Abstract
Introduction
Podocyte loss or functional impairment is a key event in the pathophysiology of nephrotic syndrome (NS). NS can further progress to chronic kidney disease (CKD) and/or end-stage renal disease (ESRD). This study aimed to explore the potential role and underlying mechanism of TRIM37 in NS.
Methods
Bioinformatics analysis was performed on the GEO dataset GSE97709 to analyze the association between TRIM37 expression and CKD. Adriamycin (ADR)-induced nephropathy models in mice and ADR-exposed human podocytes were used to investigate the function of TRIM37. TRIM37 overexpression experiments were conducted
Results
Both ADR-treated mice and ADR-exposed podocytes showed decreased TRIM37 expression.
Discussion
The findings demonstrate that TRIM37 attenuates ADR-induced nephropathy by inhibiting mitochondrial apoptosis and podocyte injury through the PTEN/Akt signaling pathway. Collectively, these results suggest that TRIM37 may serve as a potential therapeutic target for NS.
Introduction
Nephrotic syndrome (NS) represents a significant clinical challenge in nephrology, characterized by the hallmark features of massive proteinuria, hypoalbuminemia, edema, and hyperlipidemia. 1 The development of NS is primarily attributable to a pathological increase in the permeability of the glomerular filtration barrier (GFB) to plasma proteins, clinically manifested as proteinuria. 2 Podocytes, highly specialized and terminally differentiated epithelial cells lining the outer aspect of the glomerular capillary loop, are integral components of the GFB. Their intricate structure, including foot processes interconnected by the slit diaphragm, is crucial for maintaining size and charge selectivity. 3 Critically, the limited regenerative capacity of terminally differentiated podocytes means that their loss or dysfunction constitutes a major barrier to recovery from proteinuric injury and represents a critical event driving the progression towards glomerulosclerosis in NS.4,5 Podocyte injury, manifesting as foot process effacement, detachment, or apoptosis, is now recognized as a central pathophysiological mechanism underlying many forms of NS, including those initiated by toxic insults. 6 Mitochondrial dysfunction and the activation of intrinsic apoptotic pathways within podocytes have emerged as key molecular drivers of this injury and subsequent proteinuria. 7
The Tripartite motif (TRIM) family of proteins is characterized by the presence of RING, B-box, and coiled-coil domains, and many function as E3 ubiquitin ligases regulating diverse cellular processes via ubiquitin-dependent signaling. 8 TRIM37, a member of this family, exhibits canonical E3 ligase activity and plays multifaceted roles in various pathological contexts. It has been implicated in oncogenesis, where it acts as an oncogenic coactivator for AP-2γ, promoting the ubiquitination and degradation of tumor suppressors like p14ARF and thereby driving breast cancer progression.9,10 Beyond oncology, TRIM37 contributes significantly to injury responses in multiple organs, with emerging parallels to pathways relevant in renal pathophysiology. It exacerbates liver ischemia/reperfusion injury by ubiquitinating IκB kinase γ (IKKγ) and facilitating its nuclear translocation, leading to dysregulated NF-κB signaling, a pathway also central to renal inflammation and podocyte damage in NS.11,12 TRIM37 also plays a detrimental role in the central nervous system, where it is a critical mediator of striatal degeneration in Huntington’s disease by interfering with autophagy regulation, 13 and promotes intracerebral hemorrhage (ICH)-induced brain injury through ubiquitination and regulation of PPARγ stability. 14 Additionally, TRIM37 has been linked to ischemic injury responses in the heart. 15 TRIM37 has been well documented to participate in apoptosis, autophagy, and stress responses across various disease models involving epithelial and endothelial cells. However, its specific role and functional importance in the kidney, especially in podocytes or NS, remain entirely unexplored. This represents a significant gap in understanding the ubiquitin-mediated regulation of podocyte health and disease. Given the critical role of podocyte injury, particularly apoptosis driven by mitochondrial dysfunction, in the pathogenesis of NS, and considering TRIM37’s established function as an E3 ubiquitin ligase regulating stress responses, apoptosis, and cell survival pathways in other disease contexts, we hypothesize that TRIM37 may play a pivotal regulatory role in protecting podocytes against nephrotoxic injury. Adriamycin (ADR; doxorubicin) is a widely used chemotherapeutic agent whose dose-limiting nephrotoxicity reliably induces a well-characterized experimental model of NS, primarily driven by podocyte apoptosis and mitochondrial damage. 16
Therefore, we employed both in vivo ADR-induced nephropathy models and in vitro ADR-treated podocytes to systematically investigate the functional significance of TRIM37 in NS pathogenesis. The preliminary data indicated interactions between TRIM37 and key apoptosis regulators. We hypothesized that TRIM37 could alleviate ADR-induced podocyte injury and mitochondrial apoptosis by regulating the tumor suppressor phosphatase and tensin homolog (PTEN). Specifically, TRIM37 might promote PTEN ubiquitination and subsequent degradation, which in turn modulated downstream pro-apoptotic signaling pathways essential for podocyte survival.
Materials and methods
Animal model of ADR-induced nephropathy
Male C57BL/6 mice aged between six and 8 weeks old (20 ± 2 g,
Real-time PCR
Real-time PCR primers.

Biochemical analysis
The levels of creatinine (Cr), blood urea nitrogen (BUN), total cholesterol (TC) and triglyceride (TG) in serum were analyzed using commercial kits from the Nanjing Jiancheng Bioengineering Institute (China) in accordance with the manufacturer’s instructions.
Histological analysis
Renal tissues were subjected to fixation in 4% paraformaldehyde for 24 h. Thereafter, the tissues were dehydrated in a series of graded ethanol solutions, cleared in xylene, and embedded in paraffin. The embedded tissues were sectioned at 5 µm using a rotary microtome (Leica, Germany). The sections were then stained with hematoxylin and eosin (HE), Periodic acid-Schiff (PAS), and Masson’s trichrome staining. HE staining (Solarbio) was utilized to evaluate the pathological changes in renal tissues. PAS staining (Servicebio, China) was utilized to assess glycogen accumulation in renal tissues. Furthermore, Masson’s trichrome staining (Solarbio) was utilized to evaluate fibrosis within the renal tissues. Photographs of the stained histological sections were captured using a microscope (Olympus).
Immunofluorescence
The paraffin-embedded tissue sections were dewaxed using xylene and then rehydrated using a series of graded ethanol solutions. The sections were immersed in a citrate-EDTA solution (Beyotime) to retrieve the antigens. The sections were incubated with goat serum to block non-specific binding sites. Thereafter, the samples were subjected to an overnight incubation with primary antibodies against podocin (Proteintech, 20384-1-AP), nephrin (Proteintech, 22912-1-AP), desmin (Proteintech, 16520-1-AP), synaptopodin (Proteintech, 21064-1-AP), TRIM37 (Proteintech, 13037-1-AP), and PTEN (Proteintech, 60300-1-Ig) at 4°C. This was followed by an incubation with secondary antibodies (Invitrogen, USA, A27039; Abcam, USA, ab6785) for 1 h at room temperature. Photographs were captured under an Olympus microscope (Japan).
TUNEL
The sections were dewaxed twice with a duration of 15 min for each cycle, utilizing xylene. Thereafter, a process of rehydration was conducted by employing a sequence of graded ethanol solutions, ensuring a gradual dilution of the samples. Thereafter, the sections were incubated with 0.1% Triton X-100. The sections were then washed three times with PBS, after which they were incubated with TUNEL staining solution (50 µL; KeyGEN BioTECH, China) for 1 h at 37°C. The sections were then stained with DAPI (Beyotime), after which they were mounted using anti-fluorescence quenching solution (Solarbio), and photographs were captured under an Olympus microscope (Japan).
Cell culture and treatment
Human renal podocytes were purchased from Wuhan Procell Biotechnology Co., Ltd. (CP-H075). The cells were identified by PCK immunofluorescence (>90%) by the Laboratory of Wuhan Procell Biotechnology Co., Ltd. in February 2025 and STR profiling also performed by Laboratory of Wuhan Procell Biotechnology Co., Ltd. in February 2025. These cells possessed three key characteristics: adherent growth pattern, epithelial-like morphology and a restricted passage capacity of 1 to 2 generations. The cells utilized in this study were verified to be mycoplasma-free through PCR detection by our laboratory independently in February 2025. The cells were maintained in complete culture medium for human renal podocytes at 37°C in a 5% CO2 incubator. The cells were infected with TRIM37-overexpressing lentivirus (lenti-TRIM37-OE), PTEN-overexpressing lentivirus (lenti-PTEN-OE), or lentiviral vector (lenti-vector) (GenePharma) at a multiplicity of infection of 30. Following a 48-h infection period, renal podocytes were treated with 0.4 μg/mL ADR for a 24-h period prior to further analysis.
Flow cytometry
The cell suspension containing 2 × 105 cells was centrifugation at 300× g and then resuspended in 500 μL of binding buffer. Following this process, 5 µL of Annexin V-FITC and 5 µL of PI (KeyGEN BioTECH). The mixture was blended and then incubated at 25°C for 15 min. Cell apoptosis rate was analyzed by flow cytometry (BD Biosciences, USA).
CCK-8 assay
The cells were seeded in a 96-well culture plate, with an approximate density of 2 × 103 cells per well, and then cultivated overnight in an incubator containing 5% CO2 at 37°C. Subsequently, 10 µL of CCK-8 solution (Solarbio) was administered to each well, and the plate was then incubated for a further 2 h. The OD value at 450 nm was subsequently measured using a microplate reader (BioTek, USA).
Determination of mitochondrial membrane potential
The cells were initially washed once with PBS and subsequently immersed in 1 mL JC-1 staining solution (Beyotime) for a period of 20 min at a temperature of 37°C. Subsequent to this process, the cells were washed twice with staining buffer and photographs were captured using an Olympus microscope (Japan).
Co-IP assay
The Co-IP protocol was performed according to the manufacturer’s instructions with modifications based on established methods. 18 The cells were lysed in ice-cold IP lysis buffer (50 mmol/L Tris-HCl, pH 7.4, 150 mmol/L NaCl, 1% Triton X-100) supplemented with 1% protease inhibitor, and then the cell lysates were subjected to centrifugation (10000× g, 5 min, at 4°C) to obtain the supernatant samples. Protein concentration in the supernatants was quantified using a BCA protein assay kit (Beyotime), and the protein concentration of each sample was adjusted to 2 mg/mL with IP lysis buffer to ensure equal protein input (500 μg/well). A primary antibody against TRIM37 (500 μL; 13037-1-AP, Proteintech) or IgG was incubated with protein A/G magnetic beads (Beyotime) and the mixture was blended thoroughly for 1 h. To obtain antigen-antibody complex, pretreated protein A/G magnetic beads were added to the supernatant samples and incubated at 4°C overnight. Then, the magnetic beads were washed with ice-cold IP lysis buffer for 3–5 times to remove non-specifically bound proteins and eluted via boiling in basic elution buffer at 95°C for 10 min. Finally, the samples were subjected to western blot analysis. IgG controls were included in all Co-IP assay to exclude non-specific binding signals.
CHX-chase assay
The cells were seeded in a 6-well culture plate, with an approximate density of 5 × 105 cells per well, and then cultivated in an incubator containing 5% CO2 at 37°C to reach 70%–80% confluence. After removing the culture medium and washing twice with pre-warmed PBS, the cells were treated with Cycloheximide (CHX, Sigma). CHX was initially dissolved in dimethyl sulfoxide (DMSO) to prepare a 100 mg/mL stock solution and the working solution (100 µg/mL, Sigma) was subsequently diluted in complete medium (final DMSO concentration <0.1%). The cells were then harvested for indicated time points (0, 2, 4, and 6 h) and 0 h time point was defined as the state immediately after CHX addition. Subsequently, the cells were lysed using RIPA lysis buffer (Beyotime), and the cell lysates subjected to western blot analysis mentioned above to observe the half-life of PTEN.
Ubiquitination assay
The cells were seeded in a 6-well culture plate at a density of 5 × 105 cells per well and cultured to reach 80%–90% confluence under standard conditions. Prior to MG132 treatment, the cells were washed once with pre-warmed PBS to remove residual culture medium. For the proteasome inhibition step, MG132 (Selleck) was first dissolved in DMSO to prepare a 20 mmol/L stock solution and the MG132 working solution (20 μmol/L) was freshly diluted in complete medium before use (final DMSO concentration <0.1%). The cells were exposed to 20 μmol/L MG132 for a period of 6 h, immediately placed on ice, washed twice with ice-cold PBS, and harvested. In sequence, the cells were subjected to lysis using IP lysis buffer and the cell lysates were immunoprecipitated with anti-TRIM37 antibody (Proteintech, 13037-1-AP). The immunoprecipitate was eluted via boiling at 95°C for 10 min, and subjected to western blot analysis using an anti-ubiquitin antibody (Abcam, ab134953).
Western blot
Whole cell lysates were prepared using RIPA lysis buffer (Beyotime) and the protein concentration was then quantified using the BCA Protein Assay Kit (Beyotime). An equal amount of protein was separated using SDS-PAGE, before being transferred to a PVDF membrane. Following blocking with 5% non-fat milk, the membrane was probed with primary antibodies against TRIM37 (Proteintech, China, 13037-1-AP), Bax (Abcam, USA, ab32503), Bcl-2 (Abcam, ab182858), cleaved caspase-3 (Proteintech, 25128-1-AP), cleaved PARP (Proteintech, 60555-1-Ig), cytochrome C (Abcam, ab133504), PTEN (Proteintech, 60300-1-Ig), p-Akt (Proteintech, 66444-1-Ig), and Akt (Proteintech, 10176-2-AP) at 4°C overnight. Following washes, the membrane was incubated with HRP-conjugated secondary antibodies (Abcam) for 1 h. The protein bands were then developed with ECL reagent (Solarbio), and the band intensities were analyzed using the ImageJ software. To ensure experimental reproducibility, blot band intensities were quantified from at least three independent biological replicates for in vitro experiments and six for in vivo. For each target protein, the linear range of detection was validated by analyzing serial dilutions of protein samples, which confirmed that the measured band intensities were within the linear response range of the ECL detection system.
Statistical analysis
Data are presented as mean ± standard deviation (SD). Statistical analyses were performed using GraphPad Prism software 8.0. Normality was assessed using the Shapiro-Wilk test and homogeneity of variance was confirmed using Levene’s test. Statistical differences between two groups conforming to normal distribution and homogeneous variance were analyzed using Student's t-test. One-way ANOVA followed by Tukey’s test was used to analyze statistical differences between multiple groups. Tukey’s test inherently incorporates a multiple comparison correction to control the family-wise error rate at α = 0.05, and no additional post-hoc adjustments were applied. All experiments were repeated in at least three independent experimental sessions. For in vitro experiments, each assay was conducted in at least three independent cell culture experiments, and all experiments were repeated in at least three independent experimental sessions. For in vivo experiments, each assay was performed with three technical replicates and six biological replicates. Investigators were kept blinded throughout the entire process of data collection and analysis. A value of
Results
TRIM37 overexpression improves renal function and ameliorates renal injuries in ADR-induced nephropathy
The analysis of the data from the GEO dataset GSE97709 revealed a marked reduction in TRIM37 expression in patients diagnosed with CKD relative to the healthy control group (Figure 1(a)). In mice treated with ADR, the levels of TRIM37 mRNA and protein were significantly decreased. Furthermore, the injection of lentiviruses overexpressing TRIM37 resulted in a marked elevation of TRIM37 expression in the renal cortex of ADR-treated mice (Figure 1(b) and (c)). The ADR-treated mice exhibited a significant increase in proteinuria when compared with the control group. The overexpression of TRIM37 led to a reduction in proteinuria in ADR-treated mice (Figure 1(d)). Furthermore, the levels of Cr, BUN, TC and TG were elevated in the serum of ADR-treated mice. However, TRIM37 overexpression led to a reduction in these parameters in ADR-treated mice (Figure 1(e)). Gross pathology images showed no marked difference among the groups (Figure 1(f)), indicating that ADR-induced damage was primarily at the microscopic level. The results of HE staining revealed renal tissue structure is intact in the control group. Obvious pathological changes, specifically disordered glomerular and tubular structures as well as mesangial expansion (rather than inflammatory infiltration), were found in the renal tissues of ADR-treated mice, which were ameliorated following TRIM37 overexpression (Figure 1(g)), suggesting that TRIM37 overexpression mitigated ADR-induced structural damage to renal parenchyma. Masson staining revealed the presence of fibrosis in ADR-treated mice, which was attenuated by TRIM37 overexpression (Figure 1(h)), implying that TRIM37 exerted a protective effect against ADR-mediated renal fibrosis. As demonstrated by PAS staining, ADR treatment induced a significant increase in glycogen deposition in renal tissues, and this pathological alteration was alleviated by TRIM37 overexpression (Figure 1(i)), indicating that TRIM37 may regulate renal glycogen metabolism disturbed by ADR exposure. TRIM37 expression is reduced in CKD and its overexpression ameliorates renal injury in ADR-treated mice. (a) TRIM37 expression levels were analyzed using data from the GEO dataset GSE97709, comparing CKD patients and healthy controls. (b) The mice were administered a dose of 10 mg/kg ADR, followed by injection with TRIM37-overexpressing lentivirus or lentiviral vector. Four weeks later, renal tissues were excised. The gene levels of TRIM37 in the renal cortex were quantified by real-time PCR. (c) The protein levels of TRIM37 in the renal cortex were quantified by western blot analysis. (d) The levels of urinary protein in mice were measured weekly. (e) The serum levels of Cr, BUN, TC, and TG were assessed. (e) Gross pathology images. (g) Renal histopathology was examined by HE staining (original magnification 200×, bar = 100 μm). Yellow arrow: mesangial expansion; Blue arrow: disordered glomerular; Green arrow: disordered tubular. (h) Renal fibrosis was assessed by Masson staining (original magnification 200×, bar = 100 μm). Green arrow: collagen fiber accumulation. (i) Glycogen deposition in renal tissues was evaluated using PAS staining (original magnification 200×, bar = 100 μm). Black arrow: deposited glycogen. CKD: chronic kidney disease; ADR: Adriamycin; Cr: creatinine; BUN: blood urea nitrogen; TC: total cholesterol; TG: triglyceride; OE: overexpression. ## indicates 
TRIM37 overexpression alleviates podocyte injury and inhibits cell apoptosis in ADR-induced nephropathy
To investigate the effect of TRIM37 overexpression podocyte injury in ADR-induced nephropathy, the expression levels of several regulators associated with podocyte injury were evaluated by immunofluorescence. As demonstrated in Figure 2(a)–(d), the expressions of podocin, nephrin, and synaptopodin were significantly reduced, while desmin expression was augmented in ADR-treated mice. However, TRIM37 overexpression partially restored their expression in ADR-treated mice. To further investigate the effect of TRIM37 overexpression on cell apoptosis in ADR-induced nephropathy, the TUNEL assay was performed. As presented in Figure 2(e), the ADR-treated mice exhibited a significant increase in TUNEL-positive cell rate in the renal cortex in comparison with the control mice. The overexpression of TRIM37 led to a reduction in TUNEL-positive rate in ADR-treated mice. Furthermore, the expression levels of Bax, cleaved caspase-3, and cleaved PARP were elevated, while that of Bcl-2 was reduced in the renal tissues of ADR-treated mice. However, TRIM37 overexpression partially restored their expression in ADR-treated mice (Figure 2(f)). ADR treatment increased cytoplasmic cytochrome C levels but decreased mitochondrial cytochrome C levels in the renal cortex of ADR-treated mice. TRIM37 overexpression was found to inhibit the cytoplasmic translocation of cytochrome C in ADR-treated mice (Figure 2(g)). TRIM37 overexpression inhibits podocyte injury and apoptosis in ADR-induced nephropathy. Immunofluorescence staining was performed to assess the expression levels of podocyte injury markers ((a) podocin, (b) nephrin, (c) desmin, and (d) synaptopodin) in renal tissues (bar = 50 μm). (e) Apoptosis was evaluated by TUNEL assay in the renal cortex (bar = 50 μm). (f) Western blot analysis was conducted to measure the expression levels of apoptosis-related proteins (Bax, Bcl-2, cleaved caspase-3, and cleaved PARP) in renal tissues. GAPDH was used as a loading control and Band intensities were normalized to Control. (g) The subcellular distribution of cytochrome C was examined by western blot analysis to assess its cytoplasmic and mitochondrial levels in the renal cortex. GAPDH was used as the cytosolic protein loading control, while VDAC1 served as the loading control for mitochondrial protein. Band intensities were normalized to Control. ADR: Adriamycin; OE: overexpression. ## indicates 
TRIM37 overexpression alleviates podocyte injury in vitro
To verify the involvement of TRIM37 in podocyte injury, human renal podocytes were exposed to ADR to mimic ADR-induced nephropathy TRIM37 overexpression ameliorates podocyte injury 
TRIM37 overexpression inhibits mitochondrial apoptosis of podocytes in vitro
Subsequently, the involvement of TRIM37 in the mitochondrial apoptosis of podocytes was verified. The results of the TUNEL assay demonstrated that exposure to ADR significantly increased the TUNEL-positive cell rate in comparison with the control cells. TRIM37 overexpression led to a reduction in the TUNEL-positive cell rate in ADR-exposed podocytes (Figure 4(a)). This observation was further confirmed through the utilization of a flow cytometric analysis (Figure 4(b)). Furthermore, exposure to ADR markedly elevated the expression levels of Bax, cleaved caspase-3, and cleaved PARP were elevated, while reduced that of Bcl-2. TRIM37 overexpression partially restored their expression in ADR-exposed podocytes (Figure 4(c)). As demonstrated in Figure 4(d), exposure to ADR resulted in a loss of mitochondrial membrane potential in podocytes. The overexpression of TRIM37 led to a partial blocking of the loss of mitochondrial membrane potential in podocytes exposed to ADR. Furthermore, exposure to ADR significantly elevated the cytoplasmic level of cytochrome C, while decreasing the mitochondrial level of cytochrome C in podocytes. In addition, TRIM37 expression was found to inhibit the cytoplasmic translocation of cytochrome C in ADR-exposed podocytes (Figure 4(e)). TRIM37 modulates mitochondrial apoptosis in podocytes. (a) TUNEL assay was performed to assess apoptosis in podocytes (bar = 50 μm). (b) Flow cytometric analysis was conducted to quantify apoptotic cells. (c) Western blot analysis was used to measure the protein levels of Bax, Bcl-2, cleaved caspase-3, and cleaved PARP. GAPDH was used as a loading control and Band intensities were normalized to Control. (d) Mitochondrial membrane potential was evaluated using the JC-1 fluorescent probe (bar = 50 μm). (e) Cytochrome C distribution was analyzed by separating cytoplasmic and mitochondrial fractions, followed by western blot analysis. GAPDH was used as the cytosolic protein loading control, while VDAC1 served as the loading control for mitochondrial protein. Band intensities were normalized to Control. ADR: Adriamycin; OE: overexpression. ## indicates 
TRIM37 directly binds to PTEN and regulates its expression via the ubiquitin pathway
The results of Co-IP assay demonstrated that TRIM37 directly bound to PTEN in podocytes (Figure 5(a)). Immunofluorescence staining assay demonstrated the co-localization of TRIM37 with PTEN in podocytes (Figure 5(b)). Additionally, TRIM37 overexpression significantly decreased PTEN expression in podocytes (Figure 5(c)). In ADR-exposed podocytes, PTEN expression was elevated. TRIM37 overexpression greatly reduced PTEN expression in ADR-exposed podocytes (Figure 5(d)). Furthermore, the half-life of the PTEN protein was found to be diminished following TRIM37 expression (Figure 5(e)). Additionally, TRIM37 overexpression was found to promote the ubiquitination of PTEN in podocytes (Figure 5(f)). As demonstrated in Figure 5(g), the p-Akt/Akt ratio was reduced in ADR-exposed podocytes. However, TRIM37 overexpression significantly elevated the p-Akt/Akt ratio in ADR-exposed podocytes. TRIM37 directly binds to PTEN and regulates its expression via the ubiquitin pathway. (a) Co-IP assay was performed to assess the interaction between TRIM37 and PTEN in podocytes. (b) Immunofluorescence staining was conducted to examine the co-localization of TRIM37 and PTEN in podocytes (bar = 50 μm). (c) Following TRIM37 overexpression, the protein levels of PTEN were quantified by western blot analysis. (d) Following ADR treatment and TRIM37 overexpression, the protein levels of PTEN were quantified by western blot analysis. (e) The half-life of PTEN protein was measured in podocytes following TRIM37 overexpression using CHX assay. GAPDH was used as a loading control and band intensities were normalized to time point 0. (f) Ubiquitination assay was performed to assess PTEN ubiquitination levels in TRIM37-overexpressing podocytes. (g) Following ADR treatment and TRIM37 overexpression, the phosphorylation status of Akt was analyzed by western blot analysis. GAPDH was used as a loading control and band intensities were normalized to Control in panels C, D, and G. ADR: Adriamycin; OE: overexpression; CHX: cycloheximide. ## indicates 
PTEN overexpression partially reverses the effects of TRIM37 overexpression in ADR-exposed podocytes
To confirm the TRIM37/PTEN regulatory axis, podocytes were infected with TRIM37-overexpressing lentivirus with or without PTEN-overexpressing lentivirus, then exposed to ADR. The results demonstrated that PTEN overexpression partially blocked the effects of TRIM37 overexpression on podocyte injury (Figure 6(a)–(d)) and cell viability (Figure 6(e)). Furthermore, the inhibitory effect of TRIM37 overexpression on cell apoptosis was partially reversed by PTEN overexpression (Figure 6(f)–(h)). In addition, the effects of TRIM37 overexpression on mitochondrial membrane potential (Figure 6(i)), the translocation of cytochrome C (Figure 6(j)), and the Akt pathway (Figure 6(k)) were partially blocked by PTEN overexpression. PTEN overexpression partially reverses the effects of TRIM37 overexpression in ADR-exposed podocytes. (a–d) Podocytes were infected with the recombinant lentiviruses and then exposed to ADR. Subsequent analysis of podocyte injury was conducted through the utilization of immunofluorescence staining (bar = 50 μm). (e) The viability of the podocytes was assessed. (f) The measurement of apoptosis of podocytes was conducted by flow cytometry. (g) The measurement of apoptosis of podocytes was conducted by TUNEL staining (bar = 50 μm). (h) Western blot analysis was used to measure the protein levels of Bax, Bcl-2, cleaved caspase-3, and cleaved PARP. Band intensities were normalized to Control. (i) Mitochondrial membrane potential was evaluated using the JC-1 fluorescent probe (bar = 50 μm). (j) Translocation of cytochrome C was examined by western blot analysis. GAPDH was used as the cytosolic protein loading control, while VDAC1 served as the loading control for mitochondrial protein. Band intensities were normalized to Control. (k) The activation of the Akt pathway was analyzed by western blot analysis. GAPDH was used as a loading control and band intensities were normalized to Control in panels H and K. ADR: Adriamycin; OE: overexpression. ## indicates 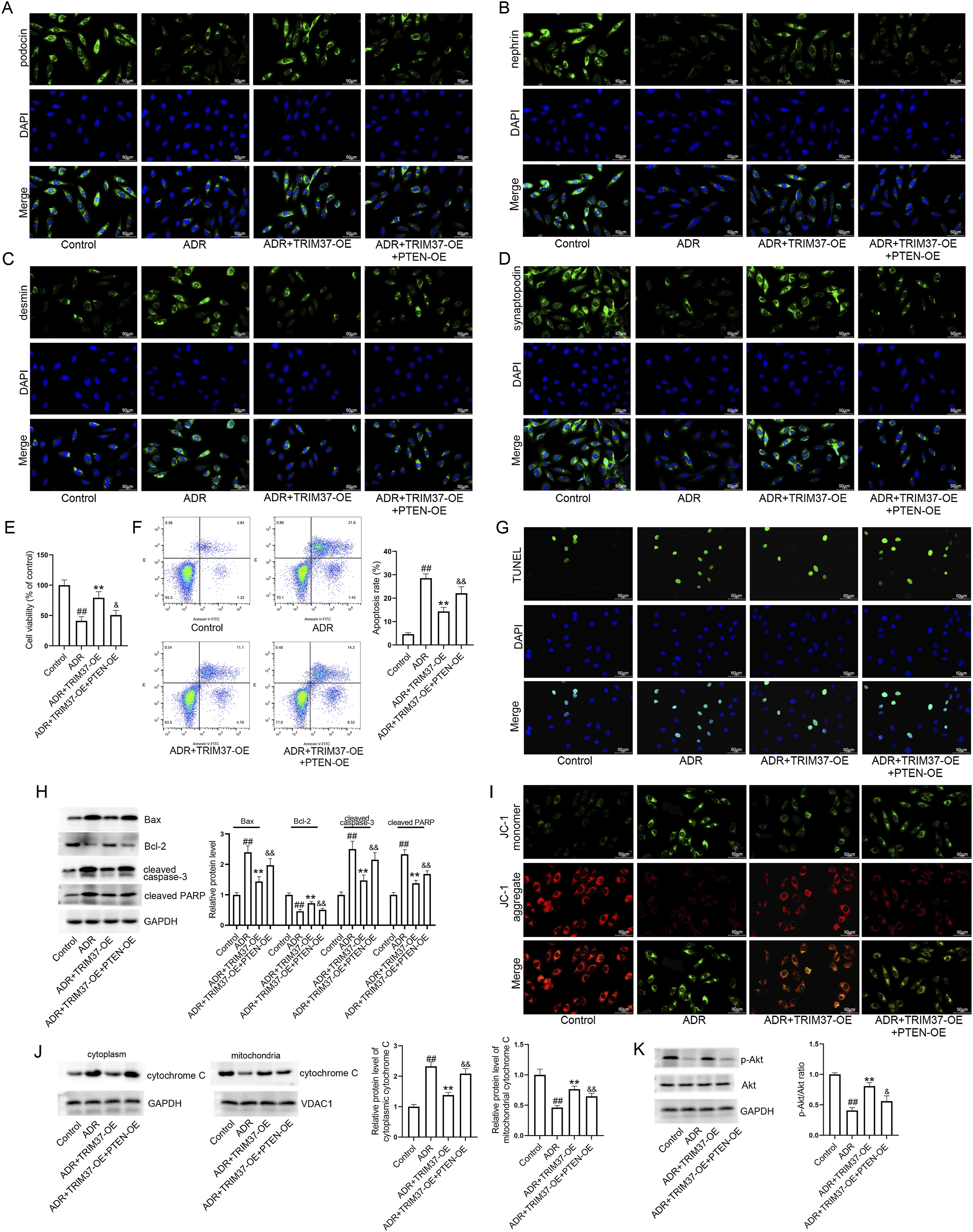
Discussion
It has been established that podocytes represent a pivotal component in the maintenance of the glomerular filtration barrier.
19
Podocytes have a crucial function in preserving normal glomerular structure and function; therefore, podocyte injury has been demonstrated to result in proteinuria, glomerulosclerosis and ultimately, NS.
20
Inhibition of podocyte injury has emerged as a potential therapeutic strategy for the prevention of ADR-induced nephropathy.21,22 It was demonstrated that injection with ADR led to a significant reduction in the expression of podocin, nephrin and synaptopodin, whilst concomitantly increasing desmin expression, in the renal cortex of mice. However, TRIM37 overexpression partially restored podocyte markers. The podocytes subjected to ADR treatment are employed as an
It is widely recognized that podocyte apoptosis serves as a pivotal driver in the progression of proteinuria associated with various chronic kidney diseases.
24
Alterations in mitochondrial membrane permeability are intimately linked with mitochondria apoptosis.
25
The knockdown of TRIM37 promotes the apoptosis of ovarian cancer cells, thus indicating that TRIM37 serves an anti-apoptotic role in this particular type of cancer.
26
Our data demonstrated a substantial enhancement in the apoptosis rate within the renal cortex of mice treated with ADR. TRIM37 overexpression resulted in a decline in the apoptosis rate in ADR-treated mice. The anti-apoptotic effect was also confirmed in the
As the primary executioner caspase in apoptosis, caspase-3 is modulated by various cellular components under both physiological and pathological conditions.
29
In the process of apoptosis, the cleavage of PARP, which occurs at a highly conserved evolutionary site, is mainly mediated by caspase-3, demonstrating PARP cleavage’s significant role in apoptosis.
30
Furthermore, we ascertained that the expression levels of cleaved caspase-3 and cleaved PARP were diminished in response to TRIM37 overexpression in both
Ubiquitination is a post-translational modification that involves the covalent attachment of ubiquitin to target proteins. This process plays a critical role in regulating various cellular processes, including cellular localization, protein stability, and protein activity. 32 The promotion of PTEN poly-ubiquitination by TRIM27 is a contributing factor to the development of esophageal cancer. 33 TRIM37 binds to PTEN and regulates the PI3K/Akt pathway, thereby promoting the growth of human T-cell acute lymphocytic leukemia cells. 34 In pancreatic cancer cells, TRIM37 facilitates chemoresistance and maintains stemness by ubiquitinating PTEN and activating the Akt/GSK-3β/β-catenin pathway. 35 As indicated by earlier research, TRIM37 has been shown to have direct interactions with PTEN in two distinct types of cancer cells, specifically T-cell acute lymphocytic leukemia cells and pancreatic cancer cells.34,35 Co-IP assay results demonstrated the occurrence of a direct interaction between TRIM37 and PTEN in podocytes. Furthermore, the results of the immunofluorescence assay indicated the co-localization of TRIM37 and PTEN within podocytes. TRIM37 overexpression decreased the half-life of the PTEN protein, promoted the ubiquitination of PTEN, and decreased its expression in podocytes, which is in accordance with previous findings.34,35 The PTEN protein is a lipid phosphatase that functions as a negative regulator of the PI3K/Akt pathway.36,37 TRIM37 overexpression activated the Akt pathway in ADR-exposed podocytes. It has been documented that the Akt pathway is activated by TRIM37 in pancreatic cancer cells.34,35 PTEN overexpression partially blocked the effects of TRIM37 overexpression on cell viability, podocyte injury, mitochondrial apoptosis, and the Akt pathway. The findings indicate that TRIM37 may alleviate ADR-induced nephropathy by promoting the ubiquitin-dependent proteasomal degradation of PTEN and activating the Akt pathway.
Limitations
However, it should be noted that this study has certain limitations. Only one podocyte cell line and one ADR-induced mouse model is used, lacking validation with human kidney biopsy samples and additional experimental models, which should be addressed in future research to better confirm the therapeutic potential of TRIM37 in NS. Meanwhile, future studies should include TRIM37 knockdown or knockout experiments to confirm the specificity of the observed effects.
Conclusions
In conclusion, TRIM37 expression was significantly lower in both
Footnotes
Ethical considerations
All animal studies were approved by the Ethics Committee of General Hospital of Northern Theater Command Hospital (No. 2025-40), and were performed in strict accordance with the NIH Guide for the Care and Use of Laboratory Animals (8th edition).
Consent to participate
No human participants were involved, and thus clinical trial registration was not required.
Author contributions
Yu Xin: Methodology, Visualization, Data curation, Software, Writing-original draft; Xue Yang: Methodology, Visualization, Data curation, Software, Validation; Yanning Zhang: Conceptualization, Validation, Supervision, Writing-review and editing; Yanhua Jin: Conceptualization, Funding acquisition, Supervision, Writing-review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Liaoning Province Applied Basic Research Plan Projects (2022JH2/101500043).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data supporting the findings of this study are available within the article and its supplementary materials, or from the corresponding author upon reasonable request.
