Abstract
Background
Cyclophosphamide (Cyp) is associated with various organ toxicities. The study aimed to investigate the efficacy of zofenopril (Zf), thymoquinone (Thym), and their combination in Cyp-induced cardiotoxicity.
Methodology
Thirty rats were divided into five groups of six rats each. They received the following treatment orally for 19 days: Control (Con) and Cyp groups: normal saline. Zf: Zf 15 mg/kg, Thym: Thym 80 mg/kg, and Zf + Thym: a combination of both. A single dose of Cyp 200 mg/kg intraperitoneally (IP) was given on day 17 of the experiment to all the groups except the Con. Cardiac, inflammatory, and apoptotic biomarkers, including troponin T, lactate dehydrogenase (LDH), CK-MB, hs-CRP, nuclear factor kappa B (NF-κB), caspase-3, and total antioxidant capacity (TAC), along with lipid profile and histopathological lesions, were assessed.
Results
Cyp resulted in cardiotoxicity as manifested by a significant increase in troponin T, CK-MB, caspase-3, hs-CRP, suppression of TAC level, and marked histopathological alterations in cardiac tissues. Zf, Thym, and their combination significantly reduced CK-MB levels. NF-κB level was significantly decreased by Thym, while the combination of Zf and Thym significantly elevated TAC. hs-CRP was significantly reduced only by Zf. Caspase-3 were significantly lowered by both Zf and Thym individually, as well as by their combination.
Conclusion
Zf and Thym provided cardioprotection against Cyp-induced cardiotoxicity through distinct mechanisms. Zf exhibited anti-inflammatory effects, evidenced by a significant reduction in hs-CRP, along with anti-apoptotic activity. Thym significantly suppressed NF-κB expression. Their combination enhanced antioxidant capacity, however, no superiority over individual treatments was observed concerning their other actions.
Introduction
Zofenopril is a sulfhydryl (H2S) releasing angiotensin-converting enzyme inhibitor (ACEI). It is a highly lipophilic molecule and could readily reach cardiovascular tissues to exert its potential protective effects. Numerous studies are focused on the role of the H2S group, an active metabolite of Zf, in protecting the cardiovascular system. It exerts anti-remodeling actions, improves left ventricular function, and ameliorates ventricular hypertrophy and fibrosis.1,2 Donnarumma et al. 3 demonstrated that the potential cardioprotective activity of Zf in murine and swine models of myocardial ischemia-reperfusion injury was linked to the release of H2S and increased nitric oxide bioavailability. Additionally, another study revealed that the release of H2S from Zf provided an additional cardioprotective mechanism independent of ACE inhibition. 4 In general, H2S has been recognized as a potent cardioprotective signaling molecule, effectively mitigating the pathological effects of myocardial ischemia-reperfusion injury and heart failure, also improving left ventricular remodeling and reducing cardiac fibrosis in spontaneously hypertensive rats.5–7 Furthermore, another study revealed that zofenoprilat protects the coronary endothelium from doxorubicin-induced apoptosis. 8 In a clinical setting, the efficacy and safety of Zf and its long-term follow-up demonstrated that Zf was more effective than ramipril in reducing cardiovascular mortality and hospitalizations in patients with left ventricular dysfunction after acute myocardial infarction, with benefits persisting for up to 5 years.9,10
Cyclophosphamide (Cyp), a widely used chemotherapeutic agent, is related to dose-dependent cardiotoxicity. 11 Multiple mechanisms are involved in Cyp-induced cardiotoxicity, including oxidative stress, inflammation, and apoptosis, with nuclear factor-kappa B (NF-κB) acting as a key player in driving inflammatory responses and caspase-3 mediating apoptotic pathways.12,13 Moreover, Cyp downregulates antioxidant genes of catalase, glutathione peroxidase, superoxide dismutase, and increases lipid peroxidation. 14
Acrolein, an active metabolite of Cyp, induces oxidative stress by reducing glutathione, depleting antioxidant enzymes like catalase, and activating caspases and NF-κB. Caspase activation triggers apoptosis, while NF-κB moves to the nucleus, enhancing the transcription of interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), the pro-inflammatory cytokines. Furthermore, acrolein initiates cardiac cell apoptosis through the extrinsic apoptotic pathway.11,15 Furthermore, findings from a previous study revealed an elevation of serum creatine phosphokinase isoenzyme (CK-MB), lactate dehydrogenase (LDH), free and esterified cholesterol, and triglycerides significantly. In addition, Cyp markedly upregulated the mRNA expression of apoptotic genes, including p53 and Bax. 14 Various substances have been explored for their protective effects against Cyp-induced cardiac damage; mainly, they exert cardioprotective effects by inhibiting free radical formation, reducing inflammatory cytokines, and suppressing apoptosis.13,16,17 These findings highlight the potential of antioxidant, anti-inflammatory, and anti-apoptotic strategies in mitigating Cyp-induced cardiotoxicity.
Great efforts have been made for detoxification and amelioration of Cyp-induced cardiotoxicity. The management of cardiovascular diseases associated with Cyp-induced toxicity involves multiple drugs, and these are often accompanied by side effects and drug interactions. Therefore, the investigators are searching for alternative management strategies to lessen the cardiotoxicity associated with the use of Cyp. The use of inhibitors of ACE with distinctive characteristics such as powerful antioxidant activity, high lipophilicity, and high tissue penetration might be of value with fewer side effects, more cost-effectiveness, and easy accessibility.
Additionally, thymoquinone (Thym), the main active component of Nigella sativa, is known for its powerful antioxidant, anti-inflammatory, and anti-angiogenesis properties. 18 Many experimental and clinical studies reported the importance of the use of Thym as a cardioprotective agent.19–21 Several studies also provide supportive evidence for the potential use of this substance in managing chemotherapy-associated cardiovascular complications. Accordingly, this study was designed to evaluate the efficacy of Zf and Thym, both individually and in combination, against cyclophosphamide-induced cardiotoxicity.
Materials and methods
Chemicals and drugs
The supplier of cyclophosphamide was Baxter Oncology GmbH, Frankfurt am Main, Germany. Zofenopril was from A.Menarini Pharmaceutical Industry, Florence, Italy. While thymoquinone was obtained from Glentham Life Sciences-United Kingdom. NF-κB, caspase-3, Troponin T, and TAC were determined by the ELISA kits for rats from the Bioassay Technology Laboratory in Shanghai, China. While hs-CRP, LDH, and lipid profile were measured by the automatic analyzer SIEMENS Dimension EXL 200 using kits from Siemens Healthcare Diagnostics Inc., USA. CK MB (IU/L) was measured using the automatic analyzer cobas c311.
Ethical consideration
The approval of the Ethics and Research Registration Committee of the College of Pharmacy-University of Sulaimani was obtained for the protocol of this study with a Registration number (PH98-23 on 24.05.2023). All procedures were carried out following the Principles of Laboratory Animal Care, 22 and the study adhered to the ARRIVE guidelines to ensure ethical conduct and transparent reporting of animal research. Animals were randomly assigned to treatment groups using a random number generator to minimize selection bias. Blinding was maintained throughout the experimental procedures: both the investigators conducting biomarker analyses and the histopathologist evaluating tissue sections were blinded to group allocations to ensure objective and unbiased data analysis and interpretation. All animal-related procedures, including housing, handling, dosing, and monitoring, were conducted in full compliance with the ARRIVE guidelines, including adherence to the 3Rs principles (Replacement, Reduction, and Refinement). The present study was designed as an exploratory/preliminary investigation to investigate the efficacy of Zf, Thym, and their combination in Cyp-induced cardiotoxicity in rats. Accordingly, no formal sample size calculation was performed. Instead, the number of animals per group was determined based on common practice in similar experimental studies, while also adhering to the 3Rs principle to minimize animal use.23,24 Animals were monitored daily for signs of distress or discomfort, and humane endpoints were established to minimize suffering. At the end of the experiment, animals were humanely sacrificed under anesthesia using an intraperitoneal (IP) injection of ketamine at a dose of 75 mg/kg and xylazine at 10 mg/kg, following approved ethical protocols. 25
Experimental design, groups, and treatment protocol
This experimental study included 30 female rats. Six animals per group were chosen based on established practices in similar toxicological studies, balancing scientific validity with ethical considerations. 26 They were purchased from the animal house of the College of Pharmacy-University of Sulaimani, “self-funded”. The rats were housed in plastic cages and allowed to acclimate for 1 week under standardized conditions, including a 12-h light-dark cycle, a temperature of 22 ± 1°C, and a relative humidity of 50 ± 5%.
The animals were divided into five groups, each of six rats (n = 6), and named as follows: (1) Control (Con) group: Animals received normal saline (1 mL) orally once daily for 19 days. On the 17th day, they received a 1 ml intraperitoneal injection of normal saline. (2) Cyclophosphamide (Cyp) group: Animals received normal saline (1 mL) orally once daily for 19 days. On the 17th day, they received a single intraperitoneal injection of Cyp 200 mg/kg body weight prepared in normal saline. (3) Zofenopril (Zf) group: Animals received Zf 15 mg/kg orally for 19 days. On the 17th day, they received a single intraperitoneal injection of Cyp 200 mg/kg body weight prepared in normal saline. (4) Thymoquinone (Thym) group: Animals received Thym 80 mg/kg orally for 19 days. On the 17th day, they received a single intraperitoneal injection of Cyp 200 mg/kg body weight prepared in normal saline. (5) Zofenopril + Thymoquinone (Zf + Thym) group: Animals received a combination of both Zf 15 mg/kg and Thym 80 mg/kg for 19 days. On the 17th day, they received a single intraperitoneal injection of Cyp 200 mg/kg body weight prepared in normal saline.
The dose and duration of Cyp,17,27 Zf,3,23,24,28 and Thym 18 were selected depending on previous studies with modifications. A fresh Thym solution was prepared by dissolving the required dose in a minimal volume of olive oil to achieve a homogenous solution, freshly prepared and administered orally by gavage tube. While the Zf solution was prepared by dissolving the required dose in a 0.5% carboxymethyl cellulose (CMC) solution to form a uniform solution, which was freshly prepared and administered orally by gavage tube. At the end of the experiment, the rats were sacrificed, and the blood and cardiac tissue were collected for biochemical, inflammatory, and total antioxidant capacity evaluations.
Measurement of body weight
The body weight of each rat was measured and recorded before and after the experiment. The initial weight was then compared to the weight recorded on the final day of the experiment to assess changes over time.
Assessment of inflammatory, oxidative stress status, and apoptotic marker caspase-3
hs-CRP was measured by an automatic analyzer SIEMENS Dimension EXL 200 using kits from Siemens Healthcare Diagnostics Inc., NF-κB, TAC, and caspase-3 were determined by using the ELISA kits for rats.
Assessment of cardiac and biochemical markers
The cardiac tissue Troponin T level was determined using an ELISA Kit following the manufacturer’s instructions. CK-MB was measured using the automatic analyzer Cobas c311. LDH and lipid profile were measured by the automatic analyzer SIEMENS Dimension EXL 200 using kits from Siemens Healthcare Diagnostics Inc.
Histotechnique procedure
The histological protocol commenced at the experiment’s endpoint. The rats were humanely euthanized using an intraperitoneal (IP) injection of ketamine at a dose of 75 mg/kg and xylazine at 10 mg/kg, after a fasting period of no less than 10 h. Following euthanasia, a necropsy was performed, during which tissue samples were collected for histological analysis.
Briefly, heart samples were collected, placed in tissue cassettes, and fixed in 10% neutral buffered formaldehyde for approximately 48 h. Subsequently, tissue sections underwent dehydration through a different concentration of ethanol (50%, 60%, 70%, 90%, and 100%), followed by two to three cycles of xylene clearance. The processed heart tissue was then infiltrated and embedded in paraffin blocks using an automated wax embedder at 60°C–70°C.
Paraffin-embedded tissues were sectioned into 5 µm slices using a semi-automated rotary microtome. The sections were mounted on glass slides and dried on a hot plate tissue holder. The slides were then deparaffinized, the cleaning process was performed with xylene for 30 min, then dried at 50°C for 5 min in a hot oven. Harris’s hematoxylin and eosin solution was used to stain the tissue sections, cleared with xylene, cover-slipped, and examined under a bright-field light microscope.
Semi-quantitative lesion scoring
Lesion scoring was assessed semi-quantitatively using image analysis software (AmScope 3.7) with a microscope eyepiece camera (MD500, 2019). Tissue samples were examined under a light microscope (NOVEL XSZ-N107T, China). Within the interstitial regions of heart sections, infiltrated inflammatory cells were counted in in randomly chosen four fields under high power magnification (100X), then the mean average was calculated statistically in percentage, whereas vascular Vascular congestion was assessed in micrometers (µm) and statistically evaluated as mean percentage. Additionally, inflammatory exudate zones dispersed within the interstitial connective tissue were quantified as the mean percentage of the estimated area in square micrometers. All calculated values were expressed as the following lesion scoring and grading scores: 0%–10% as No lesions, 10%–25% as Mild, 25%–50% as Moderate, 50%–75% as Severe, and 75%–100% as Critical.
Statistical analysis
Statistical analyses were carried out using GraphPad Prism version 10.4.1 (GraphPad Prism Software LLC, CA, USA). Results are expressed as mean ± standard error of the mean (SEM), and 95% confidence intervals (CIs) were provided. Normality of data distribution was assessed using the Shapiro-Wilk test. Differences between groups were evaluated using one-way ANOVA, followed by Tukey’s or Bonferroni’s multiple comparison test, and two-way ANOVA confirmed by Tukey’s multiple comparison test. Effect sizes were calculated using Eta squared (η2) for ANOVA, which is indicated as having no effect if 0 ≤ η2 < 0.01, a minimum effect if 0.01 ≤ η2 < 0.06, a moderate effect if 0.06 ≤ η2 < 0.14, and a strong effect if η2 ≥ 0.14.29,30 A p-value <0.05 was considered statistically significant.
Results
Survival rate and effect of zofenopril and thymoquinone on body weight
All the animals survived throughout the experimental period, with no mortality observed in any of the study groups; therefore, no unexpected deaths or suffering occurred. As depicted in Figure 1, a significant increase in body weight has been observed in the Con group, i.e., a group that received normal saline for 19 days (p < 0.0003). No significant changes in body weight have been noted in the Cyp-group and Cyp-induced cardiotoxicity rats receiving Zf, Thym, and their combinations (p > 0.05). Effect of Zf and Thym on body weight in Cyp-induced cardiotoxicity in rats. Data are presented as mean ± standard error of the mean (SEM) (n = 6) and statistically analyzed using two-way ANOVA, confirmed by Tukey’s multiple comparison test. ***p < 0.0003 compared the body weight of the last day of the experiment with the baseline (First day). Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone, gm: gram.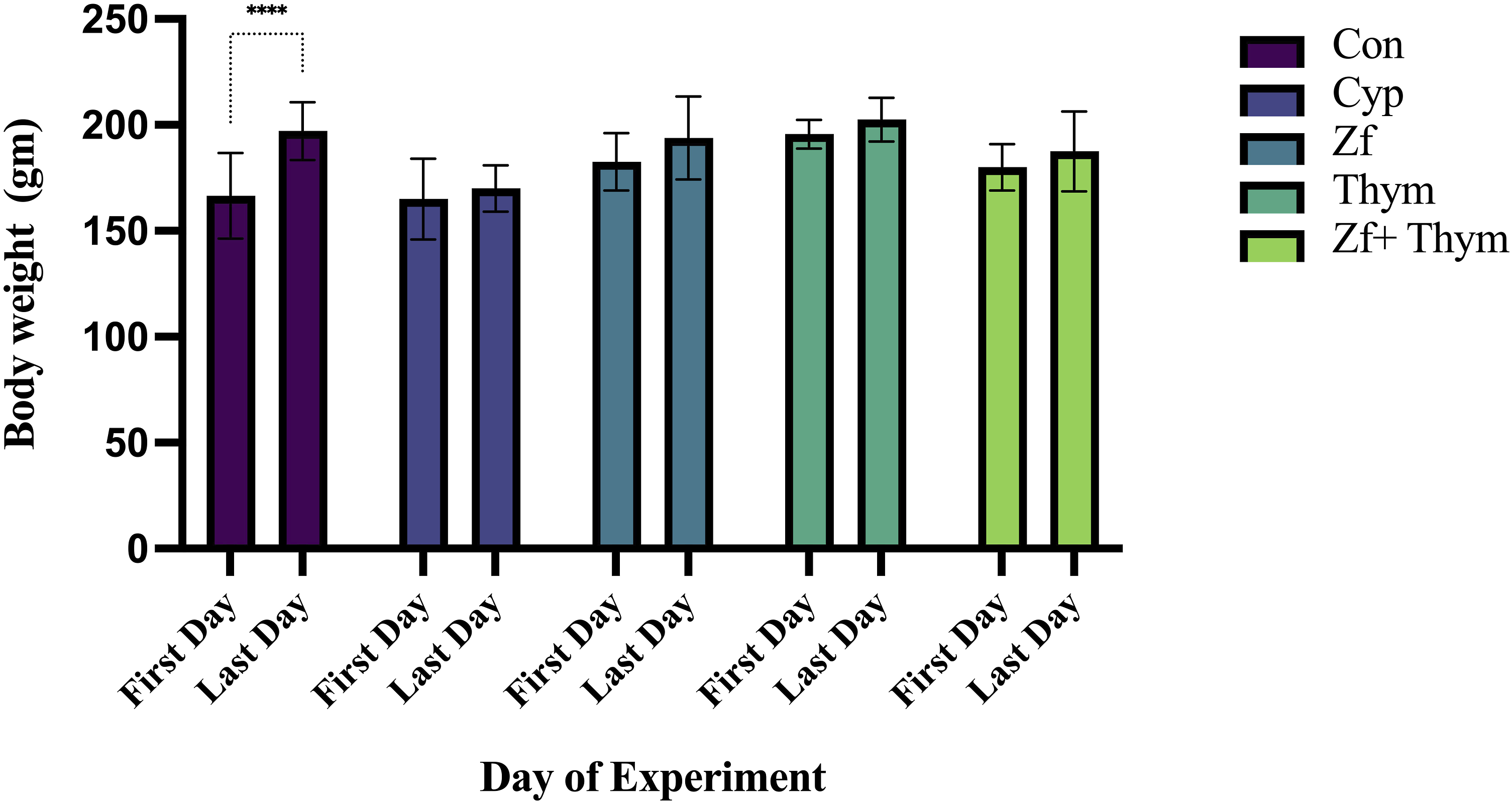
Effect of zofenopril and thymoquinone on lipid profile in Cyp-induced cardiotoxicity in rats
As shown in Figure 2(a)–(d), serum cholesterol level was significantly elevated in the Cyp-exposed group compared to the Con group (p = 0.001), Zf (p = 0.015) and Thym (p = 0.04) treated groups, but non-significantly different when compared to the combination treatment group Zf + Thym (p = 0.052) (Figure 2(a)). Additionally, TG was notably increased in the Cyp-treated group when compared to the Con group, and Zf halted this elevation and resulted in a reduction of serum TG level in a significant manner (p = 0.03), while Thym and the combination of Zf + Thym non-significantly reduced serum TG (p > 0.05) (Figure 2(b)). There was a significant increase in the serum level of LDL in the Cyp-treated group versus the Con group (p = 0.0002). Treatment with Zf and Thym resulted in a non-significant lowering of this component of lipid profile (p > 0.05). However, the Zf + Thym combination synergistically reduced LDL in a significant manner (p = 0.0025) (Figure 2(c)). In Figure 2(d), non-significant changes in HDL have been noted in all groups. (a)–(d) Effect of Zf and Thym on lipid profile in Cyp-induced cardiotoxicity in rats. (a) Cholesterol, (b) Triglyceride, (c) Low-density lipoprotein, (d) High-density lipoprotein. Values are presented as mean ± standard error of the mean (SEM) (n = 6) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.47, 0.34, 0.58, and 0.3 for panels (a), (b), (c), and (d), respectively. Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). In panel (a): *p = 0.015, 0.04 when comparing the Cyp-treated group with Zf and Thym, respectively. **p = 0.001 when comparing the Cyp-treated group with Con. In panel (b): *p = 0.03. p < 0.05 was considered significant. Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone. Effect of Zf and Thym on heart tissue homogenate level of troponin T. Data are presented as mean ± SEM (n = 6) and statistically analyzed using one-way ANOVA confirmed by Tukey’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.28. Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). p < 0.05 was considered significant. *p = 0.03 when comparing the Cyp-treated group with the Con group. Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone.

Effect of zofenopril and thymoquinone on cardiac biomarkers in Cyp-induced cardiotoxicity in rats
As shown in Figure 3, the cardiac biomarker troponin T in heart tissue homogenate was significantly elevated in the Cyp-exposed group (p = 0.03). Although the reduction in troponin T levels did not reach statistical significance (p > 0.05) by Zf and Thym, treatment with Zf, Thym, and their combination appeared to attenuate the release of this biomarker from cardiac tissue, suggesting a potential protective effect.
Additionally, the effect of Zf and Thym on CK-MB and LDH in serum was assessed. A significant elevation in the serum level of CK-MB was observed in the Cyp-exposed group (p = 0.001) compared to the Con group. Treatment with Zf, Thym, and their combination resulted in a significant reduction in the level of CK-MB biomarker versus the Cyp-exposed group (p = 0.0002, 0.02, 0.0004, respectively) (Figure 4(a)). The reduction in the level of CK-MB in all treated groups was comparable to the Con group (p > 0.05). In Figure 4(b) serum LDH level was non-significantly elevated (p > 0.05) compared to the Con group. Additionally, no significant difference in LDH levels was observed between the treated groups and the Cyp-exposed group (p > 0.05). (a and b). Effect of Zf and Thym on the serum level of (a) CK-MB and (b) LDH. Results are presented as mean ± SEM (n = 6) and statistically analyzed using one-way ANOVA, confirmed by Tukey’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.59 and 0.14 for panels (a) and (b), respectively. Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). p < 0.05 was considered significant. *p < 0.02 when compared Cyp-treated group with Thym, **p < 0.001 when compare Cyp-treated group with Con, ***p = 0.0002 when compared Cyp-treated group Zf, ***p = 0.0004 when compared Cyp-treated group with Zf + Thym group. Cyp: Cylophosphamide, Zf: zofenopril, Thym: thymoquinone, CK-MB: creatine phosphokinase isoenzyme, LDH: lactate dehydrogenase.
Effect of zofenopril and thymoquinone on inflammation and oxidative stress induced by Cyp in rats
As shown in Figure 5(a), an apparent increase in NF-κB levels was observed in the Cyp-exposed group in heart tissue homogenate; however, this change was not statistically significant (p > 0.05), indicating that Cyp did not significantly affect NF-κB expression. Treatment with Thym reduced this inflammatory biomarker significantly (p = 0.003). Meanwhile, NF-κB in Zf and Zf + Thym treated groups was not changed. (a and b) Effect of Zf and Thym on (a) NF-κB and (b) TAC. Results are presented as mean ± SEM (n = 6) and statistically analyzed using one-way ANOVA confirmed by Bonferroni’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.4 and 0.5 for panels (a) and (b), respectively. Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). p < 0.05 was considered significant. In (a) **p = 0.003 when compared Cyp-treated group with Thym. In (b) *p = 0.04 when comparing the Cyp-treated group with con, ***p < 0.0002 when comparing the Cyp-treated group with the Zf + Thym group. Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone, ns: non-significant, NF-κB: nuclear factor kappa-B, TAC: total antioxidant capacity.
In Figure 5(b) TAC was significantly reduced in the Cyp-treated group compared to the Con group (p = 0.04). Treatment with Zf and Thym individually slightly restored antioxidant capacity in a non-significant manner; meanwhile, their combination significantly increased serum TAC level (p = 0.0002).
The effect of Zf and Thym on inflammatory hs-CRP is shown in Figure 6. A significant increase in the serum level of hs-CRP was observed in the Cyp-exposed group compared to the Con group (p = 0.015). Treatment with Zf resulted in a significant reduction in the level of hs-CRP compared with the Cyp-treated group (p = 0.02). No significant changes have been observed in hs-CRP in Thym and Zf+Thym groups. Effect of Zf and Thym on hs-CRP. Values are presented as mean ± SEM (n = 6) and statistically analyzed using one-way ANOVA confirmed by Tukey’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.48. Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). p < 0.05 was considered significant. *p = 0.015 when comparing the Cyp-treated group with Con. While *p = 0.02 when comparing the Cyp-treated group with the Zf group. Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone, ns: non-significant, hs-CRP: high sensitivity C-reactive protein.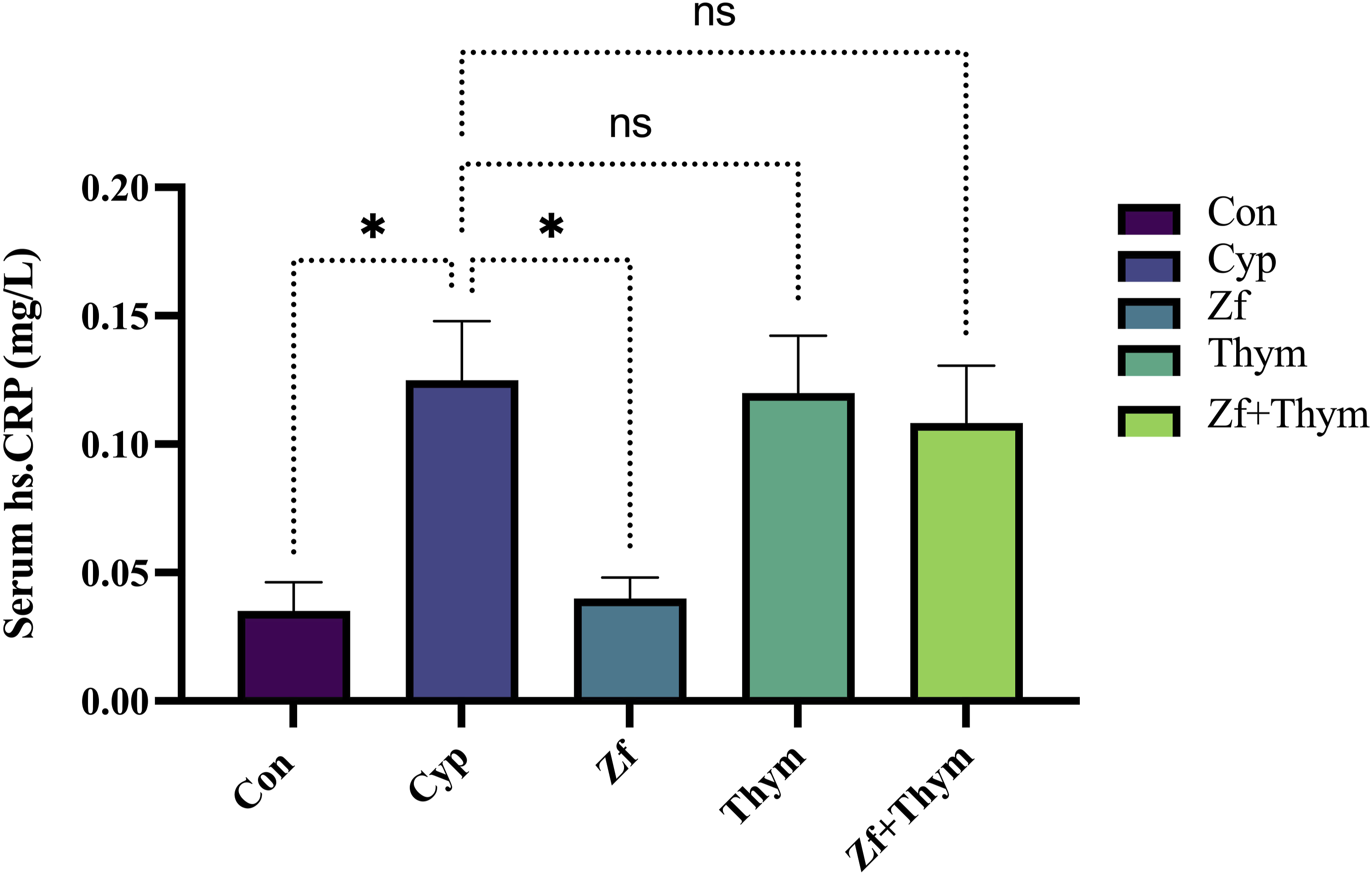
Effect of zofenopril and thymoquinone on apoptotic biomarker in Cyp-induced cardiotoxicity in rats
In the Cyp-exposed group, there was a significant increase in the serum level of apoptotic biomarker caspase-3 compared to the Con group (p = 0.01). Treatment with Zf, Thym, and their combinations resulted in a significant reduction in caspase-3 level (p = 0.001, 0.01, 0.001 respectively) in comparison to the Cyp-exposed group (Figure 7). Effect of Zf and Thym on serum caspase-3. Values are presented as mean ± SEM (n = 6) and statistically analyzed using one-way ANOVA confirmed by Bonferroni’s multiple comparison test. Eta-squared (η2) for ANOVA was calculated as a measure of the effect size with values of η2 = 0.49, Effect sizes were interpreted based on the following cutoff points: no effect (η2 < 0.01), minimal (0.01 ≤ η2 < 0.06), moderate (0.06 ≤ η2 < 0.14), and strong (η2 ≥ 0.14). p < 0.05 was considered significant. *p = 0.01 when comparing the Cyp-treated group with the Con and Thym groups. While **p = 0.001 when comparing the Cyp-treated group with Zf and Zf + Thym groups. Con: control, Cyp: cyclophosphamide, Zf: zofenopril, Thym: thymoquinone.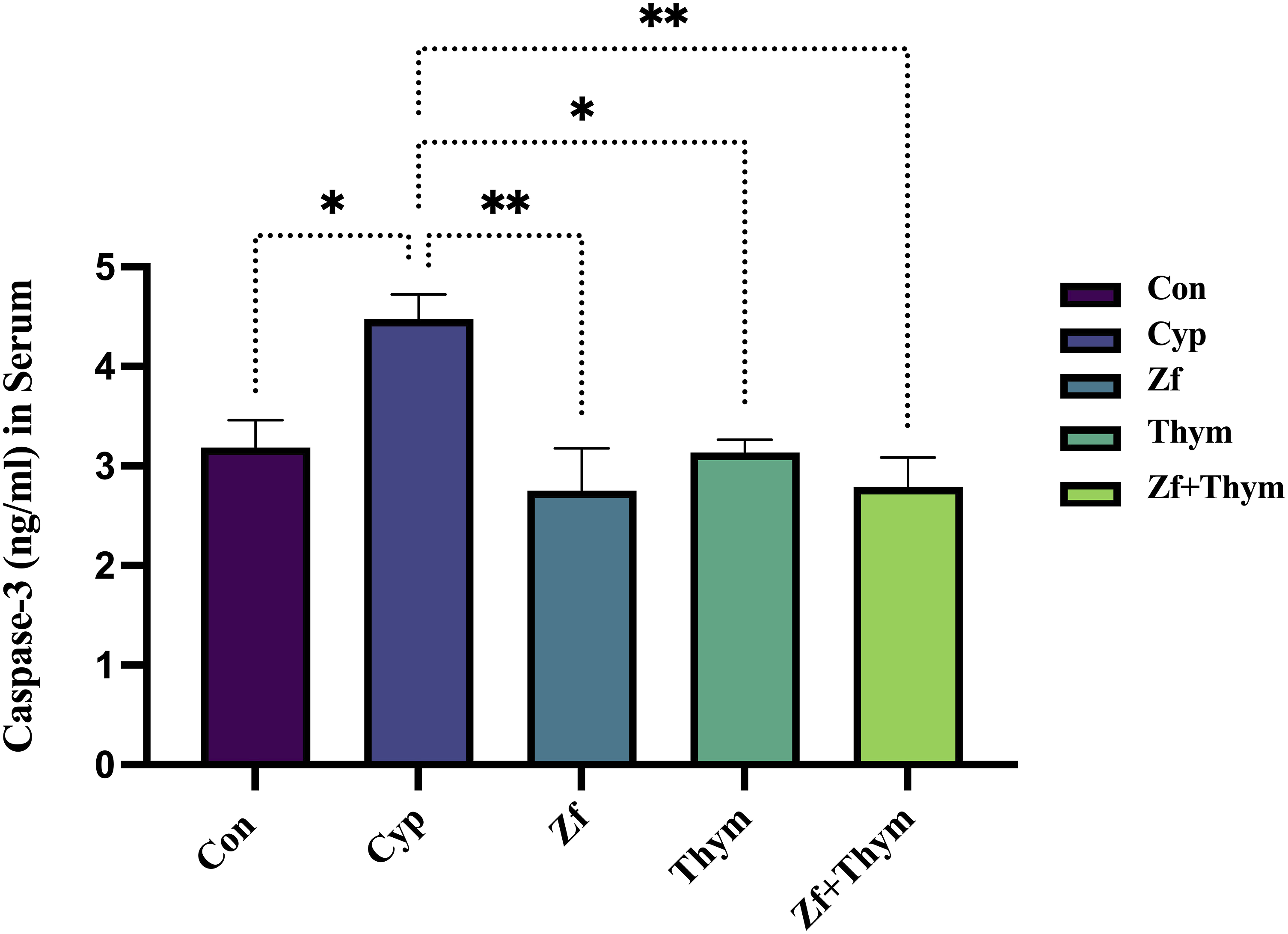
Histopathology findings
Semi-quantitative evaluation of heart histological sections.


Photomicrograph of the heart from groups; Con: exposes characteristically structured myocardial cells (MC), the section reveals some brunches of the coronary artery (BV) together with low-grade blood engorgement and areas of cardial mesenchymal connective tissue (CT). Cyp: shows significant infiltration of inflammatory cells (IC), mainly lymphocytes and macrophages within the cardiac stromal connective tissue. Presence of infiltrated adipocytes, fatty infiltration (FI) among the myocardial cells (MC), together with the presence of significant vascular congestion (yellow arrow). Zf: expressed a moderate grade of vascular congestion (VC) of some blood vessels (BV) in the given section. Myocardial cells (MC) reveal a distinctive association with their acidophilic cytoplasm. Thym: displayed a clear longitudinal section of vascular congestion (VC), together with the presence of inflammatory exudates and significant infiltration of inflammatory cells (yellow arrows) within the intestinal connective tissue, the section also reveals typical cross and longitudinal sections of myocardial cell (MC). Zf + Thym: shows low-grade vascular congestion (VC) within the cross-sectional blood vessels (BV). The section also reveals scattered infiltration of inflammatory cells (yellow arrow) within the interstitial connective tissue, with typical eosinophilic myocardial muscle cells (MC) arranged in a longitudinal manner. H&E. Scale bars: 4 mm. 100X. Con: Control; Cyp: Cyclophosphamide; Zf: Zofenopril; Thym: Thymoquinone; Zf + Thym: Zofenopril + Thymoquinone.
Discussion
Chemotherapy-induced cardiotoxicity poses a significant challenge to its clinical application. Cyclophosphamide is among the most effective chemotherapeutic agents used for various cancer types; however, its clinical use is severely restricted due to the risk of cardiotoxicity. Identifying novel protective agents to mitigate this harmful side effect while enhancing therapeutic efficacy is crucial.13,17 This experimental study aimed to investigate the potential cardioprotective effects of Zf and Thym individually and in combination against Cyp-induced cardiotoxicity, as well as to explore the underlying protective mechanisms. Exposure to Cyp is known to cause several pathological alterations in the heart, which are accompanied by elevated serum levels of cardiac enzymes such as CK-MB and LDH, alongside specific cardiac injury biomarkers like troponin T. These pathological changes in cardiac tissue have been previously documented in acute Cyp-induced cardiotoxicity models.31,32 The results from this study revealed that a single dose of Cyp administration induced cardiac cell damage as indicated by an increase in the CK-MB, hs-CRP, and a specific marker for cardiac damage; troponin T level, which has high specificity for cardiac injury and cardiomyocyte necrosis.33,34 These biomarkers are often correlated with histopathological alteration of cardiac tissue as depicted in previous studies.13,15
Excessive free radicals generated by Cyp toxicity cause significant damage to myocardial membranes, leading to the rapid leakage of these enzymes into the bloodstream and resulting in elevated serum levels, 31 accompanied by toxic effects on cardiac tissue, demonstrated by histopathological alterations as reported by an earlier study. 13 Treatment with Zf and Thym led to a non-significant reduction in serum cardiac injury biomarkers except CK-MB, and the addition of Thym did not produce a statistically significant additive effect. Although these changes were not statistically significant, they may indicate a modest cardioprotective effect. The slight effect observed with Zf and Thym could be attributed to the severity of Cyp-induced cardiac injury, as well as the limited dosage and treatment duration of Zf and Thym, which may have been inadequate to significantly reduce troponin T levels. In the present study, NF-κB has been evaluated to assess the extent of Cyp-induced myocardial inflammation. Although NF-κB levels were slightly elevated, the increase was not statistically significant, suggesting a relatively mild or early-stage inflammatory response in the heart. This contrasts with findings from previous studies, which reported significant NF-κB activation following Cyp administration.27,35 Myocardial inflammation is a well-established pathological consequence of Cyp-induced cardiotoxicity, and the NF-κB signaling pathway is a key regulator of inflammation and plays a pivotal role in the expression of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β.35,36 Its activation has been strongly associated with myocardial injury in response to cardiotoxic agents like Cyp. Therefore, while our findings showed a trend toward increased NF-κB expression, the absence of statistical significance may indicate a less severe inflammatory reaction in our experimental model, potentially influenced by factors such as dosage and duration of the treatment.
Notably, pretreatment with Thym exhibited a significant amelioration in NF-κB activation following Cyp administration. However, non-significant changes have been found with pre-treatment with Zf and its combination with Thym.
Furthermore, a significant reduction in the TAC parameter of the animals exposed to Cyp was observed in the present study, in line with previous studies highlighting the significant role of oxidative stress in mediating Cyp toxicity,11,37 leading to membrane lipid peroxidation, DNA damage, and cell death. 15 The excessive production of reactive oxygen species (ROS) can trigger various inflammatory mediators in cardiac tissue, resulting in direct DNA damage and the initiation of programmed cell death,38,39 as indicated by the elevated caspase-3 enzyme levels in this study. The interplay between inflammation and oxidative stress status contributes to severe cardiac inflammation observed in Cyp toxicity. 40
Although Zf treatment led to a slight, non-significant decrease in NF-κB levels and a modest elevation in TAC, these changes may indicate mild cardioprotective activity, potentially mediated through its antioxidant and anti-inflammatory properties. This observation is consistent with previous studies that reported the protective effects of Zf against Cyp-induced nephrotoxicity. 41 Additionally, captopril, an H2S-containing ACEI, has been shown to significantly reduce cardiac inflammation in spontaneously hypertensive rats by lowering pro-inflammatory cytokines such as IL-1β and IL-6 and increasing anti-inflammatory mediators like IL-10, primarily through inhibition of the NF-κB signaling pathway. Despite the relatively modest impact of Zf in our model, the comparable response pattern suggests a potential mechanistic overlap and highlights the need for further investigation.42,43
Our finding was also supported by the study of Bozacali et al., which demonstrated that Zf could protect both ischemia/reperfusion injury and doxorubicin-induced cardiotoxicity more effectively than enalapril and valsartan. In this study, the additive effect of Zf is explained by the presence of the SH-group, which is responsible for scavenging the ROS generated by doxorubicin. 24 On the other hand, Sacco et al. stated that Zf could protect against cardiotoxicity induced by doxorubicin without interfering with its chemotherapeutic effect. 44
Histopathological alteration of cardiac tissue induced by Cyp is associated with interstitial inflammatory cells infiltration, vascular congestion, and formation of inflammatory exudates, which is consistent with Cyp-cardiotoxicity demonstrated in the recent animal studies as multiple interstitial myocardial hemorrhages, multifocal myofibre necrosis, congestion, and degeneration of myocardial tissue.13,45
In the present study, the cellular and biochemical alterations accompanying the Cyp-cardiotoxicity are linked to the disturbance of lipid profile, generation of free radicals, inflammation, and apoptosis, this finding is consistent with the earlier study conducted by Ahmed et al. 17 who stated that Cyp administration causes a significant elevation in the levels of LDH, troponin-I, lipid peroxidation, triglycerides, IL-6, TNF-α, and caspase-3. Additionally, Cyp decreases antioxidant capacity as shown by a reduction in TAC level, which is attributed to an abnormal lipid profile. 46
In the current study, some of these abnormal biochemical changes induced by Cyp, such as cholesterol and TG, were restored by Zf and Thym. Moreover, we showed that Cyp administration decreased TAC level in comparison to the negative control group. A previous study showed that oxidative stress arises when the production of ROS surpasses the cellular antioxidant defense capacity. 47 Therefore, in the current era of combination therapies and prolonged survival for cancer patients, incorporating antioxidants to support cancer treatment should be considered, particularly as the interaction between plant-derived bioactive compounds and pharmaceutical drugs has emerged as an important research focus, given its potential to enhance, diminish, or have no extra effect on therapeutic outcome. 48
Use of herbal medicine and polyphenol compounds, including thymoquinone, 18 silibinin, 49 quercetin, 50 and resveratrol, 51 have been shown to exert pleiotropic activity in various preclinical and clinical studies. Several studies suggested the value of these antioxidants in synergizing the effects of other medications. In this study, Thym has been added as adjuvant therapy in the amelioration of Cyp’s adverse cardiac effects and improvement of lipid profile. It has been found that Zf attenuated the release of cardiotoxicity biomarkers, particularly CK-MB and hs-CRP, in a significant manner while mitigating troponin T and LDH non-significantly. Additionally, a combination of Zf + Thym produced a significant reduction (p < 0.05) in the percentage of infiltrated inflammatory cells in histological analysis, along with a notable decrease in the inflammatory exudate and vascular congestion in rats treated with this combination compared to the Cyp group, accompanied by a significant enhancement in TAC. Non-significant improvements of other biochemical markers or synergism were observed in the combined regimen of Zf and Thym. This observation may be explained by the two distinct biologically relevant activities of Thym, namely its antioxidant and prooxidant properties, as reported in earlier studies. 52 A research on antioxidant interactions shows that compounds with comparable activities may lead to additive or even sub-additive effects, particularly when they target the same oxidative signaling pathways. 53 Additionally, another peer-reviewed meta-analysis examined fixed-dose combination analgesics in acute pain and migraine, comparing observed outcomes against theoretical additive expectations, and it has been found that most combination therapies produced effects consistent with additive predictions, with no evidence of synergy. 54 Therefore, the lack of observed synergistic cardioprotection may be attributed to overlapping mechanisms of action between Zf and Thym, such as shared anti-inflammatory and antioxidant pathways, including suppression of NF-κB activation, reduction of oxidative stress, and modulation of apoptotic signaling. These overlaps could result in a pharmacodynamic ceiling effect, where maximal protective benefit is already achieved by monotherapy, limiting additional benefit from combination therapy. The cardioprotective effect of Thym individually has been reported in several previous studies, particularly against chemotherapy-induced cardiotoxicity. Research indicates that Thym supplementation effectively mitigates cardiac toxicity associated with cyclophosphamide, 19 methotrexate, 55 5-fluorouracil, 56 and doxorubicin. 20 Its protective mechanisms in these studies involve restoring antioxidant defenses, reducing oxidative stress, and modulating inflammatory pathways. It notably lowers serum cardiac biomarkers such as CK-MB, LDH, and aspartate aminotransferase (AST) while enhancing antioxidant enzymes, including catalase, superoxide dismutase, and glutathione peroxidase. Additionally, it suppresses pro-inflammatory cytokines such as TNF-α and IL-2. Histopathological analyses further confirmed that Thym administration prevents or alleviates chemotherapy-induced cardiac tissue damage. These findings support Thym’s potential as an adjuvant therapy to mitigate chemotherapy-related cardiotoxicity in our study.
The current study has certain limitations. One limitation of this study is that the selected dose was determined based on a previous study, thus eliminating dose-response assessment; consequently, the potential for a more precise effect at different dose levels remains unexplored. Conducting a full dose-response curve analysis would provide a clearer understanding of the optimal therapeutic dose and its corresponding mechanism of action. Second, the role of the sulfhydryl group in Zf was not assessed in exploring its mechanism of action. Additionally, the present study did not investigate the underlying molecular mechanisms, including mRNA and protein expression analyses (e.g., qPCR and Western blotting), or evaluations of signaling pathway activation and receptor expression, which are intended to be addressed in future studies. Despite some limitations, this study is, to the best of our knowledge, the first to examine the combined use of two agents with different mechanisms to reduce chemotherapy-induced cardiotoxicity. The novelty of this work lies in the dual assessment of Zf and Thym in a Cyp-induced cardiotoxicity model in rats, an approach not previously documented in the literature. Although the individual cardioprotective effects of each agent are supported by earlier research, our study contributes new evidence on their combined impact on key biomarkers associated with cardiac injury, inflammation, oxidative stress, and apoptosis. Additionally, the current in vivo results provide valuable preclinical evidence supporting the potential clinical application of combining Zf with Thym for cardioprotection against chemotherapy-induced cardiotoxicity.
Conclusion
Zofenopril and thymoquinone provided cardioprotection against Cyp-induced cardiotoxicity through distinct mechanisms. Zf exhibited anti-inflammatory effects, evidenced by a significant reduction in hs-CRP, along with anti-apoptotic activity. Thym significantly suppressed NF-κB expression, indicating its anti-inflammatory potential. Their combination enhanced antioxidant capacity, however, they did not show superior efficacy over individual treatments concerning their other actions. Furthermore, histopathological analysis confirms that Zf and Thym administration prevent Cyp-induced cardiac tissue damage as they improved histopathological outcomes by amelioration of lesion severity, as reflected by decreased number of inflammatory cells, reduction of area of exudate infiltration, and blood vessels congestion in the cardiac tissues, which supported the biochemical results. The findings of this study collectively indicate that the protective effect of Zf and Thym versus Cyp-induced cardiotoxicity may be linked to their antioxidant and anti-inflammatory properties. Further research is required to elucidate the underlying mechanisms, to establish clinical significance, and to develop guidelines for safe co-administration. The findings of the current study suggest that incorporating safe, plant-derived agents like Thym could further enhance therapeutic outcomes. Nonetheless, interpretation for clinical use should be approached with caution due to limitations including interspecies variability, reliance on a single-dose regimen, and the lack of chronic treatment assessment. Further clinical research is needed to validate these effects and establish optimal dosing strategies for patients receiving cardiotoxic chemotherapy.
Footnotes
Acknowledgments
The authors gratefully thank the support of the College of Pharmacy-University of Sulaimani for providing the necessary facilities and technical support from the animal house for conducting this study.
Ethical considerations
The approval of the Ethics and Research Registration Committee of the College of Pharmacy-University of Sulaimani was obtained for the protocol of this study with a Registration number (PH98-23 on 24.05.2023). All the procedures were performed following the Principles of Laboratory Animal Care. 22 All the authors complied with ARRIVE guidelines.
Author contributions
SKA: Execution of experimental work, biochemical analyses, interpretation of data, and documentation of results. BMR: Execution of experimental work, biochemical analyses, and interpretation of data. KHS: Execution of experimental work, interpretation of data, and documentation of results. NNM: Execution of experimental work, biochemical analyses, and interpretation of data. BHM: Design and execution of experimental work, writing the manuscript, statistical analysis, interpretation of results, and documentation of findings. HHO: Execution of experimental work, conducting histological analyses, and interpretation of results. All Authors read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All the data that support the findings of this study are included in the article.
