of disease activity. Independent variables included: patient demographics, such as age and ethnicity; validated patient-reported outcomes (PROs), including FACIT-F and EQ-5D scores as well as time since diagnosis, hospitalizations, flare status, pain, adherence and disease progression.
Results
Demographics
Demographics of patients included in the Adelphi Lupus Disease-Specific Programme
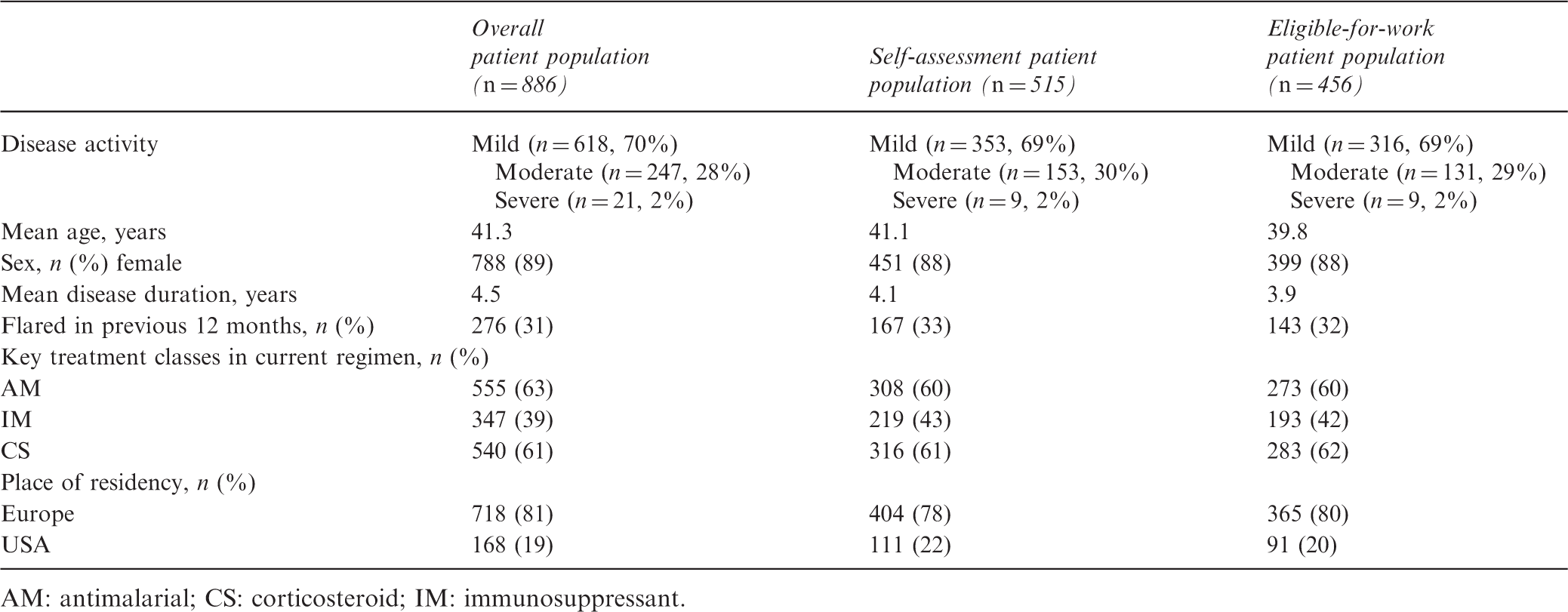
AM: antimalarial; CS: corticosteroid; IM: immunosuppressant.
Disease activity and treatment regimen (overall patient population)
Some 36% of surveyed patients were currently receiving one medication class; the majority (56%) two or more of CS, IM or AM. In the overall patient population, AM were most commonly utilized (63%); in the self-assessment population and eligible-for-work populations CS were most common (61% and 62%; Table 1). Distribution of patients with ‘mild’, ‘moderate’ or ‘severe’ disease activity was similar within each of the three different patient populations. Moderate agreement between patient and physician assessments of disease activity was evident, reflected by kappa analysis (κ = 0.5643).
Number of key treatment classes received by patients, according to severity of disease activity (overall patient population). Disease activity was physician-defined. aNo treatment data for one patient
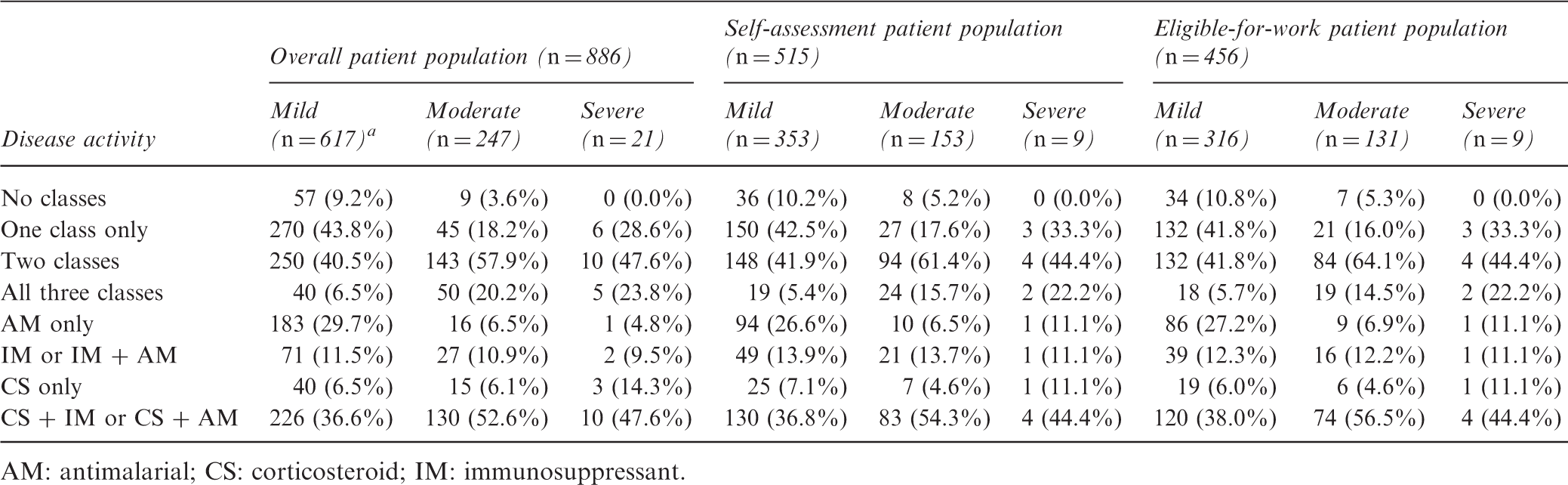
AM: antimalarial; CS: corticosteroid; IM: immunosuppressant.
Patient- and physician-reported satisfaction with current SLE treatments (overall patient population)
Most physicians reported they were satisfied with patients’ current treatment regimens (79.0%; n = 700). However, physicians were less likely to report satisfaction if treatment regimens included CS (70.7% vs. 90.7% if not; p < 0.0001; Figure 1). Although most patients were satisfied with their current treatment regimens (73.3%; n = 337), those receiving CS were less likely to report satisfaction (67.6% vs. 80.8% not receiving CS; p = 0.0016).
Physician satisfaction with patients’ treatment regimens, according to whether or not the patient was prescribed corticosteroids (CS) (overall patient population, n = 885).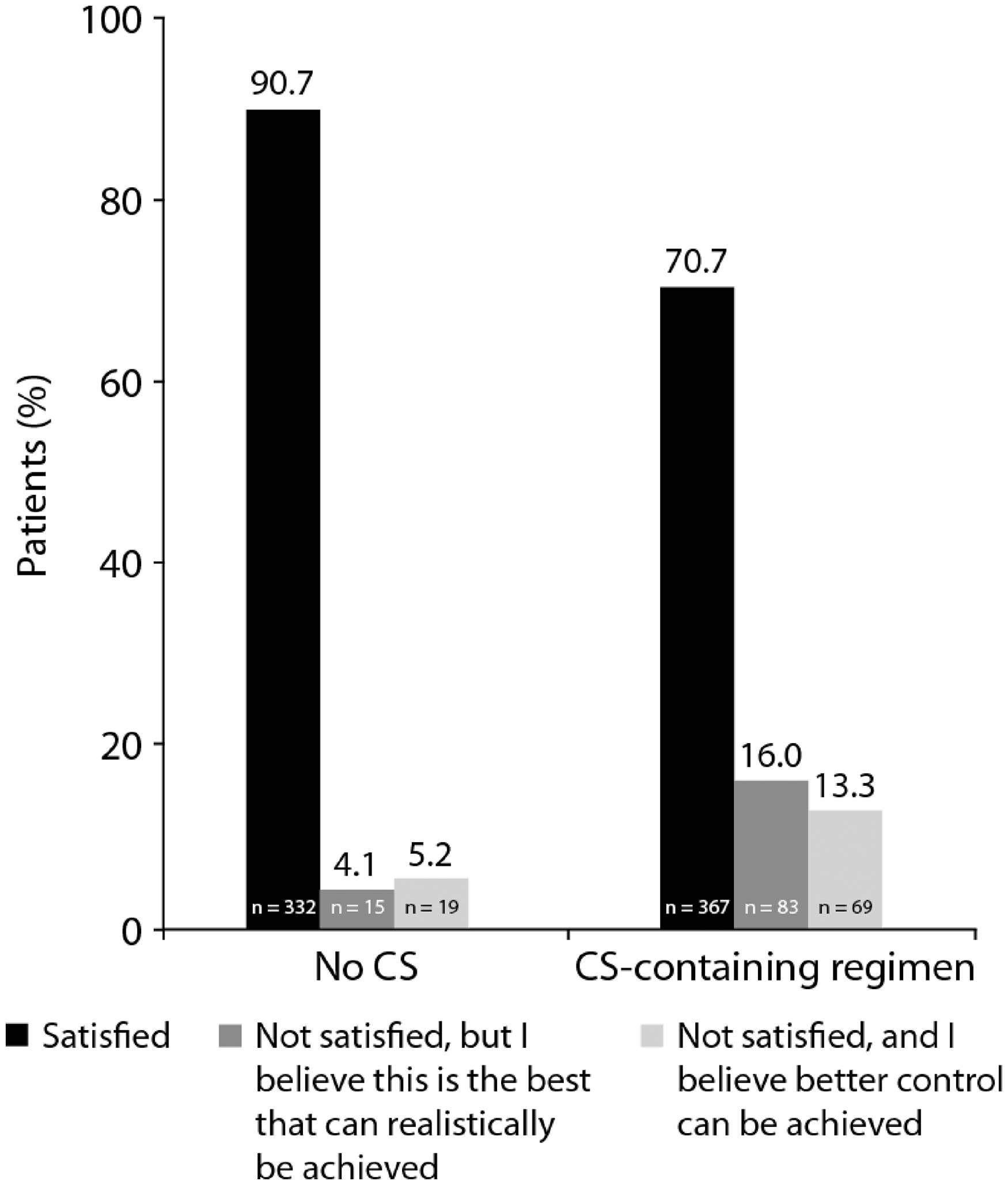
Health status and fatigue (self-assessment population)
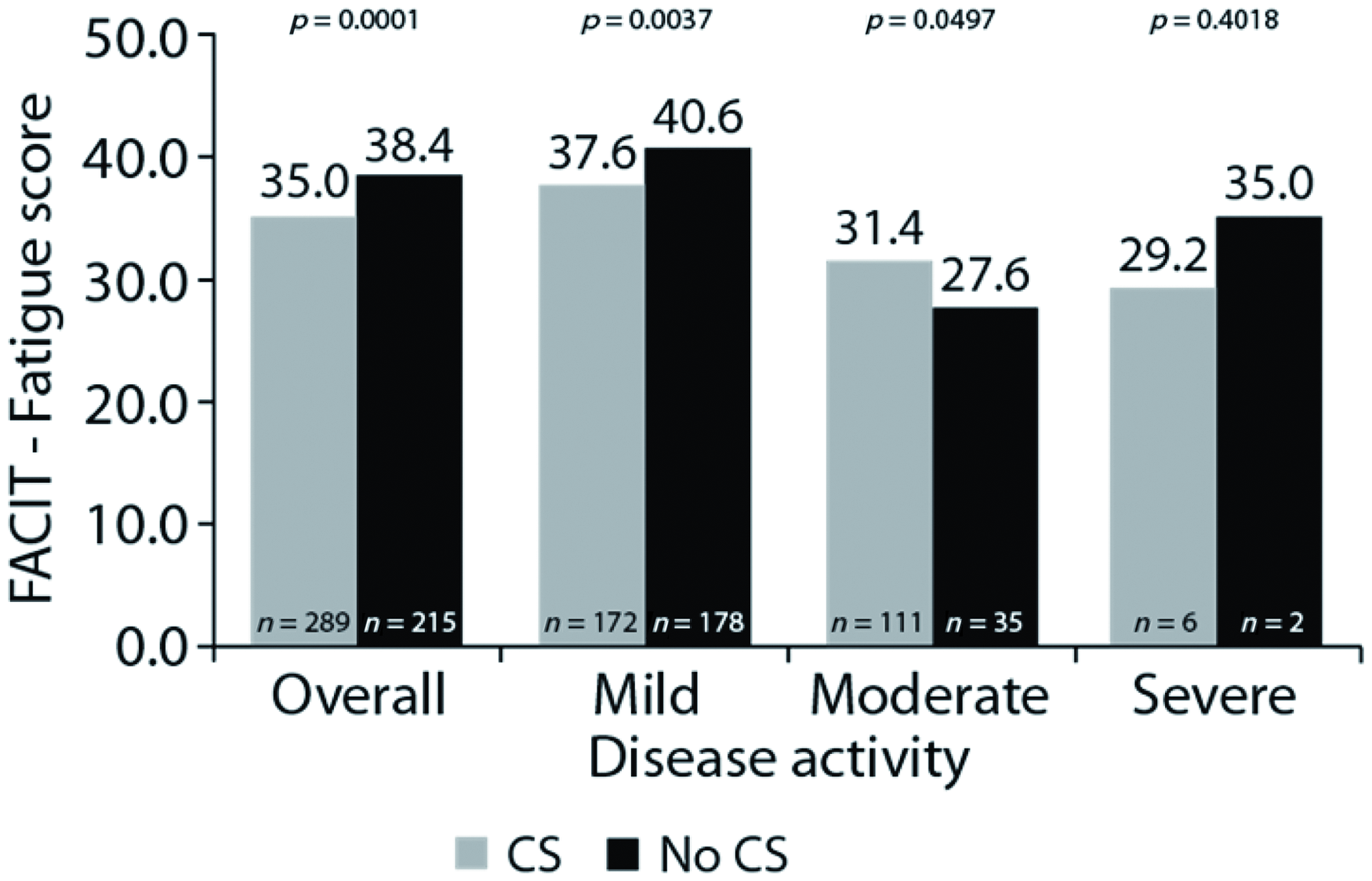
Among 515 patients who completed EQ-5D, a substantial burden of SLE (and its associated medications) on health status was evident. Patients receiving CS reported lower EQ-5D scores (0.786 (n = 292) vs. 0.819 (n = 215); p = 0.0019) and greater fatigue levels (FACIT-F score 35.0 (n = 289) vs. 38.4 (n = 215); p = 0.0001). Effects of CS treatment on EQ-5D and FACIT-F score were significant in patients with mild-to-moderate (all p < 0.05), but not severe disease activity (Figures 2 and 3).
EQ-5D scores for patients receiving corticosteroids vs. not receiving corticosteroids, stratified by disease activity. FACIT-Fatigue scores for patients receiving corticosteroids vs. not receiving corticosteroids, stratified by disease activity.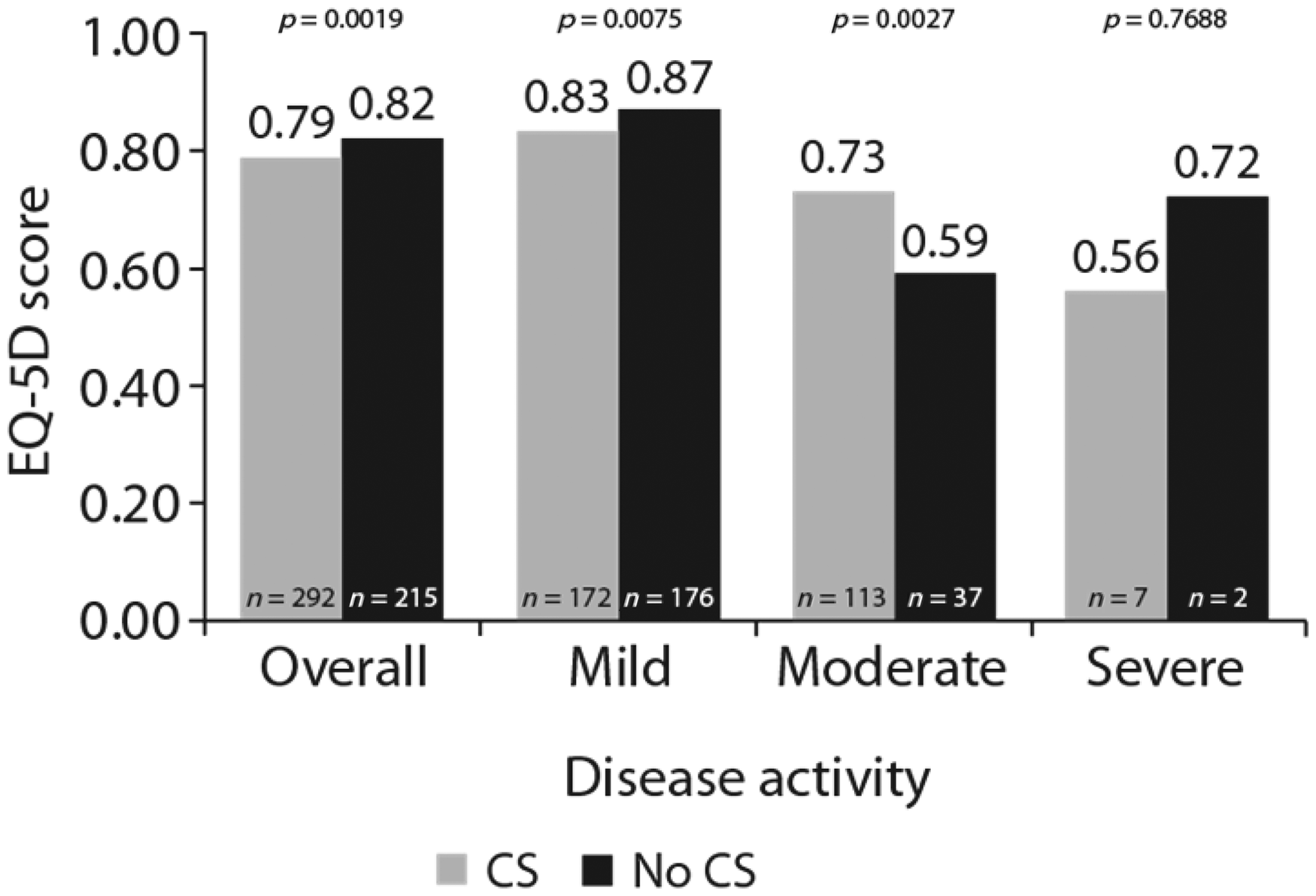
Productivity (eligible-for-work population)
Patient characteristics associated with unemployment included older age and higher levels of fatigue (odds ratios and 95% CIs for variables associated with employment; n = 450). Of the eligible-for-work population (n = 456; Table 1), 56.4% were employed or self-employed. Patients with moderate or severe disease activity were less likely to be employed than those with mild activity (47% and 44%, respectively, vs. 59% ‘mild’ (n = 456); p = 0.0574).
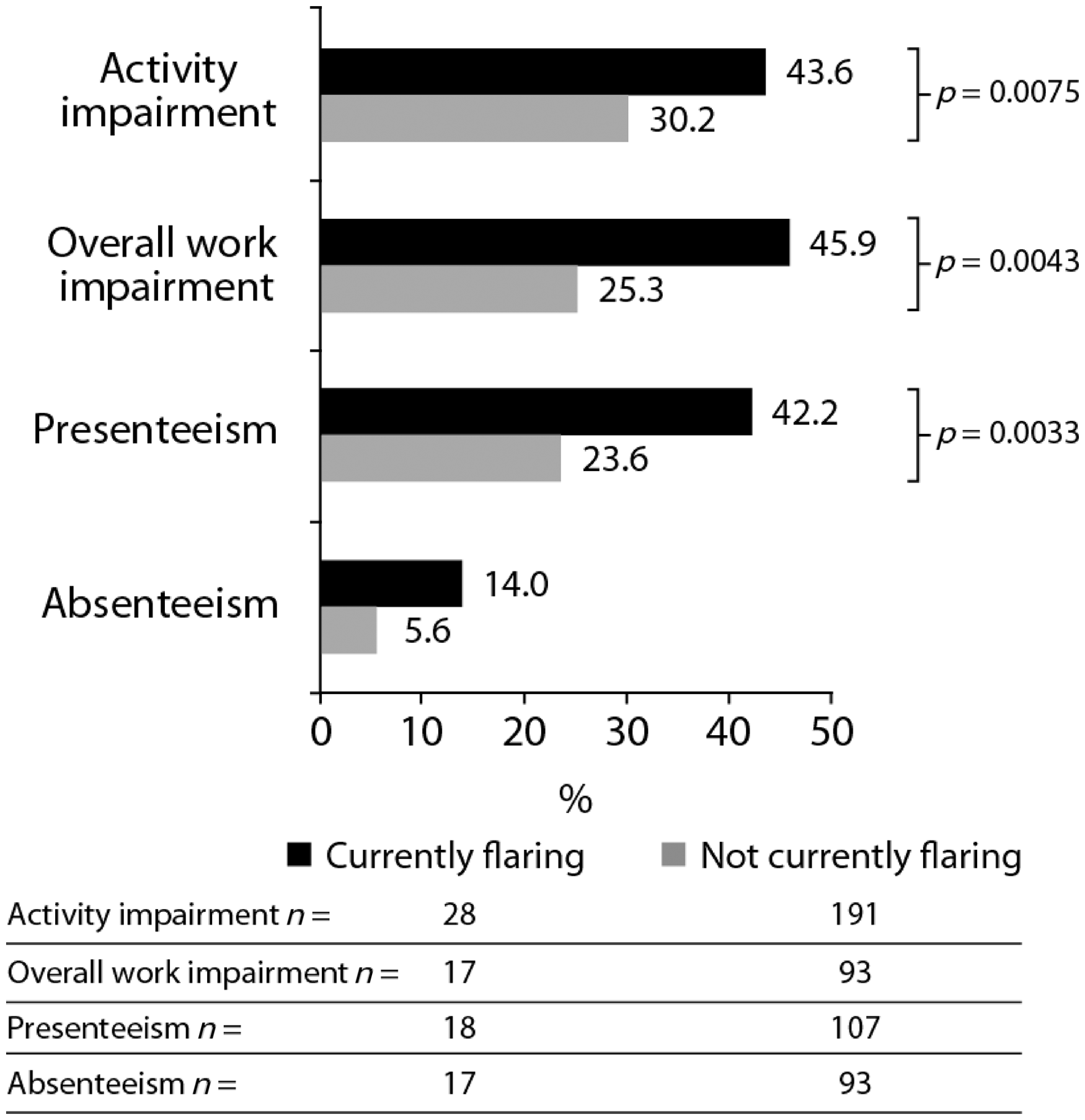
Moderate-to-severe disease activity was also associated with greater impairment in productivity, assessed by the WPAI domains of ‘activity impairment’ (impairment while carrying out regular everyday activities outside work, such as household chores, shopping, child care, and exercise; 40.7% vs. 21.4%; p < 0.0001), ‘presenteeism’ (impairment while at work; 32.5% vs. 18.8%; p = 0.0001; Figure 4), and ‘overall work impairment’ (combination of presenteeism score and time spent away from work due to disease; 36.4% vs. 21.2%; p = 0.0003). Coefficient estimates for variables associated with WPAI-Lupus overall percentage of work impairment revealed that associated characteristics included high levels of fatigue and current experience of a disease flare (n = 206). Furthermore, current flares were also associated with greater impairments in productivity (p < 0.01; Figure 5), by WPAI domains of ‘activity impairment’ (43.6% vs. 30.2%), ‘overall work impairment’ (45.9% vs. 25.3%) and ‘presenteeism’ (42.2% vs. 23.6%).
Percentage of impairment in productivity in eligible-for-work patients, measured by the four WPAI-Lupus domains, by severity of disease activity. Percentage of productivity impairment, as measured by the four WPAI-Lupus domains, for flaring patients (currently flaring vs. not currently flaring).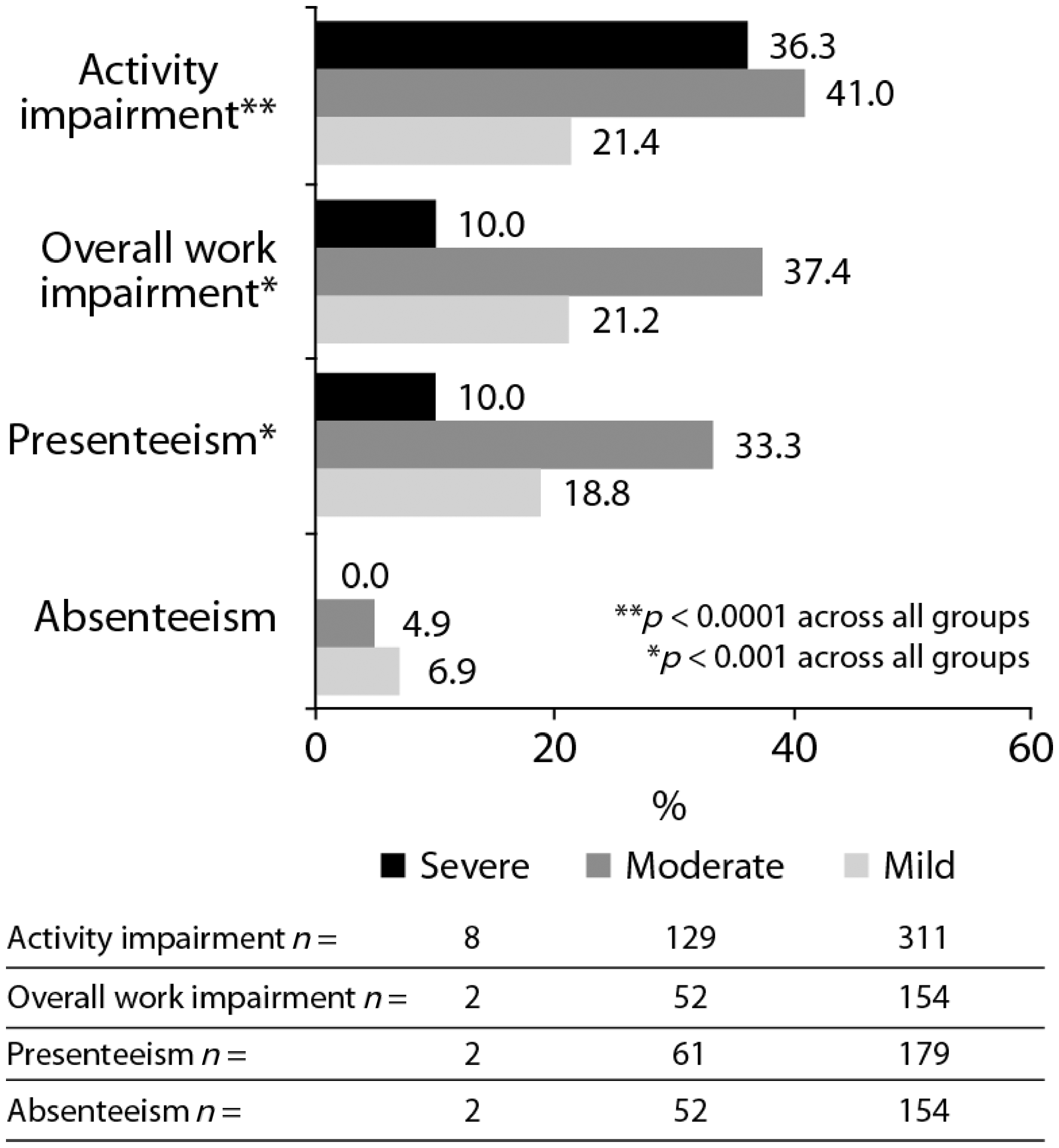
Discussion
This analysis of the Adelphi Lupus DSP confirms that disease activity and treatment significantly reduce patients’ health status. More severe activity and current disease flares were associated with prescription of two or more medication classes, and specifically CS use. Treatment regimens including CS were associated with less patient and physician satisfaction, lower health status (EQ-5D) and more fatigue (FACIT-F). Impairment in work/productivity was associated with higher disease activity, current flares and more fatigue.
This survey also demonstrated that approximately one-third of patients receiving treatment had moderate-to-severe disease activity, yet most patients and physicians reported satisfaction with treatment. Since current therapy for SLE follows an ‘add-on’ paradigm, where more therapies are added as disease activity increases, this indicates a surprising finding: substantial number of physicians as well as patients appear to accept moderate-to-severe disease activity despite use of multiple medication classes. Further, it suggests that patients and physicians have become resigned to poorly controlled disease activity. Indeed, most physicians reporting dissatisfaction with their patients’ treatment regimens indicated they believed this was the best that could be realistically achieved with currently available therapies – highlighting the apparent inadequacies of current treatment regimens.
The reported effects of CS treatment on patients’ health status and fatigue were also surprising. In patients with mild disease activity significant worsening was reported, compared with significant benefit in patients with moderate disease activity, yet no effect in patients with severe disease activity. This latter result may reflect the fact that a low number of patients with severe disease activity were included in the survey. Alternatively, adverse effects of CS treatment may be more apparent in patients with less severe disease activity, and/or may outweigh perceived benefits of treatment. In contrast, adverse effects of CS treatment may appear relatively less burdensome in patients with severe disease activity and already impaired HRQoL.
This analysis of the Adelphi Lupus DSP data confirms that SLE disease activity has a negative effect on patient-reported fatigue, health status, productivity (both at work and in daily activities outside of work) and employment status, particularly in those with moderate-to-severe disease activity. In the eligible-for-work population, 45% of patients surveyed were unemployed, which is consistent with the 46% unemployment found in a structured telephone cohort survey in the US, 15 although higher than the 33% reported in a more recent general literature review of 26 publications on SLE and employment. 14 Fatigue is an aspect of HRQoL that is particularly pronounced in SLE, 11 and high levels of fatigue in this survey were associated with work impairment as well as unemployment. In addition to employment status, consideration should be given to the effect of SLE on presenteeism and productivity outside work. Although many European studies have shown SLE may result in loss of employment, 26 –30 only a few have shown that SLE reduces overall productivity outside of paid employment. 31,32 As SLE occurs predominantly in women of childbearing age, frequently responsible for childcare and housework, productivity losses outside of paid employment are particularly important to understand and quantify. 33 –35
This analysis has limitations commonly associated with survey-based/observational studies. As the DSP survey was distributed via physicians, it did not include patients who had not recently consulted a physician, which may have skewed the population. Physician assessment of disease activity was by their professional judgment; use of a validated disease activity instrument may have been more accurate, although would also have been more time-consuming. Not unexpectedly, few patients with severe disease activity were included. Overall, more data were collected from treating physicians than from direct patient reports, and severity of disease activity and presence/absence of flares were physician not patient-reported. In further studies, it would also be valuable to analyze patient characteristics and HRQoL stratified by CS dose.
The Adelphi DSP study confirms the substantial burden of active SLE on patients. Moderate and severe disease activity, flares, lower health status and more fatigue are ubiquitous among patients with SLE, and currently available therapies frequently add to this burden. New therapies, and new treatment regimens, are needed to reduce the negative effect of this disease on patients’ lives.
Footnotes
Funding
This work was supported by UCB Pharma, who provided financial support for the data analysis of the survey.
Conflict of interest
Vibeke Strand has received consulting fees from UCB Pharma, Abbott Immunology, Amgen, Anthera, AstraZeneca, Biogen Idec, Biotest, Bristol-Myers Squibb, Genentech/Roche, GlaxoSmithKline, Incyte, Idera, Janssen, Lilly, Medimmune, Novo Nordisk, Novartis Pharmaceuticals Corporation, Pfizer Inc., Rigel Pharma, Sanofi and Takeda.
Catrinel Galateanu, Daphnee S Pushparajah and Enkeleida Nikaï are employees of UCB Pharma.
Jennifer Sayers is a consultant for UCB Pharma and employee of Adelphi Real World Ltd.
Robert Wood is a consultant for UCB Pharma and employee of Adelphi Real World Ltd.
Ronald van Vollenhoven has received consulting fees or honoraria from Abbott, Bristol-Myers Squibb, GlaxoSmithKline, Human Genome Sciences, MSD, Pfizer, Roche and UCB Pharma; research support or grants from Abbott, Bristol-Myers Squibb, GlaxoSmithKline, Human Genome Sciences, MSD, Pfizer, Roche, and UCB Pharma.
Acknowledgments
The authors would like to thank the network of physicians and patients who participated in the DSP survey. The authors would also like to thank Tania Kapoor and Niall Harrison of Darwin Healthcare Communications for provision of medical writing support, funded by UCB Pharma.
