Abstract
Introduction
Adipose-derived mesenchymal stem cells (ADSCs) are promising candidates for regenerative therapies, but their clinical application is limited by cellular aging. This study investigated the effects of hydrogen on ADSC senescence and myogenic differentiation, along with the underlying molecular mechanisms.
Methods
ADSCs were treated with hydrogen gas. Senescence was assessed using β-galactosidase staining, proliferation assays, measurements of mitochondrial oxidative stress, and protein expression analysis. Differentiation capacity was evaluated through MyHC immunofluorescence, MYOD expression profiling, and quantification of myogenic regulatory factors. Additionally, the key molecular pathway of hydrogen’s action was investigated by pharmacologically inhibiting PRDX6.
Results
The findings showed that hydrogen treatment reduced senescence and increased differentiation capacity, as evidenced by higher proportions of MyHC-positive cells, increased myogenin levels, and decreased Muscle RING finger protein1 (MuRF1) expression. Molecular investigations revealed activation of the PRDX6/SIRT1/PGC-1α axis, accompanied by elevated NQO-1 expression. Importantly, pharmacological inhibition of PRDX6 largely eliminated the protective effects of hydrogen on cellular aging, disrupted differentiation, and caused mitochondrial dysfunction.
Discussion
These results suggest that hydrogen can regulate ADSC behavior via PRDX6-driven activation of SIRT1/PGC-1α signaling, offering potential approaches to improve stem cell quality for regenerative medicine.
Introduction
Musculoskeletal injuries remain a significant concern for both athletes and the general population, often leading to impaired mobility and reduced quality of life. Recent advancements in medical research have increasingly turned toward cell-based therapies, with stem cells emerging as a promising option for managing muscular trauma. These undifferentiated cells possess strong regenerative potential due to their ability to differentiate into multiple lineages, providing new possibilities for expediting tissue repair and supporting full functional restoration of damaged muscle tissue. 1
Adipose-derived stem cells (ADSCs), a type of multipotent mesenchymal stromal cell, can be readily isolated from adipose tissue. These progenitor cells exhibit the capacity to differentiate into various lineages, including adipocytes, osteoblasts, chondrocytes, and myocytes. Due to their ease of acquisition and differentiation potential, ADSCs have been widely recognized for their therapeutic relevance in regenerative medicine, especially in soft tissue repair, musculoskeletal regeneration, and the treatment of muscle degeneration.2,3 Inducing efficient myogenic differentiation in ADSCs is essential for their application in musculoskeletal recovery. However, like other stem cell types, ADSCs are vulnerable to senescence during extended in vitro culture and natural aging in vivo. Senescent ADSCs exhibit reduced proliferation, impaired differentiation, and increased secretion of inflammatory cytokines, all of which hinder their clinical utility. 4
Hydrogen, a colorless and odorless gas known for its non-toxic profile, has attracted attention as a novel therapeutic agent. Studies have demonstrated its antioxidant, anti-inflammatory, and anti-apoptotic properties. Evidence suggests that hydrogen improves mitochondrial function, reduces oxidative stress, and slows cellular aging.5–7 However, its specific effects on ADSC senescence and myogenic differentiation, as well as the molecular pathways involved, remain poorly defined.
Peroxiredoxin 6 (PRDX6), a key member of the peroxiredoxin family, plays an essential role in regulating redox homeostasis and related signaling pathways. Recent findings suggest that PRDX6 significantly influences cellular changes associated with aging.8,9 Our team’s proteomic analysis revealed a pronounced decrease in PRDX6 levels in human skeletal muscle affected by advanced age-related atrophy. This study was designed to investigate PRDX6’s involvement in mediating the effects of hydrogen on ADSC aging and differentiation by selectively suppressing PRDX6 expression.
Methods
Cell culture
The stem cells used in this study were derived from the same lineage described in our previous publication. ADSCs were obtained from the adipose tissue of five male Sprague-Dawley rats that were 2 weeks old, and their identity as stem cells, including surface markers and multilineage differentiation capacity, was confirmed as previously reported. 10 Cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2. The culture medium was refreshed every 48–72 h until the cells reached 80%–90% confluence. Cells from passages three to five were selected for all experiments.
Cell viability assay
ADSCs were seeded in 96-well plates at a density of 5 × 103 cells per well and allowed to adhere for 24 h before treatment. The CCK-8 solution was prepared by diluting the reagent in serum-free medium at a 1:10 ratio. After washing with PBS, 100 μL of the CCK-8 mixture was added to each well, followed by incubation at 37°C for 2 h. Absorbance was measured at 450 nm using a microplate reader. Each experiment was performed in at least three independent biological replicates.
Experimental groups
ADSCs displayed notable differences in terms of cell viability and growth. On day 1, no statistically significant differences were detected across the four experimental groups (Control, 20% H2, 50% H2, and 70% H2) (p > 0.05). However, by days 2 and 3, the 70% H2 group showed significantly enhanced proliferation when compared to the remaining groups (p < 0.01). Given these results, further investigations were carried out using a hydrogen concentration of 70%. ADSCs were randomly divided into five groups: an untreated control group; a group treated with D-Galactose (D-G) alone and non-targeting siRNA; a group receiving combined treatment with 70% H2, D-G and non-targeting siRNA; a PRDX6-silenced group transfected with siRNA targeting PRDX6 and D-G exposure; and a group receiving combined treatment with 70% H2, D-G and siPRDX6.
siRNA transfection
ADSCs were plated in 6-well plates at a density of 5 × 104 cells per well. Once cells reached 50%–60% confluence, they were transfected with siRNA against PRDX6 (siPRDX6) or non-targeting control siRNA (siNC) according to the manufacturer’s protocol.
Senescence-associated β-galactosidase staining
ADSCs from each group were seeded into 6-well plates at an initial density of 5 × 104 cells per well. After 72 h, cells were fixed with 4% paraformaldehyde for 15 min at room temperature. Following three washes with phosphate-buffered saline (PBS), the cells were incubated with senescence-associated β-galactosidase staining solution (Beyotime) at 37°C in a CO2-free chamber for 12–16 h. Staining was assessed using bright-field microscopy.
LDH release assay
ADSCs were seeded in 96-well plates at a density of 5 × 103 cells per well and incubated for 24 h to allow for adhesion prior to treatment. Extracellular LDH levels in the culture supernatant were quantified using the LDH Release Assay Kit (Beyotime), following the manufacturer’s instructions for reagent preparation and colorimetric measurement procedures.
Myogenic differentiation induction and detection
Myogenic differentiation was induced in ADSCs from all five experimental groups using 5-azacytidine. After a 7-days induction period, cells were fixed with 4% paraformaldehyde for 15 min at room temperature. Immunofluorescence staining was performed to detect MHC and MYOD expression. Cells were incubated sequentially with primary antibodies (rabbit anti-MHC and anti-MYOD), followed by DyLight 594-conjugated goat anti-rabbit IgG secondary antibodies. Nuclei were counterstained with DAPI.
Expression levels of MYOG and MuRF1 were assessed by immunoblotting. Cells were lysed in RIPA buffer supplemented with protease and phosphatase inhibitors. Protein concentrations were determined using the BCA assay. Equal amounts of protein (40 μg) were loaded onto 10% polyacrylamide gels, separated by electrophoresis, and transferred to PVDF membranes. Membranes were blocked at room temperature for 60 min in 5% skim milk in TBST and then incubated overnight at 4°C with primary antibodies. After multiple washes with TBST, membranes were incubated with horseradish peroxidase-conjugated secondary antibodies for 60 min. Protein bands were detected using enhanced chemiluminescence reagents and quantified by densitometry with ImageJ. We confirm that the WB images utilized in our manuscript are authentic and unaltered, with no splicing or nonlinear image adjustments applied. Any brightness/contrast modifications were uniformly applied across the entirety of each image.
Western blot analysis of P53, P21, PRDX6, SIRT1, and PGC-1α
Western blotting was also used to evaluate the expression of P53, P21, PRDX6, SIRT1, PGC-1α, and NQO-1. The procedures mirrored those used for myogenic marker analysis, except for the use of the following primary antibodies: mouse anti-P53, rabbit anti-P21, rabbit anti-PRDX6, mouse anti-SIRT1, mouse anti-PGC-1α, and mouse anti-NQO-1.
Determination of ATP content
Cellular ATP levels were measured using a commercially available detection kit (Beyotime), following the manufacturer’s protocol. Bioluminescence was recorded using a luminometer. ATP values were normalized to total protein content for each sample.
Statistical analysis
All data are presented as mean ± standard deviation. Comparisons between groups were performed using one-way analysis of variance, followed by Tukey’s honest significant difference test for post hoc analysis, conducted in GraphPad Prism (version 9.0). A significance threshold of p < 0.05 was applied to all statistical tests. Each experiment was performed at least three times.
Results
Hydrogen therapy attenuates cellular aging in ADSCs
Cell viability assays showed that hydrogen treatment effectively reversed the decline in survival caused by DG exposure in ADSCs (Figure 1(a)). SA-β-Gal staining further indicated that hydrogen reduced cellular aging in DG-treated ADSCs. Western blot analysis revealed lower levels of P53 and P21 in cells receiving hydrogen compared to those treated with DG alone (Figure 1(b), (c)). Additionally, hydrogen significantly reduced marker of mitochondrial oxidative stress relative to DG-only conditions (Figure 1(c)). H2 exhibits the potential to reverse the aging process of DG-induced ADSCs. (a) Cell viability of DG-treated ADSCs. (b) Expression of key proteins in cellular senescence, including P53 and P21. (c) Cellular senescence was assessed using the SA-β-gal staining method; mitochondrial ROS content was determined by MitoSOX Red; immunostaining of P21 was performed in ADSCs. Data are expressed as mean ± standard deviation (SD), the experiment independently repeated three times (n = 3). *p < 0.05, vehicle versus DG; #p < 0.05, DG versus DG + H2, and one-way analysis of variance was employed to compare differences across multiple groups.
Hydrogen therapy enhances myogenic differentiation potential in ADSCs
Immunofluorescence detection of MHC showed increased MHC expression in the hydrogen-treated group compared to the DG-treated group (Figure 2(a)). Western blot results confirmed higher MYOG protein levels and a significant decrease in MuRF1 expression in the hydrogen group (Figure 2(b)). Together, these results indicate that hydrogen promotes myogenic differentiation in ADSCs. H2 exhibits the potential to reverse the decline in the myogenic differentiation ability of ADSCs induced by DG. (a) Immunostaining of MHC in ADSCs. (b) Expression of key proteins in myogenic differentiation, including MYOG and MuRF1. Data are expressed as mean ± standard deviation (SD), the experiment independently repeated three times (n = 3). *p < 0.05, vehicle versus DG; #p < 0.05, DG versus DG + H2, and one-way analysis of variance was employed to compare differences across multiple groups.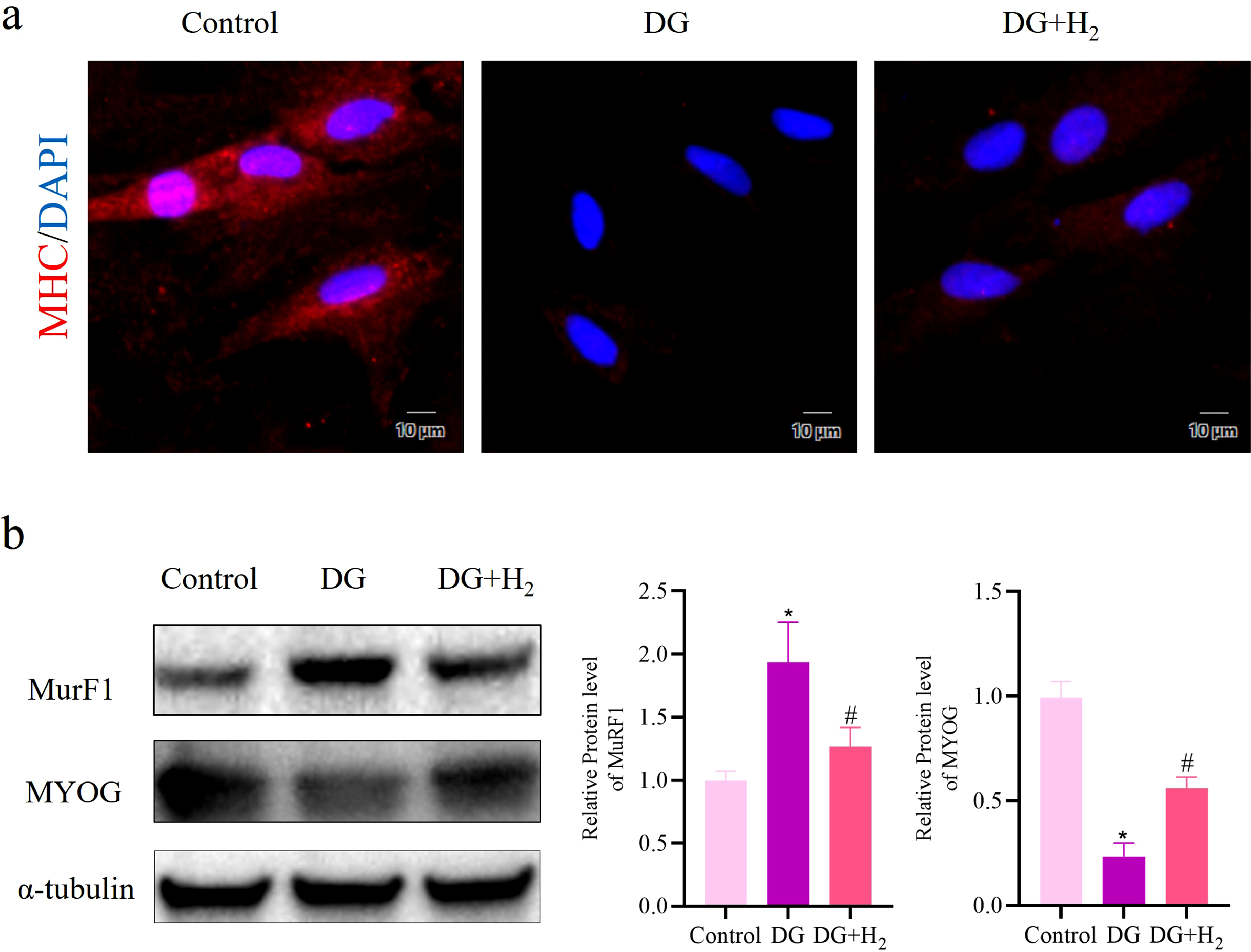
Inhibition of PRDX6 significantly attenuates the hydrogen-mediated benefits on senescence and myogenic differentiation of ADSCs and triggers mitochondrial dysfunction.
Western blot analysis showed that the expression level of PRDX6 was reduced by approximately 69% after knockdown treatment, as shown in Figure 3(a). The expression of P53 and P21 was more strongly restored in the siPRDX6 + DG group, reversing the hydrogen-induced suppression of these proteins, with statistical significance (p < 0.05, Figure 3(a)). In addition, LDH release assays and 8-OH-dG quantification demonstrated that PRDX6 silencing combined with DG treatment increased oxidative damage and weakened the protective effect of hydrogen, as reflected in comparative analysis (p < 0.05, Figure 3(b), (c)). H2 exhibits the potential to reverse the aging process of DG-induced ADSCs by targeting PRDX6. (a) Expression of key proteins in cellular senescence, including P53 and P21. (b) LDH level in DG-treated ADSCs. (c) Cellular levels of 8-OH-dG in mtDNA from DG-treated ADSCs. Data are expressed as mean ± standard deviation (SD), the experiment independently repeated three times (n = 3). *p < 0.05, Control versus DG + siNC; #p < 0.05, DG + siNC versus DG+siNC+H2; **p < 0.05, DG + siNC versus DG + siPRDX6; ##p < 0.05, DG+siNC+H2 versus DG+siPRDX6+H2, and one-way analysis of variance was employed to compare differences across multiple groups.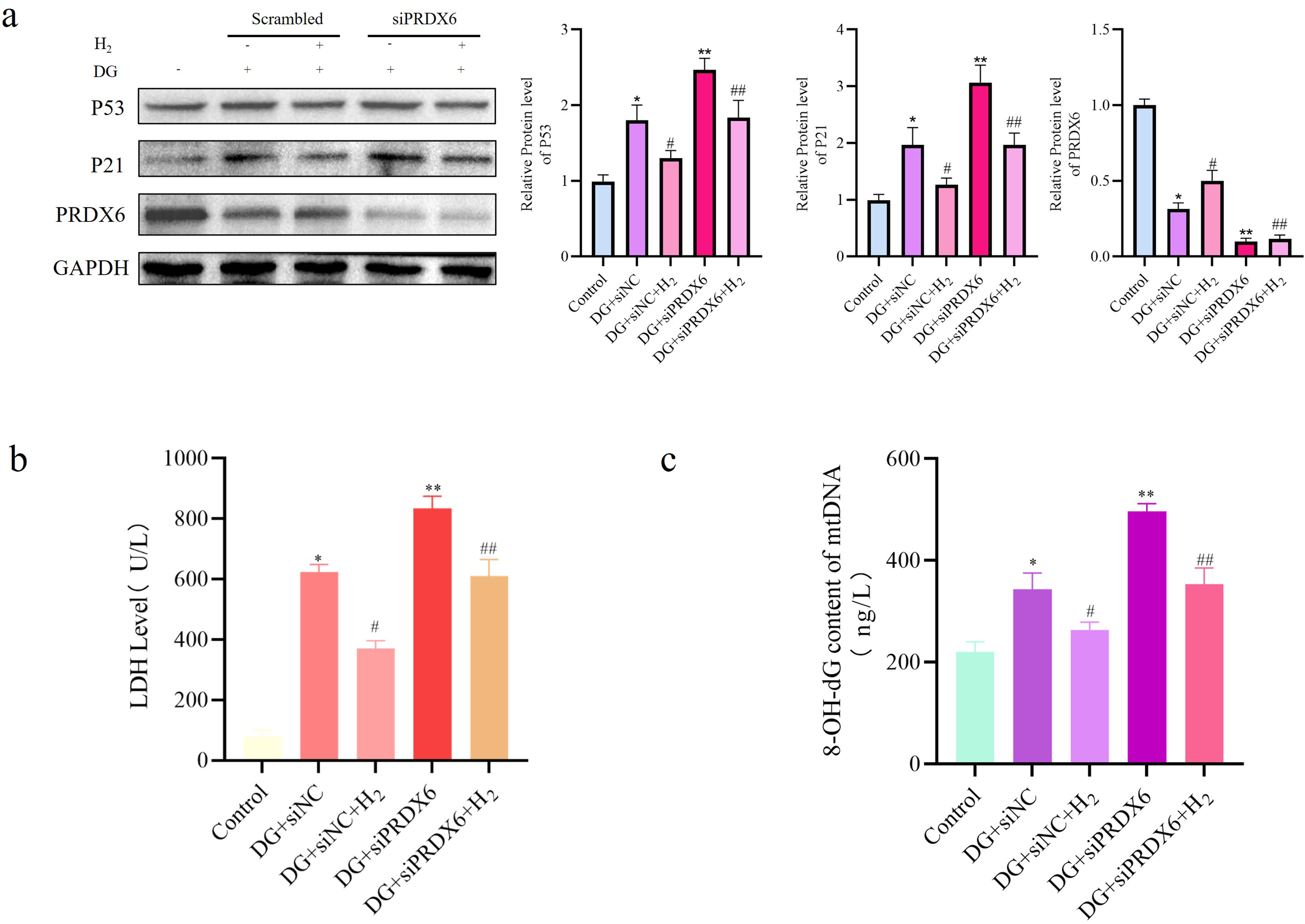
Regarding myogenic differentiation, western blot results showed further decreases in MYOG levels and increased MuRF1 expression in the siPRDX6 + DG group, reversing the changes induced by hydrogen (p < 0.05, Figure 4(a)). Immunofluorescence staining revealed a clearer reduction in MYOD expression in the siPRDX6 + DG group, effectively negating hydrogen’s stimulatory effects on MYOD (Figure 4(b)). These results suggest that PRDX6 knockdown largely eliminates the beneficial impact of hydrogen on both senescence and myogenic differentiation in ADSCs. H2 has the potential to reverse the decline in myogenic differentiation ability of adipose-derived stem cells caused by DG by targeting PRDX6. (a) Expression of key proteins in myogenic differentiation, including MYOG and MuRF1. (b) Immunostaining of MYOD in ADSCs. Data are expressed as mean ± standard deviation (SD), the experiment independently repeated three times (n = 3). *p < 0.05, Control versus DG + siNC; #p < 0.05, DG + siNC versus DG+siNC+H2; **p < 0.05, DG + siNC versus DG + siPRDX6; ##p < 0.05, DG+siNC+H2 versus DG+siPRDX6+H2, and one-way analysis of variance was employed to compare differences across multiple groups.
Western blotting also indicated that mitochondrial regulatory proteins PGC1α, SIRT1, MFN1, and NQO-1 were further downregulated in the siPRDX6 + DG group, effectively reversing the increases seen in the hydrogen-treated condition (p < 0.05, Figure 5(a)). Supporting results from mitochondrial fluorescence staining and ATP content measurements showed that PRDX6 knockdown intensified mitochondrial dysfunction under DG exposure and mostly eliminated hydrogen’s protective effect on mitochondrial stability (p < 0.05, Figure 5(b), (c)). These findings indicate that PRDX6 downregulation worsens mitochondrial decline during cellular aging. H2 reverses mitochondrial dysfunction induced by DG via targeting PRDX6. (a) Expression of key proteins involved in the preservation of normal mitochondrial function, including PGC-1α, SIRT1, MFN1, and NQO-1. (b) Mitochondrial content in DG-treated scrambled control and si-PRDX6 cells, as assessed by MitoTracker Green staining. (c) ATP concentration in DG-treated scrambled control and si-PRDX6 cells. Data are expressed as mean ± standard deviation (SD), the experiment independently repeated three times (n = 3). *p < 0.05, Control versus DG + siNC; #p < 0.05, DG + siNC versus DG+siNC+H2; **p < 0.05, DG + siNC versus DG + siPRDX6; ##p < 0.05, DG+siNC+H2 versus DG+siPRDX6+H2, and one-way analysis of variance was employed to compare differences across multiple groups.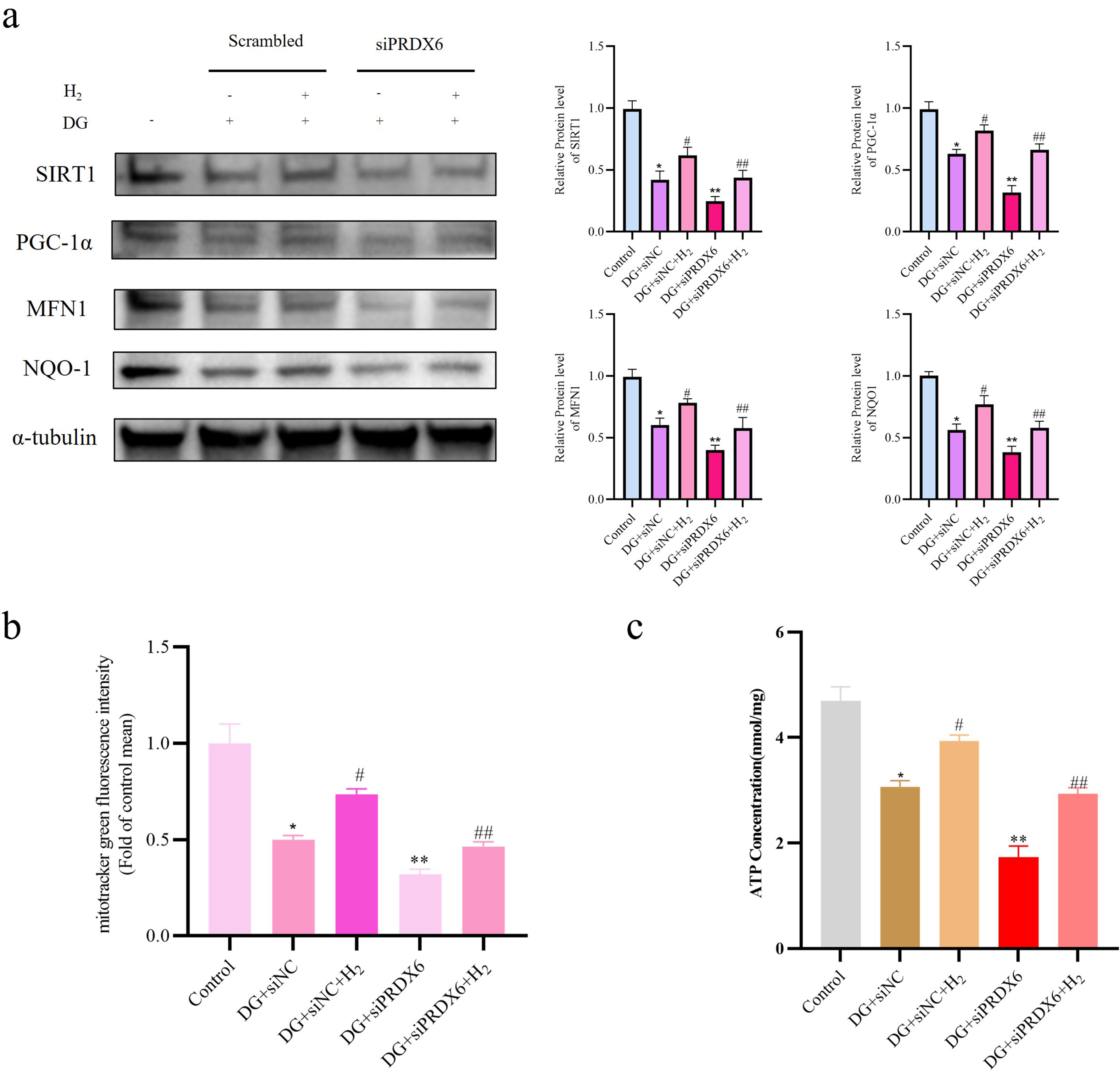
Discussion
Our study demonstrated that hydrogen treatment effectively slows aging in ADSCs, extends their proliferative capacity, and improves their ability to undergo myogenic differentiation. The intervention simultaneously decelerates cellular aging mechanisms in these stem cells, potentially prolonging their functional longevity, while substantially boosts their capacity for specialized muscle tissue development. This improvement in differentiation is particularly relevant for clinical applications, especially in the context of cell-based repair strategies for neuromuscular disorders. These findings contribute to the development of new therapeutic approaches in muscle regeneration. Previously, we reported that hydrogen reduces the cytotoxic effects of 5-azacytidine and synergistically improves ADSC myogenic differentiation through the p38 mitogen-activated protein kinase pathway. In the current study, we further demonstrate that hydrogen delays ADSC aging by activating the SIRT1/PGC-1α axis via PRDX6, thereby extending their proliferative potential and enhancing differentiation capacity. Unlike our earlier work, this study focuses more specifically on identifying the molecular targets through which hydrogen affects stem cell function and myogenesis.
To evaluate hydrogen’s anti-aging effects, we performed SA-β-Gal staining, which showed a significant reduction in senescent cell counts in the hydrogen-treated group compared to controls, indicating a reversal of aging characteristics in ADSCs. Western blot analysis further confirmed reduced expression of P53 and P21 following hydrogen exposure. 11 Given the known role of mitochondrial reactive oxygen species in promoting cellular aging, 12 our findings showed that hydrogen effectively suppressed mitochondrial ROS accumulation. Taken together, these results show that hydrogen restores ADSC vitality and supports cell proliferation through multiple molecular pathways.
In examining myogenic differentiation, we quantified key markers including MHC, MYOG, and MuRF1, which are central to muscle lineage commitment. 13 The hydrogen-treated group showed a 24% increase in MHC-positive cells. Western blot analysis revealed higher MYOG expression and decreased MuRF1 levels. These combined findings demonstrate that hydrogen enhances the myogenic differentiation potential of ADSCs through specific molecular mechanisms.
Through mechanistic investigation, we conducted detailed analyses of the signaling pathways involved. By combining gene silencing, immunoblotting, and fluorescence-based imaging techniques, our results showed that hydrogen activates the PRDX6/SIRT1/PGC-1α signaling cascade in ADSCs. As a key antioxidant enzyme, PRDX6 plays an essential role in reducing reactive oxygen species and protecting cells from oxidative injury [9]. SIRT1, a NAD+-dependent deacetylase, regulates multiple cellular processes including metabolism, stress response, and proliferation through its deacetylation of various substrates. 14 PGC-1α functions as a central transcriptional regulator of mitochondrial biogenesis and activity. It supports ATP production by driving the expression of nuclear genes involved in mitochondrial oxidative phosphorylation and mitochondrial genome maintenance.15,16 This signaling axis exerts widespread protective effects on ADSC function. PRDX6 reduces ROS levels, restoring redox balance and creating favorable conditions for SIRT1 activity, which in turn delays senescence and supports metabolic regulation. Simultaneously, PGC-1α activation contributes to mitochondrial expansion and improved energy production, necessary for sustained cell growth and lineage commitment. Immunoblotting confirmed increased expression of PRDX6, SIRT1, and PGC-1α in hydrogen-treated samples, consistent with these functional observations.
To determine whether PRDX6 is essential for hydrogen’s beneficial effects, we used siRNA to silence PRDX6 in ADSCs. Loss of PRDX6 largely eliminates hydrogen’s effects on reducing senescence and enhancing myogenic differentiation. This was accompanied by increased levels of damage-associated markers LDH and 8-OH-dG,17,18 as well as a drop in MYOD-positive cells. At the molecular level, MYOG expression declined and MuRF1 levels rose, reflecting impaired myogenic differentiation. Mitotracker green fluorescence and ATP detection outcomes further supported these results. These findings indicate that hydrogen initiates a cascade by targeting PRDX6, which then reduces ROS levels and activates SIRT1. SIRT1 subsequently regulates PGC-1α, leading to reduced senescence and improved myogenic differentiation in ADSCs.
Limitations and future directions
1. Our current research lacks detailed functional validation of regenerative potential. In future studies, we aim to incorporate traction force microscopy to measure the contractile forces of engineered tissues, fusion index analysis to assess myoblast fusion into multinucleated myotubes, and in vivo engraftment models to evaluate therapeutic outcomes in regenerative settings. 2. The present study employed gene knockdown only. Future work should include rescue experiments and dose–response evaluations to reduce the risk of off-target effects and to better define translational relevance. 3. Profiling of ROS and mitochondrial stress was limited to MitoSOX Red staining and ATP quantification. To achieve a more complete assessment of mitochondrial status, future research should include measurements of mitochondrial membrane potential (e.g., JC-1 staining), other oxidative stress-related indicators (such as MDA, SOD and CAT, etc.),19,20 mitophagy markers, and mtDNA integrity. 4. While PRDX6 was identified as a mediator, its upstream regulators or interactions with parallel pathways were not investigated. Exploring potential links with other redox-sensitive factors such as Nrf2 or AMPK may provide additional mechanistic insight
21
and should be considered in subsequent studies.
In summary, this study shows that molecular hydrogen reduces ADSC senescence and promotes myogenic differentiation through activation of the PRDX6/SIRT1/PGC-1α signaling pathway. These findings hold promise for applying ADSCs in regenerative therapies, particularly for treating muscle-related conditions such as dystrophies and age-associated muscle loss. Hydrogen may improve both the viability and function of ADSCs, increasing the effectiveness of cell-based interventions. However, further investigations are needed to map the broader molecular framework between hydrogen signaling and PRDX6, and to explore interactions with additional regulatory networks in ADSC biology.
Footnotes
Ethical considerations
The ADSCs were isolated from the adipose tissue of rats, The animal experiments were performed under the approval of the Institutional Animal Care and Use Committee of Yangzhou University (Approval No. DWLL-202,112-201)]
Consent to participate
All authors have read and approved the final version of this manuscript.
Author Contributions
Z.P, F.WY, L. Y and W. JC contributed to the conception and design of the study. ZP, H.YM, L. WK performed the experiments. Z. P and L. Y analyzed the data. Z. P drafted the manuscript. All authors contributed to the manuscript writing and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Yangzhou Green Yang Golden Phoenix Plan: LYJF00044 and Jiangsu Province’s Double Innovation Plan: JSSCBS20230496.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
