Abstract
Spinal cord injury (SCI) is a destructive event in central nervous system (CNS) with the hallmark of deficits in neuronal function. Phoenixin-14 (PNX-14) is a reproductive peptide that also has neuroprotective effects. However, the role of PNX-14 in SCI has not yet been studied. In this study, we firstly investigated the effects of PNX-14 on the recovery of neurological dysfunction and microglial polarization in a SCI mice model. We demonstrated that PNX-14 improved the recovery of neurological dysfunction with increased Basso Mouse Scale (BMS) scores, reduced lesion area volume and Evans blue (EB) dye extravasation. PNX-14 alleviated neuronal apoptosis and neuroinflammation in mice underwent SCI. In vitro co-culture assay proved that PNX-14 protected neurons injury in response to LPS- activated BV-2 cells. PNX-14 suppressed the LPS- induced microglia M1 phenotype polarization with decreased expression of M1-associated markers (CD16 and iNOS) and increased expression of M2-associated markers (CD206 and Arg1). PNX-14 also suppressed LPS- caused decrease in anti-inflammatory cytokines TGF-β, IL-10, and IL-13, as well increase in pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 in BV2 cells. PNX-14 treatment caused increased PTEN expression and decreased p-Akt expression in BV2 cells against LPS induction. While inhibition of PTEN by SF1670 reversed the effects of PNX-14 on LPS- induced phenotypic transition of BV2 cells. Taken together, we found that PNX-14 exerted protective effects on neurological dysfunction and inflammation in SCI mice through modulating microglial polarization via PTEN/Akt signaling pathway.
Keywords
Introduction
Spinal cord injury (SCI) is a destructive event with the hallmark of deficits in neuronal function. 1 SCI commonly causes major motor disabilities, and autonomic and sensory dysfunctions. 1 Although extensive researches have been conducted to develop therapeutic approaches for SCI, none of them have exerted a meaningfully functional recovery in patients with SCI. To date, a more thorough understanding of the pathophysiology is still needed to be established for developing effective therapeutic targets to intervene SCI damage.
A cascade of destructive events is found to be associated with the pathophysiology of SCI including ischemia, oxidative stress and inflammation responses, profound activation of apoptotic-associated pathways, and mechanical death of neurons.2,3 There is a general consensus that inflammation after SCI is complex with dual effects. Although inflammation has several positive effects, the extensive inflammatory response with deregulated immune cells infiltration also contribute to neural degeneration after SCI.4,5 Inflammation is orchestrated by diverse inflammatory cytokines and numerous cell types. In particular, microglia are resident immune cells in CNS that respond rapidly to perturbations. It is well known that microglia are very plastic cells with two phenotypes, either protective (anti-inflammatory) or cytotoxic state (pro-inflammatory), which can be influenced by the microenvironment of the injured CNS following SCI. 6 Due to their profound roles in CNS, modification of microglia is a focus of therapeutic research for SCI.
Phoenixin (PNX) is a newly identified reproduction-associated peptide with two active isoforms, namely PNX-14 and PNX-20. 7 Initially, PNX is demonstrated as a reproductive peptide. Further studies have found that PNX also exerts a wide range of functions in inhibiting visceral pain, improving memory retention, and cardiac damages, inducing anxiety and eliciting pruritus. 8 Recently, both PNX-14 and PNX-20 have been found to have neuroprotective effects against neuroinflammation and ischemia/reperfusion-induced cerebral injury. PNX-20 also has the property to regulate the microglia-mediated neuroinflammation via modulating NLRP3 inflammasome. 9 However, the role of PNX-14 in regulating microglial activation has not yet been studied.
For this purpose, we investigated the effects of PNX-14 on the recovery of neurological dysfunction and microglial polarization in a SCI mice model and the underlying mechanisms associated with its regulatory property. We found that PNX-14 exerted protective effects on neurological dysfunction, inflammation, and microglial polarization in SCI mice and LPS- induced BV-2 microglia through regulating PTEN/Akt signaling pathway.
Materials and Methods
In vivo animal studies
The establishment of SCI mouse model was performed as described previously.
10
Briefly, after anesthesia with 50 mg/kg sodium pentobarbital via intraperitoneal injection, mice were subjected to excision of the T9 lamina. Then the T7 and T11 spinous processes were clamped to stabilize the spine. The dorsal surface of the cord was exposed, a 1.3 mm rod was used to compress the spinal cord to moderate contusion injury. For the animal studies, 60 male C57BL/6J mice weighting 18–35 g (8 weeks old; Beijing Vital River Laboratory Animal Technology Co., Ltd, Beijing, China) were separated into three groups (
On day 14 post injury, blood samples were collected by the heart punctures, and the serum samples were prepared. Afterwards, the spinal cord segments were extracted after the incision of SCI surgery was opened. The lesion area was monitored and recorded, and then parts of the spinal cord samples were then fixed in paraformaldehyde (4%, w/v) at 4°C, while the remaining parts of the spinal cord samples were stored at −80°C until processed for western blot analysis.
Functional recovery tests
The motor function of the mice was quantified at 1, 3, 7, 14, 21, and 28 days after sham or SCI surgery using the Basso Mouse Scale (BMS) for locomotion capacity. As described by previous studies , 10 scores were recorded to reflect the functional recovery.
Measurement of blood-spinal cord barrier permeability
The extravasation of Evans blue (EB) assay was performed to determine the integrity of the blood-spinal cord barrier. 11 Briefly, 2% EB dye solution was intravenously injected to the mice on 7 and 14 days post SCI injury. The mice were transcardially perfused with 4% paraformaldehyde. The spinal cord segments were immediately weighed and processed for determining the quantity of EB extravasation (expressed as μg/g of tissues) through detecting the absorbance at a wavelength of 620 nm by a spectrophotometer (Shimadzu Corporation, Japan).
Immunofluorescence analysis
The spinal cord samples fixed with paraformaldehyde were processed into sections (5 μm thick), followed by incubation with anti-cleaved caspase 3 (1:200; Abcam, Cambridge, MA) for 10 h at 4°C. After being washed for four times, the sections were incubated with AlexaFluor 488 secondary antibody (1:200; Invitrogen, Carlsbad, CA) for 1 h at 37°C. Then, the sections were washed and finally sealed with a coverslip. The sections were analyzed by confocal fluorescence microscope (Nikon, Japan) and the images were captured.
BV-2 cells culture and treatment
The BV-2 cells (ATCC, Manassas, VA) were cultured in high-glucose DMEM (Sigma-Aldrich, Missouri, USA) supplemented with 0.1% antibiotics (Sigma) and 5% FBS (Gibco, Grand Island, NY). BV-2 cells were pretreated with or without 10 nM PNX-14 or 1 μM SF1670 for 2 h before stimulation with 0.1 μg/mL LPS for another 24 h. The supernatant samples of BV-2 cells from different groups were collected as conditioned media in co-culture assay.
Neuronal co-culture assay
A co-culture model of neuronal cells and microglia was applied for the investigation of the protective effect of PNX-14. 12 After incubation with differentiation media (0.5% horse serum and 100 ng/mL NGF) for 1 week, the differentiated PC-12 cells (ATCC; 1000 cells/well) were incubated with conditioned media for 18 h. The PC-12 cells in control group were cultured with 0.1 μg/mL LPS to normalize the effect of LPS.
CCK-8 assay
The PC-12 cells viability was assessed by CCK-8 assay with a CCK-8 kit (Dojindo Kumamoto, Japan). After incubation with 10 μL of CCK-8 solution for 2 h, the PC-12 cells from each group were subjected to detection of absorbance at 450 nm using microplate reader (Tecan, Durham, NC).
Western blot analysis
The spinal cord samples, PC-12 cells and BV-2 cells were homogenized in ice-cold lysis buffer. Then protein concentrations in the extracts were detected using a Pierce BCA protein assay (Thermo Fisher Scientific, Waltham, MA). Subsequently, equal amounts of total proteins were separated using SDS‐PAGE and transferred to membranes, followed by western blotting assay described elsewhere. The antibodies against iNOS, CD16, CD206, Arg1, PTEN, Akt, and p-Akt (1: 500; Abcam) and secondary antibodies conjugated with HRP (1: 3000; Abcam) were applied for the immunoblotting. Targeted protein bands were finally detected with an enhanced chemiluminescence reagent (Thermo Fisher Scientific) and the intensity was semi-quantified with Image J software.
ELISA
The concentrations of TGF-β, IL-10, IL-13, TNF-α, IL-1β, IL-6 and IFN-γ in serum samples or cell culture medium were measured by ELISA using appropriate kit (R&D Systems, Minneapolis, MN). Absorbance at 450 nm was measured by a microplate reader (Tecan).
Data processing
All data were processed using SPSS version 13.0 software (SPSS Inc., Chicago, IL) with analysis of variance (ANOVA) method for comparisons among multiple groups. Data with normal distribution are presented as means ± SD.
Results
PNX-14 promoted neurological function recovery in rats underwent SCI
The BMS scores in SCI group and PNX-14 treatment group were markedly reduced after SCI injury, while the BMS scores were gradually improved after 7 days post SCI injury. Mice treated with PNX-14 exhibited higher BMS scores than those in SCI group, implying that PNX-14 improved the motor function of mice with SCI injury (Figure 1(a)). The results in Figure 1(b) demonstrated that the lesion area volume was markedly reduced in mice treated with PNX-14 on both 7 and 14 days after SCI injury. Measurement of EB extravasation showed that significant increase in EB content was observed on both 7 and 14 days after SCI injury. While PNX-14 treatment group obtained significant less EB content than that in SCI group, which suggested that PNX-14 attenuated the increased BSCB permeability (Figure 1(c)). PNX-14 promoted neurological function recovery and spinal cord injury in rats underwent SCI. (a) BMS scores at 0, 1, 3, 7, 14, 21, and 28 days (b) Lesion area volume on 7 and 14 days after SCI. (c) Evans blue (EB) dye extravasation on 7 and 14 days after SCI (***, 
PNX-14 alleviated neuronal apoptosis in rats underwent SCI
Immunofluorescence staining assay proved that the proportion of apoptotic neurons was higher in SCI group than that in sham group after 14 days post SCI injury, while PNX-14 treatment group exhibited lower proportion of apoptotic neurons (Figure 2(a)). The expression levels of cleaved-caspase-3 and bax were markedly increased, while expression of bcl-2 was decreased in SCI group relative to sham group. But the changes in cleaved-caspase-3, bax and bcl-2 expression were attenuated by PNX-14 treatment after 14 days post SCI injury (Figure 2(b)). PNX-14 alleviated neuronal apoptosis in rats underwent SCI on 14 days after SCI. (a) Immunofluorescence staining for cleaved-caspase-3, the proportion of apoptotic neuron. (b) Western blot for cleaved-caspase-3, bax, bcl-2 expression (***, 
PNX-14 mitigated neuroinflammation and promoted microglia polarization to M2 phenotype in rats underwent SCI
ELISA confirmed that the increased serum contents of TNF-α, IL-1β, IL-6, and IFN-γ in SCI group were reduced after PNX-14 treatment after 14 days post SCI injury (Figure 3(a)). Significantly phenotypic transition of the microglia from the M2 (anti-inflammatory-like) to the M1 (pro-inflammatory-like) was identified in SCI group, as shown by increased expression of CD16 and iNOS, and decreased expression of CD206 and Arg1. While PNX-14 treatment reversed SCI-induced phenotypic transition of the microglia after 14 days post SCI injury (Figure 3(b)). PNX-14 mitigated neuroinflammation and microglia polarization in rats underwent SCI on 14 days after SCI. (a) ELISA for serum contents of TNF-α, IL-1β, IL-6, and IFN-γ. (b) Western blot for CD16, iNOS, CD206, and Arg1 expression (***, 
PNX-14 protected neurons injury in response to LPS-activated BV-2 cells
From CCK-8 assay, the cell viability of PC-12 cells was decreased after co-culture with conditioned media from LPS- induced BV-2 cells, which was attenuated by PNX-14 treatment (Figure 4(a)). The LPS- induced BV-2 cells caused increase in expression of cleaved caspase-3 and bax, as well decrease in bcl-2 expression in PC-12 cells, which could be attenuated by PNX-14 treatment (Figure 4(b)). PNX-14 protected PC12 neurons injury in response to LPS-activated BV-2 cells. (a) CCK-8 for 
PNX-14 regulated the microglial polarization in LPS- induced BV2 cells
The LPS stimulation caused increase in the expression of M1 associated markers (CD16 and iNOS) and decrease in the expression of M2 associated markers (CD206 and Arg1) were altered in PNX-14 treated BV-2 cells (Figure 5). PNX-14 suppressed the microglia M1 phenotype polarization in LPS-induced BV2 cells. Western blot for CD16, iNOS, CD206, and Arg1 expression (***, 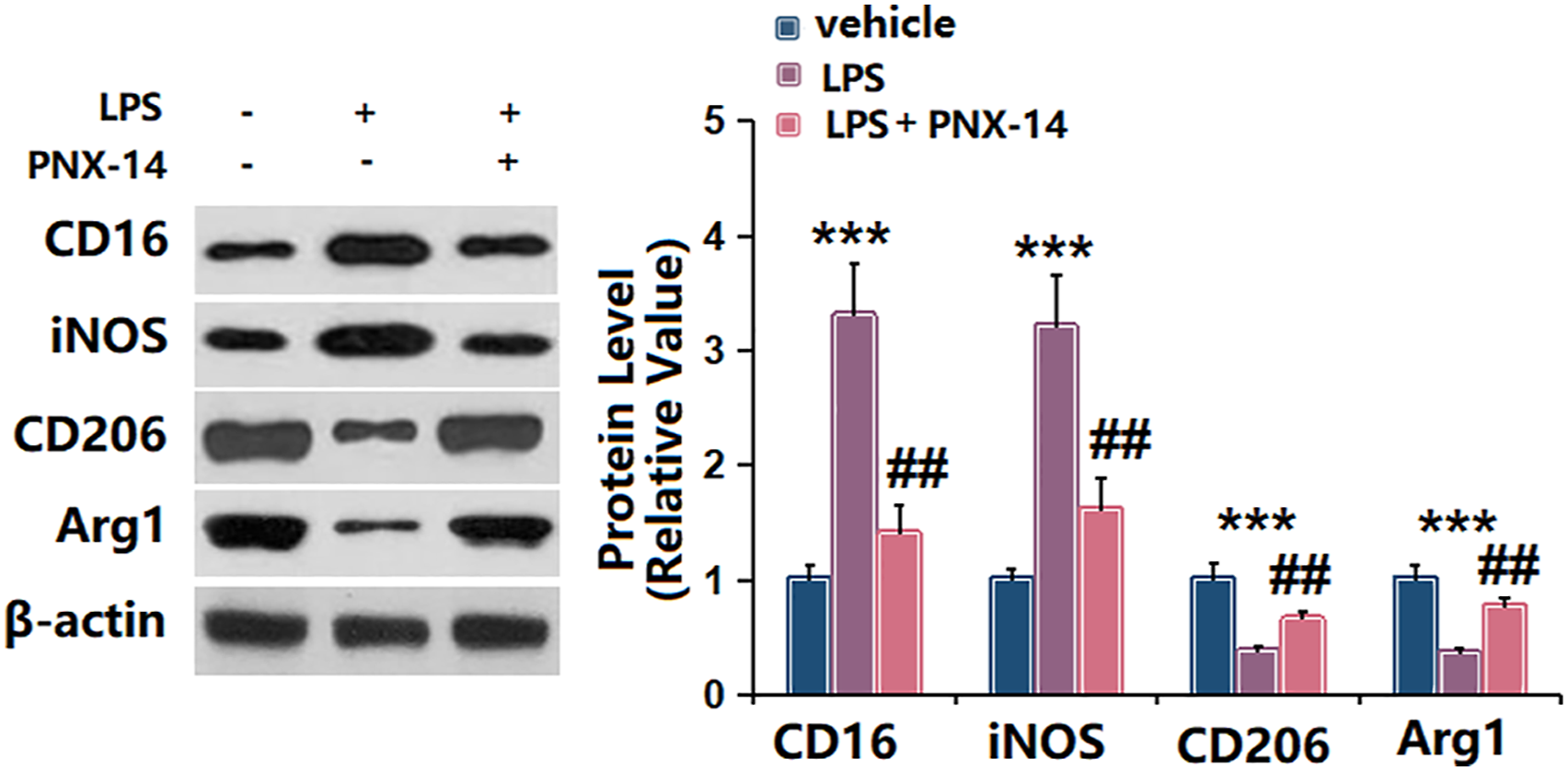
PNX-14 suppressed LPS- induced inflammation in BV2 cells
The secretion levels of anti-inflammatory cytokines TGF-β, IL-10, and IL-13 were lower, while pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 were higher in LPS- induced BV-2 cells than those in control BV-2 cells. However, these changes caused by LPS were suppressed by PNX-14 (Figure 6). PNX-14 suppressed LPS-induced inflammation in BV2 cells. ELISA for anti-inflammatory cytokines TGF-β, IL-10, and IL-13 as well as pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 (***, 
PNX-14 regulated PTEN/Akt signaling in LPS- induced BV2 cells
In Figure 7, the results showed that PTEN was expressed at a lower level, while p-Akt was expressed at a higher level in LPS- induced BV2 cells. PNX-14 treatment caused increased PTEN expression and decreased p-Akt expression in BV2 cells against LPS induction. PNX-14 regulated PTEN/Akt signaling in LPS-induced BV2 cells. Western blot for PTEN, p-Akt, and Akt (***, 
Inhibition of PTEN by SF1670 reversed the effects of PNX-14 on LPS- induced phenotypic transition of BV2 cells
Treatment with SF1670, a PTEN inhibitor, caused significant decrease in PTEN expression and increased p-Akt expression in PNX-14- treated BV2 cells (Figure 8(a)). SF1670 also reversed PNX-14- caused phenotypic transition of BV2 cells from the M1 to the M2 phenotype as shown by increased expression of CD16 and iNOS, and decreased expression of CD206 and Arg1 (Figure 8(b)). The PNX-14- caused increase in TGF-β, IL-10, and IL-13 levels, as well decrease in TNF-α, IL-1β, and IL-6 levels were reversed by SF1670 (Figure 8(c)-(h)). Inhibition of PTEN by SF1670 reversed the effects of PNX-14 on BV2 cells. (a) Western blot for PTEN, p-Akt, and Akt, (b) Western blot for CD16, iNOS, CD206, and Arg1 expression. (c–h) ELISA for anti-inflammatory cytokines TGF-β, IL-10, and IL-13 as well as pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 (***, 
Discussion
Immediately after SCI, shearing and mechanical damages in spinal cord tissues and cells cause disruption of tissue homeostasis and initiate the inflammatory phase. These injuries also cause the activation of glia, such as microglia, astrocytes, pericytes, fibroblasts, and Schwann cells, followed by secretion of toxins and cytokines and subsequent release of chemotactic cellular adhesion molecules.
13
Afterwards, immune cells, neutrophils and monocytes, are infiltrated and migrated to the injury sites, by which drives a robust inflammatory response and underlies the pathological processes in the injured CNS after SCI.
14
During this phase, the profound functions of microglia/macrophages have been established. Microglia are resident immune cells in CNS and patrol the environment through phagocytosing potential pathogens and producing supportive and growth factors. Microglia act as a sensor for pathological changes in the CNS for the defense of diverse events.
15
It is evident that microglia/macrophages possess very novel plastic property that can achieve pro-inflammatory (M1) phenotype/anti-inflammatory (M2) phenotype conversion, in response to both in vitro and
PNX-14 exhibits protective effects against LPS- induced endoplasmic reticulum (ER) stress and inflammasome activation in mouse cerebral astrocytes. 16 PNX-14 has a cerebroprotective effect in a MCAO rat model and inhibits ischemia/reperfusion-induced microglial injury in vitro. 17 PNX-14 improves inflammation and permeability in brain vascular endothelial cells in response to oxygen-glucose deprivation/reoxygenation (OGD/R) exposure. 18 In addition to neuroprotective effect, previous studies have found that PNX-14 exerts an anti-inflammatory role in various pathological conditions. PNX-14 ameliorates myocardial injury and improves cardiac function in a streptozotocin-induced diabetes mice model through regulating severe oxidative stress and inflammation. 19 PNX-14 significantly ameliorates obesity and fatty liver in experimental mice, associated with its anti-oxidative and anti-inflammatory activity. 20 Here we demonstrated that PNX-14 improved the recovery of neurological dysfunction with increased BMS scores, reduced lesion area volume and EB dye extravasation. PNX-14 alleviated neuronal apoptosis, neuroinflammation, and promoted microglial M2 polarization both in vivo and in vitro.
PTEN is a phosphatase frequently altered in many types of diseases, especially in cancer.21, 22 Mounting evidences indicate that PTEN participate in regulating protein synthesis, cell cycle, survival, growth, migration, and DNA repair signaling. 23 PTEN has the ability to antagonize PI3K/Akt signaling by inhibiting PIP3-dependent processes, thereby inhibiting cell growth, survival, and proliferation progression. 24 Of note, Akt signaling occupies a critical node for microglia/macrophages activation and M1/M2 polarization. 25 Recently, it has been proven that PTEN/Akt is involved in the microglia polarization. Inhibition of histone deacetylases modulates inflammation and promotes the microglia polarization towards M2 phenotypein white matter injury (WMI) animal model via GSK3β/PTEN/Akt axis. 26 Activated microglia-mediated inflammatory pain is related to increased spinal cells apoptosis, which is probably mediated by PTEN/PI3K/Akt signaling. 27 Our results proved that PNX-14 regulated PTEN/Akt signaling in LPS-induced BV2 cells. Moreover, inhibition of PTEN by SF1670 reversed the effects of PNX-14 on LPS- induced phenotypic transition of BV2 cells, indicating that PNX-14 exerted its roles through the PTEN/Akt signaling.
In summary, we found that PNX-14 exerted protective effects on neurological dysfunction, inflammation and microglial polarization in SCI mice and LPS-induced BV-2 microglia. Further studies showed that PTEN/Akt signaling pathway was involved in the regulatory property of PNX-14.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the “Natural Science Foundation of Shanxi Science and Technology Department (Project code:201601D011104)”.
