Abstract
Diabetic retinopathy (DR) is a common diabetic complication that severely impacts the life quality of diabetic patients. Recently, cellular senescence in human retinal endothelial cells (HRECs) induced by high glucose has been linked to the pathogenesis of DR. Fluorometholone (FML) is a glucocorticoid drug applied in the treatment of inflammatory and allergic disorders of the eye. The objective of the present study is to investigate the protective function of FML on high glucose-induced cellular senescence in HRECs. The in vitro injury model was established by stimulating HRECs with 30 m
Introduction
Diabetic retinopathy (DR) is a clinical complication of injury on the retina induced by diabetes and it is one of the most common causes of blindness in adults. 1 As diabetes progresses, the probability to develop DR increases significantly, by over 54% approximately 10–20 years after the patients are diagnosed in type 2 diabetes. 2 Effective prevention can be achieved on the majority of DR patients by diagnosis and intervention at an early stage. 3 The inadequate understanding of the pathophysiology of DR, however, limits the therapy of DR. Recently, cellular senescence in human retinal endothelial cells induced by a high concentration of glucose was reported to be involved in the pathogenesis of DR. According to Menaka, miR-34a mediated cellular senescence in retinal endothelial cells contributes to retinal microvascular injury and DR. 4 The development of DR has been linked to the cellular senescence-induced vascular dysfunction, in both DR animal models.5–6
Multiple processes regulate cellular senescence, which is an important physiological process. Pro-inflammatory cytokines, including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-8 (IL-8), play a pivotal role in cellular senescence. Previous studies revealed that these cytokines could directly increase the activity of p53 and p21 signaling to promote cellular senescence 7 Activated Akt is an important signaling reported to be involved in the regulation of multiple cellular physiological processes, including cell senescence. In addition, it is reported that cell senescence induced by Angiotensin II is mediated by the inactivation of Akt. 8 p53 is a multifunctional transcriptional factor that plays a critical role in the progression of DNA injury and repairment, cell growth and differentiation, and cell cycle and apoptosis. 9 Recently, it has been reported that p53 impacts the progression of cell senescence by regulating the expression level of multiple downstream genes. In the p53 signaling pathway, p21 and Rb both play an important role in the progression of the cell cycle and ultimately regulate the development of cell senescence. 10 Therefore, targeting key factors in human retinal endothelial cells, such as Akt, p53, and p21, could be a viable treatment for clinical DR. Vascular endothelial growth factor (VEGF) is considered a major mediator of intraocular neovascularization, responsible for pathologic angiogenesis in the development of DR. 11 In patients with diabetes, persistent hyperglycemia leads to ischemic changes in the retina, ultimately stimulating the release of VEGF, thereby promoting neovascularization. 3 Tissue factor (TF) is widely expressed on the surface of various cells. Recently, it has been demonstrated that overexpression of TF results in increased transcription of VEGF, promoting pathologic angiogenesis in DR. 12 Thus, expression of VEGF mediated by TF has become an important target for the treatment of DR.
Fluorometholone (FML) belongs to the glucocorticoid drugs developed by Pfizer for the treatment of acute herpes simplex virus keratitis, fungal infections of the eye tissue, cowpox and chickenpox infections, viral corneal and conjunctival infections, and tuberculosis. 13 It can be used as anti-inflammatory and anti-allergic agent and also when the acute infections are well controlled by proper anti-infective agents first. A recent study revealed that FML significantly reduced the expressions of TNF-α, IL-6, and IL-1β at both gene and protein levels in LPS-challenged keratocyte cell models. 14 However, whether FML exerts protective benefits in DR is still unknown. In the present study, we aim to explore the potential protective effects of FML against DR by investigating the impacts of FML on the cell senescence in human retinal endothelial cells induced by high glucose.
Materials and methods
Cell culture and treatments
This study was approved by the Ethical Committee of Shandong University. HRECs (ACBRI-181) were purchased from Cell Systems, Inc. and cultured in the endothelial cell medium (Lonza, Switzerland) containing 5% FBS, 100 μg/mL penicillin, and 100 μg/mL streptomycin, which were cultured in the condition of 5% CO2 and 37°C in a humidified atmosphere.
15
Fluorometholone (FML) was dissolved in dimethyl sulfoxide (DMSO). Glucose was directly dissolved in a cell culture medium. To assess the effects of FML, cells were stimulated with 0.005%, 0.01%, 0.05%, 0.1%, 0.5%, and 1% FML for 24 h with DMSO as the vehicle group. To assess the protective effect of FML against high glucose, cells were co-stimulated with high glucose (30 m
Dihydroethidium staining
The level of intracellular reactive oxygen species (ROS) was determined with the DHE staining assay. Briefly, after necessary treatment, HRECs were loaded with 10 μ
Lactate dehydrogenase release assay
In brief, HRECs were seeded on a 96-well plate followed by different treatment strategies, incubated for 24 h. Subsequently, the collected supernatants were added with the CytoTox 96 Reagent (Promega, Wisconsin, USA), followed by incubation with the stop solution. Lastly, the percentage of LDH release was calculated with the following formula: LDH releases (%) = (experimental LDH release − spontaneous LDH release)/maximum LDH release.
Caspase-3 activity determination
The activity of caspase-3 in HRECs was measured using a colorimetric assay kit (#ab39401, Abcam, USA). Cells were washed and lysed, after the specified treatment. After centrifugation at 14,000×g, the supernatant was used for caspase-3 activity measurement following the manufacturer’s instructions. The assay was repeated 3 times.
Real-time polymerase chain reaction assay
The extracted total RNAs using RNAiso plus reagent (TAKARA, Tokyo, Japan) were used to synthesize cDNA with the Primescript RT reagent Kit (TAKARA, Tokyo, Japan) and added with specifically designed primers followed by RT-PCR amplification and detection with the ABI PRISM 7500 system (Applied Biosystems, MA, USA) using the SYBR Premix EX Taq. Lastly, the relative expression level of target genes was determined using the 2-△△t method with GAPDH for the normalization.
Enzyme-linked immunosorbent assay
The production of TNF-α, IL-6, IL-8, TF, and VEGF in human retinal endothelial cells was determined using the commercial kits (Elabscience, Wuhan, China). Briefly, the standards were diluted to five gradient concentrations and were added to a 96-well plate along with the supernatants collected from each cultural medium. After incubation for 30 min at 37°C, the medium was removed and the wells were washed using the washing solution. Subsequently, the wells were added with conjugate reagents followed by incubation for 30 min at 37°C. After washing, the tetramethyl benzidine (TMB) solution was added for coloration at 37°C for 15 min, followed by adding the stop solution to terminate the reaction. Lastly, the absorbance at 450 nm was measured using the microplate reader (Mindray, Shenzhen, China).
Western blot assay
Human retinal endothelial cells were cultured with lysis buffer to extract total proteins, which were further quantified using the bicinchoninic acid (BCA) kit (Sigma, Missouri, USA). The proteins were then loaded and separated with a 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), before being transferred to the polyvinylidene fluoride (PVDF) membrane (Sigma-Aldrich, Missouri, USA). The PVDF membranes were subsequently incubated with 5% skim milk and washed using the TBST buffer. Then, the primary antibodies against TF (1:1000, EarthOx, California, USA), VEGF (1:2000, EarthOx, California, USA), p53 (1:3000, EarthOx, California, USA), p21 (1:2000, EarthOx, California, USA), Rb (1:2000, EarthOx, California, USA), Akt (1:2000, EarthOx, California, USA), p-Akt (1:1000, EarthOx, California, USA), and tubulin (1:8000, EarthOx, California, USA) were used for incubation with the membranes, followed by changing the solution with the secondary antibody (1:1000, EarthOx, California, USA) at room temperature for 2 h. Lastly, the bands were exposed to the ECL solution (Sigma, Missouri, USA) and the relative expression level of target proteins was determined with Image J software.
SA-β-Gal staining assay
Briefly, the cell culture medium was removed and cells were washed using the PBS buffer, followed by fixation with 4% Paraformaldehyde Fix Solution for 10–15 min. After adding dyeing solution, dyeing supplements, and X-gal solution into each well, the cells were incubated at 37°C overnight. After removing the mixing solution, 70% glycerin was added. Lastly, the images were taken under the microscope (Keyence, Tokyo, Japan).
Telomerase activity measurement
The telomeric repeat application protocol (TRAP) assay was used for the measurement of telomerase activity using the TeloTAGGG Telomerase PCR ELISA PLUS kit (Roche, Mannheim, Germany). Briefly, the 3′-end of the biotin-labeled synthetic P1-TS-primer was combined with the telomeric repeats (TTAGGG), which were amplified using PCR together with the internal standard using the primer P1-TS and the anchor-primer P2. The products of the PCR were denatured, followed by hybridized to digoxigenin (DIG)-labeled detection probes specific for the telomeric repeats. Lastly, anti-DIG-HRP was used to detect the immobilized amplicons, followed by detecting the telomerase activity using PCR.
Statistical analysis
All the data were expressed with mean±SD and the data analysis was conducted using the GraphPad software. The Shapiro–Wilk test was used to test normal distribution and Levene’s test was used to test the homogeneity of variances.
The data between the two groups were analyzed using Student’s t-test and the data among groups were analyzed with the ANOVA method, followed by the Duncan’s multiple range test. p < 0.05 was considered a significant difference in the present study.
Results
Cytotoxicity of fluorometholone in human retinal endothelial cells
Firstly, the optimal concentration of FML was determined using the LDH release assay. Cells were stimulated with 0.005%, 0.01%, 0.05%, 0.1%, 0.5%, and 1% FML for 24 h, respectively. The molecular structure of FML was shown in Figure 1(a). Compared to the vehicle group, the release of LDH was maintained around 5% as the concentration of FML increased from 0.005% to 0.1%, which was significantly elevated to 11.4% and 15.6% by stimulation with 0.5% and 1% FML. Therefore, 0.05% and 0.1% were utilized as the incubation concentration of FML in the subsequent experiments. Cytotoxicity of fluorometholone in human retinal endothelial cells (HRECs): (A) Molecular structure of fluorometholone and (B) cells were stimulated with 0.005%, 0.01%, 0.05%, 0.1%, 0.5%, and 1% fluorometholone (FML) for 24 h. Cytotoxicity was measured with LDH release (†, ††, p < 0.05, 0.01 vs. vehicle, $, p < 0.05 vs. high glucose+0.05% FML).
Fluorometholone suppressed high glucose-induced generation of intracellular reactive oxygen species
As intracellular ROS are an important biomarker of oxidative stress, the effect of FML on their high glucose-induced generation was tested. As shown in Figure 2, high glucose treatment increased the generation of intracellular ROS to approximately 3.6-fold and later reduced to 2.5- and 1.7-fold by 0.05% and 0.1% FML, respectively. This finding indicates that FML can attenuate high glucose-induced oxidative stress in HRECs. FML suppressed high glucose-induced generation of intracellular ROS in HRECs. Cells were stimulated with high glucose (30 m
Fluorometholone inhibited high glucose-induced production of TNF-α, IL-6, and IL-8 in HRECs
To investigate the anti-inflammatory effect of FML in HRECs under hyperglycemia, cells were stimulated with high glucose (30 m Fluorometholone inhibited the high glucose-induced production of TNF-α, IL-6, and IL-8 in HRECs. Cells were stimulated with high glucose (30 m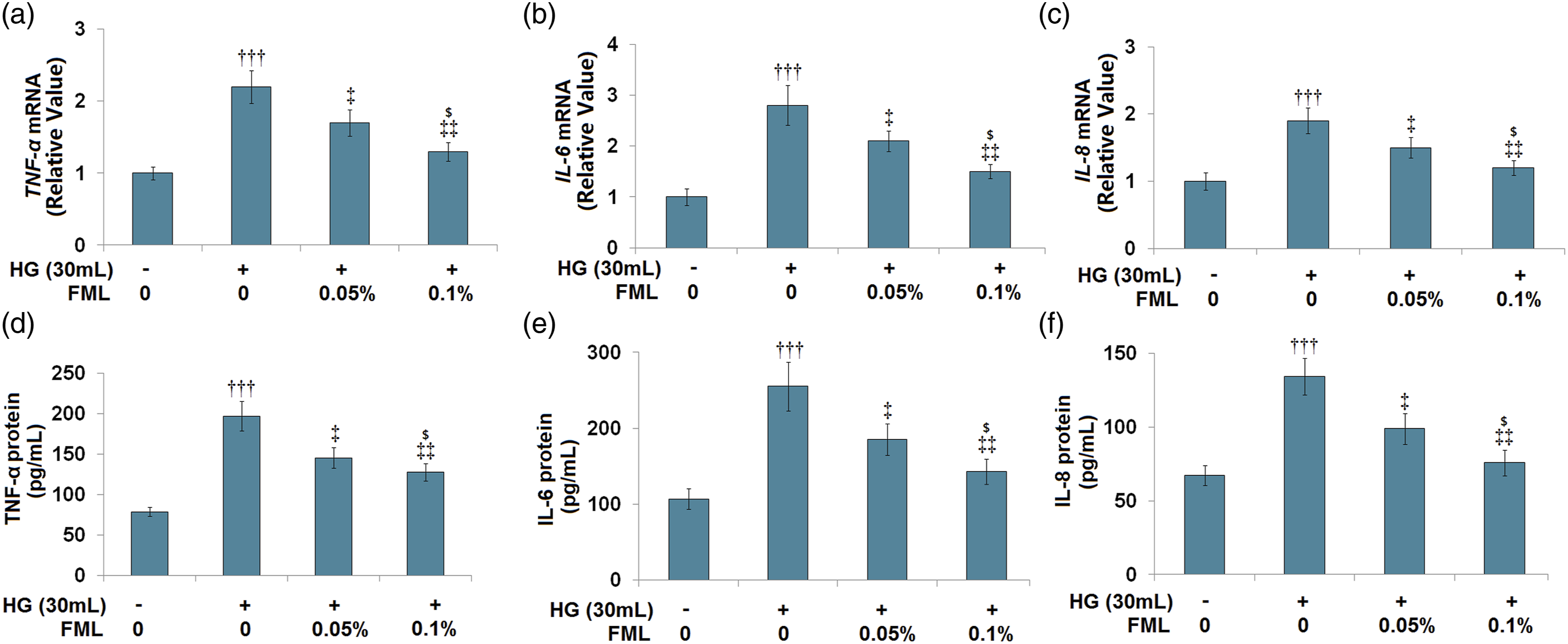
Fluorometholone (FML) inhibited high glucose-induced production of TF and the gene and protein expression levels of TF and VEGF in treated HRECs were then determined. As shown in Figures 4(a) and (b), the elevated expression levels of both TF and VEGF in high glucose-treated HRECs were significantly lowered by the introduction of FML in a dose-dependent manner. In addition, the secretions of TF (Figure 4(c)) in the vehicle, high glucose, 0.05%, and 0.1% FML groups were 56.5, 123.7, 92.8, and 71.6 pg/mL, respectively. As shown in Figure 3(d), compared to the vehicle group, the production of VEGF was significantly elevated from 83.7 pg/mL to 185.7 pg/mL and greatly suppressed to 142.1 pg/mL and 115.5 pg/mL by the introduction of 0.05% and 0.1% FML, respectively. These data indicate that the upregulation of TF and VEGF in HRECs induced by high glucose was dramatically reversed by FML. Fluorometholone inhibited the high glucose-induced production of TF and VEGF in HRECs. Cells were stimulated with high glucose (30 m
Fluorometholone ameliorated cellular senescence in high glucose-challenged human retinal endothelial cells
To evaluate the effects of FML on cellular senescence in high glucose-treated HRECs, cells were stimulated with high glucose (30 m Fluorometholone ameliorated cellular senescence in high glucose-challenged HRECs. Cells were stimulated with high glucose (30 m
Fluorometholone increased telomerase activity in high glucose-challenged human retinal endothelial cells
Declined telomerase activity was regarded as an important biomarker for cellular senescence. The level of cell senescence was then verified by measuring the telomerase activity. As shown in Figure 6, compared to the vehicle group, the telomerase activity was significantly decreased from 26.5 IU/L to 15.3 IU/L by stimulation with high glucose and greatly elevated to 20.5 IU/L and 24.2 IU/L by the introduction of 0.05% and 0.1% FML, respectively, verifying the promising protective effects of FML on HRECs from cellular senescence. Fluorometholone increased telomerase activity in high glucose-challenged HRECs. Cells were stimulated with high glucose (30 m
Fluorometholone reduced p53, p21, and Rb in high glucose-challenged HRECs
Subsequently, the senescence-related signaling was examined. Cells were stimulated with high glucose (30 m Fluorometholone reduced p53, p21, and Rb in high glucose-challenged HRECs: (A) Representative bands of p53, p21, and Rb; (B) quantification of p53 protein; (C) quantification of p21; and (D) quantification of Rb (†††, p<0.005 vs. vehicle, ‡‡, p < 0.01 vs. high glucose).
The protective effects of fluorometholone against high glucose-induced cellular senescence were mediated by Akt
To explore the potential mechanism underlying the effects of FML, cells were firstly stimulated with high glucose (30 m The protective effects of fluorometholone against high glucose-induced cellular senescence are mediated by Akt: (A) Cells were stimulated with high glucose (30 m
The protective effects of fluorometholone against high glucose-induced apoptosis
To further investigate the potential benefits of FML in high glucose-challenged HRECs, the activity of caspase-3, an important biomarker of cell apoptosis, was examined. Cells were stimulated with high glucose (30 m The protective effects of FML against the high glucose-induced increase in activity of caspase-3. HRECs were stimulated with high glucose (30 m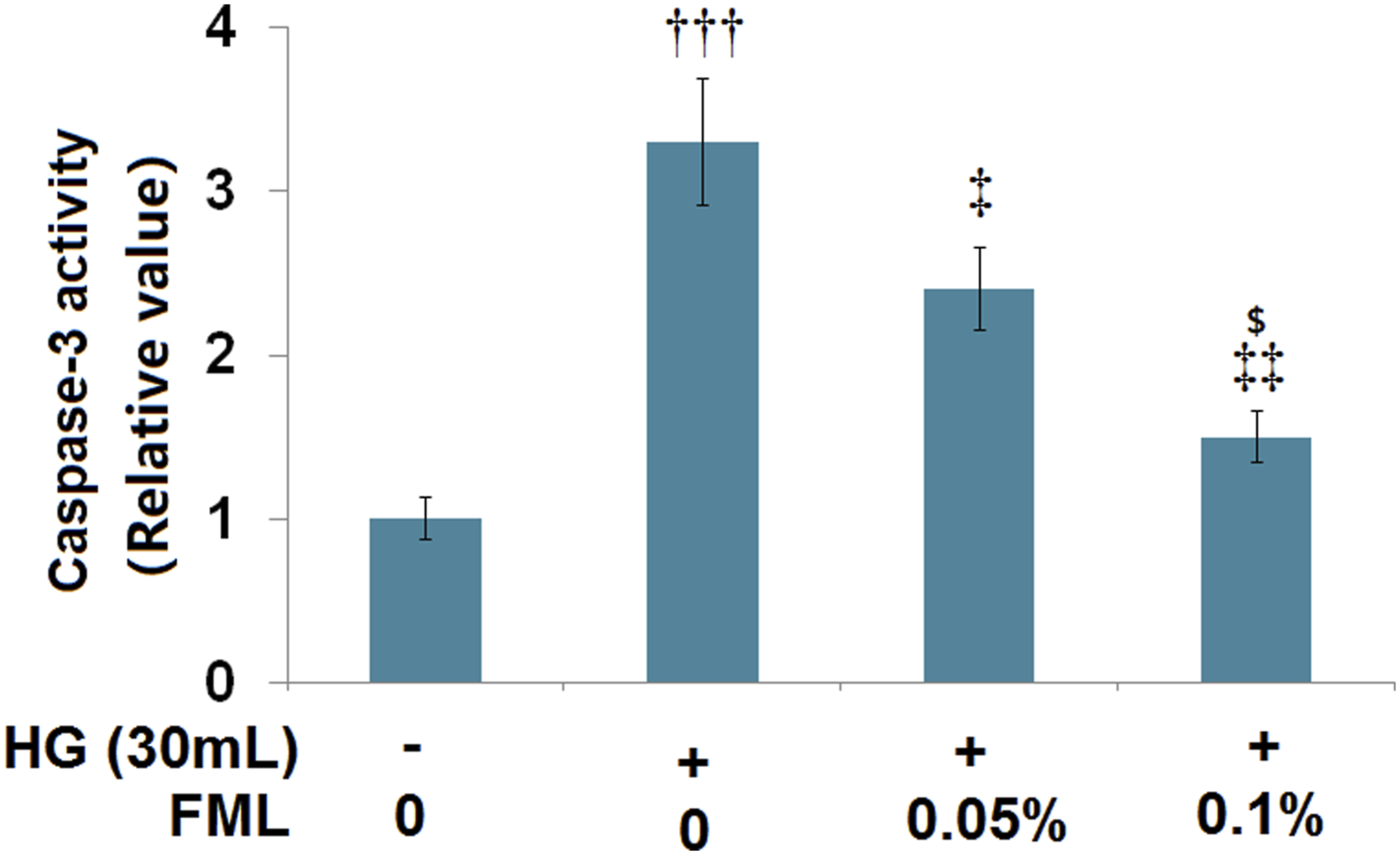
Discussion
The synthesis of diglyceride can be facilitated by hyperglycemia and diglyceride, which is the activator of all subunits of VEGF, and a potential vascular leakage factor mainly secreted by the vascular endothelial cells, pericytes, and retinal pigment epithelium cells.
17
The phosphorylation of tight junction proteins, such as ZO-1 and occludin, induced by the highly produced VEGF trigger structural changes, contributing to the damaged blood-retinal barrier, and retinopathy. The production of VEGF can be passively regulated by the tissue factor (TF),
18
which is regarded as an important inducer of cellular senescence
19
and apoptosis
20
in endothelial cells. In this study, we simulated hyperglycemia by incubating HERCs with 30 m
Among the cellular senescence-related mechanisms, p53 and Rb pathways have been extensively studied and have been shown to be involved in the pathogenesis of cell senescence in different cell types. 23 p53 is an important nuclear phosphoprotein closely related to the abnormal cellular proliferation and is reported to promote the cellular level of ROS to induce cellular damage. 24 The activation of p21 can be induced by p53 and the activity of p53 can also be regulated by the feedback of p21, such as downregulating the activity of CDK2 to induce the arrest of DNA replication. 25 Rb/p16 is another cellular senescence signaling pathway. Rb arrests the cell cycle at the G1 phase by suppressing the activity of CDK2 to inhibit protein transcription and cell proliferation. 26 In the present study, we found that the cellular senescence in HERCs was significantly induced by stimulation with high glucose, which was accompanied by the activation of p53/p21 and Rb signaling pathway and significantly reversed by the introduction of FML, indicating that FML ameliorated the cellular senescence in HERCs induced with high glucose by inhibiting the p53/p21 and Rb signaling pathway.
The PI3K/Akt pathway is involved in the regulation of multiple physiological processes, such as the cell cycle, the inhibition of apoptosis, anti-oxidation, angiogenesis, the activity of telomerase, and the synthesis of proteins.
27
Recently, the regulation of Akt phosphorylation on the activity of telomerase by inhibiting the phosphorylation of human telomerase catalytic subunit (hTERT) has been widely reported.28,29 By activating the PI3K/Akt pathway in the endothelial cells overexpressed with hTERT, significant anti-senescence and anti-oxidation properties have been observed.
30
In the present study, we found that the Akt pathway in HERCs was significantly suppressed by treatment with high glucose and further activated by FML, indicating an activation effect of FML on the Akt pathway. Furthermore, the protective effects of FML against high glucose-induced cellular senescence were significantly abolished by the co-administration of a PI3K inhibitor, indicating that FML might exert the anti-senescence effects by activating the PI3K/Akt pathway. A representative sketch of the underlying molecular mechanism is shown in Figure 10. A representative sketch of underlying molecular mechanism.
The treatment options for DR are still very limited. Clinicians typically utilize laser photocoagulation and a few accessible drugs (VEGF inhibitors and other inflammatory inhibitors) to arrest the progression of the problems in the early stages of DR.30,31 This study provided promising data that FML could attenuate the high glucose elicited senescence of retinal endothelial cells. Vascular degeneration is one of the major pathological features of DR. The effect of FML on endothelial senescence implies that this compound could have the potential to prevent the degeneration of retinal endothelial cells. FML eye drops are typically prescribed to treat eye inflammatory conditions, it is approved to be an easy and safe choice, these data supports that FML could have beneficial effects in the diabetic retinal vascular cells, and it could have a prophylactic role in the prevention of retinopathy in diabetic condition.
The limitation of the current approach has been addressed. All test results in this study are based on the isolated HERCs in vitro in dishes, and the high glucose-induced senescence of HERCs is suitable to investigate the molecular mechanism of DR, 32 but it may be an ideal model to mimic the pathology of DR. DR is a complex condition ranging from mild and moderate to the advanced stage. Pathologically, its development often involves many neurological changes and their interaction with vascular components. 33 Recently, several DR animal models have been established. 34 Future in vivo studies in a DR animal model and clinical studies are warranted to investigate the effectiveness of FML in the prevention of DR.
Taken together, these findings reveal that FML inhibited high glucose-induced cellular senescence through the Akt/p53/p21 signaling pathway in HERCs. This study implies FML compound may have the potential in the modulation of diabetic retinopathy.
Footnotes
Author contribution
Xuemei Zhou and Dejing Shi contributed to the conceptions of the study, data analysis, and manuscript preparation.
Xuemei Zhou, Lifeng Wang, Zhongwei Zhang, Jing Liu, Qun Qu, and Yuanyuan Zu performed the experiments.
All the authors have read and approved the submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the fund from Weihai Municipal Hospital, Cheeloo College of Medicine, Shandong University (FH-2021-JY03).
Ethical approval
This study is approved by the ethical committee of Shandong University.
Data availability
Data are available upon reasonable request to the corresponding author.
